Abstract
Introduction
Interstitial Lung Diseases (ILD) affect the lung parenchyma and are often complicated by respiratory failure (RF) and impaired physical activity. High Flow Nasal Cannula (HFNC) has proved effective in other disease entities with RF. The aim of this study is to investigate the effect of domiciliary HFNC in ILD on dyspnea and walking distance.
Methods
A 6 weeks cross-over study with domiciliary HFNC-treatment/6 weeks’ observation in ILD-patients requiring ambulatory oxygen therapy or with newly prescribed (within 12 months) long term oxygen therapy. Patients were advised to use HFNC 8 h/day, recommended night-time use. Body phletysmography; 6-min walk test (6MWT) including BORG-score, oxygen saturation (SO2) at start, minimum SO2 and time to recovery after 6MWT; arterial blood gasses; modified Medical Research Council (mMRC)-score; quality of life, by the St George Respiratory Questionnaire (SGRQ) and QoS, by the Richards-Campbell Sleep Questionnaire (RCSQ) were investigated at baseline; six weeks and 12 weeks.
Results
10 patients were included; one later withdrew consent. Patients used HFNC between 8-<1 h/day. There were no differences in lung function; blood gasses; SGRQ or RCSQ over the observational period). Walking distance improved significantly (393–441 m p = 0.049) as did time to recovery (3.4–2-2 min, p = 0.001). When correcting for HFNC use (hours/day) significant improvement was also seen in mMRC-score (p = 0.035) and minimum saturation during 6MWT (p = 0.01).
Conclusion
Despite a very heterogenous group and no effect on quality of life and -sleep, the study indicates an improvement in dyspnea and physical ability of HFNC in ILD patients.
Keywords
Introduction
Interstitial lung diseases (ILD) is the common denomination of a heterogenous group of more than 200 rare diseases, characterized by disperse fibrotic and/or inflammatory abnormalities of the lung parenchyma. 1 The incidence is estimated to be three to nine per 100.000/year. 2 A common clinical finding in ILD-patients is dyspnea, caused by multiple pathophysiological changes, amongst these decreased lung elasticity. 3 This results in reduction in lung compliance and through increased respiratory rate, a work overload of the thoracic muscles. 4 Increased dyspnea is commonly associated the reduction of the single breath diffusion capacity of carbon monoxide (DLCO). 5 Symptoms often progress, from mild dyspnea over significant oxygen desaturation on exercise, resulting in exercise limitation, to persistent respiratory failure. 6 Both in exertional desaturation and persistent respiratory failure oxygen treatment is indicated, either as ambulatory oxygen treatment (AOT) or long-term oxygen treatment (LTOT) respectively. 7 Concomitantly with increases dyspnea a progressive decrease in exercise capacity is seen. 8
High-flow, humidified, nasal cannula with optional supplementary oxygen delivery (HFNC) has evolved in recent years, with a growing body of evidence of reduced respiratory resistance; decreased work load of breathing; improved pulmonary compliance; and recruitment and mucus clearance in adults.9–11 HFNC is primarily established in acute and critical care settings for treating mild to moderate acute hypoxic failure 12 and ventilator weaning. 13 However, there is increasing evidence that domiciliary HFNC is beneficial in chronic respiratory diseases, primarily in obstructive lung diseases. Here reduction in number of exacerbations prolonged time to first exacerbation; 14 improved Forced Expiratory Volume in the first second (FEV1) and mucus retention challenges, 15 in addition to a reduction in respiratory rate, 16 PaCO2 17 and increased exercise performance 18 has been shown, in addition to improved health-related quality of life (HR-QoL) and -sleep quality.14,19
Patients with ILD experience exertional hypoxemia that can be refractory to conventional oxygen therapy and may experience discomfort and drying of mucosal tissue at high air flows and may therefore benefit from the heated and humidified air. Theoretically, patients may also benefit from the minor PEEP effect HFNC offers, 20 which in addition to the heat and humidification may offer patients an accurate and adequate oxygen supply which may mitigate severe hypoxemia. In ILD, literature is sparse. However, in the acute setting, there is good tolerability of HFNC in patients with ILD. 21 In an out-patient setting these authors are aware of two studies: a Japanese study showing a significant increase in endurance time, when compared to training whilst oxygenated through a venturi mask system, although no effect was seen on exercise capacity 22 and a German study on patients with interstitial pulmonary fibrosis, showing improved inspiratory capacity. 23 There are, however, no studies investigating the effect on patients’ symptoms and quality of life, which has been shown to improve in other patient groups with chronic respiratory diseases.
Hence, the primary aim of this study is to investigate the effect of HFNC on patients’ sensation of dyspnea and health-related quality of life (HR-QoL). Secondary outcomes were to investigate sleep quality, lung function, walking distance, including desaturation and time to recovery after exercise.
Methods
In this prospective cross-over study patients with ILD were recruited from the respiratory out-patient clinic at Aalborg University Hospital, Denmark, in the period 1 January 2019, till 30 June 2021. Recruitment was delayed due to the COVID-19 pandemic, with a temporary stop for recruitment from April 1 till 30 November 2021, as research personnel was included in primary care during that period. The local ethical committee was advised of the delay.
Patients were informed according to the Helsinki Declaration and written informed consent was obtained prior to inclusion. The study was approved by the Nordic Jutland Region Ethical Committee (N-20180070) and registered at ClinicalTrials.gov (NCT038166722).
Inclusion criteria were: i) Diagnosed with any ILD according to diagnostic workup recommended in international guidelines ii) prescribed AOT or LTOT <12 months prior to inclusion iii) age >18 years old and iv) ability to understand oral- or written information and to give informed consent. Exclusion criteria were: i) life expectancy less than 3 months ii) pneumonia or exacerbations of ILD less than 3 months prior to inclusion.
At the baseline visit age, sex, body mass index (BMI), time since ILD diagnosis (months), time with LTOT, smoking status (present/previous smoker for >6 months/never smoker), 24 pack years, 25 oxygen flow, in liters per minute, and number of exacerbations in the preceding year were recorded. A body plethysmography, registering total lung capacity (TLC), forced vital capacity (FVC), residual volume (RV) and inspiratory capacity (IC), as well as diffusion capacity of the lung of Carbon Monoxide (DLCO) was performed. Furthermore, a six-minute walk test (6MWT) was performed, according to the ATS guidelines, 26 monitoring peripheral oxygen saturation (SO2) with Vyntus® Walk (CareFusion, Hoechberg, Germany), as well as recording of time to SO2-recovery to baseline after 6MWT and performing the BORG scale 27 at the beginning and the end of the procedure. In addition, modified Medical Research Council (mMRC) score, 28 HR-QoL by the St George Respiratory Questionnaire (SGRQ) 29 and quality of sleep (QoS) by the Richards-Campbell sleep questionnaire (RCSQ) 30 were registered. Lastly, an arterial blood gas was drawn on ongoing oxygen therapy. The patients were allowed 30 min’ rest before the arterial blood gas was drawn. From the blood gas pH, PaO2, PaCO2 (both kPa) and SaO2 were registered.
By the Randomizer App (Microsoft 2019) patients were then randomized to either a 6 weeks’ observational period or 6 weeks’ treatment with HFNC as add on to previously prescribed therapy at baseline (hereafter referred to as period 1, (P1)). In those randomized to HFNC, a flow of at least 30 L/minute (L/min) and a temperature of 37o Celsius were target settings at baseline. Initially, flow was titrated to patients’ highest acceptable flow. Meanwhile, the patients’ oxygen flow was kept at the prescribed level, for both LTOT/AOT therapy, allowing 15 min for oxygen saturation steady state to occur. Following this, oxygen flow was titrated, if necessary, till the patients reached the target saturation prescribed by the treating physician, again allowing a 15 minute-period for steady state to occur. After 30 minutes’ treatment on stable conditions in the outpatient clinic, another arterial blood gas analysis was performed. Patients were instructed in the use of HFNC whilst using it for the first time in the outpatient facility. In line with recommendations
31
from previous studies patients were instructed in 8 h’ diurnal use, preferably during the night. After 6 weeks the procedures of the diagnostic work up; Body plethysmography, 6MWT, mMRC, SGRQ, RCSQ and arterial blood gas were repeated and the patients previously designated to the control group were now treated with HFNC, and those in the HFNC-group were in the control group. For those in the HFNC-group, the procedure for adjusting oxygen administration described above was performed, and recommendations for use also as previously described. This period is hereafter referred to as period 2 (P2). The study procedure is summed up in Figure 1. Flow diagram of study design.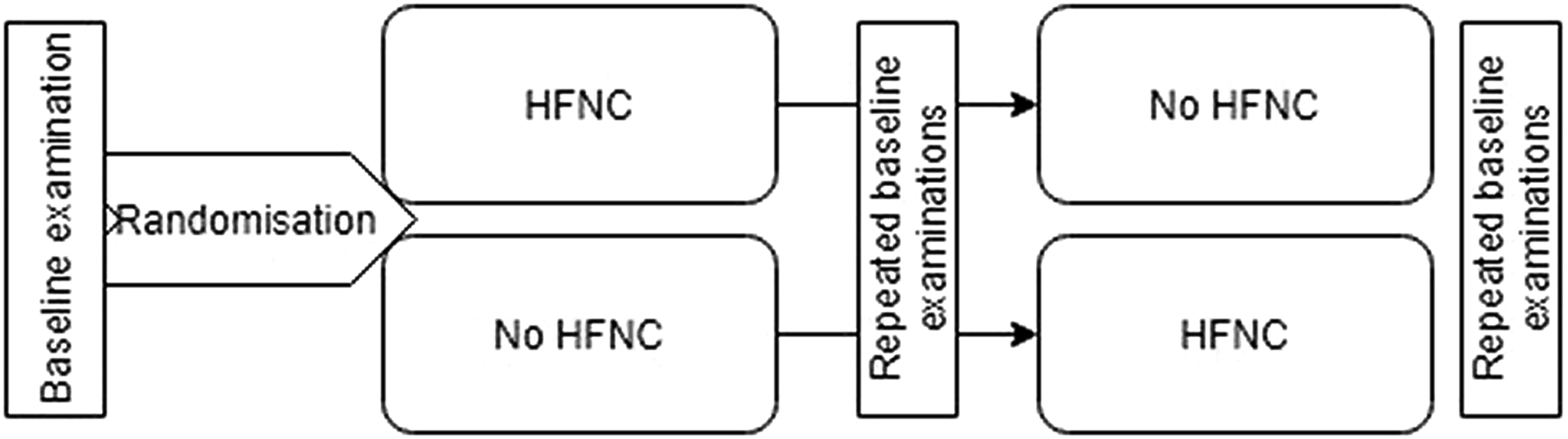
At the end of the 6-week treatment period patients were asked of their self-perceived effect of HFNC.
During study participation patients were free to use all usual healthcare services and medications.
The High Flow device used in this study was a myAIRVOTM 2 (Fisher&Paykel Healthcare, Auckland, New Zealand). The device and utensils were delivered to the patients and serviced by VitalAire (AirLiquide, Silkeborg, Denmark), who also re-instructed the patients in the use of HFNC in their homes. All patients were offered to continue treatment with domiciliary HFNC after end of treatment.
Statistics
Baseline information was described in actual numbers, percentages or mean (standard variation), whichever applied. A paired t-test was used to compare data before and after HFNC-treatment and, in addition, binary logistic regression analysis, adjusting for time of use of HFNC, was applied. Kolmogorov-Smirnov’s test was used to compare SGRQ- and RCSQ data before and after HFNC. Data analysis was performed using IBM®SPSS®statistics, version 27 (IBM, New York, USA).
Results
Diagnoses of patients included in the study.

HFNC was used an average 6.5 h/24 h, (±1 h), range<1-8 h/24 h. All patients used a flow of 30 L/minute during treatment and all patients used the device at a temperature of 37oCelcius. Two patients on LTOT increased the oxygen flow rate during HFNC use, both with one L/minute. Six patients used supplementary oxygen whilst using HFNC; one patient using AOT used one L of oxygen during HFNC treatment. None of the patients required extra domiciliary back up due to difficulties with HFNC use.
Baseline characteristics, lung function, blood gasses and six-minute walk test of the study population, at baseline and after 6 weeks’ use of High Flow Nasal Cannula (HFNC). Results presented in mean (SD) where nothing else stated.

M/F: male/female; AOT: ambulatory oxygen; LTOT; Long term oxygen treatment; BMI: Body mass index; mMRC: modified Medical Research Council score; %: in per cent of expected value; FEV1: Forced expiratory volume in the first second; FVC: Forced Vital Capacity; IC Inspiratory Capacity; TLC: Total Lung Capacity: RV; Residual Volume; DLCO: diffusion capacity of the lung of carbon monoxide; PaO2: Partial arterial Pressure of Oxygen; PaCO2: Partial arterial pressure of carbon diaoxide; SO2: Oxygen saturation.
Regression analysis of independent variables, corrected for hours of use of High Flow Nasal Cannula.

a6-min walk test.
HR-QoL, measured by SGRQ was on average 38.4 (±5) before HFNC and 40.0 (±5) after HFNC (p = 0.7). In controls, P1, SGRQ was 38.0 (±5.3) and for control, P2, 38.1 (±3.9). There were no significant differences in SGRQ between baseline and after HFNC (p = 0.3), nor between baseline and control periods (P1; p = 0.8 and p2; p = 0.6). There was a large diversity in results, patients in between (Figure 2(a)). The development in St George’s Respiratory Questionnaire (SGRQ) (a) and Richard-Campbell’s sleep questionnaire (RCSQ) (b) before and after High Flow Nasal Cannula (HFNC) treatment in individual patients.
QoS, measured by the RCSQ, an average score of 76.5 (±22.0) at baseline and 75.6 (±13.1) after HFNC was seen. Controls in P1 had an average score of 75.6 (±19.2) and controls in P2 had an average score of 87.7 (±10.8). There was no significant difference between RCSQ before and after HFNC (p = 0.5), nor between baseline and P1 (p = 0.9), however controls in P2 had significantly better sleep quality, compared to baseline (p = 0.042). As with SGRQ, large inter-personal variation was seen in QoS (Figure 2(b)).
Figure 3 shows patients’ self-perceived effect of HFNC-treatment. Three of four patients who indicated increased ease of breathing also increased their walking distance after HFNC. Patients 2, 5, seven and eight in Figure2(a) and (b) felt an ease in the work of breathing. The four patients who found the treatment beneficial chose to continue treatment after the end of study. Patient statements about perceived effect of High Flow Nasal Cannula treatment, in percent of the total population.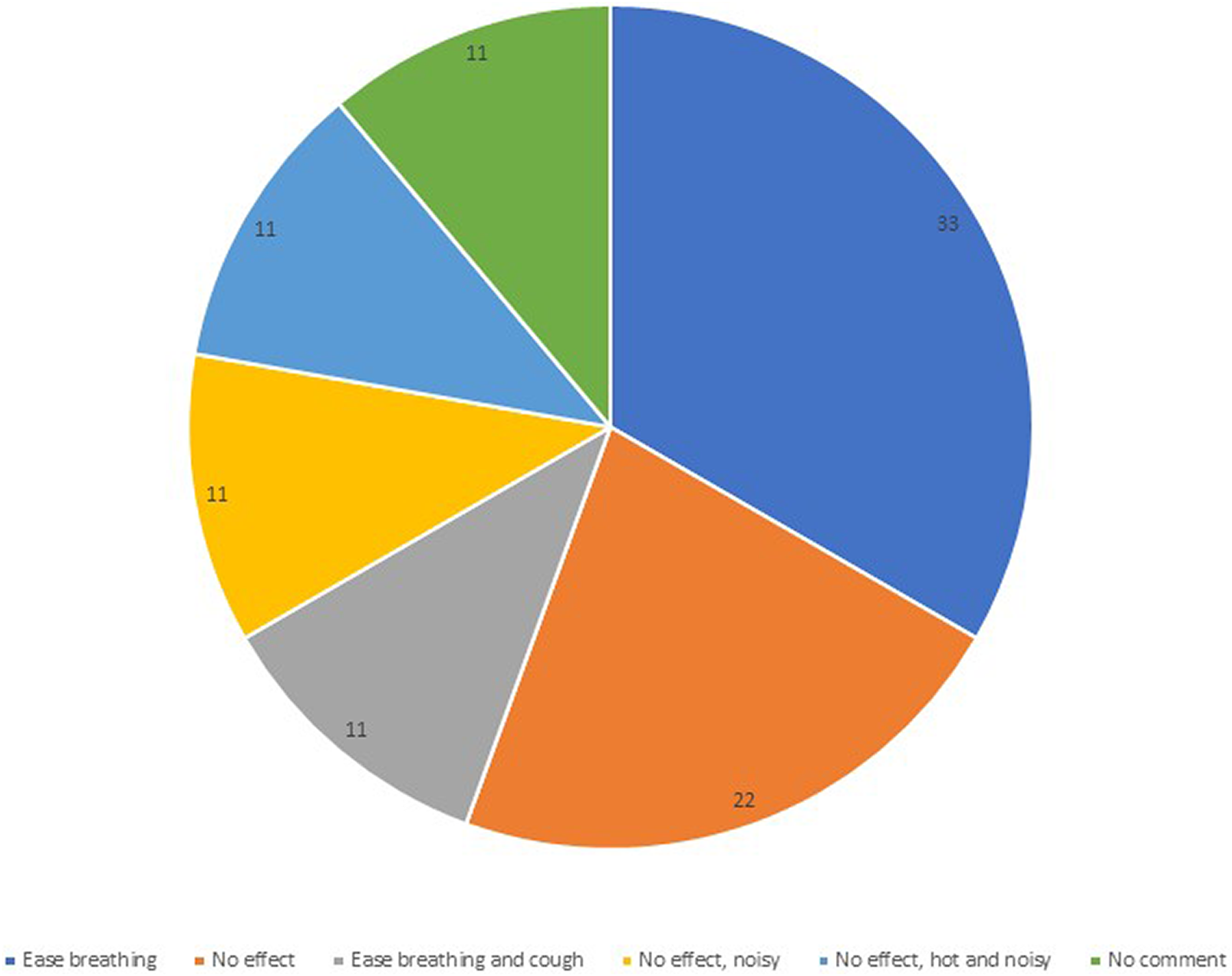
At the 1-week interview none of the patients indicated difficulties in using the device and the service partner had no unscheduled visits to the patients during the study period. There were only minor adverse events, i.e. experience of mild rhinorrhea in the initial phase of the treatment, only experienced by patients in LTOT-treatment and three patients were initial bothered by the heat from the device, but all customized. One patient experienced an exacerbation during the treatment period which did not require hospitalization.
Discussion
This study indicates that domiciliary HFNC may increase walking distance and shorten recovery after physical activity, and corrected for hours of use of HFNC, improve minimum oxygen saturation during 6MWT and mMRC-score.
We found significant increase in walking distance and reduced recovery time after exercise after the use of HFNC, the significance increased after correction for hours of use of HFNC. This is in consistency with recent findings by Arizono et al. and Chikhanie et al.,32,33 however in contrast to the findings of Suzuki et al. 22 In all three trials HFNC was only used during exercise and as such the study setups differ from that of the present study. However, in the study by Suzuki et al., the authors themselves do conclude that the missing effect during endurance testing, which was used in this study, should not rule out the effect in another study set up. 22 As such, the type of physical testing performed in these studies could influence results, which should be kept in mind, when designing future studies.
HFNC has previously been shown to decrease inspiratory resistance and expiratory resistance, reduce the trans-diaphragmic tension and, in general, to decrease the respiratory work load34–36 in patients with chronic obstructive pulmonary disease. Although the pathophysiology is greatly different between the two diseases, ILD-patients would hypothetically also benefit from a decrease in inspiratory resistance and reduced trans-diaphragmatic tension, however, whether this can explain our findings is outside the scope of our study. Mechanistic studies in ILD patients are needed to investigate this.
Dyspnea, measured by mMRC-score improved after HFNC-treatment, when correcting for the actual use of HFNC. Literature is sparse on domiciliary HFNC-treatment in ILD, and as such, this is, to these authors knowledge, the first study to indicate a beneficial effect of HFNC on other than exertional dyspnea. The reduction in respiratory work by HFNC in ILD 22 described above is hypothetically also the underlying mechanism for the amelioration seen in mMRC-score. Further, larger, and longer studies are needed to confirm this.
In this study we found no changes in lung function after 6 weeks’ treatment with HFNC. Patients with ILD are mainly affected on the static lung function parameters 1 and time since diagnosis was on average more than 4 years in the included patients. As such chronic changes in the pulmonary tissue may well have occurred.
Interestingly, we found a very variable effect of HFNC on patients’ QoS, and QoS did not seem to influence patients’ perceived effect of HFNC. Previously, HFNC-treated patients with COPD have expressed improved QoS. 19 Although a recent study concluded that patients with ILD have poor sleep quality, 37 there may be differences between the two disease entities. Furthermore, the RCSQ is not validated in domiciliary HFNC treatment, but the score was chosen as it included “noise” as a parameter affecting sleep. 30 Noteworthy is also that there was no consistency between a feeling of easier breathing and QoS in these patients. Neither were there any concordance between RCSQ and SGRQ. Both the area of perceived effect and effect on sleep in both HFNC and other home ventilatory treatment modalities need further investigation to be fully understood.
We found no amelioration in HR-QoL in this study. As for QoS, there was a large diversity in the effect of HFNC on the patients’ HR-QoL, measured by the SGRQ despite the patients’ interviews at the end of the study indicated that about half of the patients felt an amelioration of their work of breathing after HFNC.
A specific version of the SGRQ now exists for IPF, 38 still not published at the time of the study initiation, and this may affect results.
Only mild side effects were seen from the use of HFNC, similar to those seen in other patients groups treated with domiciliary HFNC. 14 Only one patient had an exacerbation during the study period and given that previous studies have found that between 5-19% of ILD patients experience an exacerbation per year, it is not more than expected. 39 This is in line with a previous qualitative study where patients with severe COPD expressed the ease of use of HFNC. 19 One patient used HFNC very little. The patient had not complained of any difficulties at the 1-week interview, nor had he made any complaint to the service company. When asked directly the patient had made private decisions for when to use the device, which lead to very little use. All in all, these observations indicate that HFNC is safe and easy to use in a domiciliary setting.
Limitations are multiple in this study, some of those mentioned above. First and foremost, it is, however, the study size and duration. However, this was a pilot study, to explore potential future focus in clinical studies on domiciliary HFNC in ILS. Although results are merely indications, the results are valuable for designing future studies and indicate, that within this heterogenous group of patients, phenotypes exist to whom treatment with HFNC would be beneficial. Patients in this study had milder disease (need of AOT or only use of lower oxygen flows at LTOT use). It is not known whether results are applicable in patients with more severe disease.
The cost of HFNC should of course be taken into consideration when considering this as a long-term treatment for patients. To an even higher extent than for pharma, the cost for medico-devices varies from country to country 40 which makes this assessment difficult. Furthermore, to these authors’ knowledge, there are no studies on cost-effectiveness of LT-HFNC in ILD-patients. However, in COPD, long term studies of cost-effectiveness do indicate an economical advantage of LT-HFNC-treatment.41–43 This should, however, be investigated specifically in ILD.
We do not have any knowledge on the patients’ rehabilitation status. Theoretically, this could bias the study, as concomitant rehabilitation could improve patients’ physical ability in line with the improvement we have seen in this study, as previously indicated in a study by Chihara et al. 44 However, during COVID lock down there was no organized rehabilitation available for the patients and it was not accessible to patients again till Autumn 2021. Therefore, we do not consider it of major importance in this study. However, further studies should consider this parameter, as an exclusion criterion or a stratification parameter, depending on the aim of the study, in order to avoid bias.
In addition, there are methodological limitations. There is no clear evidence of how long domiciliary HFNC should be used in any patient group. Improvement in exacerbation rate has been seen for less than 2 hours per day, 15 and stabilization of HrQoL seen with six to 8 hours of use. 14 In COPD adherence to HFNC improved with nighttime use. 45 However, it is not known whether this also applies for ILD patients. The importance of avoiding side flow to achieve optimal effect of HFNC is well known, 46 however no other measure for optimal flow than securing saturation is available at present. It is therefore at the prescribing doctor’s discretion to ensure this, and it may affect outcome.
In conclusion, this study indicates that domiciliary HFNC treatment has potential for reducing dyspnea, increasing walking distance and diminishing recovery time in ILD, although no effect is seen on lung function, oxygenation at rest, HR-QoL, and QoS. Further and larger long-term studies should be initiated to investigate the effect of LT-HFNC on physical ability and HR-QoL in ILD.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fisher&Paykel Healthcare.
