Abstract
Background and objective
Many bronchiectasis patients suffer dyspnea, decreased exercise tolerance, and low body mass index. Chest wall muscles play a special role in respiratory movement and make up parts of skeletal muscles. This study aimed to examine the chest wall muscle thickness and their relationship with disease severity in bronchiectasis.
Methods
We retrospectively included 166 patients with bronchiectasis and 62 patients with pneumonia as comparators. The thickness of chest wall muscle as determined in chest CT, pulmonary function, and Bronchiectasis Severity Index (BSI) score were recorded. We compared the thickness of the chest wall muscle in two groups and assessed the relationships among chest wall muscle thickness, pulmonary function, and BSI score.
Results
Chest wall muscle thickness of the anterior midclavicular line and posterior exterior scapula were thinner in bronchiectasis patients than comparators both above the aortic arch level and at the aortic arch window level. Muscle thickness of the posterior interior scapula above the aortic arch level was significantly thinner in bronchiectasis patients. Chest wall muscle thickness at the anterior midclavicular line both the above aortic arch level and at the level of the aortic arch window were related to diffuse capacity in bronchiectasis patients. Anterior chest wall muscle thickness above the aortic arch was found to be a risk factor of disease severity.
Conclusion
Anterior chest wall muscles in the upper and middle chest were thinner in bronchiectasis patients than in comparators, and had relationship with spirometry and diffuse compacity factors. We provide another method to conveniently assess bronchiectasis severity.
Non-cystic fibrosis bronchiectasis is a chronic inflammatory lung disease characterized by permanent bronchial dilatation, mucociliary dysfunction, and progressive structural damage. 1 Representative symptoms are chronic cough, sputum production, and recurrent exacerbations which are major causes of morbidity and mortality, 2 and they cause further decline in respiratory function and poor quality of life. 3 In Germany 4 and the United States, 5 the annual incidence of hospitalization is around 9.4 and 16.5 per 100,000 population, respectively. The disease burden is continuously increasing.
Many bronchiectasis patients suffer low body mass index (BMI), and the magnitude of this weight discrepancy has a relationship with disease severity. Chronic respiratory diseases often involve sarcopenia. Whether this is true bronchiectasis is unknown. In our clinical practice, we observed that the chest wall muscles as observed under chest computed tomography appeared thinner in bronchiectasis patients, but only a few studies have focused on this. Some studies have shown that pectoralis muscle mass may be an indicator of sarcopenia and has associations with all-cause mortality in chronic obstructive pulmonary disease. 6 We hypothesized that the mass of chest wall muscles may be decreased in bronchiectasis patients and chest wall muscle mass reduction may be related to disease severity. To simplify the complexity of measuring chest muscle mass and make it convenient for adoption in clinical settings, we chose chest wall muscle thickness as an indicator of chest wall muscle mass.
Method
Study design and subjects
This was a retrospectively case-control study. Participants were recruited from Beijing Jishuitan Hospital from January 2016 through December 2017.
The inclusion criteria were as follows: a diagnosis of non-cystic fibrosis bronchiectasis confirmed by high-resolution computed tomography, age ≥18 years, clinical stability (without exacerbations in the last 4 weeks), and completion of all clinical tests. Bronchiectasis was diagnosed according to the criteria in the 10th revision of the International Statistical Classification of Diseases and Related Health Problems(ICD-10). 7
The exclusion criteria were as follows: a history of cancer, uncontrolled thyroid disease, any other metabolic disease affecting the mass of muscles, chronic obstructive pulmonary disease, asthma, primary immotile cilia syndrome, and primary immunoglobulin deficiency.
The comparator group comprised patients with community-acquired pneumonia treated during the same period without any history of cancer, chronic obstructive pulmonary disease, asthma or other diseases affecting muscle mass.
Measurements
Demographic data in this study included gender, age, smoking history, BMI, underlying diseases, albumin level, and blood urea nitrogen.
The thickness of chest wall muscle was measured. To simplify the procedure and render our findings convenient enough for clinical use, we set three bedding planes as focused measure levels: a slice above the aortic arch, aortic arch window (before the appearance of pulmonary artery in axial direction), and the 12 thoracic vertebra.
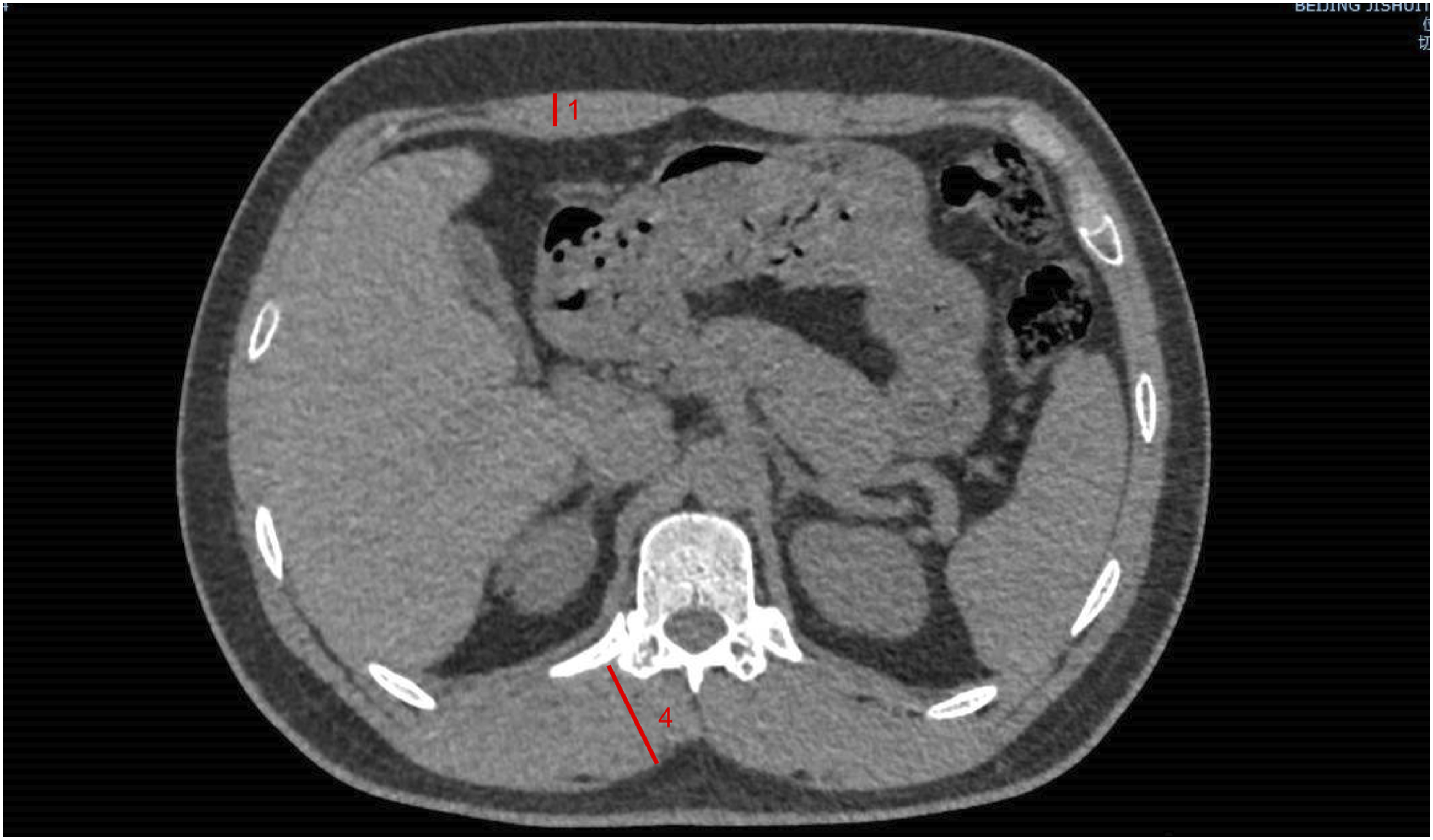
In each focused slice, we measured anterior chest wall muscle thickness at the bilateral midclavicular line (including pectoralis major and pectoralis minor muscle), interior scapula and exterior scapula muscle thickness in posterior chest wall, and erector spinae muscle thickness(Figures 1, 2 and 3). Then we measured the area of a cross section of pectoralis muscle from above the aortic arch(Figure 1). Above the aortic arch, 1 anterior chest wall muscle thickness at the midclavicular line, 2 interior scapula and 3 exterior scapula muscle thickness in posterior chest wall, 4 erector spinae muscle thickness. The blue area represents the cross section of pectoralis muscle from above the aortic arch. Aortic arch window, 1 anterior chest wall muscle thickness at the midclavicular line, 2 interior scapula and 3 exterior scapula muscle thickness in posterior chest wall, 4 erector spinae muscle thickness. Twelve thoracic vertebra level, 1 anterior chest wall muscle thickness at the midclavicular line, 4 erector spinae muscle thickness.

Bronchiectasis patients underwent pulmonary function tests in the sitting position according to guidelines. 8 Spirometric parameters, static lung volumes, and transfer factors were recorded. Results were compared with standard reference values. 9
Bronchiectasis Severity Index (BSI) 10 scores were used to classify the severity of bronchiectasis.
Statistical analysis
Statistical analyses were performed in SPSS (version 22,NY,USA). A two-sample independent t test was used to compare normally distributed data, a Mann Whitney U test was used to compare non-normally distributed data, and a Chi-square test was used to compare enumeration dates. Spearman’s correlation was used to assess correlations between thickness of chest wall muscle and severity of bronchiectasis, and Pearson’s correlation was used for correlations between chest wall muscle thickness and pulmonary function test results. Logistic regression analysis was used to confirm whether chest wall muscle thickness was risk factor in bronchiectasis patients in the presence of other nutrition-related factors. BMI, albumin, and BUN were included in Logistic regression analysis. Statistical significance was set at P < 0.05 (two-tailed).
The studies were approved by local research ethics committees. Patients provided written informed consent for their data to be reanalyzed. At no point in this work do we expose any identifying data for any patient.
Results
Baseline clinical data
Baseline characteristics of bronchiectasis versus comparators.

SD: standard deviation; CHD: coronary heart disease; BMI: body mass index.
Compares chest wall muscle thickness between bronchiectasis and comparators.

*p<0.05;**p<0.005.

Chest CT in bronchiectasis patients, above the aortic arch.

Chest CT in bronchiectasis patients, aortic arch window.

Chest CT in bronchiectasis patients, twelve thoracic vertebra level.
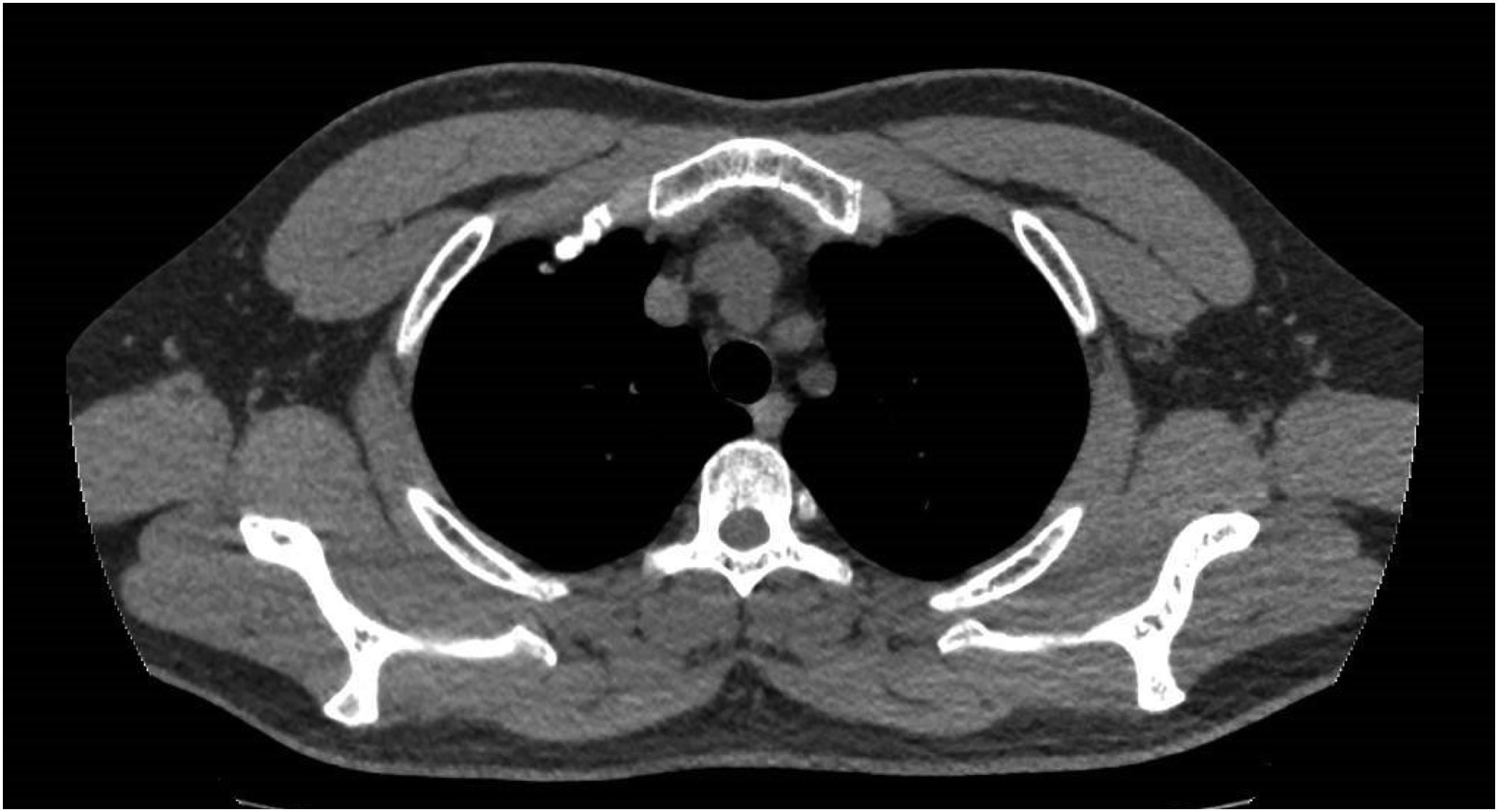
Chest CT in comparators, above the aortic arch.

Chest CT in comparators, aortic arch window.

Chest CT in comparators, twelve thoracic vertebra level.
Correlation between chest wall muscle thickness and pulmonary function test in bronchiectasis patients.

DLCO: gas transfer factor measurement; FEF75%: forced expiratory flow at 75% pf the vital capacity; FEV1: forced expiratory volume in one second; FVC: forced vital capacity; FEF25-75%: forced expiratory flow from 25% and 75% of vital capacity; FEF25%: forced expiratory flow at 25% pf the vital capacity; FEF50%: forced expiratory flow at 50% pf the vital capacity; RV: residual volume; TLC: total lung capacity; VC: vital capacity.
*: p<0.05;**p<0.005.
Correlation between chest wall muscle thickness and disease severity (BSI) in bronchiectasis patients.

*p<0.05;**p<0.005.
5. Pectoralis muscle thickness showed a strong relationship with cross section area (Pearson’s correlation 0.82, P = 0). There was a difference in pectoralis muscle area between bronchiectasis patients and pneumonia controls (25.53 ± 12.97 vs 29.49 ± 10.57, P = 0.041).
Logistic regression of bronchiectasis severity, considering anterior chest muscle thickness and other nutrition related factors.

Average anterior chest muscle thickness above the aortic arch = (chest muscle thickness of anterior right midclavicular line above the aortic arch + that of anterior left midclavicular line)/2; BMI: body mass index.
Discussion
The chest wall muscle was thinner in non-CF bronchiectasis patients than in comparators, especially the anterior part of upper and middle chest. To the best of our knowledge, no study has investigated the effect of chest skeletal muscle in bronchiectasis or established any clinically significant relationships. The chest wall muscle depletion level was related to disease severity and some important pulmonary function factors, such as FEV1, small airway function, and diffuse capacity. This indicates that we should pay special attention to chest wall muscle mass in non-cystic fibrosis bronchiectasis patients. Assessing the thickness of chest wall muscles in CT patients is simple and convenient. It is possible to use this indicator in daily clinical settings.
Bronchiectasis is a chronic respiratory disease. We found chest muscle thickness to be lower in bronchiectasis patients than in comparators, which suggested that bronchiectasis patients may suffer from sarcopenia. Various techniques were used to evaluate the skeletal muscle mass and to diagnose sarcopenia. X-ray absorptiometry (DXA) is a reference method for assessing body composition 11 but it has seen limited use because it involves extra radiation exposure and considerable expense. DXA requires dedicated machines and is not available in many hospitals. Chest computed tomography (CT) is widely used in routine clinical practice. Bronchiectasis patients often undergo chest CT during the course of disease. Chest CT–derived chest wall muscle thickness is a reproducible measure of muscle mass, and it can be observed without additional radiation exposure or cost.
Muscle integrity reflects general well-being and nutritional status. In chronic obstructive disease (COPD), muscle mass and cross-sectional area are correlated with the severity of lung disease. 12 Pectoralis muscle area has been found to be adversely associated with disease severity as measured by GOLD stage, lower resting oxygen saturation, BODE score, quality of life score, and exercise capacity. 12 One recent report indicated that lower erector spinae muscle mass is associated with poor dyspnea score, higher BMI, worse emphysema, low FEV1, and high mortality in COPD patients. 13 Among cancer patients, lower pectoralis muscle area was found to be associated with increased mortality. 14 Consistent with pectoralis muscle mass studies in COPD and cancer patients, we found chest wall muscle thickness to be related to pulmonary function, and pectoralis muscle thickness above the aortic arch was found to be a risk factor for bronchiectasis severity.
In previous studies, quantitative assessments of the pectoralis muscle area were obtained from a single axial slice above the aortic arch. We also chose the slice above the aortic arch as one of our studied positions. Because few studies pay attention to muscle mass reduction or sarcopenia in bronchiectasis, we included several other slices in order to identify sample most indicative of chest wall thickness. To make the measurement process simpler and more convenient, we chose chest wall muscle thickness as our research object. Several other studies have already measured muscle thickness. Emmanuel Vivier and his colleagues 15 measured diaphragm and pectoral thicknesses by ultrasound and found pectoral atrophy to be associated with prolonged weaning from mechanical ventilation and longer ICU stays. Muscle thickness measured on a computed tomography image was used to assess transversal psoas muscle thickness (TPMT) at the level of the umbilicus16. Abdominal muscle thickness has been measured in cystic fibrosis patients, 16 and the training effect on respiratory muscles has been assessed. 17
We chose pneumonia patients as comparators in our study. It would be more persuasive to choose healthy people as controls. But the pneumonia patients included in our study didn’t have any other underlying diseases which would influent they skeletal muscles significantly. As shown in the results, chest muscle thickness of middle-upper part was thinner in bronchiectasis patients compared with sick pneumonia patients. We may speculate that the chest muscle thickness in healthy controls would be thicker than sick pneumonia patients.
As a retrospective study, it was hard to reconfirm complications in pneumonia group, but the pneumonia patients included as comparators were in-patients. We completed thorough examinations for in-patients in clinical practice. The diagnosis and complications were convictive.
Our study has some limitations. Because this is a retrospective study, there are some inherent limitations such as recall bias and selection bias. However, we tried to use comparative objective factors. The main factors used in the study were thickness of the chest wall muscle, pulmonary function test, and disease severity. The thickness of chest wall muscle was remeasured on computed tomography of the chest. Pulmonary function test and BSI were recorded in the digital medical records. All patients included were continuously brought into the study. We did not assess whole-body muscle mass, and more test methods should be used to ensure the accuracy of chest wall muscle mass measurement. Dual-energy x-ray absorptiometry (DXA) is considered the gold standard technique in clinical practice to estimate fat-free body mass, 11 but it is not available in many hospitals and can be costly. Bio-electrical impedance analysis (BIA) and skinfold thickness measurement (STM) are more readily available to health care teams, but there are no good coefficients obtained in truck fat-free mass assessment with BIA and DXA. Measuring chest wall muscle thickness may be less conventional, but it is helpful in clinical practice. Our study also involved a relatively small sample. Prospective multi-center studies may confirm the significance of measuring the chest wall muscle thickness. What’s more, we didn’t included other parts of chest wall muscle mass in our study, e.g. intercostal muscle measurement. We intent to measure the intercostal muscle in our future work.
Conclusion
Anterior chest wall muscle thickness of the upper and middle chest in bronchiectasis patients was lower than in comparators, and it was related to spirometry and diffuse compacity factors. We here provide another method of conveniently assessing bronchiectasis severity.
Footnotes
Acknowledgements
Author’s Note
The work should be attributed to Capital Medical University
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
