Abstract
Uptake of nutritional supplementation during pulmonary rehabilitation (PR) for people with chronic obstructive pulmonary disease (COPD) has been limited by an absence of rigorous evidence-based studies supporting use. The objective was to report and summarise the current evidence supporting the use of nutritional supplementation to improve outcomes during PR in stable COPD patients. A systematic search was conducted up to 7 August 2019 (registration number CRD42018089142). The preferred reporting items for systematic reviews and meta-analyses guidelines were used. Six databases were included: Medical Literature Analysis and Retrieval System Online or MEDLARS Online, Allied and Complementary Medicine Database, the Cochrane Database of Systematic Reviews, Excerpta Medica dataBASE, Cumulative Index of Nursing and Allied Health Literature and Web of Science. This systematic search generated 580 initial matches, of which 22 studies (917 COPD participants) met the pre-specified criteria and were included. Sixteen of 19 studies that used nutritional supplements in addition to PR did not show additional benefit compared to PR alone when measuring exercise capacity. Nutritional supplements significantly increased body weight in 7 of 11 studies. Body mass index increased significantly in two of six studies. Handgrip strength did not improve, while quadriceps muscle strength significantly improved in 3 of 11 studies. Four of eight studies showed a significant improvement in inspiratory muscle function. Only 2 of 14 studies demonstrated a significant improvement in quality of life with supplementation in addition to PR. There remains insufficient evidence on the effect of nutritional supplementation on improving outcomes during PR in patients with COPD due to heterogeneity in supplements, outcome measures and PR programmes. Therefore, controversy remains and further research is needed.
Keywords
Introduction
Patients with chronic obstructive pulmonary disease (COPD) experience daily symptoms, reduced exercise capacity and susceptibility to exacerbations, resulting in reduced health-related quality of life. 1 –3 The international Global Initiative for Chronic Obstructive Lung Disease (GOLD) strategy document summarises current approaches to COPD management. 1 Cost-effective treatment approaches for COPD, described in the ‘value pyramid’ 4 include smoking cessation, influenza vaccination and pulmonary rehabilitation (PR). Multiple high-quality randomised controlled trials (RCTs) and meta-analyses have demonstrated that PR is an effective management strategy in COPD, since it improves exercise performance, reduces dyspnoea, reduces the risk of exacerbation and improves health-related quality of life. 5 –10
Exercise limitation is one of the most common problems for COPD patients and this may be compounded by reduced muscle mass and malnutrition. Some COPD patients may lose body weight and skeletal muscle mass, which leads to muscle weakness and dysfunction, impacting functional ability and quality of life. 11 Muscle disuse, caused by a prolonged sedentary lifestyle and voluntary immobilisation, leads to further muscle deconditioning and thus reduced muscle strength and endurance. 12 It has also been postulated that COPD is associated with a myopathy, which may be driven by systemic inflammation. 12 Being underweight is associated with an increased risk of mortality in COPD and weight loss predicts mortality and morbidity in chronic lung disease patients. 8,13 Therefore, patients with COPD are at risk of significant morbidity and mortality as a result of changes in body composition and nutritional and metabolic status.
It has been suggested that healthy older adults require additional nutrients compared with younger adults to preserve bone and lean mass. For instance, it is recommended that young adults require 0.7 g of protein/kg body weight per day while the recommendation for older adults is 1.2–1.5 g protein/kg body weight/day, especially for people with conditions that require higher levels of protein, such as COPD. 14 Nutritional supplements have been used to overcome malnutrition in patients with COPD. It has been suggested that nutritional support integrated with exercise training may improve exercise activity, decrease mortality and improve muscle strength in undernourished COPD patients. 15,16 A meta-analysis of nutritional supplementation for stable COPD by Ferreira et al. included 17 randomised clinical trials and concluded that nutritional supplements increased muscle mass and body weight and improved respiratory function and exercise tolerance in COPD patients who were poorly nourished. 17,18 Additionally, Collins et al. demonstrated in a meta-analysis of nutritional support and functional capacity in COPD that nutritional supplements improved weight and handgrip strength in COPD patients. 19 Both reviews only included randomised clinical trials and it was not necessary for participants to be engaged in PR. We hypothesised that an integrated approach of exercise training and nutritional support might be the best way to seek functional improvements. However, the uptake of nutritional supplementation during PR, where the potential benefit may be greatest, has been limited by the absence of rigorous evidence-based studies supporting use. The objective of this systematic review was to report and summarise the current evidence for using nutritional supplementation during PR in stable COPD patients to enhance PR outcomes.
Methods
Search strategy
The preferred reporting items for systematic reviews and meta-analyses guidelines were used for this systematic review, with Prospero registration number CRD42018089142. 20 The search was conducted up to 7 August 2019 using Medical Literature Analysis and Retrieval System Online or MEDLARS Online, Excerpta Medica dataBASE, Allied and Complementary Medicine Database, the Cochrane Database of Systematic Reviews, Cumulative Index of Nursing and Allied Health Literature and Web of Science database (Supplemental Material Tables A1 to A5). The search strategy and terms used in this systematic review are described in Supplemental Material. The bibliography of eligible articles and existing systematic reviews in the field were also screened.
Inclusion criteria
The PICO (P: population, patient, problem; I: intervention; C: control, comparison or comparator; O: outcome) criteria for included studies appear in Table 1. Studies were included in the systematic review if they met all of the following criteria: studies of patients with a confirmed diagnosis of COPD; no evidence of recent exacerbation, as described in the individual studies; patients enrolled on a PR or other exercise training programme and patients receiving nutritional supplementation (caloric, non-caloric, powder, liquid, capsule or tablets) during PR.
PICO criteria used for the inclusion of studies.

COPD: chronic obstructive pulmonary disease; PICO: P—population, patient, problem; I—intervention; C—control, comparison or comparator; O—outcome.
Exclusion criteria
We excluded book chapters, systematic reviews (but screened the reference lists), non-English manuscripts, conference abstracts with no full-text and non-full text articles.
The main outcomes of interest were to investigate the impact of nutritional supplementation during PR programmes on exercise function, body composition, peripheral muscle strength, respiratory muscle function and quality of life.
Data collection
Three authors (AMA, JRH, and SM) screened the titles and abstracts to exclude irrelevant studies. Full texts of the relevant studies were read by the first author (AMA) to evaluate if they fulfilled the inclusion criteria. The reference lists of included studies and excluded systematic reviews were also screened; two additional studies were found, and the senior authors (JRH and SM) discussed eligibility. Disagreements between authors were resolved by discussion.
Quality assessment
The first and seventh authors (AMA and JRH) performed risk of bias assessment using the Cochrane risk of bias tool to assess randomised studies, which comprises seven questions, and the modified Newcastle–Ottawa scale to assess cohort studies, which is also made up of seven questions. 21,22 For the randomised trials, we scored each of the seven domains as 0 (low risk of bias) or 1 (high risk of bias or bias unclear). There was, therefore, a total score between 0 and 7 in which a higher score equates to a higher risk of bias. For cohort studies, each of the seven domains was scored from 0 (high risk of bias) to 3 (low risk of bias) and we took a mean of the domains to result in a score between 0 and 3, where a higher score represents a lower risk of bias.
Synthesis of results
The main purpose of this systematic review was to report and summarise the current evidence of using nutritional supplementation during PR in stable COPD. A meta-analysis was not attempted due to methodological heterogeneity between studies. Our discussion focuses on the studies at lower risk of bias.
Results
Initially, 580 studies were considered potentially eligible. However, after removing duplicates, 449 titles and abstracts were included. Screening the titles and abstracts resulted in 30 of 449 studies being considered for full-text reading. After reading the full text of 30 studies, 10 further studies were excluded (Supplemental Material Table A6). Screening the reference list of eligible studies revealed two further relevant studies. Thus, 22 studies in total met the inclusion criteria for the systematic review (see Figure 1).
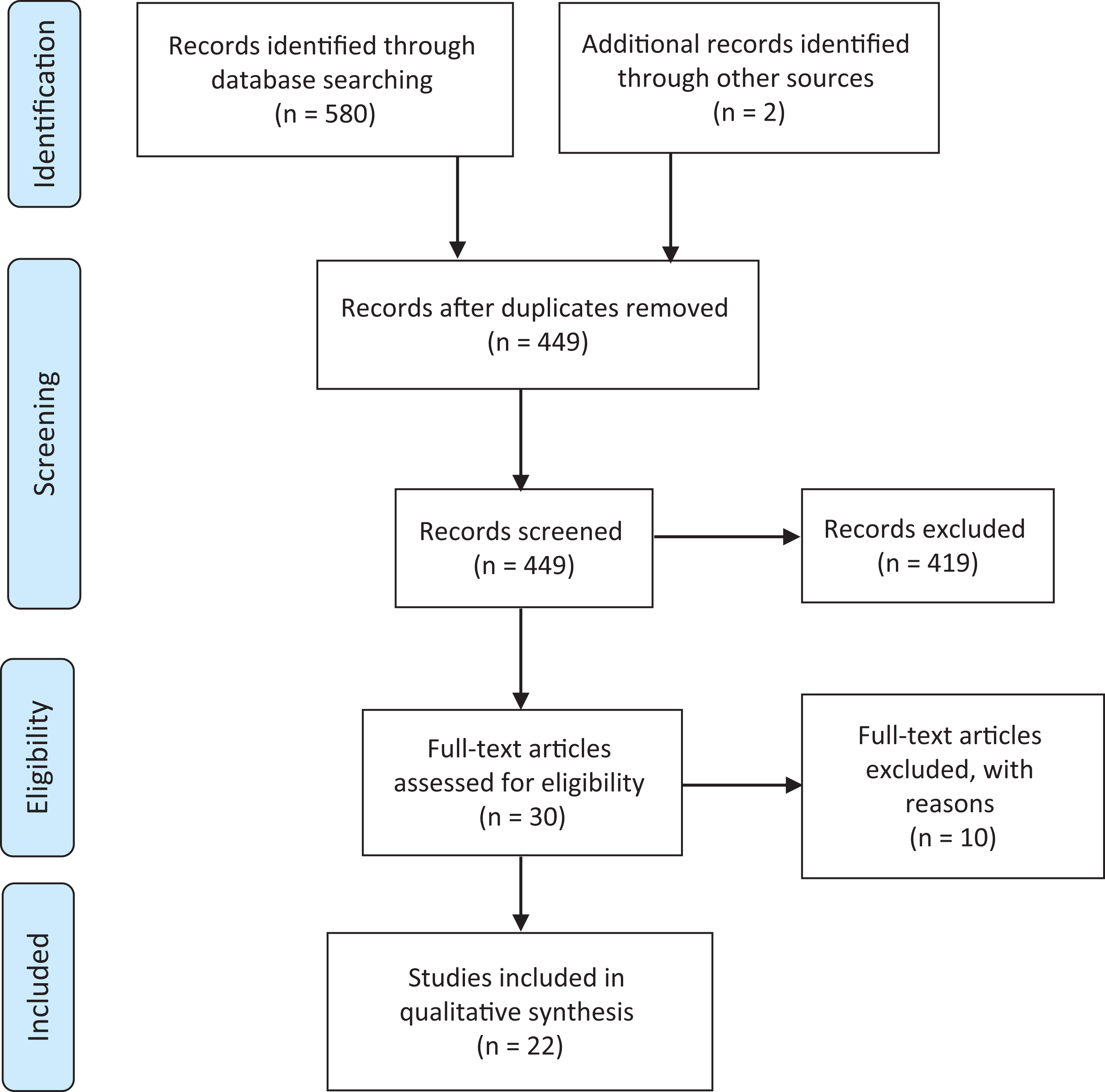
Preferred reporting items for systematic reviews and meta-analyses flow diagram.
The 22 studies comprised 5 cohort studies and 17 RCTs. The sample size and study duration varied between 8 and 80 participants and 6 weeks to 4 months, respectively. A full description of the included RCTs and cohort studies appears in Tables 2 and 3, respectively. The risk of bias assessment for RCT and cohort studies appears in Tables A7 and A8 in Supplemental Material, respectively.
Detailed description of the included RCT studies.

I: intervention group; P: placebo or control group; 12MWT: 12-minute walk Test; 6MWD: 6-minute walk distance; BM: body mass; BMC: bone mineral content; BMI: body mass index; CRQ, Chronic Respiratory Disease Questionnaire; CWT: constant work rate test; ESWT: endurance shuttle walk test; FEV1: forced expiratory volume in 1 second; FFM: fat-free mass; FM: fat mass; FMI: fat mass index; IBW: ideal body weight; ISWT: incremental shuttle walking test; MEP: maximum expiratory pressure; FFMI: fat-free mass index; MIP: maximum inspiratory pressure; MMC: mid-arm muscle circumference; PEF: peak expiratory flow; QAP: quadriceps average power; QuadCSA: quadriceps cross-sectional area; SGRQ: St. George’s Respiratory Questionnaire; SMM: skeletal muscle mass; UI: International unit; LBM: lean body mass; LBMI: lean body mass index; SMI: skeletal muscle mass index; BCM: body cell mass; BMC: bone mineral content; ASM: appendicular skeletal muscle mass; EQ-5D-3 L: EuroQoL five-dimensions Questionnaire; W, Watt.
Detailed description of the included cohort studies.

I: intervention group; P: placebo or control group; 12MWT: 12-minute walk test; 6MWD: 6-minute walk distance; BMI: body mass index; CRQ: Chronic Respiratory Disease Questionnaire; FEV1: forced expiratory volume in 1 second, FFM: fat-free mass; FFMI: fat-free mass index; FM, fat mass; MIP: maximum inspiratory pressure; PR: pulmonary rehabilitation; SGRQ: St. George’s Respiratory Questionnaire.
Exercise capacity
Data on exercise function, performance, capacity or endurance were reported in 19 studies using the endurance shuttle walking test (ESWT), incremental shuttle walking test, 6-minute walk test (6MWT), 12-minute walk test, treadmill and incremental or constant work-load cycle ergometry. Seventeen studies found that using nutritional supplements such as high carbohydrates, vitamin D, creatine, or
There was only one study with positive findings at the lowest risk of bias (1/7), in which Sugawara et al. reported increases in 6-minute walk distance by 19.7 ± 24.7 m with this addition of supplement (less than the minimum clinically important difference). In this RCT, the intervention group received ready-to-drink oral nutritional supplement (ONS) twice a day composed of 200 kilocalories, 60% carbohydrates, 15% protein, 25% fat, 248 μg of omega-3 PUFAs 0.6 with vitamins A, C and E and a 12-week exercise programme while the control group underwent a 12-week exercise programme only.
28
There were four RCTs with a similar low risk of bias, which demonstrated no benefit of supplementation. van de Bool et al.
23
reported that using a high carbohydrate supplementation once a day (125 mL of 9.4 g protein, 28.1 g carbohydrate and 4.1 g fat, leucine, n-3 PUFA and vitamin D) over a period of 4 months within an outpatient PR did not show any significant improvement in exercise performance measured by cycle endurance time or 6MWT compared to the control PR group, who received flavoured non-caloric aqueous solution as a placebo. Similarly, the study by Paulin et al. found that using vitamin B12 for 8 weeks during outpatient, PR did not show any significant improvement in exercise performance or endurance compared to PR alone.
24
Borghi-Silva et al. reported that using
Body composition
Seventeen trials measured body composition including body weight, fat-free mass (FFM), fat-free mass index (FFMI) and body mass index (BMI).
Body weight was one of the most frequent outcomes measured before and after giving nutritional supplementation; 11 studies measured body weight in COPD patients with normal BMI. Seven studies reported that body weight increased significantly following nutritional supplementation compared to the placebo groups, 26,37,39,28,29,42,44 and the study by Broekhuizen et al. 41 compared two nutritional supplement regimes, respifor versus ensini, fortimel and nutridrink, which found that both interventions significantly increased body weight. Four studies reported that body weight did not significantly improve in the intervention groups when compared to the placebo groups. 30,32,34,35 Of the RCTs in which body weight significantly increased, there was only one study, by Sugawara et al., that had a low risk of bias. 28 This study reported a significant increase in body weight after 12 weeks of 2.6 ± 3 kg in those receiving the ready-to-drink (ONS, described above) with the mean baseline body weight of 50.8 kg, compared to those in the placebo group with the mean baseline body weight of 54.8 kg. 28 In the study by Gurgun et al., there were significant improvements in body weight of 1.1 ± 0.9 kg, BMI 0.2 ± 1.4 kg/m2 and in FFMI (0.6 ±0.5 kg/m2) in those who received 250 mL of 83.3% carbohydrate, 30% fat and 16.7% protein three times a day as an intervention. 26 Of the four studies with negative findings, one study was at low risk of bias. 34 This study found no significant difference in body weight between the creatine intervention group and the placebo group after 8 weeks.
BMI was assessed before and after using supplementation in 5 of 24 studies. 26,33,37,31,35 BMI significantly increased in the supplementation group when compared to the placebo group in three studies. 26,37,31 Two studies reported no significant difference in BMI between participants who received nutritional supplementation with PR compared to PR only. 33,35 One RCT at the lowest risk of bias showed no improvement in BMI with carnitine. 33 In contrast, Gurgun et al. reported that BMI significantly increased after receiving nutritional supplement. 26
FFM was evaluated in nine trials. 32,36,39,41,28,35,29,42,44 Three studies demonstrated that FFM increased significantly in comparison with the placebo group but these studies all had some risk of bias. 36,39,42 Two 26,31 of four studies 26,41,31,35 with some risk of bias reported that FFMI significantly increased in the supplemental group when compared to the placebo group. In contrast, the study by Broekhuizen et al. reported no significant difference in FFMI between the group who received PUFA as an intervention and the placebo group who received palm and sunflower oil with vitamin E capsule as a placebo. 35
Peripheral muscle strength
Of the 24 studies included in this systematic review, 11 studies measured quadriceps muscle strength, handgrip strength or both 23,25,27,30,32,34,36,37,28,31,35
Three studies reported that handgrip strength did not significantly improve in the intervention groups when compared to placebo. 34,37,35 Faager et al. being at lowest risk of bias reported that using carnitine for 8 weeks during PR did not significantly improve handgrip strength when compared to the placebo group who received glucose. 34 In contrast, the study by Fuld et al., which had a higher risk of bias, showed significant improvement in handgrip after using creatine three times a day for 2 weeks followed by once a day for 10 weeks. 36
Quadriceps muscle strength was assessed in 11 studies. 23,25,27,30,32,34,36,37,28,31,35 Of the 11 RCTs, only three studies with 86 participants in total demonstrated positive findings. 36,28,31 The study by Sugawara et al., which had a low risk of bias, concluded that quadriceps muscle strength increased significantly after receiving a complex nutritional supplement when compared to the placebo group. 36,28,31 However, nine studies reported that using nutritional supplementation during PR had no additional effect on quadriceps muscle strength. 23,25,27,30,32,34,37,35 van de Bool et al. with a low risk of bias reported that using a high carbohydrate supplement showed no significant improvement in quadriceps strength when compared to the placebo group. 23 Similarly, the study by Faager et al. showed that using creatine for 8 weeks in COPD patients enrolled in an 8-week PR programme did not reveal significant differences in quadriceps muscle strength compared with those who used placebo. 34
Respiratory muscle function
Respiratory muscle function was assessed in 9 of the 24 included studies, 23,27,33,36,39,28,31,35,42 of which 3 were at lowest risk of bias. 23,33,28 Sugawara et al. reported that maximum inspiratory pressure (MIP) significantly improved in the interventional group (39.2 ± 38.9 cmH2O) after receiving nutritional supplement embedded in 12 weeks of PR compared with placebo (0.1 ± 24.1 cmH2O). 28 A small study by Borghi-Silva et al. showed a significant improvement in MIP (40 ± 14 cmH2O) with carnitine compared to placebo (MIP; 14 ± 5 cmH2O). 33 In contrast, a larger study by van de Bool et al. did not show a significant improvement in MIP when compared with placebo, who received glucose. 23 None of the studies that measured maximal expiratory pressure (MEP) showed a significant difference between interventional and placebo groups. 39,31,35
Quality of life
Quality of life was assessed in 14 of 24 studies. 23,25 –27,30,32,34,36,37,40,28,31,42 Eight studies used the St. George Respiratory Questionnaire (SGRQ), 25,26,34,36,41,31,42 and six used the Chronic Respiratory Disease Questionnaire (CRQ). 27,30,32,37,40,28 Overall, only two studies demonstrated a significant improvement in quality of life with supplementation in addition to PR. 36,28 Sugawara et al., which was at lowest risk of bias, measured quality of life using the CRQ and showed a significant improvement in those receiving nutritional supplement compared with placebo, which was clinically significant (6.2 ± 7.5 vs. −2.7 ± 13.1). 28 Thirteen studies showed negative findings including two RCTs at lowest risk of bias, including the study by Faager et al. using creatine supplementation and the study by van de Bool et al. using the high carbohydrate supplement. Faager et al. using creatine for 8 weeks during PR did not improve quality of life measured by SGRQ. 34 Similarly, van de Bool et al. reported that 4 months of using oral nutritional intervention did not show symptoms of anxiety and depression. 23
Discussion
This review is the first to summarise the potential effects of using nutritional supplementation during PR in patients with COPD. The studies varied in design, used differing supplements and measured different outcomes. In some, the primary purpose was to use the exercise component of PR to enhance the effect of nutrition, whereas others tested whether nutrition supplementation could enhance outcomes from PR. This results in considerable heterogeneity across studies, many of which were further limited by small sample size. It is, therefore, challenging to draw a single conclusion to address whether using a nutritional supplement has additional effects on exercise function, body composition, respiratory muscle function and quality of life during PR. We were also unable to perform meta-analysis due to this heterogeneity. Consequently, appropriately powered double-blinded RCT studies with suitable sample size using high energy/high protein nutritional supplement to investigate the effect of nutritional support in enhancing PR outcomes, and longer-term clinical outcomes, in COPD patients, are still needed. This would be particularly important in the high-risk group of COPD patients who are undernourished. This would support recommendations to incorporate nutritional support in PR management. 19,45 High protein/high energy ONS is recommended by the British Association for Parenteral and Enteral Nutrition for patients with COPD due to high energy and protein requirements 46 and PR services in different health contexts that need to consider how best to integrate nutritional assessment and, where successful, intervention into diverse methods of PR delivery.
Exercise capacity has been used to quantify the direct effect of nutrition interventions and to predict mortality and morbidity in COPD patients and other diseases. In this systematic review, the majority of studies demonstrated no improvement in exercise outcomes with nutritional supplementation in addition to PR, compared to PR alone. There were four RCTs with negative findings at low risk of bias, 23,24,33,34 which tested carbohydrate, B12, creatine and carnitine supplementation and just one small RCT with a positive finding, which used a ready-to-drink ONS twice a day composed of 200 kilocalories, 60% carbohydrates, 15% protein, 25% fat and 248 μg of omega-3 PUFAs 0.6 with vitamins A, C and E. These findings complement the meta-analysis of nutritional supplementation in stable COPD by Ferreira et al., which included 17 randomised clinical trials and concluded that nutritional supplements increased exercise tolerance in COPD patients who were poorly nourished when compared with baseline only, but which did not specifically consider use in the context of PR. 17 A meta-analysis was not possible in our review due to considerable heterogeneity in studies, as described above.
Body composition is one of the outcome measures that might be expected to improve when using nutritional supplement in COPD. Being underweight is associated with an increased risk of mortality in COPD. 13 Low body weight is observed in between 25% and 40% of COPD patients. Among these, 25% have moderate to severe weight loss and 35% have extremely low FFM. 47 In this systematic review, we found that ready-to-drink ONS during PR may increase body weight in a population with normal body weight but not with carnitine or creatine. Importantly, improvements in body weight and FFM using nutritional supplementation during PR appear to occur especially in depleted, malnourished and muscle-wasted patients (who are at highest risk). 23,26,31,29 In the meta-analysis by Ferreira et al, significant weight gain was noted compared to baseline in 11 RCTs and the meta-analysis of Collins et al. showed significant weight gain in favour of nutritional support when compared with control outside the context of a PR programme. 17,18
In recent years, researchers have paid attention to the assessment of outcomes, such as quadriceps muscle strength and handgrip strength. Handgrip strength and quadriceps muscle strength are valid measurements of peripheral muscle strength and are associated with mortality, morbidity and increased length of hospital stay.
19,48
In this systematic review, RCTs at low risk of bias did not support the concept that creatine, high carbohydrates, and
Respiratory muscle weakness in COPD patients may be due to several factors, such as acute exacerbations, systemic inflammation and malnutrition. 49 It has been suggested that nutritional supplements may improve respiratory muscle function. In this systematic review, we found two studies reporting that nutritional supplementation in addition to PR had an extra benefit in improving respiratory muscle function. This was demonstrated by measuring MIP and MEP. The effects were seen only on inspiratory measures, and the authors did not speculate on why they thought this was. Collins et al. concluded that MIP and MEP improved significantly in the intervention group when compared to usual care group. Ferreira et al. found that there was no significant difference between intervention control groups in MIP, but for malnourished patients with COPD, MIP and MEP improved significantly with nutritional support. 17,19
Quality of life may be affected through multiple mechanisms in COPD. The available evidence from this review included one small study demonstrating an improvement in QOL measured by CRQ using ready-to-drink ONS, and two studies with negative results, one of which used creatine and one of which also used ONS. The meta-analysis by Ferreira et al reported significant improvement for quality of life measured using SGRQ for patients with COPD who were malnourished. Additionally, Naz and Sahin demonstrated that protein-rich nutritional supplement significantly improved the quality of life in patients with COPD who participated at PR when compared to PR alone. 50
Strengths and limitations
To our knowledge, this is the only review that reports the effect of nutritional supplementation during PR in stable COPD. PR is an evidence-based and cost-effective intervention in COPD and thus maximising outcomes is of great interest to clinicians and patients alike. We have carefully searched the literature and registered our review in advance on PROSPERO. Three independent researchers examined the titles and abstracts for inclusion. Potential limitations include we only accessed studies in English, and the inherent variation, many of which had a risk of bias, for example, with inadequate sample size or absence of a power calculation, variation in outcomes measured, variety in study design or different PR protocols. Additionally, outcomes varied between studies, and we have not specifically considered the diversity of nutritional outcomes in this review, which focuses on clinical PR outcomes. There was significant diversity in the type, available substrate, energy imbalance or ingredients of the supplement either caloric or non-caloric and powder, liquid or tablets. We also observed a variation in the amount, contents and the duration of using supplements. Also, our review did not investigate the benefits of using nutritional supplements beyond the duration of PR, which could be important in clinical practice given that a major aim of PR programmes is to durably improve quality of life and reduce the risk of exacerbations and hospitalisations.
Conclusion
This is the first systematic review to report the value of nutritional supplementation during PR in patients with COPD. It is not possible to draw a definitive conclusion due to the heterogeneity of the supplements used, rehabilitation programmes and outcome measures. However, nutritional supplements may enhance the benefit of PR programmes, which would be of considerable benefit to those living with COPD. Not all studies showed positive results and there is a real need for further well-designed and rigorous research to address this area. This is particularly true in weight-losing and/or malnourished patients with COPD, who are at the highest risk of poor outcomes.
Supplemental material
supplemenary - Nutritional supplementation during pulmonary rehabilitation in COPD: A systematic review
supplemenary for Nutritional supplementation during pulmonary rehabilitation in COPD: A systematic review by Abdulelah M Aldhahir, Ahmed M Al Rajeh, Yousef S Aldabayan, Salifu Drammeh, Vanitha Subbu, Jaber S Alqahtani, John R Hurst and Swapna Mandal in Chronic Respiratory Disease
Footnotes
Authors’ note
John R Hurst and Swapna Mandal are joint senior authors.
Acknowledgement
We thank Steven Bembridge Medical Librarian at Royal Free London NHS Foundation Trust, UK, for his assistance and support in refining the search strategy.
Author contributions
AMA, JRH, and SM conceived and designed the study. AMA performed the initial search and data extraction, while JRH and SM checked the eligibility of the included articles. AMA and JRH performed the quality assessment for the included articles. AMA wrote the initial manuscript and YSA, JSA, SD, and AMR contributed to the writing of the manuscript. JRH, SM, and VS revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JRH, SM, and AMA are running an RCT of protein supplementation to enhance PR outcomes in COPD. The product is being supplied by Nutricia. JRH received grants outside the submitted work from pharmaceutical companies that make medicines to treat COPD.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
