Abstract
Rheumatoid arthritis (RA) is a chronic autoimmune disease primarily affecting joints but often also associated with lung involvement such as bronchiectasis (BE). The aim of the present systematic review and meta-analysis is to provide an update on the current evidence regarding the prevalence and association between RA and BE. This systematic review and meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines with literature search using the terms ‘Bronchiectasis AND Rheumatoid Arthritis’ without a date limitation on PubMed during May 2020. A total of 28 studies fulfilled the predefined criteria and were included in the present review, with 19 being cross-sectional studies. Twenty-three studies were included in the meta-analysis. The pooled prevalence estimate was 2.69% (95% CI 1.63–4.42) in clinically defined BE, and 24.9% (95% CI 19.21–31.67) in radiologic disease. Many inconsistencies exist regarding potential risk factors for BE in RA patients such as gender, RA duration and severity, as both negative and positive associations have been reported. Although very little is known about possible causative mechanisms between RA and BE, potential pathways might be antigenic stimulation from pulmonary mucus and/or systemic inflammation from joint disease affecting the lungs. At present, the available evidence of bronchiectasis in patients with RA is insufficient to identify RA-associated risk factors for the development of BE, possibly apart from duration of RA, and, consequently, also to fully explore a possible causal relationship between the two disease. However, the increased prevalence of BE in RA patients warrants further studies to explore the association between RA and BE.
Introduction
The diagnosis of rheumatoid arthritis (RA) is based on the presence of inflammation of joints, serological tests and duration of symptoms, and the disease is typically diagnosed using the American College of Rheumatology (ACR) criteria. 1 However, although RA is a chronic autoimmune disease of the joints, RA is also associated with many well-recognized extra-articular manifestations, such as involvement of the eyes, skin, gastrointestinal tract, heart and the lungs. 2 The most common involvement of the lungs in RA is interstitial lung disease, but bronchiectasis (BE) has also been reported in patients with RA as early as half a century ago. 3
BE is an irreversible and abnormal dilatation of the airways. 4 The diagnosis of BE is based on radiologic findings together with the presence of clinical symptoms such as cough, sputum production, dyspnoea and, and in a limited number of patients, haemoptysis. 4 Indeed, both RA and BE significantly increases morbidity and mortality in comparison to the background population 5 and older studies even suggest a synergistic effect between the two diseases. 6,7
BE is often found as a comorbid condition to other pulmonary diseases such as chronic obstructive pulmonary disease (COPD), severe asthma and interstitial lung disease. Previous studies have found associations between RA and both COPD and asthma, with RA diagnoses both pre- and superseding the pulmonary diseases in a series of large follow-up studies. 8,9 Despite the complex interplay suggested between pulmonary disease and RA, the mechanisms at play remain unknown.
Inflammation is a cornerstone of RA 10 pathogenesis and a viscous vortex of inflammation has been suggested in BE. 11 As such, the underlying chronic inflammatory changes seen in both RA and BE may suggest an association, or perhaps even a causal relationship, between the two diseases. A more detailed understanding of the interaction between the two diseases may pave the way for more targeted therapy for the individual patient. The aim of this systematic review and meta-analysis is to provide an estimate of the prevalence of BE in RA, as well as an update on the current knowledge of a possible association between RA and BE.
Methods
The present review was performed according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses. 12 The searches were conducted using the terms ‘Bronchiectasis AND Rheumatoid Arthritis’ on the PubMed during May 2020, without a limitation on study publication date. Studies were eligible for inclusion in the present review provided they fulfilled the following criteria: (I) Investigating the prevalence of and/or association with bronchiectasis in patients with RA, and (II) reporting observations from original studies, and none of the following exclusion criteria: (I) non-original research paper i.e. review (II) non-English paper. Studies without prevalence measurements were included to provide contextual evidence towards an association between BE and RA but were not included in crude prevalence estimations. The identified studies were screened for eligibility for this review based on title and abstract, and the full papers possibly fulfilling the criteria were then reviewed for inclusion.
Data was extracted using manual review by RW, and studies included, as well as data extracted, were independently verified by KEJH and CSU.
Variables sought in data extraction were: (I) Study population size and absolute and/or relative prevalence of BE, (II) Definition of BE (including diagnostic modality and/or criteria: Radiological disease or Clinical disease, (III) Definition of RA: diagnostic criteria used (including any serological tests), (IV) Reporting of important confounders: age, gender, smoking status, (V) Timing of disease: Reporting of BE-before-RA or RA-before-BE.
All included studies were individually assessed for risk of bias and quality using the risk of bias assessment tool for prevalence studies by Hoy et al. 13 Draft and summary scores are reported in Table 1. Furthermore, an overall assessment of quality of evidence was performed as per GRADE recommendations. 52
Overview of studies investigating the association between RA and BE.a
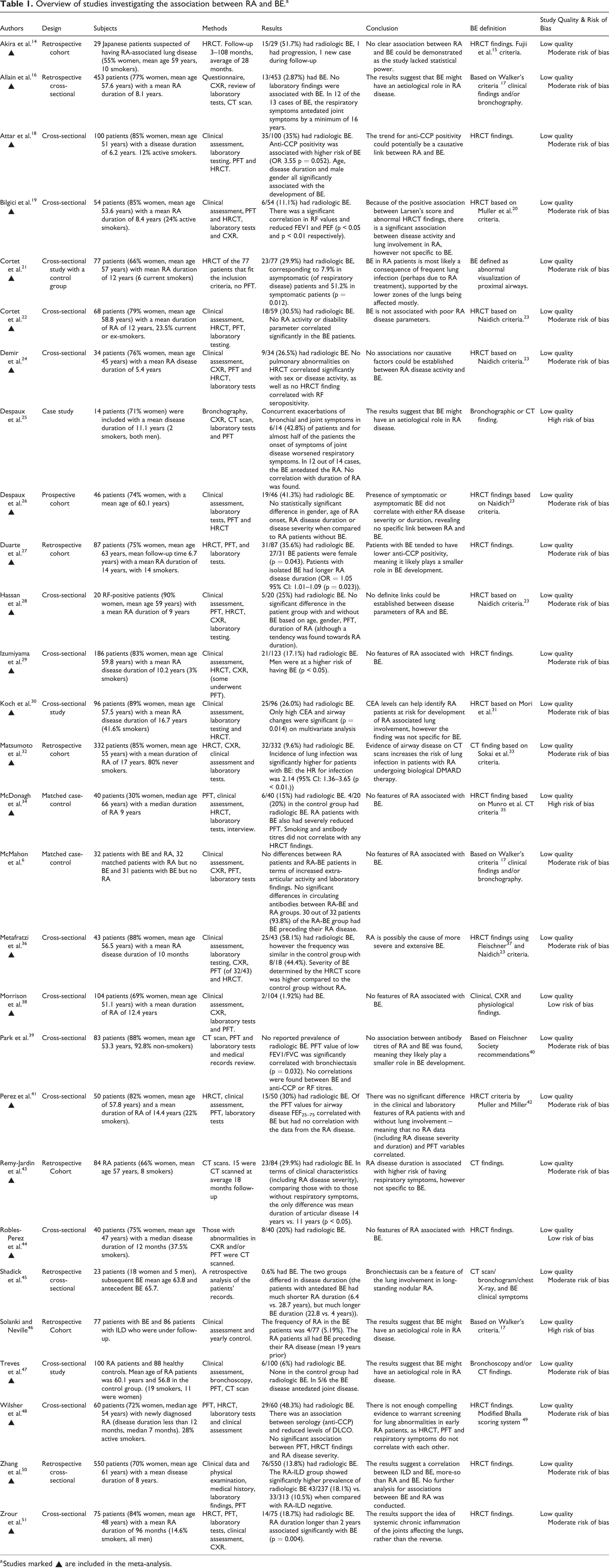
a Studies marked ▴ are included in the meta-analysis.
Finally, a meta-analysis of study prevalence measurements was performed. Studies were included if they provided relevant prevalence data, and by that case-control studies and case-series were excluded, unless they reported background prevalence from the case population. For the meta-analysis of prevalence, a generalized linear mixed model with logit transformation was used 53 and individual study confidence intervals were estimated using Clopper-Pearson exact binominal intervals. Study estimate heterogeneity was investigated using I2 and corresponding Tau 2 scores, and a likelihood-ratio test for heterogeneity. Assessment of publication bias was performed using a funnel plot and Eggers’ test. 54 All statistic calculations were performed using the Meta package version 4.9-6 for R 4.0.3 (The R Foundation, AU).
Results
The search algorithm yielded a total of 250 results, 222 of these were excluded based on the predefined criteria (Figure 1). The remaining 28 papers were included and reviewed: 19 cross-sectional studies, 6 prospective or retrospective cohort/follow-up or database studies, 2 case-control studies and 1 case-series study.
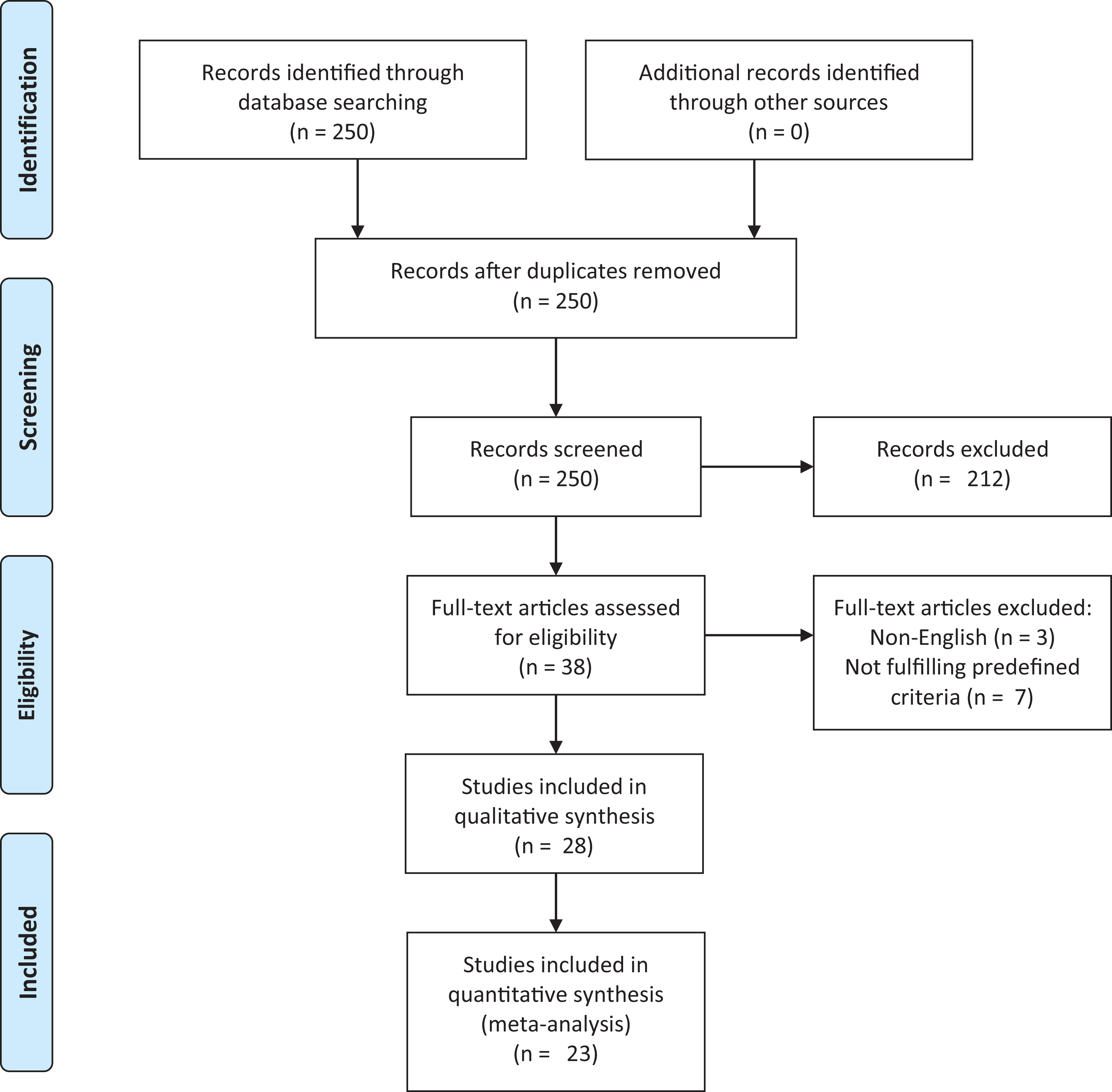
Flow-chart depicting the selection process of the included studies addressing aspects of a possible association between rheumatoid arthritis and bronchiectasis (according to PRISMA).
Individual assessments of studies are available in Supplementary Material 1, and the following results are based on narrative synthesis and meta-analysis of the individual studies.
Included populations and diagnosis of bronchiectasis
The studies included in the present systemic review encompasses 2723 patients with RA, of which 76.3% were reported as female. The crude mean age was 56.1 years (standard deviation (SD) 5.2) and an average of 20% (SD 11.3%) of included patients were reported as current or ex-smokers.
Definitions of BE used varied among the included studies, with four studies. 6,16,38,45 using clinical diagnostic criteria. The most commonly used clinical criteria was Walker’s, 17 used in three out of four studies. The study performed by Despaux et al. 25 did not specify adherence to a specific clinical protocol. In terms of radiologic diagnostic criteria, no less than eight different scoring protocols were used, with the Naidich criteria being the most commonly used. 23 Eleven studies did not use a previously published scoring protocol for radiologic diagnosis of BE (Table 1).
Prevalence of bronchiectasis in patients with rheumatoid arthritis
The reported prevalence in the included studies ranged from 1.92% 38 to 58.1%. 36 In the meta-analysis, the 23 studies marked in Table 1 were included. Five studies were excluded due to missing data on population size or ii) no usable relative measurement reported due to study design i.e. matched case-control or case-series studies. Before meta-analysis, studies were stratified by diagnostic criteria, that is Clinical or Radiologic.
In the pooled prevalence estimates, the prevalence of BE in RA was 21.1% (95% CI: 15.0%–28.9%) using a random effects model (Figure 2). Significant differences in prevalence were seen between Clinical (2.69% (95% CI: 1.63%–4.42%)) and Radiologic (24.9% (95% CI: 19.2%–31.7%)) diagnostic modalities (Q = 63.33, (p < 0.0001)).

Pooled prevalence estimates of bronchiectasis in patients with rheumatoid arthritis.
Overall, significant heterogeneity was observed in reported prevalence with an I2 of 92% and a Tau 2 of 0.94; with a corresponding significant Likelihood-Ratio test for heterogeneity (p < 0.001).
When assessed for publication bias using a funnel plot, a large spread in effect size was seen as a large spread in Hedges’ g. However, Eggers’ test performed without subgrouping was insignificant (Figure 3).
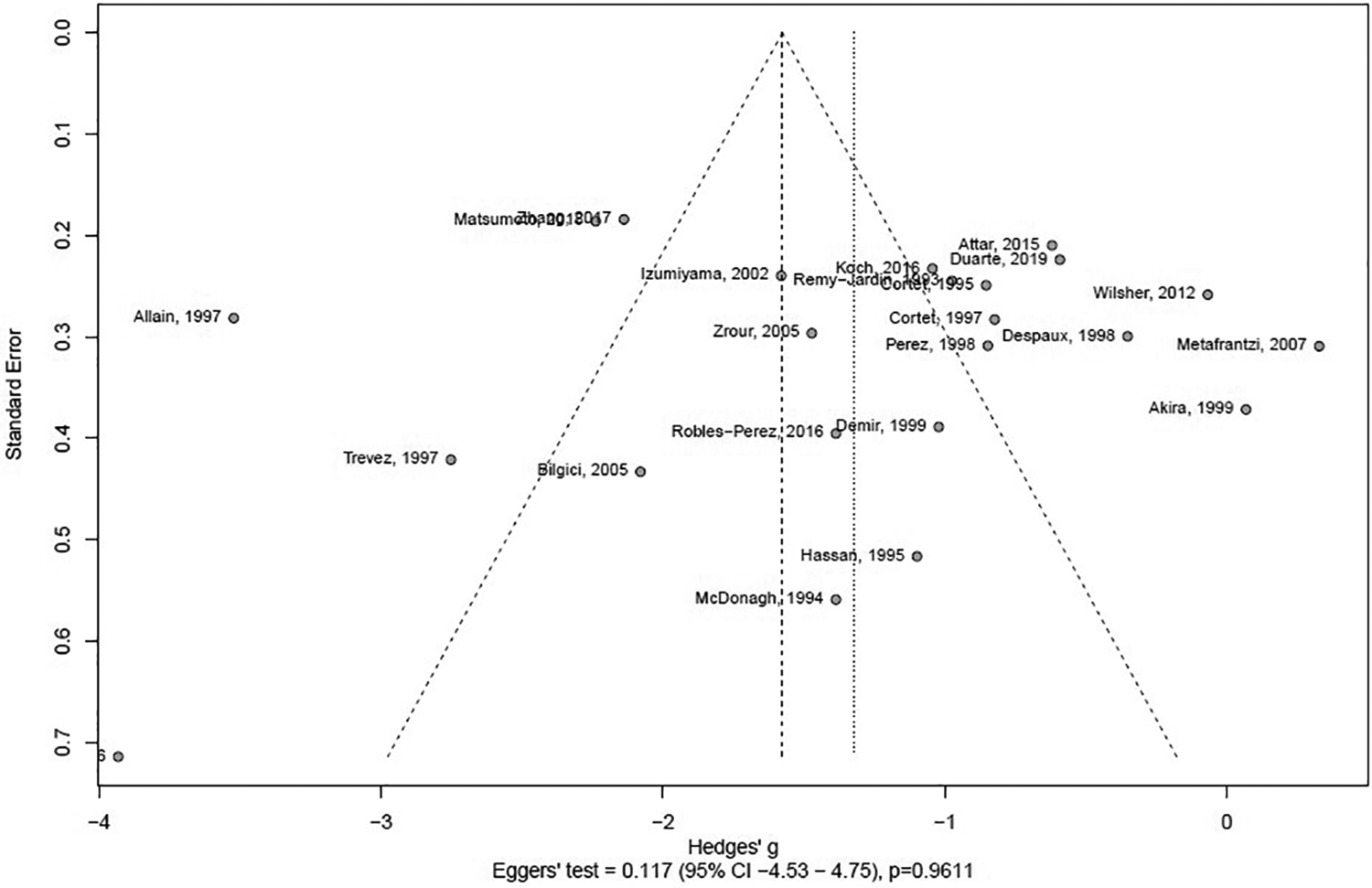
Assessment of potential publication bias of studies addressing the possible association between bronchiectasis and rheumatoid arthritis.
An overall GRADE quality of evidence-rating of ‘low quality’ was found in the present review and meta-analysis due to the observational nature of included study as well as several methodologic weaknesses including differing diagnostic criteria for BE with large effect on estimated endpoints (clinical versus radiologic disease prevalence).
Temporality and causality between rheumatoid arthritis and bronchiectasis
Five studies reported on temporal association between symptoms/diagnosis of BE and RA. 6,16,25,46,47 In all five studies, BE was found to antedate RA in 53 out of 69 cases, corresponding to 91.3%. Additionally, five studies 18,27,43,45,51 reported exposure time (to either BE or RA) to be a significant predictor of BE-in-RA, while four found inconclusive results or no association between exposure time and BE-in-RA. 25,26,28,41 Additionally, Despaux et al. 25 reported simultaneously occurring arthritic flare-ups and exacerbations of BE in 42.8% of included patients.
In terms of causality, no studies claim to present proof of causality between BE and RA. Sixteen of the included studies self-report as negative studies, 6,14,22,24,26,28,29,30,34,38,39,41,43,44,48,50 while five studies suggested associations based on biological/biochemical processes, 18,21,27,45,51 four based on (temporality of) symptoms 16,25,46,47 and three based on prevalence of radiographic findings. 19,32,36
Discussion
Prevalence of BE based on clinical findings compared to the radiologic finding of BE
The prevalence of BE in these studies varies greatly based on how BE is defined. Of the 28 studies included, only 5 used clinical symptoms to diagnose BE, the remaining used radiology, such as computed tomography, exclusively. The prevalence in the studies using clinical BE definitions varied from 0.6% 45 to 2.87%. 16 In the studies where BE was defined by CT scan the prevalence varied from 6% 47 –58.1%, 36 suggesting that the overwhelming majority of BE found radiologically does not present clinically, but longitudinal studies are needed to determine the importance of these findings. In the Dong et al. 55 pilot study for patients with early RA (average of 1 year RA disease duration and using CT for BE definition) they found a prevalence of 6%, however patients were excluded if they had existing respiratory diagnoses, and thus likely underestimating the true prevalence. In the cross-sectional study by Metafratzi et al. 36 they found a prevalence of 58.1% BE in early RA patients (without respiratory symptoms), however this was not different from their healthy control group of 18 non-smokers, but the extent of BE was greater in the RA patients when using their semiquantitative grading system. The findings from this study suggest that the origin of BE is not RA, but it is somehow exacerbated by the disease. This is further supported by the finding from Despaux et al. 25 that exacerbations of bronchial and joint symptoms coincided in 43% of patients, as well as the fact that symptoms of joint disease worsened these respiratory symptoms in over half of the study group.
In our meta-analysis, the pooled prevalence estimate ranged from 2.69% when using clinical parameters for defining BE presence, and an almost 10-fold increase to 24.9% when using purely radiological features. The differing prevalence estimates underlines the need for unified definition of BE in research when studying prevalence going forward and cements the increased prevalence, regardless of radiologic or clinical definitions, when compared to approximately 0.5% clinical BE in the background population. 5
Is there a causal relationship between RA and BE?
Early childhood lung infections and exposure to smoke and other pollutants are known risk factors of developing bronchiectasis 56 and are not unique to RA patients, however, their RA disease severity and other RA-related parameters seemingly are. Most of the studies included in this review used an RA classification system to determine disease severity of their patients, as well as blood sampling to determine antibody titres and clinical parameters such as RA disease duration. However, not all these studies then analysed their findings regarding the risk of BE. Of the ones that looked at BE specifically, Attar et al. 18 found anti-CCP positivity was associated with higher risk of BE with an OR of 3.55, (p = 0.052), and while disease severity was not significantly associated with BE development, another study showed that age, male gender and disease duration was. 29 In contrast to the findings of Izumiyama et al., 29 other studies found that female gender was significantly associated with BE in these RA patients (p = 0.043), while also finding a significant correlation with longer disease duration (OR = 1.05 95% CI: 1.007–1.095, p = 0.023), 27 but no association with any antibody titres and BE development. 39 In the study by McMahon et al. 6 in 1993 they found essentially no differences in disease severity and antibody titres between patients with RA and BE compared to patients with RA alone. The lack of differences between the two groups is contradictory to later findings but might be explained by the relatively small group consisting of only 64 patients. Additionally, in the study by Despaux et al. 26 regardless of statistical significance, the clinical significance of a decrease in lung functionality should be considered as 80% of those with radiologic BE and respiratory symptoms had abnormal PFT, whereas only 62% of those with no respiratory symptoms (but still radiologic BE) had abnormal PFT. Larsen’s score to determine RA disease severity was used in the study by Bilgici et al., 19 and they found high Larsen’s score statistically increased risk of an abnormal HRCT finding (OR 2.04 95% CI: 1.14–3.66, p < 0.01). This was however not specific to BE, as it included findings such as pulmonary fibrosis. Metafratzi et al. 36 found no such correlation between HRCT abnormalities and RA disease severity using the same scoring system. In a similar vein, Despaux et al. 26 found no such correlation to BE specifically, although they did use a different method to determine disease severity, in this case Steinbrocker Functional Classification, signalling the importance of standardized classification systems to improve comparability of future studies.
Potential causal pathways linking BE and RA
An emerging theory regarding the possible connection between RA and BE is based on antigenic stimulation from the mucus in patients with BE. This hypothesis reverses the causality, so that BE is ultimately the cause of RA. Some studies support this hypothesis by having most of their RA patients, if not all, with antecedent BE, ranging from 83.3% 47 to 100%, 46 while others have only 21.7% of their RA patients having preceding BE. 45 Allain et al. 16 had 92.3% with preceeding BE and Despaux et al. 25 and McMahon et al. 6 had 85.7% and 93.8% with preceding BE, respectively. The potential causative mechanism that could explain this has yet to be adequately established, however, it fails to explain why some RA-BE patients develop RA before their BE.
Another possible theory is based on the idea that the systemic inflammation of the joints in RA disease somehow also affects the lungs, which would in-part explain antedated RA in RA-BE. Studies have investigated the importance of the autoantibodies associated with RA, specifically RF and anti-CCP. None of the included studies have found a connection between RF and increased prevalence or severity of BE. In fact, Duarte et al. 27 found a trend that RA-BE patients had lower anti-CCP positivity when compared to those with no BE (p = 0.074), and Park et al. 39 found that there was no such association. Attar et al. 18 did, however, find an association between anti-CCP and higher risk of BE (OR 3.55, p = 0.052) as the only positive association, suggesting that these autoantibodies likely play a very small role in BE development in RA patients.
Genetic factors play a role in RA development, but very few studies have investigated this in relation to BE development. Bilgici et al. 19 found that HLA-DR1, which is associated with RA, was not significantly increased in RA-BE patients, although this might have been due to the small subject size of the study. Other genetic factors such as the cystic fibrosis transmembrane regulator (CFTR) gene mutations potentially play a role for heterozygous RA-BE patients, as a study has shown allele mutations in 20 out of 55 BE patients, 57 potentially predisposing to BE. However, Solanki and Neville 46 argued that patients with cystic fibrosis and BE, who later develop RA, are affected by a unique seronegative variant, and thus is a different mechanism of association than the one connecting isolated BE and RA.
Studies that found that longer RA disease duration was associated with the development of BE support the theory of systemic joint inflammation affecting the lungs. For example, Zrour et al. 51 found that RA duration over 2 years associated with BE (p = 0.004) and Duarte et al. 27 found that patients with isolated BE had longer RA disease duration (OR = 1.05 95% CI: 1.01–1.09 (p = 0.023)) compared to patients with ILD. It is not unlikely, however, as most of these patients were undergoing treatment for RA with immunosuppressants, that this is partly (if not entirely) because of their increased susceptibility to recurrent infection, and thus at an increased risk of developing BE, 57 and much in the same way, this would not explain why some develop BE prior to their RA.
The different diagnostic criteria for RA and BE
There are many different scoring systems, classification methods and diagnostic criteria for both RA and BE, and a major limitation during analysis of the present studies is the lack of validation and the low comparability between the different methods used. Many of the included studies did not specify how they defined diagnosis of BE, other than stating that it was identified using HRCT/CT. 18,27,29,43,44,50,51 Although explicitly defined, the rest of studies that used exclusively radiological criteria for BE diagnosis also had varying classification systems, further increasing the heterogeneity of the studies. This can also be said for RA disease severity scoring, as DAS-28, Larsen score, Stoke Index and Steinbrocker Functional Classification all measure this parameter, and simultaneously display the need for standardization.
Conclusion
Based on a meta-analysis of the currently available literature, there is an increased prevalence of both radiologic and symptomatic BE in patients with RA compared to the general population, most likely due to an increased risk of recurrent respiratory infections. Despite the many hypothesized causal pathways, such as mucus-antigenic stimulation, there is not enough evidence to suggest a single causal link between BE and RA. Standardization ranging from classification systems to diagnostic criteria of RA and BE, as well as further long-term prospective studies with unified diagnostic criteria are direly needed to uncover the risk of developing BE in
Supplemental material
Supplemental Material, sj-pdf-1-crd-10.1177_1479973121994565 - A causal relationship between rheumatoid arthritis and bronchiectasis? A systematic review and meta-analysis
Supplemental Material, sj-pdf-1-crd-10.1177_1479973121994565 for A causal relationship between rheumatoid arthritis and bronchiectasis? A systematic review and meta-analysis by Rafal Wiater, Kjell Erik Julius Håkansson and Charlotte Suppli Ulrik in Chronic Respiratory Disease
Footnotes
Authors’ note
RW and KEJH share first authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
