Abstract
Objective
We explored risk factors for cognitive frailty in older patients with chronic obstructive pulmonary disease (COPD) and diabetes mellitus to develop and verify a risk prediction model for cognitive frailty.
Methods
This was a cross-sectional study. Convenience sampling was used to randomly select 378 patients hospitalized between February 2022 and December 2023. We allocated 265 patients who visited between February 2022 and February 2023 to a modeling group to analyze risk factors for cognitive frailty and create a logistic regression model for risk prediction. Another 113 patients who visited between March 2023 and December 2023 were included in a validation group for model verification.
Results
The cognitive frailty incidence in the 265 patients was 35.09% (93/265). Regression analysis showed that age >80 years (odds ratio [OR] = 4.576), regular exercise (OR = 0.390, polypharmacy (OR = 3.074), depression (OR = 2.395) duration of COPD combined with diabetes (OR = 1.902), Family APGAR index score (OR = 0.428), and chronic pain (OR = 2.156) were factors influencing the occurrence of cognitive frailty in older patients with COPD accompanied by diabetes.
Conclusions
The constructed risk prediction model for cognitive frailty in older patients with COPD and diabetes showed good predictive value, aiding in the clinical identification of high-risk patients and facilitating timely intervention and guidance.
Keywords
Introduction
According to the eighth national population census of the Chinese National Bureau of Statistics in 2023, the number of older people aged >60 years in China has reached 297 million. This group accounts for 21.1% of the country’s population, 1 and this number is expected to reach 490 million by 2050. 2 Chronic obstructive pulmonary disease (COPD) 3 is a common respiratory disease and a serious human health concern characterized by persistent respiratory symptoms and airflow limitation. Diabetes mellitus is a common comorbidity of COPD, as well as a prevalent disease among older adults and a worldwide public health problem, with severe effects on human health. Compared with the general population, patients with COPD experience a higher incidence of diabetes (10.5% vs. 30.45%). 4
Although earlier studies have investigated cognitive impairment and physical frailty separately, increasing evidence suggests that physical frailty and cognitive impairment have closely related pathophysiological mechanisms and that their interaction may accelerate the decline in body function. 5 Therefore, the International Society for Nutrition and Aging and the International Society for Geriatrics formally proposed the concept of “cognitive frailty” (CF) in 2013 6 wherein CF is defined as the simultaneous presentation of physical frailty and cognitive impairment in the absence of dementia and other neurodegenerative diseases.
There is a limited body of research on CF in COPD, with only one study conducted by Song 7 in China demonstrating that the prevalence of CF among older individuals with COPD is 31.9%. That study identified several influencing factors including age, daily engagement in intellectual activities, presence of comorbidities, disease duration, and lung function classification. There have been numerous studies conducted on the prevalence of CF in older patients with diabetes mellitus, revealing an incidence rate of 17.1% (ranging from 8.7% to 30.9%).8,9 Influencing factors encompass educational attainment, regular physical activity, nutritional status, depressive symptoms, and glycated hemoglobin (HbA1c) levels ≥7.0%. The influencing factors of CF in these two diseases are highly heterogeneous.
The incidence of CF in individuals with COPD combined with diabetes is currently unknown. Moreover, the influencing factors of CF observed in patients with COPD or diabetes may not apply to those having COPD combined with diabetes. However, given that diabetes is one of the most common complications and comorbidities of COPD, it severely affects the prognosis of patients with COPD and increases the social and family economic burden. Furthermore, individuals with COPD and/or diabetes represent high-risk groups for CF, exhibiting a high prevalence, severe adverse outcomes, and heavy disease burden. Currently, no drugs are available to successfully treat CF. Nevertheless, CF has been shown to be reversible. Thus, the early identification and management of patients at high risk of developing CF will greatly contribute to preventing and reversing CF, reducing the occurrence of adverse outcomes, and improving the functional status and quality of life in these individuals.
Patients and Methods
Study population
In this study, convenience sampling was used to randomly select older patients with COPD and diabetes mellitus who were followed up in the Department of Respiratory Medicine and Endocrinology of the Sixth People’s Hospital of Nantong from February 2022 to December 2023. Among them, patients who visited between February 2022 and February 2023 were allocated to a modeling group, and patients who visited between March 2023 and December 2023 were included in a validation group for model verification. Patients who fulfilled the following inclusion criteria were enrolled: (1) age ≥60 years; (2) meeting the diagnostic criteria for COPD 10 or having a previous history of COPD; (3) satisfying the diagnostic criteria for diabetes 11 or an earlier diagnosis of diabetes; (4) relatively complete clinical data that do not affect the statistical analysis; and (5) clear consciousness and normal communication skills. Patients were excluded if they met any of the following criteria: (1) a diagnosis of dementia or other neurological diseases; (2) unable to walk independently; or (3) severe visual impairment. This study was approved by the Ethics Committee of The Sixth People’s Hospital of Nantongeon January 25, 2022 (no. NTLYLL2022006). All patient details have been de-identified for this study. We obtained written informed consent from all included patients.
Research methods
Sample size estimation
This cross-sectional study was conducted in accordance with the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guidelines. 12 Twenty-two independent variables were included. According to the formula for calculating the optimal model sample size, 13 each independent variable required 5 to 10 patients with CF. Therefore, the total number of required patients was estimated as follows: 22 × 5 ×(1 + 0.2)/0.36 = 367 patients. Finally, 378 patients were enrolled in this study. According to a 2:1 ratio, one group of patients was allocated to the modeling group and another group was included in the validation group.
Research tools
General information questionnaire
Risk factors for CF in older patients with COPD and diabetes were determined based on a literature search,7,8 expert group discussions, and a self-designed questionnaire. After the initial stage of the preliminary investigation and debugging of the questionnaires, the information collected using the questionnaire comprised four main aspects:
Social demographic data: sex, age, family monthly income per capita, marital status, and education level. The per capita monthly household income was set according to the per capita monthly income of Nantong residents in 2021, i.e., 3907 RMB. Lifestyle and behavioral data: living situation, hospitalization history in the past year, fall history during the past year, regular exercise, sleep duration per night, polypharmacy, and chronic pain. The sleep duration was defined as 6 hours based on relevant research.
7
Polypharmacy was defined as taking five or more medications per day. Chronic pain was defined as the patient stating they had chronic pain when asked by a nurse. COPD-specific data: disease course and modified Medical Research Council (mMRC) dyspnea scale
14
classification. On the mMRC scale, a score of 0 indicates difficulty breathing only during strenuous activity whereas a score of 4 indicates severe breathing difficulty to the extent that the respondent cannot leave the house or experiences difficulty breathing when putting on or taking off clothes. Diabetes-specific data: disease duration, recent HbA1c value, and duration of COPD combined with diabetes.
Frailty assessment
The Frailty Phenotype (FP) scale, developed by Fried et al., is a widely used instrument to evaluate frailty in patients. 15 The items included in this scale cover weight loss, walking speed, grip strength, fatigue, and physical activity within 1 year. A “yes” for each item is scored as 1 point, and a “no” is scored 0 points, with the total score ranging from 0 to 5 points. The FP scale was translated by domestic researchers into Chinese in 2018 and has demonstrated a content validity of 0.98 and a Cronbach’s α coefficient of 0.826. 16
Cognitive assessment
The Montreal Cognitive Assessment (MoCA) scale is a standard screening tool for assessing mild cognitive impairment. This test was developed by Nasreddine et al. 17 The MoCA scale incorporates seven cognitive dimensions, including visuospatial and executive functions, naming, attention, language, abstraction, delayed memory, and orientation. This questionnaire has a total score of 30, with higher scores indicating better cognitive function. The Chinese version of the MoCA scale has exhibited good reliability and validity, yielding a test–retest reliability of 0.86 and a Cronbach’s α coefficient of 0.82. 18
Dementia assessment
The Clinical Dementia Rating (CDR) scale, developed by Hughes et al., 19 is a popular instrument used in clinical practice to diagnose the severity of Alzheimer disease and related dementias. The CDR scale is administered as semi-structured interviews of patients and their caregivers by trained professionals. On the CDR scale, a score of 0 is considered to indicate no dementia whereas scores ≥0.5 signify dementia.
Nutritional status
The Nutritional Risk Screening 2002 (NRS-2002) 20 is a valuable tool to investigate nutritional status and encompasses three parts: nutrition score (0–3 points), disease score (0–3 points), and age score (0–1 points). A total NRS-2002 score of ≥3 represents nutritional risk.
Depression
The Geriatric Depression Scale-15 (GDS-15) is a short and easily administered questionnaire developed by Yesavage et al. 21 in 1986 that is primarily used to evaluate depression in patients. This instrument has a Cronbach’s α coefficient of 0.793 and a test–retest reliability of 0.728. The GDS-15 comprises 15 items, with each item coded as 1 point; a score of ≥8 indicates depression.
Family APGAR index
The Family APGAR index 22 is a subjective quantitative evaluation tool for measuring satisfaction among family members with their family relationships. This index includes five items, where “usually” corresponds to 2 points; “sometimes,” 1 point; and “almost never,” 0 points. A total score of 7 to 10 indicates well-functioning families, and 0 to 6 points reflects moderate-to-severely dysfunctional families. The Family APGAR index used in this study had a Cronbach’s α coefficient of 0.80.
Self-efficacy
The Self-Efficacy for Managing Chronic Disease 6-Item scale developed by Bodenheimer et al. is a useful tool to determine the efficiency of respondents with chronic diseases in their self-care activities. 23 Each item on this scale is scored 1 to 10 points, with the average score of all six items considered the total score. A higher total score demonstrates a higher self-efficacy. According to scores on the self-efficacy index, patients with chronic diseases are classified as having high (<7 points), medium (4–7 points), or low (<4) levels of self-efficacy.
Operational definition of cognitive frailty (CF)
The condition of CF was first proposed by the International Society for Nutrition and Gerontology and the International Society for Geriatrics and Geriatrics in 2013. 6 In the consensus statement of these two groups, CF is defined as the coexistence of physical frailty and cognitive impairment in older adults without a clear diagnosis of dementia and independent from other frailty dimensions (such as social, psychological, and oral frailty). The specific diagnostic criteria for CF in this study were as follows: 6 FP scale score ≥3 points, MoCA scale score <26 points, CDR scale score = 0.5 points, and no clinical diagnosis of dementia.
Data collection and quality control
In this study, five investigators (three nurses and two doctors) were trained by the project leader. Doctors were responsible for conducting the CDR scale assessments, and nurses administered the remaining scales. A face-to-face survey was conducted in the clinics and wards for diabetes and COPD follow-up. General information was acquired by consulting the electronic medical records of the included patients; all patient data were double-checked before being entering into the database. The data were stored by the project leader, and 10% of the sample was randomly selected for secondary verification to ensure accurate data collection.
Statistical methods
We used IBM SPSS 25.0 (IBM Corp., Armonk, NY, USA) and R software (www.r-project.org) for statistical analysis and nomogram construction. Measurement data with a normal distribution are expressed as mean ± standard deviation, and the t-test was used for comparisons between groups. Non-normally distributed data are presented as median and interquartile range, and the Mann–Whitney U test was used for group comparisons. Count data are reported as frequency and percentage, with the chi-square test used for comparisons between groups. Variables with a P value of <0.05 in the univariate analysis were further included in the logistic regression. A forward stepwise method was used to identify the variables to be used for model construction. The nomogram was created with R software.
Results
We randomly selected 378 patients hospitalized between February 2022 and December 2023 for inclusion in this study. In total, 265 patients were allocated to the modeling group and 113 patients were allocated in the validation group for model verification. A total of 378 participants were enrolled, with an average age of 75.12 ± 8.92 years; there were 173 men, accounting for 45.77%.
General characteristics and univariate analysis of older patients with COPD and diabetes mellitus
In the modeling group, 265 of the 295 distributed questionnaires were finally recovered, leading to an effective recovery rate of 89.83%. Among these respondents, 127 (47.92%) were men and 138 (52.08%) were women. Respondents’ age range was 60 to 87 years and the average age was 74.34 ± 7.34 years. In addition, 93 (35.09%) patients had CF, with average age 77.32 ± 6.21 years; among the remaining 172 patients who did not have CF, the average age was 72.11 ± 5.98 years. In univariate analysis, age, regular exercise, polypharmacy, nutritional status, depression, COPD combined with diabetes, score on the Family APGAR index, and chronic pain were significant (all P < 0.05), as shown in Table 1.
General characteristics and univariate analysis results of older patients with chronic obstructive pulmonary disease and diabetes mellitus.
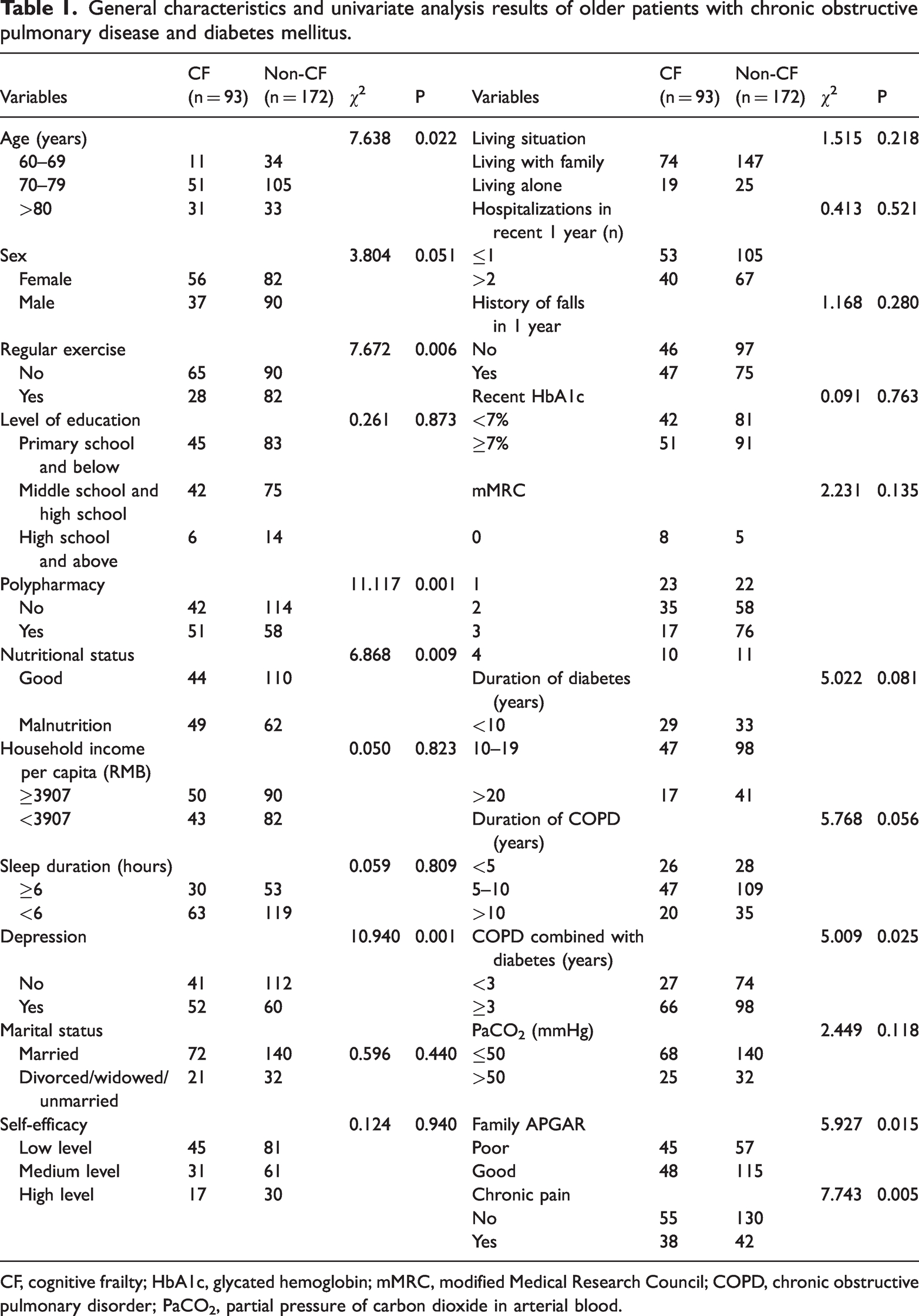
CF, cognitive frailty; HbA1c, glycated hemoglobin; mMRC, modified Medical Research Council; COPD, chronic obstructive pulmonary disorder; PaCO2, partial pressure of carbon dioxide in arterial blood.
Multivariate regression analysis of identified variables
Considering the occurrence of CF as the dependent variable, variables with P < 0.05 in univariate analysis were included in multivariate logistic regression analysis. Table 2 presents the specific values assigned to variables in this analysis. Multivariate logistic regression showed that age >80 years, regular exercise, polypharmacy, depression, duration of COPD combined with diabetes, score on the Family APGAR index, and chronic pain were significant factors (all P < 0.05), influencing CF in older patients with COPD accompanied by diabetes (Table 3).
Assignment of values to predictive categorical variables.

COPD, chronic obstructive pulmonary disorder.
Multivariate regression analysis of the identified variables.

The variance inflation factor (VIF) is generally interpreted based on values <10, and the tolerance value is 0–1.
An extremely small tolerance value indicates that the independent variable has collinearity with other variables.
COPD, chronic obstructive pulmonary disorder; OR, odds ratio; CI, confidence interval.
Construction and predictive effect of the CF prediction model in older patients with COPD and diabetes mellitus
According to multivariate regression analysis, the categorical data were assigned as follows: age >80 years = X1; regular exercise =X2; polypharmacy = X3; depression = X4; duration of COPD combined with diabetes = X5; score on the Family APGAR index = X6; and chronic pain = X7. After assignment, the risk prediction model was constructed as follows:

A nomogram for predicting the risk of CF was drawn (Figure 1). In this nomogram, each risk factor was scored according to the score scale in row 1, and the probability of CF was obtained by adding all the scores.

Nomogram model for the occurrence of cognitive frailty in older patients with comorbidities in the modeling cohort.
Validation of the risk prediction model for CF in older patients with COPD and diabetes mellitus
A total of 113 patients were included in the external validation group. The risk prediction model was evaluated by plotting a receiver operating characteristic (ROC) curve. The area under the ROC curve of the prediction model was 0.838 (95% confidence interval [CI], 0.757–0.920), with a sensitivity of 86.2%, specificity of 77.2%, and maximum Youden’s index of 0.634. As illustrated in Figure 2, the calibration curve demonstrated that the prediction curve was consistent with the actual observed curve. Moreover, the deviation from the ideal curve was small, indicating that the model could satisfactorily predict the probability of CF in older patients with COPD and diabetes (Figure 3).

Receiver operating characteristic curves for external validation of the nomogram model.
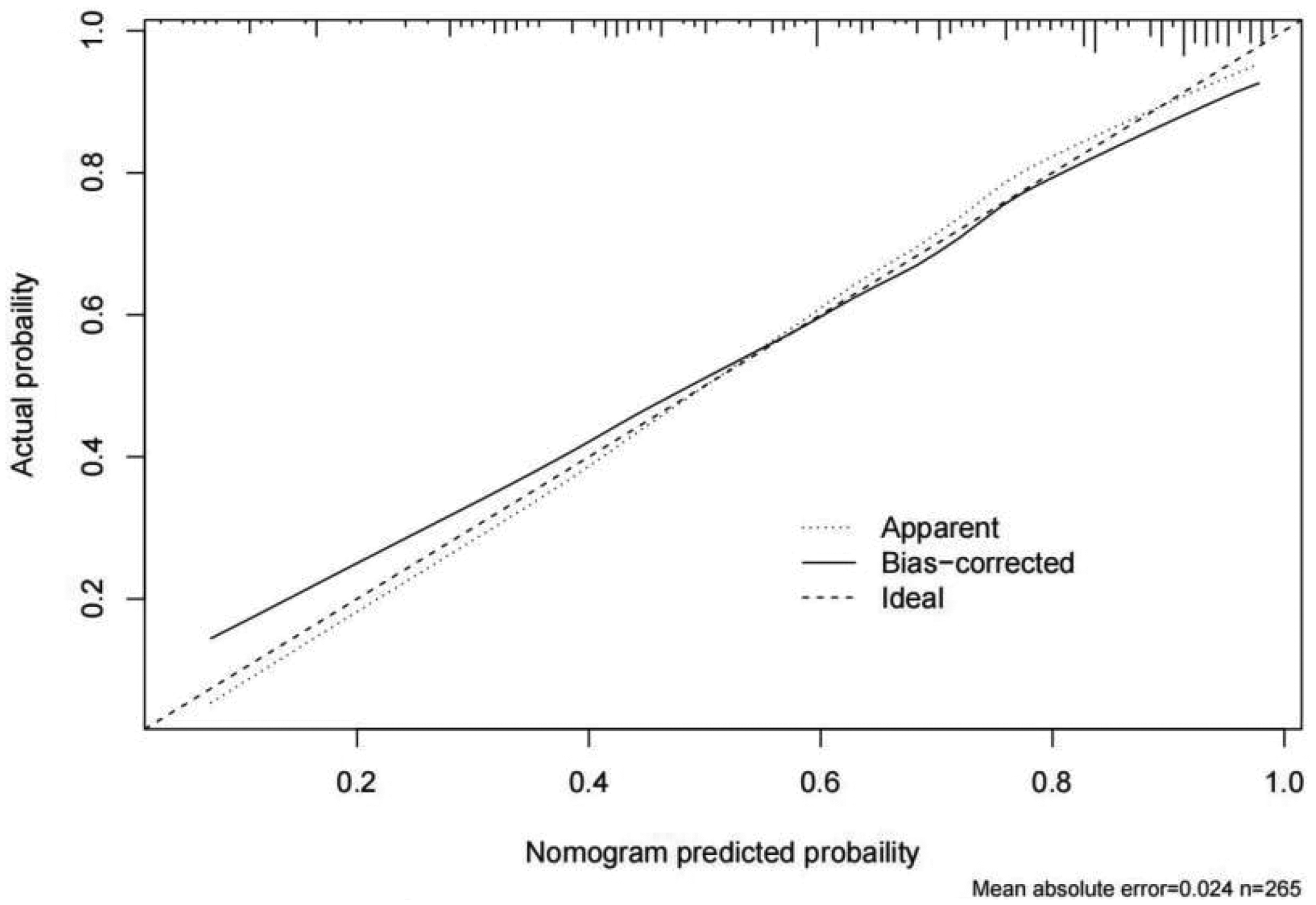
Calibration curves for internal validation of the nomogram.
Clinical benefit of the nomogram model for CF risk in older patients with COPD and diabetes mellitus
Decision curve analysis was used to determine the clinical value of the nomogram model. The analysis revealed that the nomogram curve was higher than the two extreme curves at the threshold probability interval of 0.2 to 1.0. “None” indicated that no patients had received intervention treatment for CF. Consequently, the net benefit was 0. Furthermore, the net benefit was a backslash with a negative slope, implying some clinical benefit of the model (Figure 4).

Decision curve analysis plot of the nomogram predicting risk of cognitive frailty.
Discussion
High incidence of CF in older patients with COPD and diabetes mellitus
Older patients with COPD and diabetes mellitus in the modeling group had a CF incidence of 35.09%, i.e., more than one-third experienced this condition. Consequently, the high incidence of CF in this patient population should receive clinical attention. Previous studies have shown that the probability of COPD complicated with CF was 19.3% to 31.9%7,24 and that of diabetes accompanied by CF was 8.7% to 30.9%. 8 This variation is possibly owing to the selection of patients, age, and sex as well as the variety of tools used to assess CF. Diabetes is a severe complication of COPD, with both diseases interacting with each other and inducing chronic inflammation and insufficient oxygenation. Ultimately, all these physiological conditions may adversely affect brain function and increase CF risk.25,26 The current research on CF in China is in the development stage, with a greater focus on studying individual patients with COPD and diabetes; studies on both diseases in older patients are rare. In light of this, in the present research, we aimed to investigate CF in older patients with COPD and diabetes and simultaneously analyze its influencing factors. In addition, we sought to construct a prediction model for CF in patients with COPD and diabetes mellitus and to validate its accuracy. The objective of this study was to provide a theoretical reference for the development of intervention plans targeting CF in older patients with both COPD and diabetes.
Independent risk factors for CF in older patents with COPD and diabetes
Age is a known risk factor for frailty and cognitive impairment. 27 As an individual grows older, a gradual decline is observed in the function of various body systems, including the respiratory and metabolic systems. These changes result in an increased risk of COPD and diabetes, and these affect frailty of the body and cognitive function. Our study showed that regular exercise served as a protective factor against CF, consistent with the findings of a systematic review by Tam. 28 Sarcopenia and a lack of physical activity have been shown to not only cause cognitive impairment and frailty 26 but also aggravate the progression of COPD and diabetes. 29 Conversely, regular exercise can enhance blood circulation and augment the supply of oxygen and nutrients to the brain. Moreover, routine physical exercise can aid in maintaining normal cognitive function, promoting the growth and connection between nerve cells, and contributing to the protection and improvement of brain neuronal function. Finally, patients may develop self-discipline and self-management skills by performing regular exercise, which improves the management of diabetes and COPD and consequently reduces the risk of CF.
Polypharmacy generally refers to the use of more than five drugs 30 and was reported in 41.13% of the patients in this study. Numerous studies have stressed the issue of polypharmacy.31,32 For example, the interaction or side effects of multiple drugs may increase the risk of adverse reactions and even affect cognitive function. In addition, patients with polypharmacy are more likely to have medication deviations, such as overdosing and missing doses, thereby negatively affecting frailty and cognitive function, particularly among those who have COPD complicated with diabetes. The problems associated with polypharmacy become more evident with disease progression and an increase in the number of medications. Therefore, clinical physicians and nurses should strengthen the management of medication in this patient population.
Prior studies have suggested that patients with COPD and diabetes are two to three times more likely to experience depression than the general population,33,34 with nearly half of those in the current study classified as having depression. Other researchers have demonstrated that the coexistence of depression with diabetes and coexisting depression with COPD have a prevalence of 10% to 15% 35 and 32% to 41.7%, 36 respectively. These differences may be related to variation in the depression assessment scales applied or the age of patients evaluated. Depression can also exacerbate psychological pressure on patients, and the resulting long-term psychological burden may cause impaired brain function. Moreover, patients with depression usually have concomitant sleep problems, such as insomnia or hypopnea, and such long-term poor sleep issues can affect normal brain function and aggravate CF. Furthermore, depression may lead to self-isolation among affected individuals, 37 resulting in a lack of social and emotional support and physical fatigue. Finally, patients with depression often feel physical and mental fatigue, which can also disrupt normal brain function and elevate the risk of CF.
Our study suggested that a longer duration of COPD with diabetes was linked to a greater risk of CF, potentially owing to the interaction of several other factors such as age, polypharmacy, and the physical condition and psychological factors of patients. For instance, the function of various organs, including the brain, declines gradually over time. Simultaneously, a longer duration of COPD combined with diabetes may result in a stronger cumulative effect of these chronic diseases. Based on these observations, long-term disease status may negatively affect brain function and escalate the risk of CF. Therefore, longer durations of comorbidity and polypharmacy combined with the side effects and interactions of drugs in this patient population may lead to adverse effects on cognitive and physical functioning.
Patients with better functioning family relationships are reported to have a lower incidence of CF, possibly owing to the increased emotional, social, and nutritional support that they receive. 38 Good family relationships can also provide greater opportunities for social interaction and communication, reduce social isolation, enhance the maintenance of cognitive function, and help to ensure that patients consume a balanced diet and receive adequate nutrition. An optimal nutritional status is essential for maintaining proper brain function and reducing the risk of CF. Moreover, better family relationships may help patients establish appropriate self-management mechanisms, such as ensuring regular medication intake, diet, and exercise. Patients who receive effective monitoring and care may also be able to promptly address disease recurrence or acute situations. Lastly, satisfactory family relationships can mitigate the physical and mental pressure on patients and help them to maintain stable cognitive function.
Chronic pain is another risk factor for CF identified in our study. Chronic pain can lead to the deterioration of sleep quality, with insufficient or poor sleep quality aggravating emotional problems and impairing brain function. 39 Chronic pain can also compromise the attention and concentration of patients, making it difficult to focus on cognitive tasks and worsening physical fatigue. 40 Ultimately, all these changes heighten the likelihood of CF.
Good applicability of the developed risk prediction model for CF in older patients with COPD and diabetes mellitus
An ROC curve was used to evaluate the risk prediction model constructed in this study. The results showed that the model had an acceptable predictive value with area under the ROC curve ranging from 0.7 to 0.8. An area under the curve of 0.8 to 0.9 indicates a good predictive value of the model. 41 In external validation, the area under the ROC curve was 0.838 (95% CI = 0.757–0.920), suggesting that our model had a good predictive value. Furthermore, assessment of CF using the current clinical methods usually requires 30 to 40 minutes per patient. However, patient evaluation using our model can be completed in only 6 minutes, thereby saving human clinical resources. Thus, our risk prediction model for CF may be a suitable assessment tool that may be promoted in clinical practice.
Limitations
The risk prediction model constructed in this study demonstrated good discrimination and calibration, highlighting its role as a valuable screening tool to identify high-risk patients with CF so as to provide early intervention. The present study, however, has certain limitations. (1) There may be some omissions among the included influential factors, such as the number and types of patients with comorbidities that may influence CF, which should be supplemented in subsequent trials. (2) Owing to its single-center design, this study carries a potential bias risk that necessitates future large-scale multicenter investigations.
Conclusions
In our study, CF had a high incidence rate among older patients with COPD and diabetes. The risk factors for CF occurrence in older patients with COPD and diabetes encompassed age >80 years, regular exercise, polypharmacy, depression, the duration of COPD combined with diabetes, score on the Family APGAR index, and chronic pain. It is crucial for medical personnel to focus on these influencing factors in early assessment and screening for CF, as well as to enhance education and guidance on CF to enhance the health status of older COPD patients with diabetes mellitus.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241274211 - Supplemental material for Construction and verification of a risk prediction model for cognitive frailty in older patients with chronic obstructive pulmonary disease and diabetes mellitus
Supplemental material, sj-pdf-1-imr-10.1177_03000605241274211 for Construction and verification of a risk prediction model for cognitive frailty in older patients with chronic obstructive pulmonary disease and diabetes mellitus by Meng-yao Liang, Rui Li, Li Feng and Wei-peng Qian in Journal of International Medical Research
Footnotes
Acknowledgement
The authors thank all of the medical staff in the endocrinology and respiratory departments.
Author contributions
Mengyao Liang, Rui Li, and Li Feng contributed equally to this article.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Project of Nantong Health Commission (nos. QN2023080, QN2023044, and MSZ2022055).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
