Abstract
Currently, Brazil lacks a national asthma management program and is burdened with nearly 200,000 hospitalizations due to the disease per year and approximately 5 deaths per day. The purpose of this article was to analyze the current issues surrounding severe asthma in Brazil, as the status of diagnosis and treatment is largely unknown, and to provide feasible recommendations to elicit imminent action. A panel of Brazilian medical experts in the field of severe asthma was provided with a series of relevant questions to address prior to a multi-day conference. Within this conference, each narrative was discussed and edited by the entire group. Through numerous rounds of discussion consensus was achieved. In order to overcome barriers to adequate asthma treatment, this panel recommends specific initiatives that can be implemented in the short-term to decrease the burden of severe asthma in Brazil. With increasing healthcare costs and limited resources globally, there is an opportunity to implement these recommendations in other countries in order to achieve adequate asthma care. Severe asthma is a heterogeneous and complex disease with various phenotypes that requires strict attention for diagnosis and management. Although this disease affects only a small proportion of the population with asthma, it poses a great burden to healthcare systems. Thus, barriers to diagnosis, treatment, and management should be overcome as quickly and efficiently as possible.
Introduction
Asthma affects approximately 300 million individuals worldwide and imposes a large burden for patients, healthcare systems, and society. 1 The most consistent world data on asthma prevalence has been reported in the pediatric population. 2,3 The prevalence of the disease is highest in English-speaking countries followed by Latin America 3 and has demonstrated an increasing trend in many countries. 1
The burden of asthma is a serious public health problem worldwide, particularly in low-middle income countries (LMIC). Appropriate control of the disease is the most important factor in reducing the burden that asthma represents on the healthcare system. One large Latin
American survey demonstrated that asthma is not controlled in more than 90% of patients and the use of controller medication is very low (6% of patients). 4 Several issues have been identified that contribute to a lack in asthma control, such as underdiagnosis, severity misdiagnosis, treatment nonadherence, fear of medication, lack of knowledge of treating physicians and support personnel, insufficient medication distribution, and difficult access to medications. 5 –7
A subgroup of asthma, “severe asthma,” is of particular concern. Although severe asthma represents only a small proportion (0.9% to 3.6%) of asthmatics, this subgroup has a life-threatening disease and accounts for a large amount of financial costs. 8 Severe asthma is of major concern for healthcare systems as it has shown higher associated costs than type 2 diabetes, stroke, or chronic obstructive pulmonary disease (COPD). 9 This proves to be especially true in developing countries, where limited resources can exacerbate these problems.
The purpose of this paper is to analyze the current issues surrounding severe asthma in Brazil, including barriers to the appropriate management of the disease, and provide feasible recommendations to elicit imminent action. The objective of this article was to create a practical document with standardized recommendations for the diagnosis and management of severe asthma in Brazil.
Methodology
A six-member panel of Brazilian medical experts in severe asthma (pulmonologists and allergists), selected by the Americas Health Foundation (AHF), was provided with a series of relevant questions on severe asthma to address prior to a multi-day conference. These questions covered 1) the burden and epidemiology in Brazil; 2) current guidelines for diagnosis, phenotyping, and treatment as well as diagnosis in Brazil; 3) the obstacles to effective diagnosis and treatment in Brazil; 4) treatment accessibility in Brazil; 5) recommendations for clinical management considering the different phenotypes; 6) recommendations for improving treatment access and effective treatment in Brazil. A written response to each question was initially drafted by a different member of the Panel. Within this conference, each narrative was discussed and edited by the entire group, through numerous drafts and rounds of discussion until a consensus was achieved. During the multi-day meeting of the Panel, each narrative was discussed and edited by the entire group, through numerous drafts and rounds of discussion until complete consensus was obtained.
To address the above issues, the Americas Health Foundation (AHF) identified clinicians and scientists with an academic or hospital affiliation who are experts in the field and who have published in the asthma field, specifically severe asthma, since 2014. As a result of this effort, AHF assembled a six-member panel of clinical and scientific experts from Brazil in the disciplines of pulmonology and allergy.
To better focus on the discussion, AHF staff independently developed specific questions, addressing the central issues on the subject, for the Panel to address. A written response to each question was initially drafted by a different member of the Panel. During the multi-day meeting of the Panel, each narrative was discussed and edited by the entire group, through numerous drafts and rounds of discussion until complete consensus was obtained.
Limitations of this article include 1) this panel is only representative of pulmonologists and allergists in Brazil and does not include all disciplines surrounding asthma and 2) this article primarily addresses pharmacological interventions rather than psychological or lifestyle interventions.
Search strategy and selection criteria
Manuscripts referenced in this consensus paper were identified through searches of Pub Med and Embase with the search terms “asthma”, “severe asthma”, “Brazil”, “innovative treatments in asthma” and “severe asthma in Brazil” from November 2014 to November 2019. Articles were also identified through the bibliographies of the papers identified in the search as well as from sources of the authors’ own files. Particular attention was paid to papers that reviewed or summarized the topic in question, or that were related to activities in Brazil. The final reference list was generated on the basis of the relevance to the broad scope of this consensus document.
Asthma in Brazil: Prevalence, burden, and cost of the disease
Brazil has a multiethnic population of 210 million people, with the largest public health system in the world (Unified Health System—SUS). Public healthcare services are universal and offered to all citizens for free. However, being a country of limited resources, healthcare is still underprovided and deficient in many aspects. A supplementary private system exists and requires individuals to purchase insurance and receive their care from specific healthcare providers (around 22% of the population). 10 In Brazil, asthma is underdiagnosed and probably undertreated, 11 which may be in part to unequal access to healthcare.
It is estimated that there are 20 million patients with asthma in Brazil, with a high morbidity and significant mortality. 12 The estimation of overall prevalence of asthma in Brazil is around 9%. 13 In adults, asthma has an estimated prevalence of physician-diagnosed asthma of 4.4%. 14 Most cities in Brazil have a prevalence of pediatric asthma >10%, with many cities having a prevalence >20%. 15 The prevalence of uncontrolled asthma on maximum controller treatment is estimated to be 17.4% of all adults with asthma, and only 20% of these patients have severe refractory asthma. 8 Therefore, the prevalence of severe asthma may be lower than previously described. The latest estimated prevalence is 0.9–3.6%, depending on the severe asthma definition used in the assessment. 8
Approximately five patients per day die from asthma in Brazil. 16 Between 2008 and 2013, the in-patient mortality rate increased from 0.4% to 0.5%, a 25% increase. The cost of asthma-related hospitalizations was around US$170 million in the SUS. 16 However, this calculation stipulated by the government is based on reimbursement figures so it does not reflect the “real” disease cost to healthcare providers.
Asthma also results in high costs at an individual level, direct (associated with the management of the disease) and indirect (i.e., impaired quality of life (QoL), loss of school/work, mental health disabilities). In school children with asthma, higher direct costs were associated with high hospitalization rates and frequent use of oral steroids. Indirect costs were related to high rates of school absenteeism and sedentary behavior. 17 Patients with asthma have lower health-related QoL, higher work absenteeism, and increased work and physical activity impairment. 18
Severe asthma in particular accounts for a large amount of financial costs to the patient and health-care system. 1 In Brazil, country-wide studies evaluating costs associated with severe asthma are limited. The existing data on severe asthma costs in Brazil estimate that a family’s total costs are around US$1000 a year, representing 2.5 months worth of family income. 19,20 The implementation of a structured asthma care program reduced the proportion of total family income spent in asthma treatment from 29% to 2%. 19,20 Another study found that a patient with severe asthmatic has an annual cost of US$1400, that is two to four times more when compared with patients with milder asthma. 21
Asthma control strategies
The assessment of asthma control is essential to guide asthma management. 22,23 Questionnaires such as the Global Initiative for Asthma (GINA), the Asthma Control Questionnaire (ACQ), 24 or the Asthma Control Test (ACT) 25 can be used to assess asthma control. 22 In places where spirometry may not be available, simple asthma questionnaires are especially valuable to be systematically applied to evaluate asthma control before and after treatment. If patients are not well controlled, they should be referred to specialized care. As an example of the magnitude of the uncontrolled disease in Brazil, three studies reported an estimated 40–90% of patients with uncontrolled asthma. 17,18,26
Asthma management consists of identifying and correctly diagnosing patients with the disease and subsequently initiating and optimizing treatment in order to achieve and maintain control of the symptoms. 22,23 Among patients with asthma, the subgroup with severe asthma has a high risk of complications, exacerbations, and poor QoL. Uncontrolled severe asthma contributes to high morbidity, direct and indirect costs to the public sector and family, reduced QoL, and premature mortality. 20 In Brazil, one in every five pediatric patients with uncontrolled or difficult to treat asthma would be categorized as having severe asthma. 27
Brazil does not have an updated national asthma management protocol. Several isolated asthma initiatives have emerged since the 1990s from specialized centers in several cities. In Brazil, the implementation of government-funded asthma programs and the distribution of easy-access and free basic medication (inhaled corticostertoid and short-acting beta-2 agonists) have contributed to a decrease in hospitalizations due to asthma in Brazilian public hospitals in the last two decades. Within the last 10 years, Brazil has put into effect 55 asthma programs, with only 17 specifically for severe asthma. 28,29
Primary healthcare facilities are the first point of care for patients with undertreated asthma. 11 Several initiatives have attempted to strengthen primary care settings and provide free care for a pre-determined number of individuals within a specific geographic region. 30 Despite this, a study comparing asthma diagnosis between primary care physicians and pulmonologists showed 50% of the patients were misdiagnosed in primary settings (0.53; 95% CI 0.45–0.60), 11 emphasizing the persistent need for access to specialists and a national comprehensive training and education program for primary care teams.
In 2010, a national policy for SUS approved free access to three essential asthma medications (beclomethasone, salbutamol and prednisone) and made them easily accessible throughout the country by accredited pharmacies (Programa Farmacia Popular). 31 During the first years of the program, a 36% decrease in asthma-related hospital admissions was observed. Access to free medications has been thought of as one of the reasons for this decrease. 16 Another medication distribution program, co-funded at state levels, included a combination of inhaled long-acting β-agonists and corticosteroids. Access to these medications required a prescription from the specialist, a completed medical form, and spirometry results, and could only be distributed by specific state pharmacies. These prerequisites created substantial access barriers. Despite efforts to increase medication distribution, control of the disease is still very limited (∼10%) and is associated with an excessive use of short-acting β2 agonists, low use of inhaled corticosteroids, and poor treatment adherence across the severity spectrum. 13,18,32
Two city-specific programs for asthma control included specialized care, patient and primary care physician education, and free medications (including inhaled corticosteroids). These programs resulted in hospitalization reductions of 33% and 74% respectively, as well as cost reductions to the patient and the healthcare system. 33,34 Direct asthma-related costs for a participant’s family were reduced from 24% to 2% of their annual income. 35 Comprehensive approaches, such as the ones described above, are critical for asthma control, given the reduction in the burden of disease and associated direct and indirect costs.
Definition of severe asthma
The definition of severe asthma has evolved in the last 20 years according to the understanding of the disease, its presentation forms, and pathophysiological mechanisms. Difficult to treat asthma is uncontrolled asthma despite maximum treatment (step IV-V of GINA recommendations) and in most cases the lack of control is related to modifiable factors. 22,23 Severe asthma is a subgroup of difficult to treat asthma that requires maximum treatment, after optimized management and control of modifiable factors, in order to prevent the disease from becoming uncontrolled or which remains uncontrolled despite this therapy. 22,23,36
In Brazil, the GINA strategy is the most commonly recommendation used among respiratory specialists for asthma management. Despite the existing international 22 and national guidelines for asthma, 23 data indicates inadequate application of these recommendations in the SUS, as only 24.2% of prescribed treatment were in accordance with the GINA strategy. 37 These results suggest the need for medical education programs as well as clear referral and counter-referral communication with physician specialists. 38 To date, there have been no specific protocols created for severe asthma management in Brazil. In Figure 1, an overview of the classification of asthma severity, tests recommended, and available treatments in Brazil are presented.
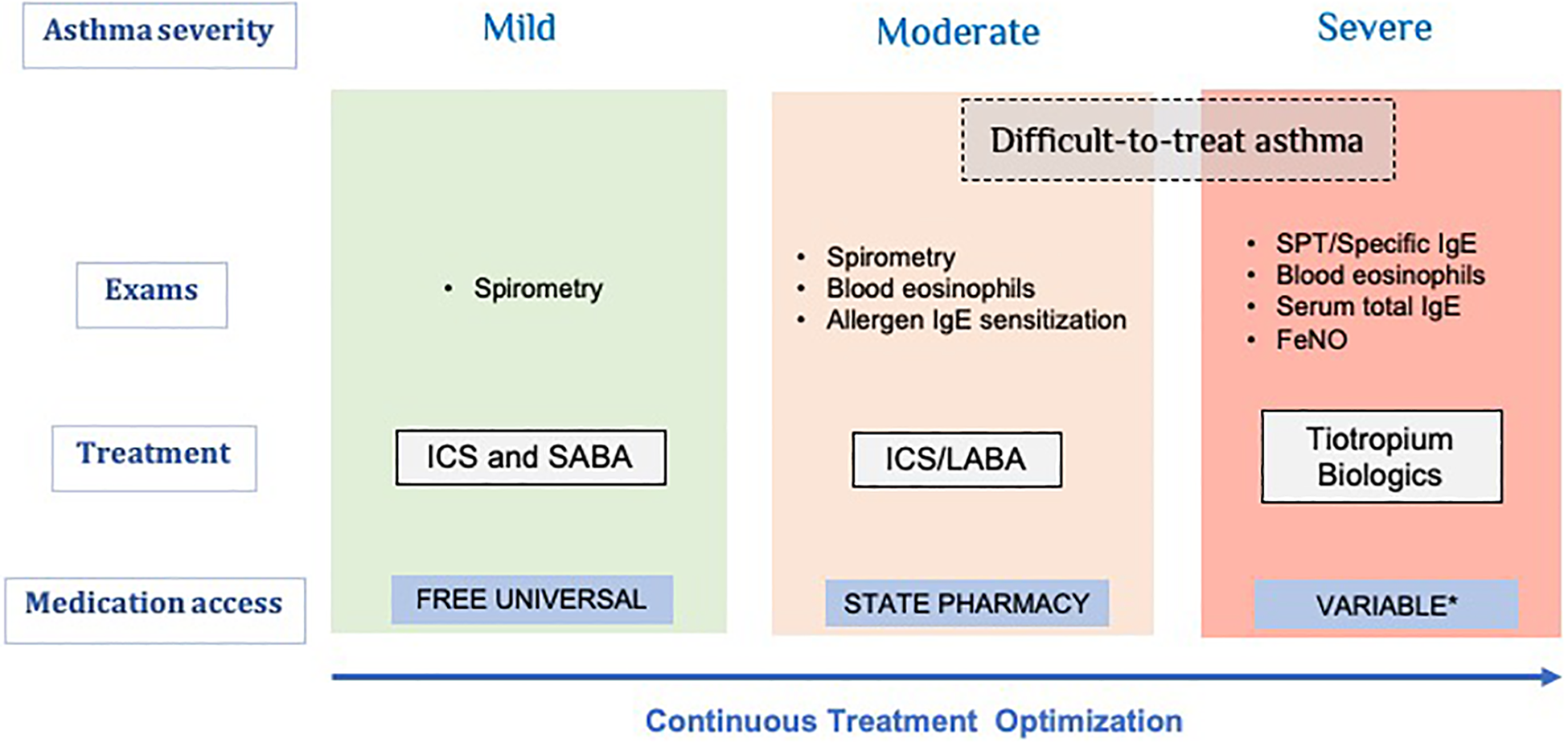
Overview of asthma classification and its management.
Severe asthma diagnosis and management
The diagnosis of asthma in general is usually confirmed by the presence of airflow limitation (FEV1/FVC ratio < 80%) with a significant response to short-acting bronchodilator. However, patients with severe asthma may have minimal or absent bronchodilator responsiveness, especially in children. In such cases, additional tests including bronchial provocation tests may be required to confirm diagnosis. Alternatively, evidence of a past history of bronchodilator response or airway hyperresponsiveness may support the diagnosis in these situations. 39 Differential diagnosis must be completed to exclude possible clinical conditions that may mimic asthma, such as vocal cord dysfunction, COPD, gastroesophageoal reflux, cardiac insufficiency, anxiety, obesity, among others. 40
The next step after confirming diagnosis is to understand why asthma may not be responding to maximum treatment. Asthma control needs to be measured objectively with a validated questionnaire. However, independent of asthma control questionnaire results, a patient with one or more severe exacerbations in the previous year should be classified as uncontrolled. Of note, it is important to managing lifestyle and psychological status is of utmost importance and should be addressed in order to improve outcomes and QoL. 41 Additionally, it is necessary to review adherence to treatment, adequate inhaled medication technique, exposure to environmental triggers, smoking, use of other medications that decrease the response to treatment, and associated comorbidities that may worsen asthma control. 36,39,42
Treatment adherence has been considered one of the major challenges in asthma worldwide and may be as low as 50% or less. 6 Poor adherence is associated with adverse outcomes, including exacerbations and increased mortality. 36 In addition, to ensure treatment response, the correct use of the inhaled medications is crucial. A recent systematic review of literature regarding errors in inhaler technique showed that the prevalence of an optimal inhalation technique was only at 31%. Hence, adherence to treatment and inhaler technique must be thoroughly assessed at every visit. 43
Several comorbidities associated with severe asthma include obstructive sleep apnea, COPD, bronchiectasis, chronic rhinosinusitis with or without nasal polyposis, gastroesophageal reflux, obesity, vocal cord dysfunction, anxiety, depression, among others, which may impair asthma control and response to treatment. 39,40,41,43 –47 These comorbidities should be systematically investigated, treated, and mitigated as they increase the risk of exacerbations and decrease QoL. 48
Given the multidimensional nature of the disease, severe asthma management should be optimized with a multidisciplinary approach that requires effective communication between all parties involved to improve outcomes. A systematic assessment should be performed across several clinical domains including airways, risk factors, and comorbidities. 39 However, unfortunately, in the Brazilian public health system, in most tertiary centers a multidisciplinary team (nurses, pharmacists, physiotherapists, psychologists and dieticians) and is not available and primary care physicians are not appropriately trained for early detection and referral of patients with severe asthma. This is an important issue to be presented and discussed, but it is not scope of this article.
Phenotyping severe asthma in clinical practice
Severe asthma is frequently described as a heterogeneous and complex disease, 42,49 –51 referring to the variability of “observable clinical characteristics of asthma” (phenotypes). 50 Adding biological information to phenotypes and information on the causative pathways underlying the phenotypes (endotypes) improves the understanding of severe asthma. 50,51 Biomarkers can help identify different phenotypes and endotypes to select treatment 52 and predict and assess treatment response. However, before considering phenotypes, endotypes, and biomarkers for severe asthma management, the diagnosis must be confirmed and all factors that may decrease the response to treatment should be eliminated or minimized. 36
Phenotyping is the natural step after establishing a severe asthma diagnosis. This is important to better understand disease course and prognosis as well as to select the right treatment for the right patient. 53,54 However, asthma phenotypes based solely on clinical characteristics are not sufficiently informative to guide a personalized treatment approach (i.e., late and early onset, obesity-associated severe asthma, allergic and non-allergic severe asthma, severe occupational asthma, etc.). Measuring biomarkers such as serum total immunoglobulin E (IgE), allergen sensitization, blood eosinophil counts, Fraction of exhale Nitric Oxide (FeNO), and induced sputum cellularity are currently used to improve severe asthma management and support treatment choice (Figure 1).
Precision treatment based on severe asthma phenotypes
The introduction of therapy with inhaled corticosteroids (ICS) has changed the natural course of the disease and has notably decreased mortality. 55 The vast majority of asthma can be controlled with ICS or with the association of low to moderate doses of ICS as monotherapy or in association with a long-acting beta2 agonist (LABA). However, until recently, for patients with severe asthma on high doses of ICS+LABA the only stepup treatment available was oral corticosteroid. Now, a new era of treatment for severe asthma has emerged: biologic agents.
Four biologic therapies have been approved and are commercially available for use in patients with severe asthma in Brazil: omalizumab, mepolizumab, benralizumab and dupilumab. The basic tests to guide the biological treatment choice are 1) total serum IgE; 2) skin prick test or specific serum IgE; and 3) peripheral blood eosinophils count. If available, FeNO and sputum eosinophil counts provide additional airway inflammation information (Table 1). Of note, all patients being considered for specialized treatments such as biologics should be reviewed by a multidisciplinary team for consensus approval of treatment.
Biologic therapies approved and commercially available for use in patients with severe asthma in Brazil.

* For patients on systemic corticosteroids.
Access to severe asthma treatments in Brazil
In order to regulate care, Brazilian government guidelines (Protocolos Clínicos e Diretrizes Terapêuticas - PCDT) have been developed. The last asthma PCDT guideline update was published in November 2013. 62 Given that these guidelines have not been recently updated, management of the disease is outdated and does not include any long-acting anti-cholinergic or immunobiological agents.
Healthcare is a constitutional right for Brazilian citizens, under which premise every Brazilian citizen should have access to ANVISA (Agência Nacional de Vigilância Sanitária) approved medications. However, in the public and private healthcare systems, access to these medications is not necessarily guaranteed and judicialization has become a common alternative to gain this access. This process delays access and increases total treatment costs associated due to increased drug price at individual level negotiation, medications used by patients that may not benefit from the requested treatment, and the additional costs of the justice system.
Wide scientific evidence exists to support the efficacy and safety of immunobiological agents (omalizumab, mepolizumab, benralizumab and dupilumab). 56 –61,63,64 One study conducted in a Brazilian asthma treatment center applied a strict protocol to treat patients with omalizumab and showed that 75% of the patients had partial or total response to treatment. Notably, out of 2,500 difficult-to-treat patients in a 3-year period, only 12 patients met the strict criteria to receive the drug. 65 Another study in severe asthmatic children treated with omalizumab showed a significant increase in control of the disease (90%) and reduced rates of hospitalization (70%). 66 Response to the medication must be strictly assessed 4–6 months after its initiation to determine whether treatment should be continued, based on the patient’s response to therapy. In the near future, there will hopefully be clinical data specific to Brazil that support the more recently approved biologics for the treatment of severe asthma.
Omalizumab (anti-IgE drug) has been commercially available in Brazil for more than a decade, but has just recently been approved by CONITEC (the governmental agency in charge of new health technology assessments). 67 This approval provided the severe asthma population access to the first biologic treatment in Brazil. The reluctance to approve new biologics may be due to a larger assumed economic impact and inadequate projections of the target population. This panel calculated that severe asthma patients who may benefit from these treatments may amount to 0.5% of the total adult asthmatic population, similar to other countries. 68 It is important to note that omalizumab will not cover the needs for all patients with distinct phenotypes of severe asthma. Consequently, the other biologics for severe asthma must be approved to improve the management of the disease in Brazil.
Areas that should be addressed when considering the importance of including new biologic agents are cost-analyzing the biologics, assessing access barriers to medication, defining a target population, determining therapeutic response criteria, and, when relevant, determining medication withdrawal criteria. Three other difficulties for adequate diagnosis and treatment of severe asthma in Brazil are:
The
The
Recommendations and conclusions
Although severe asthma corresponds to a small portion of all asthma cases, 8 diagnosing and treating these patients appropriately could alleviate costs for patients and families as well as create measurable savings for healthcare systems. 19,21 Evidence-based recommendations for severe asthma diagnosis and treatment are found in international documents and include: accurate diagnosis of the disease, adequate treatment adherence, proper inhaler technique use, environmental control, patient education about misconceptions of medications, and the recognition and treatment comorbidities. 22,36
This panel recommends specific initiatives tailored to Brazil that can be implemented in the short-term to decrease the burden of severe asthma:
Finally, this manuscript addresses issues specifically related to the lack of access to innovative treatments for severe asthma in the Brazilian public health system. However, access to these asthma therapies is a global issue; therefore, the obstacles discussed are not exclusive to Brazil. With increasing healthcare costs and limited resources, there is also an opportunity to implement the proposed recommendations in other countries in order to quickly address barriers to adequate asthma care.
Footnotes
Acknowledgments
To Elizabeth McElwee, MPH, Dr. Mariana Rico Restrepo, and Mary Herdoiza from the Americas Health Foundation (AHF) for the valuable help in the design and development of the manuscript. This manuscript was supported by a grant from the AHF, a 501(c)3 nonprofit organization dedicated to improving healthcare throughout the Latin American Region. The AHF was responsible for the development, organization and implementation of the consensus conference, along with independently selecting the experts to serve on the panel. The AHF had no role deciding the content of the manuscript and the recommendations are those solely of the panel members.
Author contributions
All authors participated and made significant contributions to the data search, drafting, and discussion of the topic and all subtopics provided in this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The organization and implementation of the consensus conference was carried out by the Americas Health Foundation (AHF), a 501(c)3 nonprofit organization dedicated to improving healthcare throughout the Latin American Region and was supported by an unrestricted grant.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. The organization and implementation of the consensus conference and manuscript was carried out by the Americas Health Foundation (AHF), a 501(c)3 nonprofit organization dedicated to improving healthcare throughout the Latin American Region and was supported by unrestricted grants from GSK. The AHF independently selected the experts to serve on the panel. The AHF had no role deciding the content of the manuscript and the recommendations are those solely of the panel members.
