Abstract
Introduction
Patients with chronic obstructive pulmonary disease (COPD) often experience severe physical limitations and psychological distress, which can lead to a deterioration in quality of life (QoL). Telemonitoring (TM) may improve QoL and reduce the number of hospitalizations and readmissions, but results from previous studies have been conflicting. The aim of this study was to assess the effect of TM on QoL in patients with moderate to severe COPD recruited during hospitalization for acute exacerbation (AECOPD).
Methods
We conducted a randomized controlled trial at Silkeborg and Viborg Regional Hospitals in Denmark. Participants were recruited during hospitalization for AECOPD and randomized to a six-month telemonitoring service in addition to standard COPD care or standard COPD care alone. Patients were followed for 24 months. QoL was measured by the Hospital Anxiety and Depression Scale (HADS), and St Georges Respiratory Questionnaire (SGRQ) at 3-, 6-, 12-, and 24-months follow-up. The main outcome was QoL at 6 months.
Results
In total, 101 patients were randomized to the TM intervention and 97 to standard care. The between-group difference in SGRQ at 6 months was −2.0 (−8.5; 4.5), in HADS-Anxiety −0.3 (−2.0; 1.4) and in HADS-depression 0.2 (−1.0; 1.4) corresponding to no significant difference in health-related QoL for patients receiving TM compared to standard care. No difference was seen at 12–24 months follow-up either.
Discussion
TM in addition to standard care did not improve QoL in patients with moderate to severe COPD. Other means of improving management and QoL in severe COPD are urgently needed.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is the third most frequent cause of death and the fifth most frequent cause of morbidity worldwide. 1 In Denmark, up to 270.000 people have clinically significant COPD, of whom 40,000 have severe disease. 2 Patients with COPD often experience severe physical and psychological distress leading to a deterioration in quality of life (QoL). A subgroup of patients experiences recurrent acute exacerbations of COPD (AECOPD). Some exacerbations are severe and result in hospitalization, which is associated with deterioration in QoL, high risk of early readmission, and increased mortality. 3 Based on data from the Danish national COPD-registry, 30-day mortality after hospitalization for AECOPD is 12–18%. 4 Disease severity, and the associated fatigue, shortness of breath and other physical limitations have an impact on perceived QoL.5,6 In addition, anxiety and depression can lead to reduced QoL in patients with COPD. 7
Technological developments, such as TM have been integrated into healthcare systems.8,9 TM can be defined as “Digitally supporting healthcare services from a distance”, and involves the use of telecommunication technology to transfer healthcare information between patients and healthcare professionals. 10 Home TM of symptoms and physiological parameters may allow hospital staff to respond to signs of a COPD exacerbation, that may require treatment, and in some cases hospitalization. Some previous studies have shown that TM as part of the management may improve the QoL for patients with COPD, 11 while other studies showed no effect of TM, and the overall the results are conflicting.12–14 Research is sparse on the effect of TM on QoL on long-term follow up and further studies are needed to evaluate the impact of TM on QoL for patients with COPD. 15
The aim of the present study was to assess the effect on QoL of the addition of TM to usual care in patients with severe and very severe COPD, recruited during hospitalization for AECOPD.
Methods
Setting
The study took place at Silkeborg and Viborg Regional Hospitals in Denmark, with a catchment area of approximately 190.000 citizens. The Danish healthcare system is tax financed and provides free coverage for all Danish residents. 16 General practitioners (GPs) initiate diagnostics and act as gatekeepers to specialized healthcare. The majority of patients with COPD have follow-up in general practice, but many patients with severe disease, and most patients with very severe disease and/or frequent exacerbations have hospital-based follow-up. 17 The primary outcome for this telemonitoring study was hospitalizations in the six-month intervention period as reported previously. 18 QoL at 6 and 12 months was a prespecified secondary outcome and is the focus of the present study.
Study design and participants
We conducted a randomized controlled trial with 6 months’ intervention and 24-months’ follow-up of patients with COPD. Patients were recruited during hospitalization for AECOPD and the inclusion took place between March 2011 and December 2016. Follow-up was completed in 2018.
We included patients who fulfilled the following criteria: - Age ≥40 years. - Diagnosis of COPD according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria.
19
- Post-bronchodilator FEV1 < 50% predicted prior to hospital admission or during hospitalization for patients with no pulmonary function test available. - Informed consent to participate.
Exclusion criteria: - A diagnosis of asthma, a terminal oncological, liver-, kidney- or heart disease, unstable heart disease or invalidating mental health disease. - Current drug or alcohol abuse. - Inability to communicate in Danish. - Inability to perform the monitoring because of cognitive impairment - Participation in other research projects.
Randomization
Randomization was performed digitally as 1:1 allocated randomization with blocks of four as a telephone randomization service provided by Aarhus University, Denmark. Patients on long-term oxygen therapy (LTOT) were allocated equally to the TM group and the control group (CG).
Standard COPD treatment
All patients received medical treatment for COPD according to national and international guidelines.20,21 Within 2 days after discharge, all patients received a home-visit by a respiratory nurse and were instructed in the medical treatment, including inhaler techniques.
The TM intervention
The intervention group received home TM (Tunstall HealthCare’s telemonitoring equipment) for 6 months in addition to standard COPD treatment. The participants were instructed in the use of the TM equipment by a respiratory nurse at a home visit within 2 days after discharge from hospital. The patients could contact the staff for additional information as needed, and in case of irregularities in the monitoring, including lack of measurements, the patient would be contacted by the study nurse. The TM consisted of measurement of oxygen saturation, heart rate, lung function and body weight, as well as symptom assessment using a questionnaire focusing on dyspnea, cough, sputum volume and color. This information was sent every weekday for the first month, and three times a week during the following 5 months. An individual normal range for each patient was established for heart rate, oxygen saturation, lung function and weight. Based on the patient’s reported measurements and symptoms, three categories were defined: green, yellow and red. Green indicated stable condition, yellow indicated a change in symptoms, and red indicated a change in physiological readings. The respiratory nurses at the hospital evaluated the incoming data and decided, in collaboration with a respiratory physician, whether additional treatment or follow-up was needed. Patients were contacted within 2 hours if the category changed from green to yellow, and within 1 hour if the category changed from green to red or yellow to red.
Prior to the initiation of the randomized controlled trial, a pilot trial was conducted including five patients who were monitored for 1 month using the same equipment to ensure the feasibility of the monitoring.
Monitoring
The data collected at baseline included demographics; smoking status; civil status; spirometry, body mass index, Medical Research Council (MRC) dyspnea score, and scores on the Hospital Anxiety and Depression Scale (HADS). All patients, both the intervention group and control group, had follow-up at the pulmonary outpatient clinic 3, 6, 12, and 24 months after inclusion. At each follow-up visit, the patients underwent spirometry, six-minute walk test, dyspnea assessment using MRC, measurements of vital signs and assessment of medication and exacerbations.
Outcomes
QoL was assessed by the patients using HADS and the St Georges Respiratory Questionnaire (SGRQ).22,23 HADS is a 14 items instrument developed to measure anxiety and depression and consists of seven items for anxiety and seven items for depression. Each item is scored on a scale with four alternatives ranging between 0–3 and a total score ranging between 0–21 with anxiety and depression assessed separately. Cut-off values are 0–7 for non-cases, 8–10 for borderline/mild cases and ≥11 for moderate and severe cases. 22
SGRQ is a disease-specific measurement for respiratory QoL, which consists of 50 items focusing on different aspects of the disease. Scores range from 0 to 100, with higher scores indicating more limitations, and a minimal clinically important difference of 4 units. 23
HADS was collected at baseline and at 3-, 6-, 12-, and 24-months’ follow-up. SGRQ was collected at 3-, 6-, 12-, and 24-months’ follow-up. All QoL data were patient reported.
Minimal clinical important difference (MCID) in COPD is −4 for SGRQ 24 and -1.8 to −1.3 for HADS-Anxiety and −1.7 to −1.5 for HADS-Depression. 25
Statistical analysis
The sample size for the study was calculated with hospitalizations for COPD exacerbation as the main focus and used the number of days in hospital for a previous cohort of 143 patients admitted to hospital with COPD exacerbation. Based on sample size estimates from previous studies of telemonitoring and QoL,26–28 the included 198 patients in our QoL study allow detection of differences above the MCID for SGRQ and HADS.
Baseline characteristics were analyzed using descriptive statistics presented as counts and frequencies when categorical and means with standard deviations when continuous, and compared using Fishers exact test when categorical and unpaired t-test when continuous.
SGRQ and HADS at each time point during follow up were compared using Student’s t-test.
Intra-group differences in mean SGRQ scores were calculated (3 months relative to 6-, 12- and 24-months follow-up). Similarly, the intra-group differences in mean HADS were calculated (baseline relative to 3-, 6-, 12- and 24-months follow-up). Next, inter-group differences of these intra-group differences were compared between the telemonitoring and the control group using Student’s t-test. Model assumptions were fulfilled.
Analyses were conducted using the intention-to-treat principle, with participants remaining in the originally assigned group and included in the analyses regardless of their level of adherence to the intervention. For the analyses of QoL, we used all available QoL data and did not use imputation of missing data. Missing data were addressed by comparing participants with and without available QoL data at 6 months with respect to age, gender, social status, FEV1, BMI and GOLD class. p-values <0.05 were considered statistically significant.
Data were analysed using STATA software version 17.0 (Stata Statistical Software, College Station, TX, USA).
Ethical considerations
Patients received verbal and written information prior to inclusion and gave written informed consent to participate. All collected data were anonymized and stored in a secured database in accordance with the provisions of the Danish Data Protection Agency. The study was approved by the Regional Committee on Health Research Ethics (file no 1–16-02–134-10). The trial was registered in ClinicalTrials.gov (Identifier NCT02615795).
Results
Patient characteristics
A total of 533 patients fulfilled the inclusion criteria of whom 222 agreed to participate in the study. One-hundred and 10 were randomized to the TM group and 112 to the control group. 24 of these patients did not fill in any QoL questionnaires, nine in the TM group and 15 in the control group. Thus, the study population consisted of 198 patients with available QoL data; 101 patients in the TM intervention group and 97 in the control group (Figure 1). CONSORT diagram.
Baseline characteristics of trial participants.

aFEV-1: Forced Expiratory Volume in the first second. Best obtained value during observation.
bGOLD: Global Initiative for Chronic Obstructive Lung Disease, classification of severity of airflow limitations. Best obtained value.
cMRC: Medical Research Council Dyspnoea score. Best obtained value.
dHADS: Hospital Anxiety and Depression scale. Assesses anxiety and depression: ≤7 are normal, ≤11 indicate significant anxiety/depression.
eSGRQ: St. George’s Respiratory Questionnaire. The lowest possible weight is zero and the highest is 100.
retrieved at 3-months follow-up.
Effect on QoL
The between-group difference in SGRQ at 6 months was −2.0 (−8.5; 4.5), in HADS-Anxiety −0.3 (−2.0; 1.4) and in HADS-depression 0.2 (−1.0; 1.4) corresponding to no significant difference in health-related QoL for patients receiving TM compared to standard care.
Between group differences in quality of life, as assessed by HADS* (at baseline, 3, 6, 12 and 24 months) and SGRQ** (at 3, 6, 12 and 24 months) for patients who underwent telemonitoring compared to usual care.
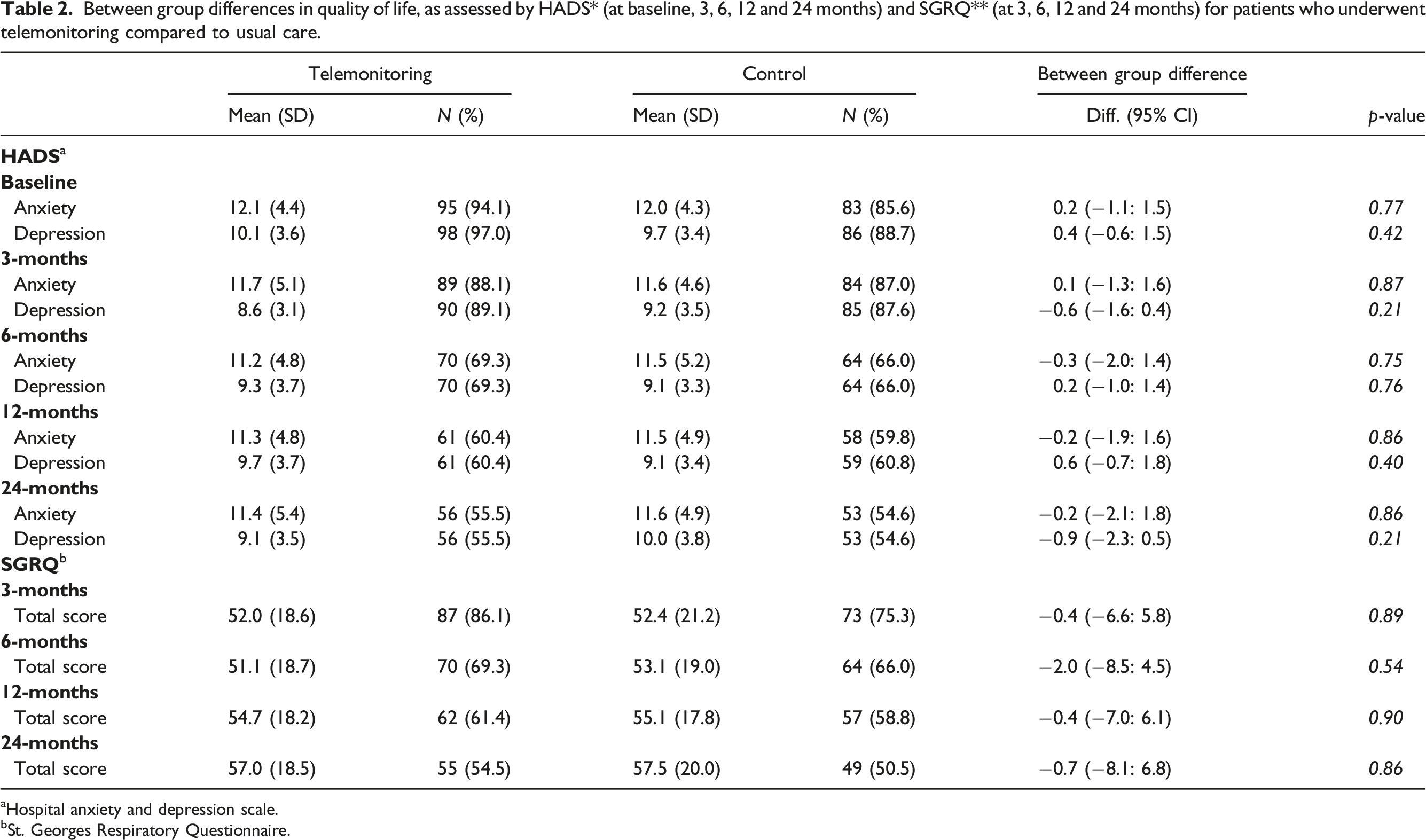
aHospital anxiety and depression scale.
bSt. Georges Respiratory Questionnaire.
Intra-group difference in quality of life as assessed by HADS* (from baseline to 3, 6, 12 and 24 months) and SGRQ** (from 3 months to 6, 12 and 24 months) for patients who underwent telemonitoring compared to usual care and difference-in-differences between groups.

aHospital anxiety and depression scale
bSt. Georges Respiratory Questionnaire
Overall, the results did not indicate a significant change between groups in health related QoL for patients who received the TM intervention, compared to those who received standard COPD treatment.
No difference was observed in baseline clinical characteristics between the participants with available QoL data and missing QoL data, neither for the entire cohort nor within the telemonitoring group or the control group in separate analyses.
Discussion
In this randomized controlled trial of patients with moderate to severe COPD, TM in addition to standard care did not improve QoL as assessed by HADS and SGRQ. No significant inter-group changes were seen in HADS and SGRQ at 3-, 6-, 12-, or 24-months’ follow-up, and only minor intra-group differences were seen, not likely to be attributed to the TM intervention.
Some previous studies have indicated that patients with COPD may benefit from TM.26,28
Koff et al. reported improvement in the total SGRQ score in favor of the intervention group after a 9-month disease management program for patients with severe or very severe COPD. 26 Patients came from primary care and pulmonary specialty clinics, had either moderate to very severe COPD, or had experienced a hospital admission within the past year. In addition to TM, the intervention group received proactive integrated care focusing on management of symptoms and medication, which may be a cause of the observed improvement in QoL rather than the TM itself. The study by McDowell et al. showed improvement in SGRQ and HADS scores after 6 months of home TM used in addition to usual care in patients with moderate to severe COPD recruited from a specialist respiratory service. 28 Patients in the intervention group had significantly more contacts the respiratory team, which may in part explain the improvement in QoL, and the QoL of participants in the intervention group was low, as assessed by baseline SGRQ score at 63.6, whereas it was 49.2 in the study by Koff et al. 26 and 52.0 in our study. Both studies showed an improvement of QoL, regardless of the different baseline SGRQ scores. This may indicate that improvement in QoL by TM are more likely if QoL at baseline is low, as seen in the study by McDowell et al. or that it may have effect when combined with other interventions as in the study by Koff et al. A study by Tupper et al. showed that only the patients with poorest QoL experienced an improvement with TM, 29 which also points toward the impact of baseline level of QoL as well as the overall standard of usual care.
Pinnock et al. conducted a 12 months’ researcher blinded study, where TM was given in addition to standard clinical care. 30 Participants were recruited through general practices if they had at least one hospitalization for COPD exacerbation during the previous year. The addition of TM service provided no improvement in QoL, and the authors attribute this to equal access to clinical care in the intervention group and CG. The study by Tupper et al., which was also conducted in Denmark, with standard care comparable to our study, showed no difference in QoL between the TM and CG. 29 A study by Vianello et al. also failed to show improvement in QoL with addition of TM to usual care, and concluded, that TM is not effective in COPD when medical services and standard care are well established. 31 In summary, existing literature with interventions focusing on more than just TM, seem more likely to improve QoL in patients with COPD, compared to studies of TM as the only intervention in addition to comparable standard care.
Patients who received TM in our study did experience some within-group differences, mainly
A significant increase in SGRQ over time, equivalent to a decline in QoL, likely to reflect increasing disease burden over time.
For the present study, we screened 533 eligible patients of whom 222 (42%) were included in the study, which is similar to other studies of telemonitoring in COPD 29, 30 reflecting rather difficult recruitment.
Strengths and limitations
The strengths of the present study were the randomized design and patient recruitment following hospital admission. The findings were based on self-reported QoL data using two standardized generic and disease-specific questionnaires. We used identical standard of care in the intervention and control groups, allowing the assessment of the impact of additional TM. Consecutive follow-up periods enabled the assessment of time factors in a potential effect of telemonitoring, as well as changes after discontinuation of the intervention.
Some limitations should be addressed. Patient recruitment during hospitalization instead of in stable phase, might have impacted our findings, if QoL was more severely affected following hospitalization. Since TM was an active treatment, neither patients nor respiratory nurses could be blinded to treatment allocation. QoL questionnaires were filled in by the patient at outpatient contacts in the presence of the respiratory nurse who were able to help with the questionnaire if needed. This may have influenced the outcome if the patients answered the questions differently in the presence of the staff. However, this may be the case for both the TM group and the CG, and the overall results showed no difference between the groups. The timing of the baseline assessment for HADS and SGRQ differed; baseline HADS was collected at discharge from hospital, allowing early QoL assessment, while SGRQ was assessed for the first time at 3 months follow up, and thus did not capture early changes after discharge.
The findings of this study are likely to be transferable to patients with moderate and severe COPD in hospital settings in similar health care systems. In line with findings of previous studies, TM did not improve QoL and our study contributes to the existing evidence base by assessing the effect of TM after long-term follow-up. It may still be relevant to consider TM as part of the treatment for patients with COPD, if other studies show beneficial effects on other parameters, since the study showed no signs of deterioration in QoL as a result of the intervention. More research is needed on the factors influencing QoL for patients with COPD.
Conclusions
This randomized controlled trial showed no improvement of patient-reported QoL based on addition of TM to best practice management of patients with moderate to severe COPD, neither on short-term nor long-term follow up.
Footnotes
Acknowledgements
The authors would like to thank the patients who participated in the trial and the staff at the participating departments at Silkeborg and Viborg Regional Hospitals. The authors also kindly acknowledge biostatistician Asger Roer Pedersen, data manager Andrew Bolas and senior researcher Charlotte Appel for their contributions to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chronic Diseases, Ministry of Health and Prevention, Denmark.
Data availability
The data that support the findings of this study are not publicly available and were used with permission from the Regional Hospital Central Jutland for the current study. Data are available upon reasonable request and with permission of the Regional Hospital Central Jutland.
