Abstract
This investigation validated the inert gas rebreathing (IGR) technique and determined IGR prognostic ability compared to invasive cardiac output measurements in patients with pulmonary hypertension. IGR compared with thermodilution cardiac output demonstrated a moderate bias. IGR technique demonstrated long-term prognostic value comparable to invasive cardiac output in pulmonary hypertension patients
Introduction
Cardiac output (CO) is a strong indicator of right ventricular function and disease severity in pulmonary hypertension (PH) and a consistent prognostic predictor.1–4 Traditional methods of CO measurement include invasive techniques using either Fick or thermodilution (TD). Limitations with right heart catheterisation (RHC)-based procedures include expense, require specialist infrastructure to be performed well and are resource intensive and additionally there can be a small but significant risk of complications. 5 Therefore, the ability to measure CO non-invasively is attractive. Although there are a number of different techniques, none have been adopted for widespread use. Inert gas rebreathing (IGR) is a simple, inexpensive non-invasive technique which can determine CO using a portable photoacoustic gas analyser. Despite the fact IGR measurements have been validated against invasive CO measurements by a number of investigations in PH patients,6–10 they have not made it into widespread use. Advantages mean IGR could be used to monitor CO in PH in the outpatient routinely and even in the community setting. Here we present data that validate the IGR technique in patients with PH and, for the first time, demonstrate that IGR has long-term prognostic value comparable to invasive CO measurements.
Method
Population characteristics.
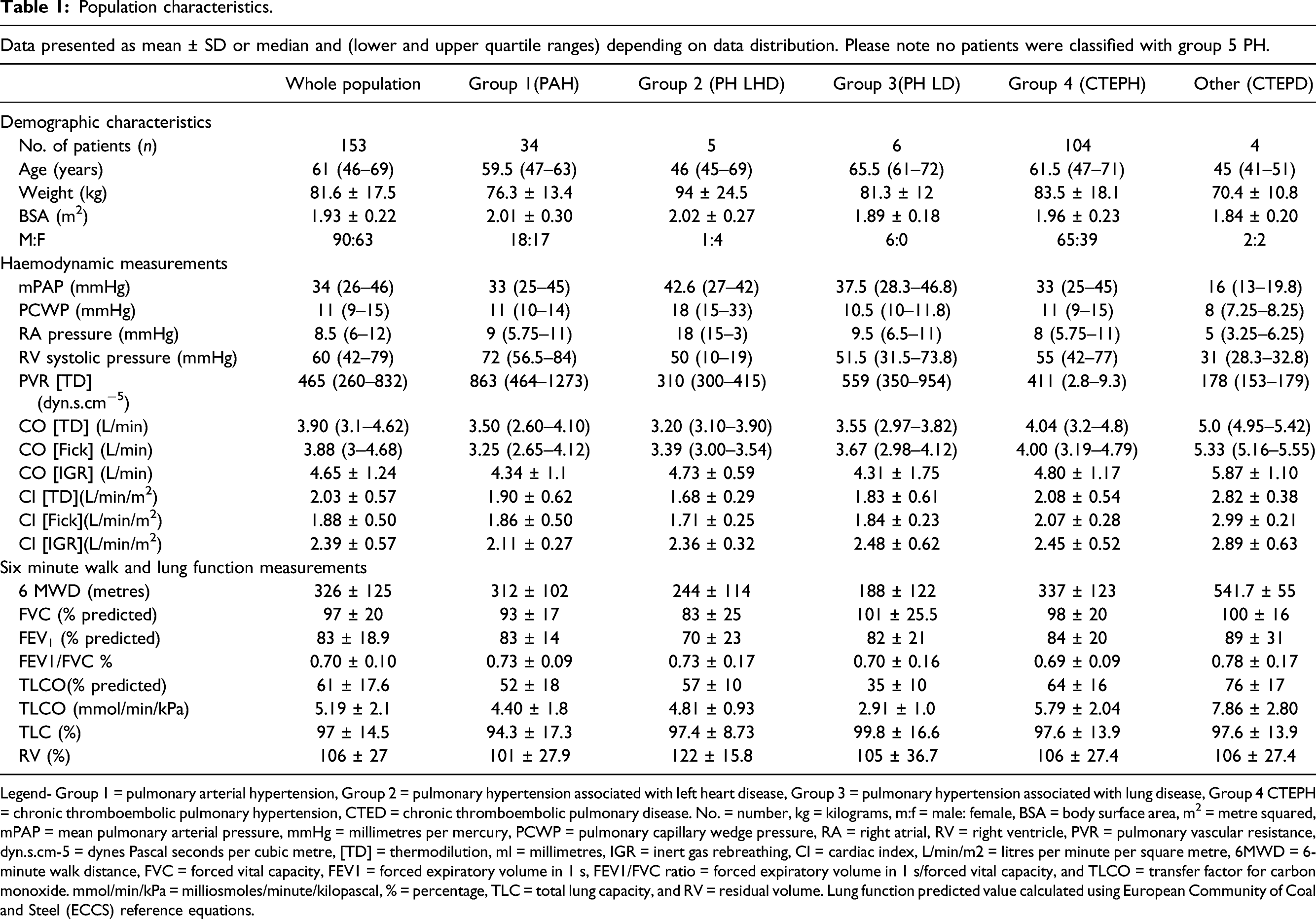
Legend- Group 1 = pulmonary arterial hypertension, Group 2 = pulmonary hypertension associated with left heart disease, Group 3 = pulmonary hypertension associated with lung disease, Group 4 CTEPH = chronic thromboembolic pulmonary hypertension, CTED = chronic thromboembolic pulmonary disease. No. = number, kg = kilograms, m:f = male: female, BSA = body surface area, m2 = metre squared, mPAP = mean pulmonary arterial pressure, mmHg = millimetres per mercury, PCWP = pulmonary capillary wedge pressure, RA = right atrial, RV = right ventricle, PVR = pulmonary vascular resistance, dyn.s.cm-5 = dynes Pascal seconds per cubic metre, [TD] = thermodilution, ml = millimetres, IGR = inert gas rebreathing, CI = cardiac index, L/min/m2 = litres per minute per square metre, 6MWD = 6-minute walk distance, FVC = forced vital capacity, FEV1 = forced expiratory volume in 1 s, FEV1/FVC ratio = forced expiratory volume in 1 s/forced vital capacity, and TLCO = transfer factor for carbon monoxide. mmol/min/kPa = milliosmoles/minute/kilopascal, % = percentage, TLC = total lung capacity, and RV = residual volume. Lung function predicted value calculated using European Community of Coal and Steel (ECCS) reference equations.
CO was measured invasively using TD and indirect Fick, the most standardly employed methods to measure CO in PH1,3–4 making it an appropriate comparison for determining utility of IGR. Patients were in the supine position for the whole procedure. A 7 F quad-lumen, balloon-tipped Swan-Ganz catheter was advanced through an 8 F introducer sheath in the right or left internal jugular vein. The TD technique to measure CO was performed using 10 ml ice-cold isotonic saline injected into the proximal port of the Swan-Ganz catheter. The mean CO was calculated from a minimum of three measurements with a maximum acceptable variation between the measurements of 10%. Any other measurements were discarded. Indirect Fick CO was calculated based on Fick’s principle according to the equation by LaFarge. 11 IGR measurements were performed at the time of the RHC procedure in the supine position and with IGR measurements recorded using a photoacoustic gas analyser (Innocor; Innovision) using the rebreathing technique from functional residual capacity. Three IGR measurements were performed on each patient with the results averaged.
Additionally, as part of a clinical review, patients performed a six-minute walk test and pulmonary function test. Bland–Altman analyses were performed to determine the level of agreement between the different CO techniques. 126 of the 153 patients were included in the survival follow-up cohort after excluding patients who had a normal mean pulmonary artery pressure (mPAP) who underwent pulmonary endarterectomy (PEA), surgery and those with chronic thromboembolic pulmonary disease.
Receiving operating curves (ROC), univariate and multiple cox proportional hazard regression analyses were undertaken to examine one- and 5-year survival rates, and CO was indexed to body surface area as cardiac index (CI). Survival was calculated from the time of the patient’s RHC procedure where IGR was performed alongside, between the years of 2008 and 2016. Patients were censored at the date of death or transplantation. Additionally, post-PEA patients with residual mPAP who were included in the follow-up analysis were censored as a survivor at date of surgery.
Results
IGR CO significantly correlated to TD CO (r = 0.79) and to Fick CO (r = 0.75) (p < .001 for both). TD, CO and indirect Fick CO measurements demonstrated a mean bias of 0.06 ± 0.84 L/min (Figure 1(a)). TD, CO and IGR CO measurements demonstrated a mean bias of-0.68 ± 0.80 (Figure 1(b)). Indirect Fick CO and IGR CO measurements demonstrated a mean bias of −0.78 ± 0.89 L/min (Figure 1(c)). Bland–Altman analysis presenting the agreement between the different CO techniques. Data presented as mean bias and upper and lower limits of agreement (calculated as ± 1.96 X SD of the bias). (A) Bland–Altman analysis presenting the agreement between TD CO to indirect Fick CO; (B) Bland–Altman analysis presenting the agreement between TD CO to IGR CO and (C) Bland–Altman analysis presenting the agreement between indirect Fick CO to IGR CO. Legend- TD CO = thermodilution cardiac output, L/min = litres per minute, IGR = inert gas rebreathing cardiac output, PH = pulmonary hypertension, PAH = pulmonary arterial hypertension, PH LD = pulmonary hypertension left heart disease, PH LD = pulmonary hypertension lung disease, CTEPH = chronic thromboembolic pulmonary hypertension, CTEPD = chronic thromboembolic pulmonary disease, ULOA = upper limit of agreement, LLOA = lower limit.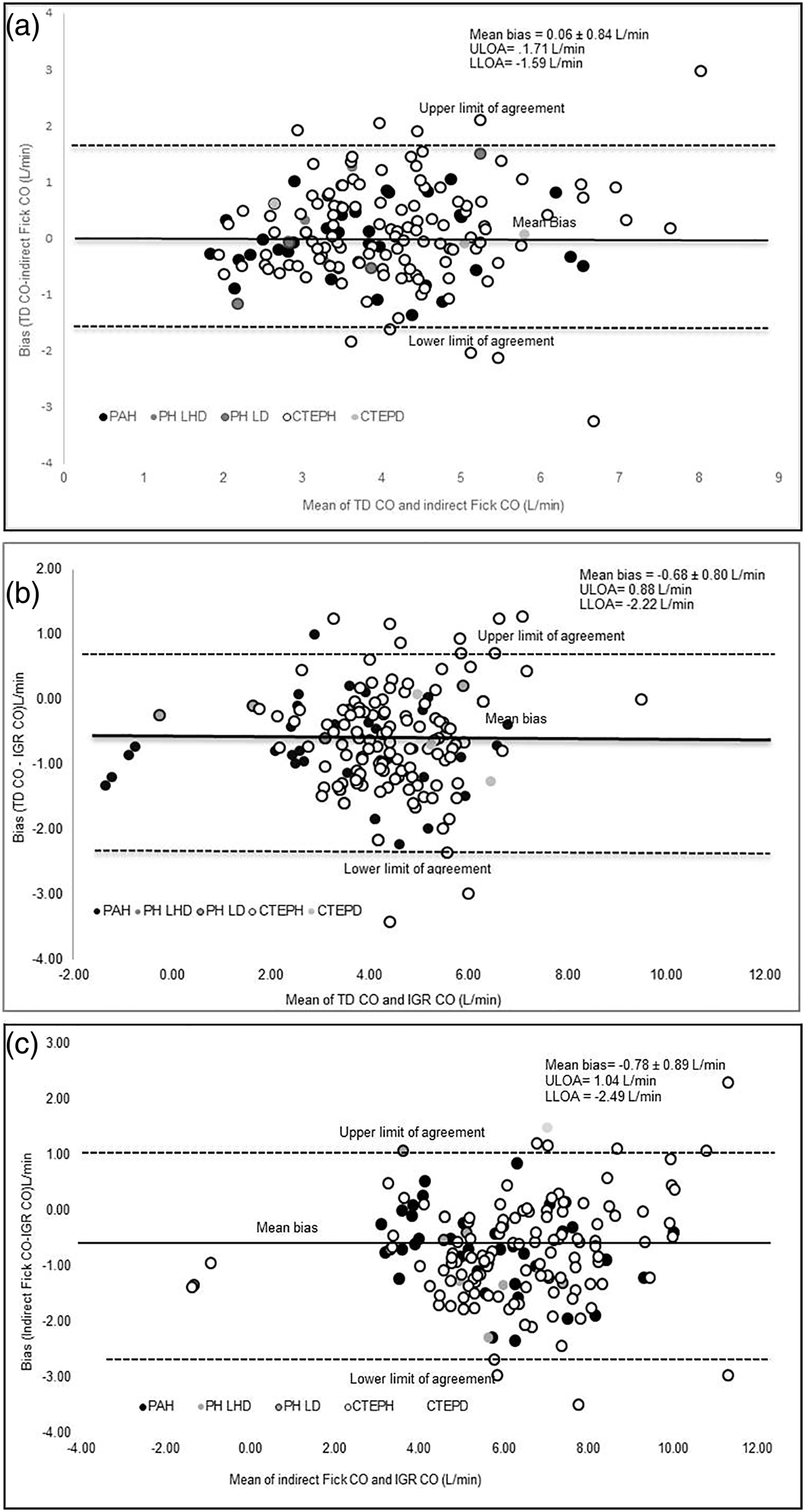
In the cohort, the median time between diagnosis and investigation RHC procedure with IGR measurements was 2 days (range: −14 to 4128 days).
Cohort 1-year and 5-year mortality was 8% and 24%, respectively. The AUC for predicting 1-year mortality for all CO methods demonstrated similar significant predictive ability (Figure 2(a)) and similarly, for predicting 5-years mortality (Figure 2(b)). The AUC for the different CO methods at predicting 1-year and 5-year mortality rate was not significantly different (p > .05). Receiving operating curves comparing different CO methods for predicting 1-year and 5-year mortality. (A) Receiving operating curves comparing different CO methods for predicting 1-year mortality and (B) receiving operating curves comparing different CO methods for predicting 5-year mortality. TD = thermodilution, CO = cardiac output CI = cardiac index, IGR = inert gas rebreathing, L/min/m2 = litres per minute metre squared.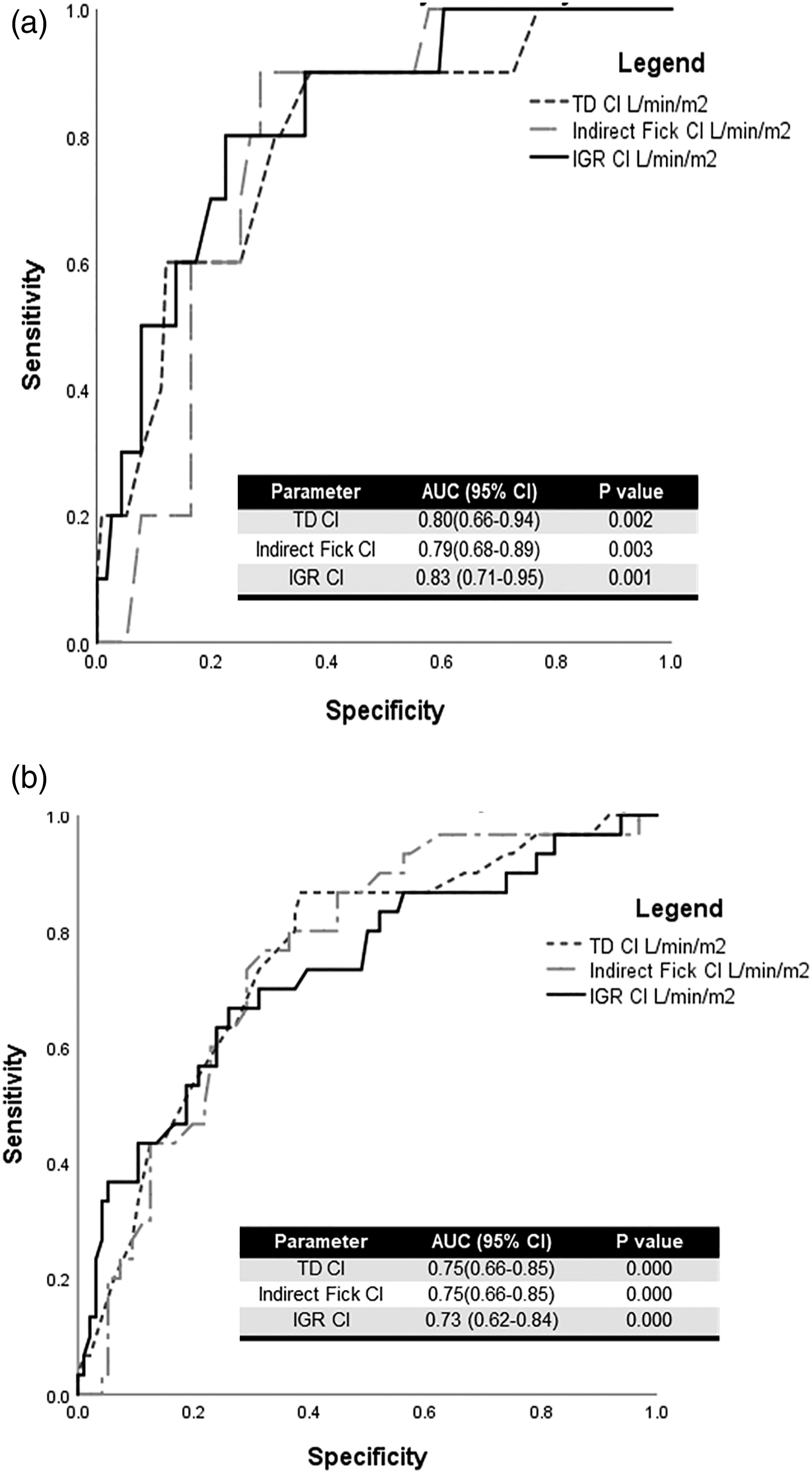
Univariate Cox proportional hazard analysis demonstrated both IGR, CI and TD CI as significant predictors of 5-year mortality along with age, pulmonary vascular resistance, mPAP, transfer factor for carbon monoxide (TLCO) and six-minute walk distance (6MWD). Multiple Cox proportional hazard regression analysis was performed with IGR CI along with demographics and inclusion of significant predictors from univariate analysis; these were mPAP, TLCO and 6MWD. The results demonstrated that independent predictors of 5-year mortality were IGR CI, 6MWD and sex.
Discussion
PH remains a disease associated with increased mortality and therefore there is emphasis on clinical parameters which can assess prognosis for monitoring and treatment. IGR CO correlates well with both TD and Fick CO but with a systematic bias. The prevalence of chronic thromboembolic pulmonary hypertension (CTEPH) in our cohort may explain our bias results because the IGR technique has demonstrated lesser accuracy in determining CO in this context. 7 In the CTEPH group, the increased heterogeneity in pulmonary blood flow in comparison to ventilation may have contributed to lesser accuracy of IGR in determining CO. 7 In our studies, there was modest systematic bias using the IGR method in our mixed populations of patients with PH; despite this, there was no reduced ability of IGR to discriminate long-term mortality. In the only other work to look at outcomes, Stadler et al. 12 found IGR technique not to be predictive of mortality in PH but only when examining PH groups separately it was predictive of mortality in group 3 PH with lung disease. Notably, IGR measurements were not taken at the same time points as invasive CO measures, but a median of 9 months later. Notably in Stadlers work, CO determined by the IGR technique only showed modest correlation (r = 0.47) to asynchronous timed diagnostic RHC CO.
Limitation of our investigation included not comparing IGR to the gold standard direct Fick method. The direct Fick method due to its impracticality is rarely the standard method of use to measure CO in PH and therefore we believe our studies are more relevant to real-world practice. The primary methods of CO measurement which we employed though have some important limitations. The TD method can be inaccurate due to a variety of factors13–15 notably demonstrated to be less reliable in patients with tricuspid regurgitation and low CO16,17 The indirect Fick method relies on estimates of total body oxygen consumption which can be inaccurate in disease.18,19 The IGR method may be less accurate in lung disease because of incomplete alveolar gas mixing and intrapulmonary shunting of blood flow causing underestimation of pulmonary blood flow. However, IGR accuracy has not always been demonstrated to be impacted by lung disease.8,20 This is not likely to affect our results as very few of our patients presented with significant lung disease. Additional limitations of this investigation include being a single-centre study with potential for selection bias as patients enrolled onto this investigation were based on those who had a planned RHC; the study was sequential and prospective and therefore no additional filtering of populations was performed. Our predominant CTEPH cohort may therefore reduce the applicability of our findings to all PH cohorts, and due to the small sized PH cohorts, we were unable to examine differences in prognostic value of IGR between different PH groups and this requires future investigation. As the innocor system has the potential to monitor PH in the outpatient or in the community setting, future investigations need to clarify the value of serial IGR measures in relation to clinical worsening of other clinical predictors.
In conclusion, cardiac output measured by inert gas rebreathing technique demonstrated long-term prognostic value comparable to invasive cardiac output in patients with pulmonary hypertension. The innocor system could be an additional tool for monitoring pulmonary hypertension in the outpatient or in the community setting but requires validation in a multi-centre study using serial measurements.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.R. has none to declare. K.B. has none to declare. R.M. has support for attending meetings/travel from GSK, Actelion and Merck. J.C. has received payment or honoraria for lectures and presentations from Actelion. K.S. has received educational funding from Actelion, Bayer, GlaxoSmithKline (GSK) and MSD and consultancy/lecture fees from Actelion. D.T. has received speaker fees and educational funding from Actelion, Bayer, GSK, Lilley, MSD and Pzier. J.P-Z. has served on advisory boards of, and received personal fees, institutional grant/research support from Actelion, Bayer, MSD and GSK. M.T. has received personal fees from Actelion, GSK and Bayer, institutional grant/research support from Actelion, Roche and Bayer and is a member of the Scientific Advisory Board of Morphogen-IX.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MT is supported by the Cambridge NIHR cardiorespiratory BRC
