Abstract
Objectives
A decrease of both diffusion capacity (DLCO) and Quality of Life (QoL) was reported after discharge in hospitalized COVID-19 pneumonia survivors. We studied three and 6 month outcomes in hospitalized and non-hospitalized patients.
Methods
COVID-19 pneumonia survivors (n = 317) were categorized into non-hospitalized “moderate” cases (n = 59), hospitalized “severe” cases (n = 180) and ICU-admitted “critical” cases (n = 39). We studied DLCO and QoL (Short Form SF-36 health survey) 3 and 6 months after discharge. Data were analyzed using (repeated measures) ANOVA, Kruskal-Wallis or Chi-square test (p < .05).
Results
At 3 months DLCO was decreased in 44% of moderate-, 56% of severe- and 82% of critical cases (p < .003). Mean DLCO in critical cases (64±14%) was lower compared to severe (76 ± 17%) and moderate (81±15%) cases (p < .001). A total of 159/278 patients had a decreased DLCO (<80%), of whom the DLCO improved after 6 months in 45% (71/159). However the DLCO did not normalize in the majority (89%) of the cases (63 ± 10% vs 68±10%; p < .001). At 3 months, compared to critical cases, moderate cases scored lower on SF-36 domain “general health” (p < .05); both moderate and severe cases scored lower on the domain of “health change” (p < .05). At 6 months, there were no differences in SF-36 between the subgroups. Compared to 3 months, in all groups “physical functioning” improved; in contrast all groups scored significantly lower on “non-physical” SF-36 domains.
Conclusion
Three months after COVID-19 pneumonia, DLCO was still decreased in the more severely affected patients, with an incomplete recovery after 6 months. At 3 months QoL was impaired. At 6 months, while “physical functioning” improved, a decrease in “non-physical” QoL was observed but did not differ between the moderate and severely affected patients.
Keywords
Introduction
As of 13 January 2022, the pandemic COVID-19, caused by the Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2), has resulted in over 312 million confirmed cases worldwide and about 5.5 million deaths. 1 Thus far, literature has focussed primarily on the epidemiology, pathogenesis, clinical characteristics and short-term outcome2,3 and relatively little is known about the longer-term consequences of the disease.
As Sars-CoV-2 primarily targets the lower airways, most patients who require medical attention suffer from respiratory symptoms, such as cough and dyspnoea.4,5 The majority of patients in need for hospitalization present with COVID-19 pneumonia, 4 radiologically characterized by wide-spread ground-glass opacities on chest computed tomography (CT) and chest X-ray.6,7 Approximately 5% of the admitted patients will develop severe respiratory failure and acute respiratory distress syndrome (ARDS), which necessitate Intensive Care Unit (ICU) admission.8,9
It has been suggested that in view of the course of disease in other coronavirus pneumonias, such as SARS-CoV and Middle East Respiratory Syndrome-CoV (MERS-CoV),3,10,11 a significant proportion of COVID-19 patients may suffer from residual pulmonary function impairment. 12 Moreover, previous studies on SARS-CoV, MERS- CoV and ARDS showed that the clinically most severely affected patients were most at risk for long-term sequelae.10,12 Literature on longer-term follow-up of pulmonary function in COVID-19 pneumonia survivors is still scarce and primarily focussed on patients following hospital admission.2,13–16 Huang et al. 16 showed that COVID-19 patients, had good physical and functional recovery after 12 months; however, the diffusing capacity for carbon monoxide (DLCO) was still impaired, particularly in ICU-admitted patients. Next to a significant impairment in carbon monoxide diffusion capacity (DLCO), several studies reported a decreased Quality of Life (QoL).2,13,15 In non-hospitalized patients long-lasting symptoms were reported that now might be addressed as post-COVID syndrome (PCS). 14
While most patients are not admitted to the hospital it is important to understand the longer term outcomes in the whole spectrum of patients. Especially, since a recent study showed that non-hospitalized patients may also suffer from a significant burden of disease that leads to long-term health problems. 17
Therefore, we studied pulmonary function and QoL in both hospitalized and non-hospitalized COVID-19 pneumonia survivors. We hypothesized that depending on the clinical severity of disease at presentation, patients will have a severe impairment of pulmonary function on the long term. The primary aim was to assess pulmonary function at 3 months, and its relation with clinical severity of disease at presentation. Secondary aims were (1) to study outcome in QoL, and its relation to clinical severity of disease at presentation; (2) to study the correlation between the pulmonary function and patient-reported (QoL) outcomes at 3 months; and (3) to study the rate of recovery in pulmonary function and QoL from three to 6 months.
Methods
The data presented were collected as part of an observational study on COVID-19, the IMPACD 1 & 2 studies (Immunological Mental Physical After effects COVID-19), performed between March 1st and June 1st 2020, i.e. the first COVID-19 epidemic wave at OLVG, a large inner city, non-academic teaching-hospital in Amsterdam, the Netherlands. All patients were presented at the emergency care department, and were analyzed with Reverse transcription-polymerase chain reaction (RT-PCR) swabs, chest X-ray or low-dose high-resolution CT (HRCT) and serology. Patients were treated according to the pre-defined COVID-care program and data were analyzed retrospectively and anonymously (IMPACD-1 study); and, consisted of admission data, QoL questionnaire (SF-36) and spirometry at 3 months. Informed consent was not required at this stage. During the 3 months outpatient visit, patients were asked to participate in a follow-up study (IMPACD-2 study), for which informed consent was obtained. The follow-up study included spirometry and SF-36 questionnaire at 6 months. Both studies had been approved by the advisory committee for scientific research of OLVG Hospital and by the medical ethics committee (MEC).
According to World Health Organization (WHO) clinical management of COVID-19 guidelines, 18 we categorised the patients into three groups: non-hospitalized “moderate” pneumonia cases (radiographic evidence of pneumonia, but no requirement for supplemental oxygen, patients were presented at the Emergency Care Department but did not stay overnight), “severe” pneumonia cases (radiographic pneumonia and respiratory frequency >30/min and/or peripheral oxygen SpO2< 93%, and therefore hospitalized) and “critical” cases (e.g. respiratory failure requiring admission at the ICU). The diagnosis of COVID-19 pneumonia was defined, as described before, 19 by (bilateral) opacities on the chest X-ray or CO-RADS scores 4 and 5 on HRCT; and, either a positive RT-PCR swab result at presentation or positive serology for COVID-19 during follow-up.
All COVID-19 pneumonia survivors (n = 317) were included in a specially designed outpatient care pathway, and were invited for a follow-up visit 3 months after the day of discharge from either the emergency care department or the day of discharge after hospital admission. At 3 months lung function, i.e. spirometry and carbon monoxide diffusion capacity (DLCO), was performed and QoL was assessed by use of the Short Form Health Survey (SF-36). 20 In case of a (previously unknown) reduced lung function, i.e. vital capacity and/or DLCO below 80% of the predicted value, patients were also invited for a 6 months follow-up visit and pulmonary function test. All other patients were referred back for follow-up to their primary care physician.
Pulmonary function testing
Spirometry and single-DLCO were performed according to the American Thoracic Society (ATS) and European Respiratory Society (ERS) guidelines, and outcomes were expressed as percentage of the predicted values.21–23 DLCO was adjusted for haemoglobin.
Assessment of Quality of Life
Health status was measured and valued using the SF-36. 19 The SF-36 consists of eight subscales: physical functioning, social functioning, role functioning, role emotional, emotional well-being, energy/fatigue, pain and general health. The scores range from 0 to 100, with higher scores indicating better QoL. SF-36 scores were compared to SF-36 outcomes of Dutch healthy adults. 20
Statistical analysis
Statistical analyses were performed using SPSS statistical software program, version 22.0. Between-group comparisons for continuous variables were tested by one-way ANOVA analysis or Kruskal-Wallis test for pulmonary function. Pre-post comparison across groups were tested with repeated measures ANOVA. Categorical variables were tested with a Chi-square test. If a statistically significant difference was obtained, a post-hoc test was performed applying Tukey or Tanhame’s T2 correction to account for multiple comparisons. Within-group differences of continuous variables between baseline and follow-up were tested with paired t-tests. Spearman’s rank correlation coefficient was used to correlate non-parametric and Pearson correlation coefficient between parametric scales of SF-36 questionnaires and pulmonary function. A p-value <.05 was considered as statistically significant.
Results
Baseline characteristics at 3 months.
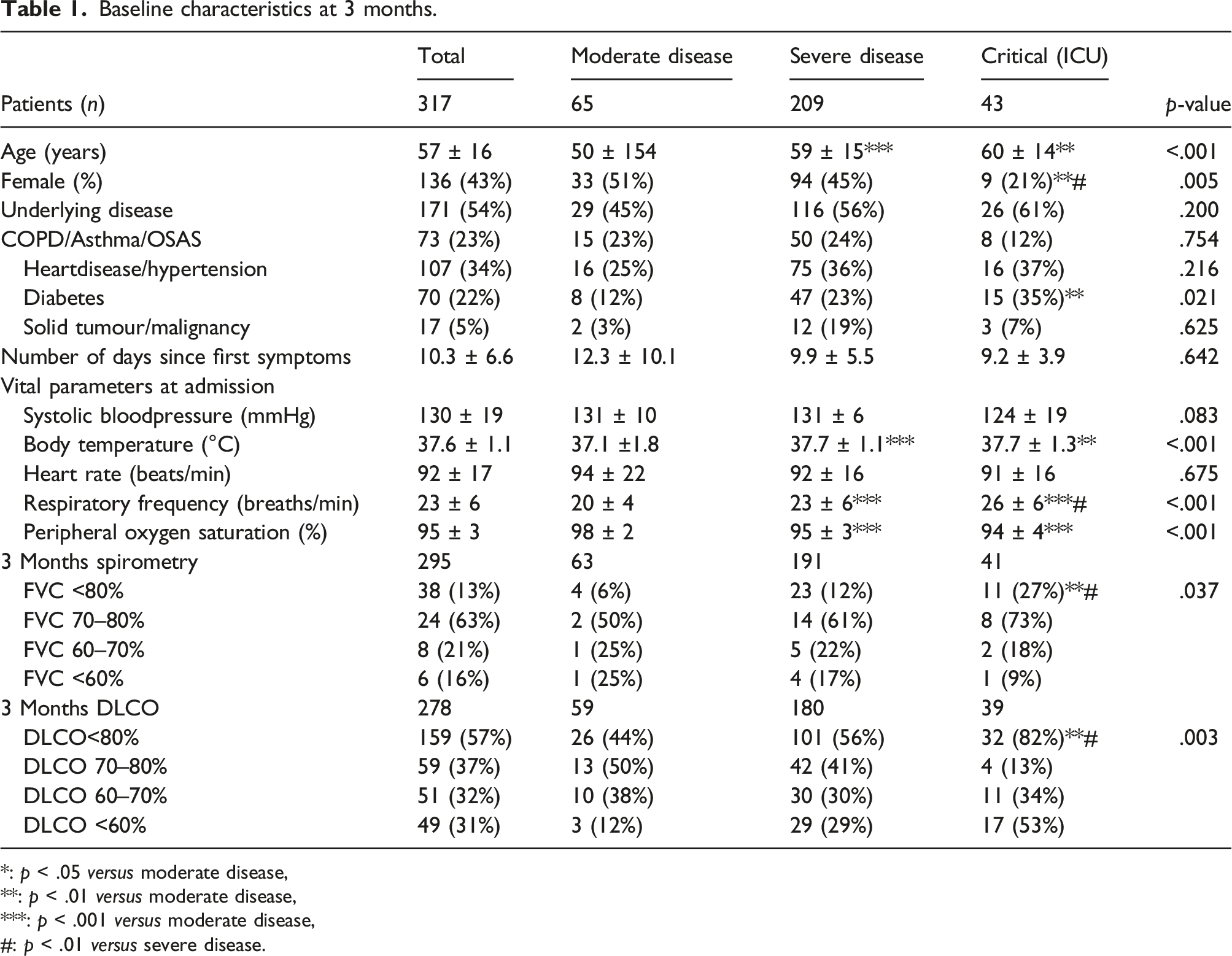
*: p < .05 versus moderate disease,**: p < .01 versus moderate disease,***: p < .001 versus moderate disease,#: p < .01 versus severe disease.
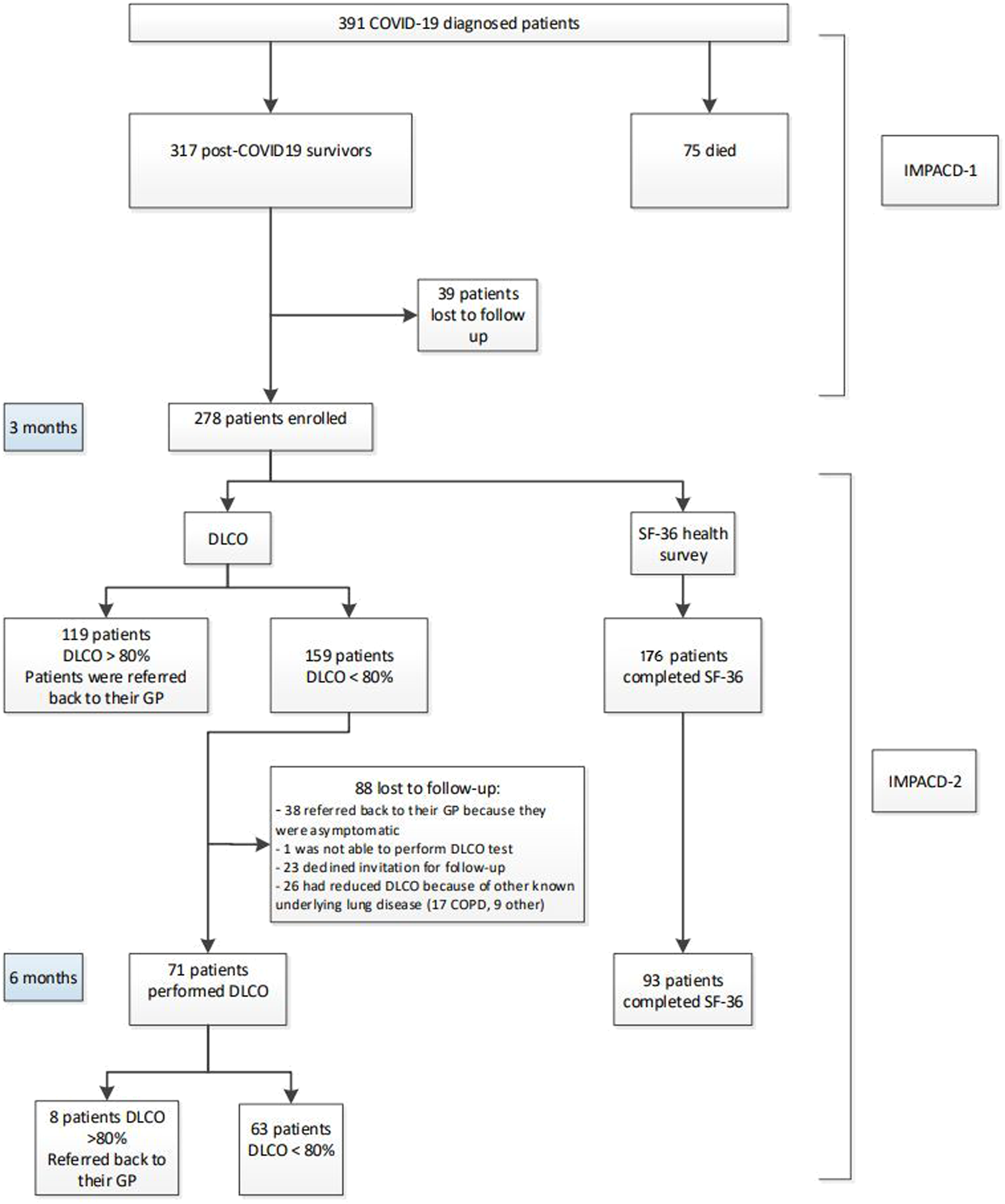
Flowchart and reasons for non-attendance at 3 and 6 months.
DLCO at 3 months
Patients who had recovered at 3 months (DLCO≥ 80%) appeared to be significantly younger (p < .001) compared to non-recovered patients. Among patients with a DLCO<80% comorbidities, i.e. diabetes and hypertension, were significantly more prevalent (Appendix, Table 3).
In all patients mean DLCO was decreased (76% ± 17%). The mean DLCO in critical cases (64 ± 14%) was significantly lower as compared to severe (76 ± 17%) and moderate (81 ± 15%) cases (p < .001; Figure 2). DLCO<80% of the predicted value was observed in more critical cases (32/39; 82%) as compared to severe (101/180; 56%) and moderate cases (26/59; 44%) (p = 0.003). Moreover, a severely impaired DLCO (<60%) was observed in 53% of critical, 29% of severe cases, and in 12% of the non-hospitalized moderate cases (Table 1). DLCO at 3 months (n = 278).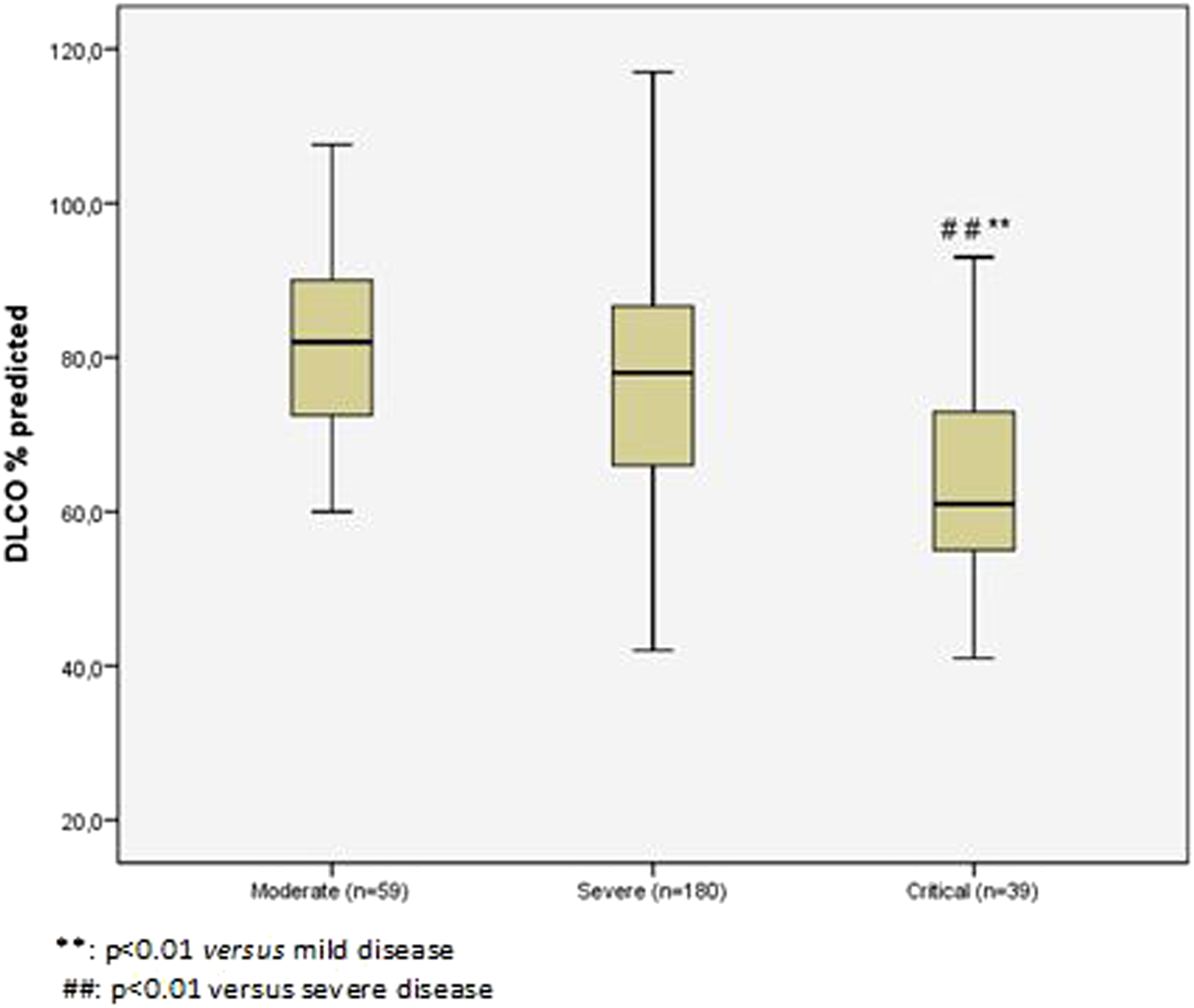
DLCO at 6 months
Of the 159 patients with DLCO <80% at 3 months, 95 patients agreed to perform 6 months follow-up (Figure 1); 23 of whom eventually declined the invitation. There were no significant differences in baseline characteristics between participants who were investigated after 6 months follow-up and those who were lost to follow-up (Appendix, Table 4). So, follow-up was performed in 71/159 patients (45%). At 6 months, a small though significant improvement in DLCO was observed: DLCO 68 ± 10% versus 63 ± 10% (p < .001; Figure 3(a)). The mean DLCO in critical cases (65 ± 11%) remained comparable to severe (68 ± 10%) and moderate (71 ± 7%) cases at 6 months (p = .242). Moreover, DLCO at 6 months had not yet normalized, i.e. ≥ 80% of the predicted value, in the vast majority (89%) of patients (Figure 3(b)) (a) DLCO at 3 months compared to 6 months (n = 71), (b) DLCO at 3 months compared to 6 months between groups (n = 71).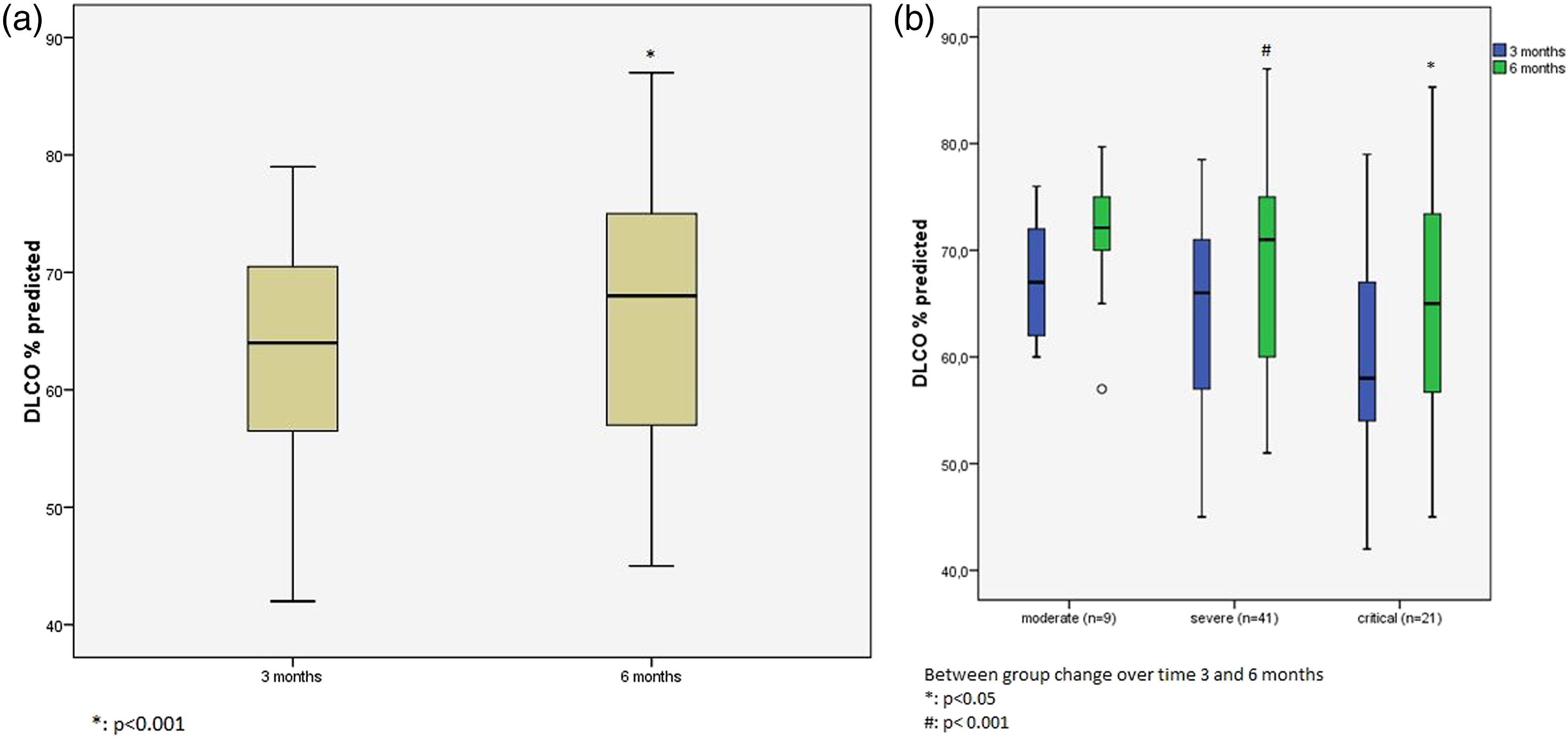
SF-36 at three months
SF-36 outcomes (n = 176) were decreased, 3 months after discharge. Significant differences were observed between the three groups with respect to two patient-reported SF-36 domains, i.e. “general health” and “health change”. Moderate cases scored significantly lower on the “general health” domain (51 ± 10) compared to severe (56 ± 13) and critical (59 ± 12) cases (p < .05; Figure 4, Table 2). In addition, compared to the critical cases (83 ± 17), both moderate (70 ± 20) and severe (71 ± 22) cases scored lower, and thus worse, on the domain of “health change”, i.e. general health compared with one year earlier (p < .05; Figure 4). There were no differences observed between the three groups in the other domains of SF-36 (Table 2) SF-36 domain outcomes at 3 months in the different subgroups. Short-Form 36 (SF-36) at 3 and 6 months after COVID-19 pneumonia. Values are presented as mean ± SD. aGroup*time interaction term was NS in all comparisons. bp < .05 versus moderate disease. cp < .05 versus severe disease.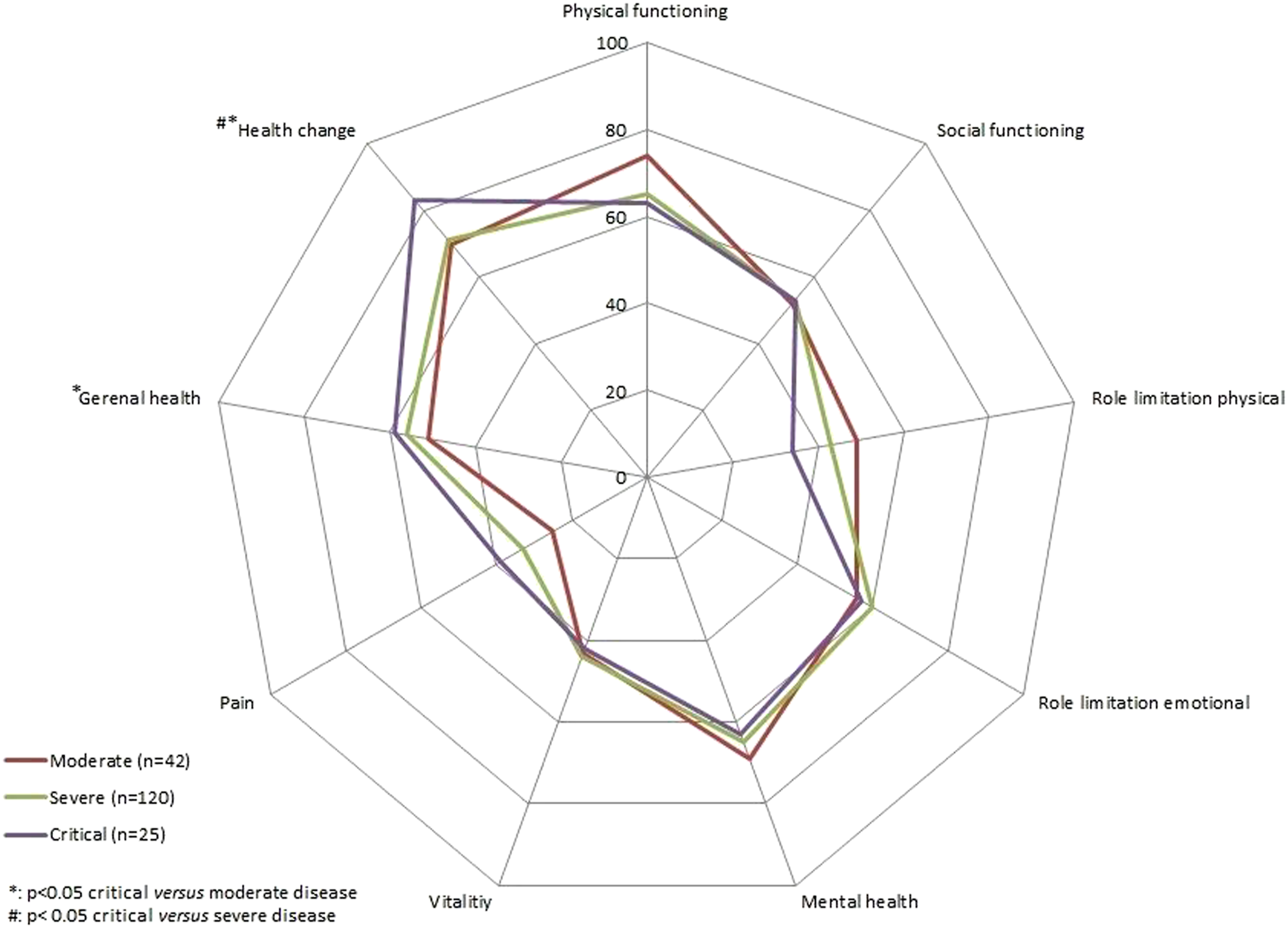

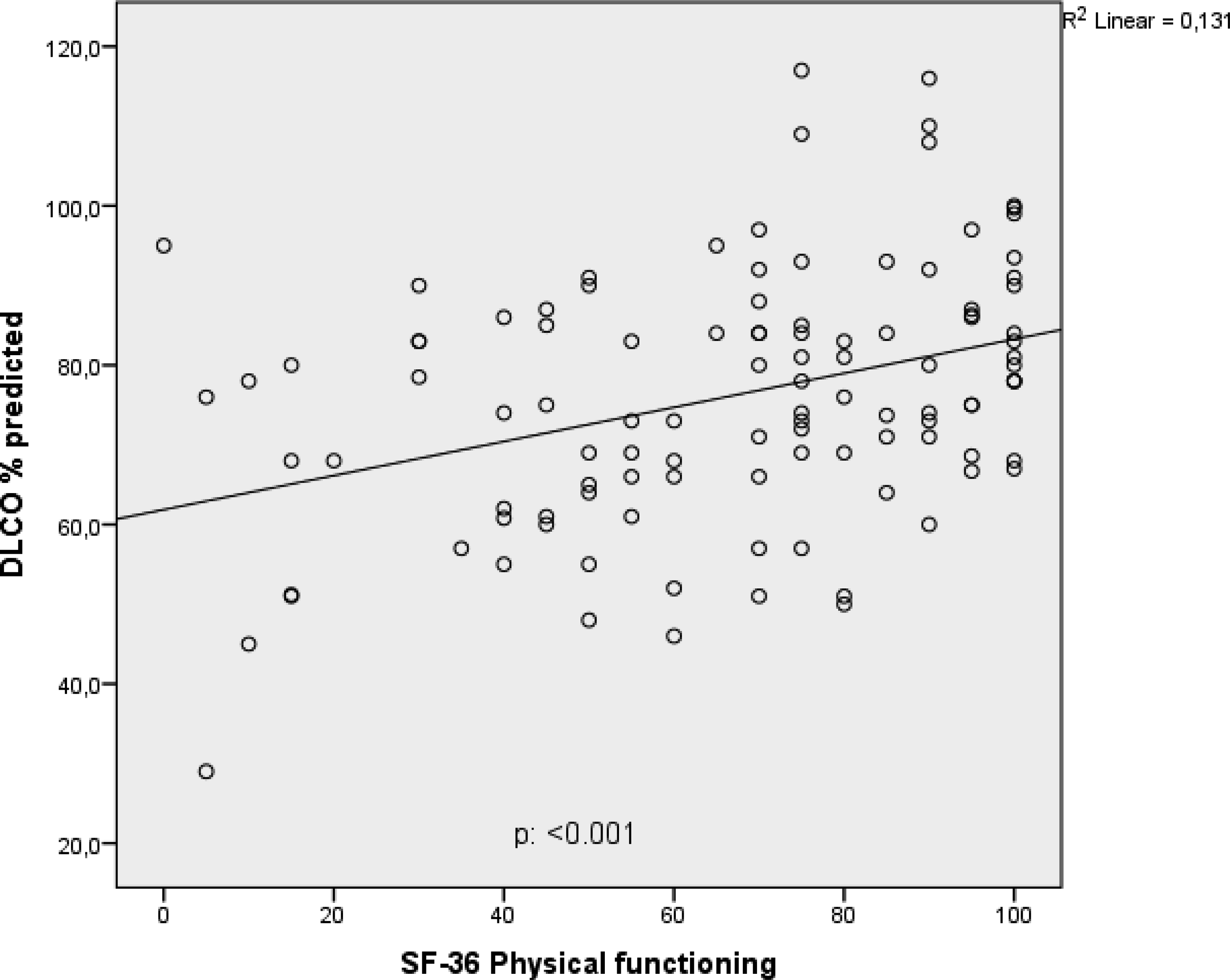
For all patients, the SF-36 domain of “physical functioning” was significantly correlated with DLCO (Figure 5). In the subgroup analysis, a correlation was found in the severe group (p < .001; Appendix, Figure 6). Positive correlation between diffusion capacity and SF-36 physical functioning.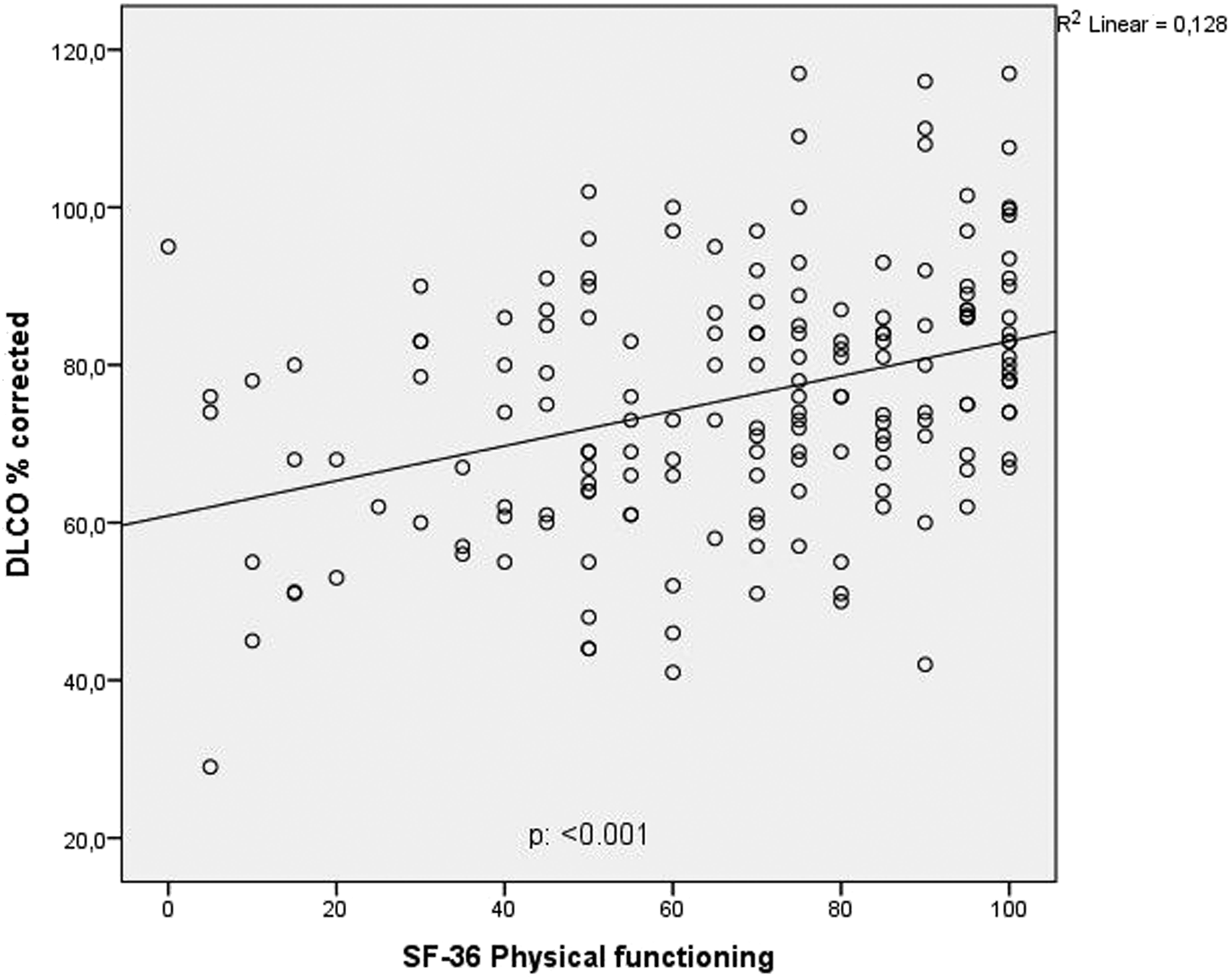
SF-36 at six months
At 6 months, we observed no significant differences between the three groups in the different SF-36 domains. All groups reported significantly better scores on the domains of “Physical functioning” and “Vitality” (Table 2). However, compared to reported scores at 3 months, all groups scored significantly worse on the domains “Social functioning”, “Mental health”, and “General health” at 6 months.
Discussion
In the present study, we showed a decreased diffusion capacity in the majority of COVID-19 pneumonia survivors 3 months after hospital discharge. The impairment was shown to be significantly more frequent and severe in the clinically most affected patients. At 6 months, in a subgroup of patients with a decreased DLCO at 3 months, a modest but significant improvement in DLCO was demonstrated; however, DLCO had not yet normalized in the vast majority of these patients.
Our study revealed that in COVID-19 pneumonia survivors the level of impairment of the pulmonary function is indeed associated with the clinical severity of disease at presentation. In particular, compared to both severe cases and cases without need for hospitalisation, the patients who required ICU admission were shown to have both more frequent and also more extensive residual pulmonary function impairment. In 53% of critical and 29% of severe cases we observed a DLCO<60%, as compared to 12% in the non-admitted moderate cases. Previously, a significant decline in DLCO was reported in 39–53% of COVID-19 survivors, 1 month after hospital discharge.5,24 In addition, two studies, performed 3 months after hospital discharge, reported a significant impairment of DLCO in 16% and 45% of patients, respectively; the latter study performed in ICU-admitted patients.25,26 In the present study, a significantly decreased DLCO in over half of all patients was observed; and, a severe decline in DLCO (<60%) was observed in 31% of all patients. Although the DLCO improved over the subsequent 3 months follow-up period, DLCO had not yet normalized in 89% of these patients at 6 months. It is worth mentioning, that these results apply only for the patients who completed the 6 months follow-up.
Also, in SARS pneumonia survivors a decreased DLCO was reported in a high percentage of patients, especially in the more severely affected patients who required ICU admission.27,28 In line with our observations, follow-up of DLCO in SARS-CoV-1 and MERS survivors showed that DLCO recovery is slow; and, decreased DLCO was observed in part of the patients over a follow-up period up to 3 years.11,29,30
The QoL reported by the patients 3 months after hospital discharge on most SF36 domains did not differ between the three groups. Interestingly, on the domains “general health” and “health change”, the scores of the non-hospitalized moderate cases were significantly lower compared to both hospital-admitted groups. One could postulate that these observations may partly be explained by the fact that moderate cases were younger, and probably more active in daily live. So, the impact of COVID-19 on their lives may have been more pronounced. Critical cases in particular were significantly older and more frequently already suffered from comorbidities.
In line with the observed improvement in DLCO, at 6 months all groups reported improvement in “Physical functioning” and “Vitality”. In contrast, however, all subgroups scored worse on the domains “Social functioning”, “Mental health”, and “General health”. Moreover, no differences were observed in SF-36 reported QoL between the three groups. So, at 6 months, we observed a striking discrepancy between the physical and non-physical outcome in patient-reported QoL. Moreover, none of the domains of the patient-reported QoL correlated with the severity of disease at presentation. Although this remarkable decrease in the non-physical QoL domains may be the mere consequence of COVID-19 pneumonia, we speculate that it may, at least in part, also be the consequence of the restrictions (lock-down) enforced during the first wave of COVID-19 in the Netherlands.
Our observations on QoL are in line with previous studies on outcome in mainly hospitalized COVID-19 survivors.17,26,31–34 In a recent study, however, it was shown that compared to hospitalized patients, non-hospitalized COVID-19 pneumonia survivors suffered from equal impairment in QoL. 17 Other studies showed, next to fatigue and reported difficulty of breathing, psychiatric symptoms like anxiety and depression 3 months after discharge; at current, these symptoms are often labelled as “long-COVID” or “post-COVID-19 syndrome” (PCS).31–35 They were mainly reported in hospitalized patients, but have also been observed after relatively moderate acute course of the disease.31,34,36 Other recently published studies among (non)-hospitalized COVID-19 patients, showed the presence of PCS in 13% to 40% of patients.34,36 Although we did not focus on PCS, 3 months after their discharge in all groups QoL was significantly impaired compared to the general population. Of interest, the non-hospitalized moderate cases reported even lower scores on the domains “general health” and “health change” than critical cases. Given the fact that pulmonary function impairment in these moderate cases was far less pronounced and DLCO did not correlate with these SF36 domains, the decreased health-related QoL in these patients is more likely to be related to PCS than the consequence of a residual physical impairment.
At 3 months, we observed a positive correlation between DLCO and the SF-36 domain of “Physical functioning”. By subgroup analysis, this correlation was also significant in the “severe” group of patients. The lack of correlation in the “moderate” cases is likely to be explained by the fact that, if present (44%), DLCO impairment was mild. In “critical” cases “physical functioning” at 3 months was not the purely consequence of COVID-19 pneumonia, but the physical impairment after COVID-19 was also caused by ICU-related complications.
Some limitations merits consideration. First, due to the lack of baseline pulmonary function test results prior to the COVID-19 pneumonia, it is not possible to draw definite conclusions on the impact of COVID-19 pneumonia on pulmonary function. Since, only a minority of patients had a chronic respiratory disease, and clinical severity of the disease was associated with the level of post-COVID-19 functional impairment, we think this does not prevent us from making more general conclusions. Because critical patients were also treated with mechanical ventilation it remains uncertain to what extent mechanical ventilation may also have attributed to the decrease in pulmonary function observed. Second, only 71 patients of 159 with DCO<80% completed assessments at 6 months, hampering the generalizability of the results. Nonetheless, the observation on the extent of the incomplete restoration of the DLCO after 6 months is still of interest. Third, the pathophysiological aetiology of the DLCO impairment was not studied and repeated imaging tests were not performed. The decrease observed in DLCO may be the consequence of COVID-19 pneumonia induced structural damage to the pulmonary parenchyma. However, previous studies in SARS and MERS survivors suggested that the decreased DLCO observed in these patients was primarily the consequence of a coronavirus-induced “small airway disease”. Due to corona-crisis related logistics, we could not perform additional lung function measurements, i.e. total lung capacity and residual volume, to study a possible small airways component. Based upon the observations presented here, however, this will be subject of future studies. Fourth, approximately half of the patients had comorbidities, which could have influenced the SF-36 scores. However, this does not prevent us from drawing conclusions on the differences observed in the moderate cases as compared to the severe and critical cases after COVID-19 pneumonia. Finally, given the -“real-life”- study design the groups were not equally matched.
Conclusion
Three months after discharge we observed an impaired DLCO in the majority of COVID-19 pneumonia survivors especially the most severely affected “critical” cases. At 6 months, DLCO had improved but not yet normalized. In line with the improvement observed in DLCO, patients reported improvement in “physical functioning” and scored significantly worse on the non-physical domains. No differences were observed in the SF-36 domains regarding the severeness of the COVID-19 pneumonia. Future research is warranted to study both the level of restoration of the functional impairment and the decreased QoL. Moreover, studies should focus on the both the etiology of the decreased DLCO and the QoL reported by non-hospitalized COVID-19 patients who suffered from moderate disease only.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix
Baseline characteristics of patients who recovered (DLCO ≥80%) versus non-recovered patients (DLCO<80%).
DLCO <80%
DLCO ≥80%
p-value
Patients (n = 278)
n = 159
n = 119
Age (years)
59.3 ± 15.2
52.11 ± 13.8
0
Female (%)
76 (48%)
40 (34%)
.032
Underlying disease
COPD/Asthma/OSAS
42 (26%)
19 (16%)
.058
Heartdisease/hypertension
64 (40%)
26 (22%)
.002
Diabetes
38 (24%)
17 (14%)
.005
Solid tumour/malignancy
8 (5%)
5 (4%)
.334
Number of days since first symptoms
9.99 ± 6.6
11.2 ± 6.9
.099
Vital parameters at admission
Systolic bloodpressure (mmHg)
128.7 ± 18.8
129.4 ± 16.2
.740
Body temperature (°C)
37.6 ± 1.1
37.6 ± 1.1
.990
Heart rate (beats/min)
91.7 ± 17.7
91.9 ± 15.0
.937
Respiratory frequency (breaths/min)
22.7 ± 5.8
22.2 ± 5.5
.442
Peripheral oxygen saturation (%)
95.3 ± 3.4
95.8 ± 3.0
.199
SF-36 (n = 176)
n = 99
n = 77
physical functioning
62.4 ± 26.6
75.3 ± 23.1
.001
Social functioning
52.7 ± 15.2
52.6 ± 15.1
.972
Role limitation physical
39.0 ± 43.3
50 ± 44.2
.092
Role limitation emotional
53.8 ± 45.2
68.4 ± 41.1
.035
Mental health
64.4 ± 13.3
68.2 ± 12.4
.112
Vitality
43.7 ± 13.2
41.8 ± 13.7
.347
Pain
34.3 ± 28.1
27.3 ± 27.7
.090
General health
56 ± 12.6
53.0 ± 11.6
.110
Health change
73.5 ± 20.9
71.1 ± 19.9
.449
Clinical characteristics of participants who showed up at 6 months follow-up and those who were lost to follow-up.
Follow-up at 6 months
Lost to follow-up
p-value
Patients (n = 159)
71
88
Age (years)
61.3 ± 13.6
57.7 ± 16.2
.137
Female (%)
33 (46%)
43 (49%)
.765
Underlying disease
COPD/Asthma/OSAS
14 (20%)
28 (32%)
.085
Heartdisease/hypertension
29 (41%)
35 (40%)
.891
Diabetes
15 (21%)
23 (26%)
.462
Solid tumour/malignancy
5 (7%)
3 (3%)
.296
Number of days since first symptoms
10 ± 6
10 ± 7
.822
Vital parameters at admission
Systolic bloodpressure (mmHg)
127 ± 19
130 ± 19
.239
Body temperature (°C)
37.5 ± 1.2
37.6 ± 1.1
.668
Heart rate (beats/min)
91 ± 17
92 ± 18
.621
Respiratory frequency (breaths/min)
22 ± 6
23 ± 6
.237
Peripheral oxygen saturation (%)
95 ± 4
95 ± 3
.968
DLCO at 3 months
63.0 ± 9.5
65.1 ± 12.1
.061
Positive correlation between diffusion capacity and SF-36 physical functioning (severe group).