Abstract
Objectives
Dysglycemia is known to be a common comorbidity of chronic obstructive pulmonary disease (COPD). However, undiagnosed dysglycemia and the associated factors remain under-reported. This study aimed to determine the prevalence and the associated factors of dysglycemia among COPD patients.
Methods
This was a cross-sectional, single-center study involving adults with established COPD (n = 186) divided into those with or without hospital admissions for acute exacerbation. Oral glucose tolerance test (OGTT) was performed in patients with no known history of dysglycemia.
Results
There were 16 patients who had overt diabetes, and 32 had prediabetes following the OGTT. Forty percent had histories of hospital admissions for COPD exacerbations. Both groups demonstrated similar 2-h post prandial glucose, glycated hemoglobin (HbA1c) and fasting blood glucose. The incidences of newly diagnosed dysglycemia were higher in both groups (40.8% vs 34.6%, p = 0.57). Cumulative days of admission (≥6 days/year) and weight (≥65 kg) were identified as predictors for dysglycemia within the study population.
Discussion
This study demonstrated a high number of overt and newly diagnosed dysglycemia among COPD patients who had no previous history of abnormal glucose. Recent acute exacerbations of COPD could have a negative impact on glycemia, although the results did not attain statistical significance. However, there is a need for adequate screening for dysglycemia, particularly among those with frequent acute exacerbations of their condition.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by progressive partially reversible airflow obstruction and airways inflammation 1 and is expected to be the third leading cause of death worldwide by 2030. 2 A population-based Burden of Obstructive Lung Disease (BOLD) collaborative study in 2016 reported the estimated prevalence of COPD in Malaysia to be 6.5%, affecting more men (8.5%) than women (4.5%). 3
Dysglycemia is defined as either diabetes mellitus (DM) or elevated blood glucose that has not attained the DM threshold, also known as prediabetes, which is further categorized as impaired fasting glucose (IFG) and impaired glucose tolerance (IGT)). The prevalence of dysglycemia has been increasing at an alarming rate, particularly in the middle- and low-income countries. 4 The Malaysian National Health and Morbidity Surveys have reported a steady rise in the overall prevalence of type 2 diabetes mellitus (T2DM) among adults above the age of 18 from 17.5% in 2015 to 18.3% in 2019.5,6 The relationship between dysglycemia and COPD has long been recognized,7,8 with the prevalence ranging between 2% to 37% among patients with COPD who were hospitalized. 9 Although this wide range reflects different study populations, one large observational study by Mannino et al. has reported a reasonable estimation of 12%. 10 The relationship between COPD and T2DM could be explained by multiple pathophysiological alterations, including inflammation and oxidative stress, insulin resistance, weight gain, and changes in metabolism of adipokines such as interleukin-6. 11
COPD patients with acute exacerbation have additional risk for impaired glucose metabolism, including overt DM and prediabetes. 9 What was more alarming was that the majority of the COPD patients were unaware of this underlying comorbidity. Moreover, DM is associated with more frequent exacerbations and poorer outcomes, including COPD patients. 11 Several studies confirmed that dysglycemia prolonged lengths of hospital stay and increased risks of death in patients with acute exacerbations of COPD .12–14 Thus, this study aimed to determine the prevalence and associated factors of dysglycemia among COPD patients attending our tertiary center in Malaysia, particularly the acute exacerbations and hospitalizations.
Subjects, materials and methods
This was a cross-sectional, single-center study conducted among a group of patients who were attending the Respiratory Clinic at a Tertiary Referral Centre in Malaysia between September 2016 to March 2017. Patients aged 30 years and above with an established diagnosis of COPD were included. Diagnosis of COPD, and exacerbation of COPD were ascertained according to the definition outlined by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines 2016 edition. 15 Exclusion criteria were diagnosis of bronchial asthma or undetermined chronic respiratory conditions, co-existing systemic illness on long-term corticosteroid therapy, malignancy, and pregnant or within a peripartum period. The study was approved by the Research Ethics Committee of our institution, and all patients gave written informed consent. The investigation was performed in conformity with the ethical principles of the Declaration of Helsinki.
Participants were subsequently categorized into a group with a recent history of acute exacerbation of COPD requiring hospital admission within the past 1 year or those with a stable disease without recent acute exacerbation. Hospital admission was defined as any hospital stay, including emergency room admission, of more than 24 h. Based on the Malaysian Practice Guidelines on the Management of Obesity, patients were classified into various categories according to their body mass index (BMI) expressed as kg/m2, that is, underweight <18.5, normal 18.5–22.9, overweight ≥23.0, pre-obese 23.0–27.4, and obese ≥ 27.5. 16 All patients had an additional 5 ml of venous blood drawn following an overnight fast prior to routine clinic appointments. Serum samples were separated from blood following centrifugation and stored at −20°C until further analysis. Those without a prior diagnosis of T2DM were subjected to an oral glucose tolerance test (OGTT), which was a 2-step venesection after 8-h fasting and at 2 h following 75 g of glucose consumption. Those with recent exacerbations had their blood samples taken at least 6 weeks after discharge. Values for the diagnosis of dysglycemia were based on the fifth edition of the Malaysian Clinical Practice Guidelines (CPG) on Management of Type 2 Diabetes Mellitus 2015, which defined IFG as fasting glucose levels between 6.1 and 6.9 mmol/l, IGT as 2 h postprandial of between 7.8 to 11.1 mmol/l, and diabetes mellitus as when serum fasting glucose was more or equal to 7.0 mmol/l and/or 2 h postprandial of more than 11 mmol/l. 17
Serum and plasma were analyzed for glycated hemoglobin (HbA1c) and glucose on automated platforms at an ISO 15,189 accredited clinical diagnostic laboratory. HbA1c was analyzed by a semi-automated analyzer D-10 (Bio-Rad Laboratories, USA), while glucose was analyzed on an automated analyzer c501 (Roche Diagnostics, Germany).
Statistical analysis
All statistical operations were performed using IBM SPSS (Statistical Package for the Social Sciences) Version 24. Comparison of continuous variables was made using independent t-test, while categorical data using chi-square. Univariate ANOVA was used to evaluate differences between two or more groups. Based on these results, logistic regression models were constructed to ascertain the effects of significant variables on the likelihood of dysglycemia. Correlations between two continuous variables were analyzed using Pearson’s correlation coefficient (normally distributed data) and Spearman’s rank correlation coefficient (non-parametric data) tests, of which Cohen’s (1988) cut-off criteria were used for interpretation of correlation strength. For variables, which were statistically significantly correlated, simple and multiple linear regressions were used. The significance level was all set at p < 0.05. The calculated sample size to reach a confidence level of 95% was a total of 212 patients. 18
Results
A total of 186 patients were recruited with a mean age of 67.9±9.1 years old, 95.2% were male patients (n = 177) and only nine were females. Approximately a third of the patients were pre-obese (30.1%), while another third was obese (33.3%), with a mean weight of 67.35 ± 16.68 kg and a mean BMI of 25.17 ± 6.08 kg/m2. Comorbidities included hypertension, dyslipidemia, and ischemic heart disease, which were reported in 61%, 43% and 31% of the study population, respectively. Almost all participants were smokers with a mean smoking duration of 46.4 ± 24.7 pack years, out of which 24.7% (n = 46) were active smokers. The median duration of COPD diagnosis was 3.0 (IQR 3.0) years, with more than half of the study cohort (55.4%) classified as GOLD D. Most of the participants (53.2%) were prescribed regular inhaled corticosteroids, mainly fluticasone (71.7%). Further details of the baseline demographics of the overall study population are presented in the Supplementary Table.
Demographic and anthropometric data within overall study cohort (n = 186). Comparison between the dysglycemia and normoglycemia groups.

Data are presented as median (interquartile range) or mean±standard deviation (SD) for continuous variables and percent for categorical variables. p-values are results of Mann-Whitney test for continuous data for between the groups, and Fisher’s exact test for categorical data. BMI, body mass index; COPD, Chronic Obstructive Pulmonary Disease; GOLD, Global Initiative for Chronic Obstructive Lung Disease ICS, Inhaled Corticosteroid; LTOT, Long Term Oxygen Therapy.
Study population who underwent OGTT (n = 130). Comparison between groups based on recent hospital admission for exacerbation of COPD.

Data are presented as median (interquartile range) or mean±standard deviation (SD) for continuous variables and percent for categorical variables. p-values are results of Mann-Whitney test for continuous data for between the groups, and Fisher’s exact test for categorical data. HbA1c, glycated Hemoglobin.
Comparison of subject sociodemographic characteristics based on recent hospital admission for exacerbation of COPD.
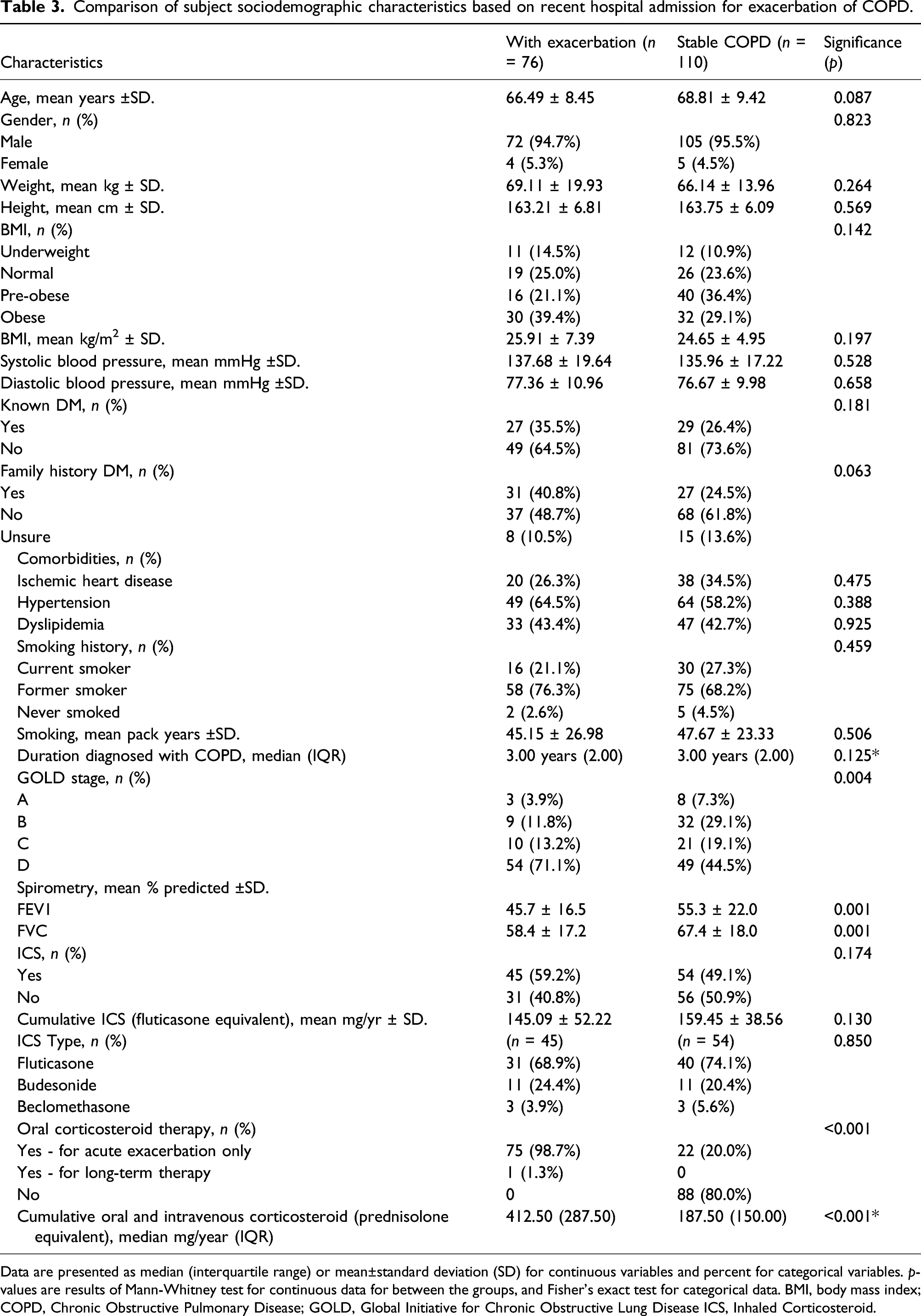
Data are presented as median (interquartile range) or mean±standard deviation (SD) for continuous variables and percent for categorical variables. p-values are results of Mann-Whitney test for continuous data for between the groups, and Fisher’s exact test for categorical data. BMI, body mass index; COPD, Chronic Obstructive Pulmonary Disease; GOLD, Global Initiative for Chronic Obstructive Lung Disease ICS, Inhaled Corticosteroid.
Correlation analyses between glycemic status, represented by HbA1c, and clinical characteristics were performed. Two variables had moderately significant positive correlations, that is, weight (r = 0.365, p ≤ 0.001) and BMI (r = 0.345, p ≤ 0.001). These remained significant within each individual group with exacerbations and those with stable disease, which may suggest that these variables may be conventional risk factors for dysglycemia and thus independent from episodes of exacerbations.
Utilizing simple logistic regression analysis, weight of ≥65 kg (OR 2.282, CI 1.26–4.12, p = 0.006), overweight with BMI 23–27.4 kg/m2 (OR 2.59, CI 1.40–4.77, p = 0.002), hypertension (OR 0.49, CI 0.27–0.89, p = 0.019), and cumulative hospital admission of more >6 days per year (OR 2.89, CI 1.04–8.03, p = 0.035) were significant risk factors for developing dysglycemia within the study cohort. Subsequent multivariate logistic regression analysis demonstrated that patients with weight ≥65 kg and with hospital admission of >6 days a year were 2.7 and 4.5 times more likely to develop dysglycemia, respectively (p = 0.026 and 0.015). The regression model was statistically significant, χ
2
(2) = 11.644, p = 0.003. It correctly classified 68.8% of the cases (>70% for good model fit). There was no multicollinearity and no interaction in this final model. By performing the Hosmer and Lemeshow test, the p-value was 0.933 (>0.05 for good model fit). The area under the ROC curve (AUC) was 75.4% (>70% is acceptable to fit), with a confidence interval 0.641–0.868, p ≤ 0.001 (Figure 1). Area under the ROC curve (AUC) of regression model for dysglycemia in COPD. Note: COPD: chronic obstructive pulmonary disease.
Discussion
The prevalence of known T2DM within our study cohort of 30.1% was significantly higher than previously reported by local data, that is, 19.2% by Ban et al., and 17% by Al Aqqad et al.19,20 This could be explained by the larger sample size of our study population but also likely due to the general increase in the prevalence of T2DM as reported in the National Health and Morbidity Survey 2015 and 2019.5,6 What was even more striking, however, was the alarmingly high rate of undiagnosed dysglycemia within this group of patients following a standard OGTT, whereby the prevalence of dysglycemia was 55.9%. This observation demands attention on the number of high-risk patients who were undiagnosed and remained unaware of the complications associated with delayed diagnosis of the disease. While there was no corresponding local data on prediabetes, Forde et al. reported 54% of dysglycemia and 4% overt T2DM within a similar study population. 13 However, the study utilized HbA1c as a diagnostic tool rather than OGTT, which was easier to perform but could be inferior in accuracy depending on the assay. Although fasting blood sugar would have been a simpler and cheaper alternative, this could result in falsely low figures as demonstrated by a negligible number of impaired fasting glucose compared to the high number of IGT within our cohort.
The prevalence of COPD patients admitted for acute exacerbations within the past year of 39.8% was notably high compared to the average within the Asia-Pacific region, which had been reported as 19%. 21 This may have been attributed to the differences in the study cohorts, whereby our participants had mainly severe and complicated COPD within the GOLD D classification. There were also the regional factors to be considered, including weather or climate changes, particularly the recurring and critical problem of haze that regularly engulfed the South Asia region. This could have influenced the rate of exacerbations and hospital admissions that we observed.
To the best of our knowledge and to date, this would be the first local data to report the prevalence of newly diagnosed dysglycemia following an OGTT among this particular group of patients. Unfortunately, we were unable to demonstrate a significant association between dysglycemia and exacerbations of COPD. However, we concur with data from Mekov et al., who reported an incidence of newly diagnosed diabetes of 21.7% and prediabetes of 30.9% among COPD patients admitted for exacerbations. 22 We opine that their findings were probably higher due to different timing of OGTT sampling, which they performed on average at the fifth to seventh-day post exacerbation compared to our study, which was done at least 6 weeks after hospital discharge. We believe that the 6-week timeline was more appropriate to detect the stable conditions of dysglycemia.
We support the finding that more patients within the dysglycemia group had been admitted within the past year due to COPD exacerbation, with more frequent admissions and longer hospital stay.12,13 As dysglycemia is associated with altered lung volumes and reduced flow rates, making patients more susceptible to lung infections on the background of impaired immune responses, there could have been more complications in this group of patients.23–25 Furthermore, systemic steroids given to most exacerbation cases aggravate hyperglycemia, thus could have contributed to the tendency for longer inpatient care to attain euglycemia. 26 However, there was a higher percentage of patients in the exacerbation group who received inhaled corticosteroid (ICS) compared to the stable group, the cumulative dose of ICS used was lower in the latter, albeit not significant. One possible explanation was the prior withdrawal of ICS and substituted to a corticosteroid-free inhalation medication or systemic therapy during the periods of exacerbations. We report similar findings by Habib et al. 27 that showed no statistically significant correlation between systemic corticosteroid use, in both oral and parenteral form, with dysglycemia. The cumulative dose of systemic steroid use was calculated from the preceding year, which could have been an underestimation. This, however, underscores the importance of a large, prospective, longer duration, controlled clinical trial to identify the risk versus benefit of this commonly used therapy.
Limitations of study and recommendations
Due to time constraint and patients’ reluctance for OGTT, the calculated sample size was not achieved. This was partly due to a number of patients who declined the OGTT (n = 58). This study included particularly high-risk COPD patients and may not represent mild diseases. The lack of data on oral corticosteroids also hindered accurate representation of the association with steroid use. Future studies should be conducted in a larger, more diverse cohort involving multiple stages of the disease. A long-term prospective, observational study would be ideal for determining the associations between COPD exacerbations, use of medications and dysglycemia in this unique elderly population.
Conclusion
In conclusion, this study revealed a near doubling of the prevalence of dysglycemia following a standard oral glucose challenge test in a group of patients with COPD with no known dysglycemia. In addition to other recognized comorbidities, patients who were overweight, had more frequent acute exacerbations, and had longer hospital stays were more likely to develop dysglycemia. Therefore, this study underscores the need for screening for dysglycemia with the use of oral glucose tolerance tests in patients with COPD requiring multiple hospital admissions.
Supplemental Material
sj-pdf-1-crd-10.1177_14799731211056348 – Supplemental Material for Prevalence and associated factors of dysglycemia among patients with chronic obstructive pulmonary disease
Supplemental Material, sj-pdf-1-crd-10.1177_14799731211056348 for P and associated factors of dysglycemia among patients with chronic obstructive pulmonary disease by Khairil K Zulkifli, Fatimah Z Mohamed Shah, Ahmad I Ismail, Thuhairah H Abdul Rahman and Rohana A Ghani in Chronic Respiratory Disease
Footnotes
Acknowledgments
We would like to thank all the medical and non-medical personnel involved in the data collection. We also would like to convey our gratitude to the patients’ advisers.
Author contributors
KKZ contributed to the data collection, data analysis, and the initial writing of the manuscript. FZMS contributed to the initial study design, data collection, and manuscript drafting. THAR contributed to the laboratory investigations, interpretation of data, and review of the draft. RAG conceived the original idea of the study and contributed to the final writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study was performed in accordance with the Helsinki Declaration. The protocol was approved by the Universiti Teknologi MARA Research Ethics committee on 20 September 2016. [REF: 600-IRMI (5/1/6)]. All participants gave informed written consent. The data underlying the results presented in the study is available from the corresponding author upon request.
Informed consent
Patients who fulfilled the inclusion criteria were approached, explained and informed regarding the main objective of the study. Confidentiality was assured to the respondents. Written consent was signed by respondents before proceeding with the study. Patients are allowed to opt-out of the study at any time should they wish to
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
