Abstract
The stigma of non-communicable respiratory diseases (NCRDs), whether perceived or otherwise, can be an important element of a patient’s experience of his/her illness and a contributing factor to poor psychosocial, treatment and clinical outcomes. This systematic review examines the evidence regarding the associations between stigma-related experiences and patient outcomes, comparing findings across a range of common NCRDs. Electronic databases and manual searches were conducted to identify original quantitative research published to December 2015. Articles focussing on adult patient samples diagnosed with asthma, chronic obstructive pulmonary disease (COPD), cystic fibrosis, lung cancer or mesothelioma, and included a measurement of stigma-related experience (i.e. perceived stigma, shame, blame or guilt), were eligible for inclusion. Included articles were described for study characteristics, outcome scores, correlates between stigma-related experiences and patient outcomes and methodological rigor. Twenty-five articles were eligible for this review, with most (n = 20) related to lung cancer. No articles for cystic fibrosis were identified. Twenty unique scales were used, with low to moderate stigma-related experiences reported overall. The stigma-related experiences significantly correlated with all six patient-related domains explored (psychosocial, quality of life, behavioral, physical, treatment and work), which were investigated more widely in COPD and lung cancer samples. No studies adequately met all criteria for methodological rigor. The inter-connectedness of stigma-related experiences to other aspects of patient experiences highlight that an integrated approach is needed to address this important issue. Future studies should adopt more rigorous methodology, including streamlining measures, to provide robust evidence.
Keywords
Background
Respiratory diseases represent a major public health concern due to their high frequency, economic burden and social impacts. 1 The majority of serious respiratory conditions are incurable. Causes are varied; however, many are considered to be largely preventable. 2 Non-communicable respiratory diseases (NCRDs), which are conditions of long duration and generally slow progression, are one of the main types of chronic conditions in the world. 3 In high-income countries, NCRDs such as asthma, chronic obstructive pulmonary disease (COPD), interstitial lung disease, cystic fibrosis, pulmonary arterial hypertension, lung cancer and occupation-related conditions are a substantial contributor to mortality and illness-related burden. 3 While the disease-related experiences of these patients may differ widely, it is important to understand whether common factors can potentially contribute to their health and general well-being.
NCRDs can significantly impact various aspects of patient outcomes. Functional capacity can become limited with symptoms including wheeziness/breathlessness, cough, fatigue, chest pain and/or tightness, or presence of blood or sputum. However, beyond physical outcomes, psychosocial well-being and quality of life can also be affected. 4 This has been demonstrated in previous studies where NCRD patients reported experiencing issues related to loss of independence, altered family dynamics, feelings of social isolation, embarrassment, guilt, fear and body image disturbances. 5 –9 Of important note is the potential ripple effect towards adverse clinical outcomes. For example, in a sample of patients with late stage lung cancer, depression was found to be associated with poor treatment adherence and prognosis. 10 This highlights the inter-relatedness of factors that influence patient outcomes and the need for an integrated approach to patient care and management in order to improve well-being, treatment and potentially survival outcomes.
Emerging research suggests that stigma-related experiences may be frequent among individuals with a number of health conditions, including NCRDs. 11 Contemporary concepts of stigma can be traced to Goffman’s work, which is described as an attribute that discredits or devalues individuals, reducing them ‘from a whole person to a tainted, discounted one’. 12 More recently the stigma concept has been reframed for health-related contexts as an adverse social judgement ‘based on an enduring feature of identity conferred by a health problem or health-related condition’. 13 Internalized or self-stigma may manifest when individuals become aware of the negative social perceptions attached to particular diagnoses and consequently anticipate prejudice or discrimination. Individuals may also carry self-blame on account of their behavior that can drive feelings of shame and guilt about their condition. 14,15 Therefore, while stigma, shame, blame and guilt may be distinct psychological concepts, they can be considered related under the term ‘stigma-related experiences’.
In the context of NCRDs, self-stigma may occur regardless of how the condition was acquired (whether through lifestyle behavior or congenital/hereditary factors). Given that tobacco is the main modifiable risk factor for COPD and lung cancer, 16 the perceived controllability of an individual’s smoking behavior may influence social reactions to disease onset. A survey of the general population showed respondents were more likely to hold lung cancer patients with a smoking history accountable for their condition compared to their non-smoking counterparts. 17 Similarly, more than one-third of people with COPD attributed their diagnosis to personal behavior. 18 Cystic fibrosis, an inherited condition, is also subjected to adverse public responses, 19 given its links to poor physical outcomes and premature mortality. 20
While mostly anecdotal, the literature suggests that stigmatizing attitudes can variably influence patient’s health and treatment behaviors. COPD and lung cancer patients have reported delays in seeking medical assistance on account of not having their concerns addressed seriously by health professionals or feeling unworthy of receiving treatment. 21 Patients with asthma have reported feeling embarrassed using treatment in public, 22 while cystic fibrosis patients describe feelings of unease at producing socially unacceptable symptoms (e.g. sputum); 23 this can contribute to issues with treatment adherence which is commonly poor in this population. 24 –26 Such experiences can act as significant barriers to achieving optimal patient care and health outcomes.
The stigma literature in NCRDs is increasing. Evidence suggests that patients with a range of NCRDs report feelings of perceived stigma, shame, blame or guilt. 20,21,27,28 These experiences may generally be a feature of the social context of disease, contributing to not only the personal barriers to seeking treatment and care, but also adherence to treatment and hence overall physical and mental health outcomes. 29 While it seems that stigma-related experiences can be a consistent experience in NCRDs, notably the aetiology, severity and familiarity of illnesses vary. Therefore, stigma-related experiences may (or may not) vary by condition. However, a comprehensive and current review of the data regarding stigma-related experiences and their broader impacts on a range of respiratory diseases is lacking.
Recently, a review investigated the impact of lung cancer stigma and nihilism on patient outcomes and concluded that perceived stigma is evident and additional research is necessary. 30 This review aims to build on this work by examining the experience of stigma in addition to shame, blame and guilt among a wider scope of NCRDs, exploring the potential impacts across the full range of patient outcomes. This will allow common features of stigma-related experiences to be identified across groups, to highlight unique or shared contributory factors to such experiences, and describe the influence on patient outcomes. Understanding the patient experience in this respect is needed to understand how to best engage and support people during provision of health care. 31 –34 To facilitate this understanding, assessing the methodological rigor of the literature is also required to enable the development of recommendations informed by strong and robust data.
Specifically, the aim of this review is to examine the current evidence regarding stigma-related experiences (inclusive of perceived stigma, shame, blame or guilt) to patient outcomes within the following NCRDs: asthma, COPD, cystic fibrosis, lung cancer and mesothelioma. The literature will be described in terms of: the number of articles relating to each NCRD and study characteristics; the prevalence of stigma-related experiences across NCRD groups; associations between reported stigma-related experiences in each NCRD group and patient outcomes across domains that were identified in the literature search; and the quality of evidence in identified articles, including whether study populations were representative of disease groups.
Methods
A systematic review was conducted using the PRISMA statement. 38
Search terms
Search terms were generated iteratively by the research team and reviewed by an experienced medical librarian. Searches were conducted in December 2015 using a selection of electronic databases to retrieve original peer-reviewed publications and unpublished studies from the grey literature. Manual searches were also conducted to ensure all relevant articles were identified. This included searches in Google Scholar, whereby the first 100 results were reviewed for relevance, and websites of organizations applicable to the NCRDs of interest. Table 1 details the search terms and sources of article retrieval.
Search strategy.

Inclusion and exclusion criteria
Articles were included for review if they involved: an adult (18 years or older) patient sample; measurement of stigma-related experiences (inclusive of perceived stigma, shame, blame and guilt); and one of the five following NCRDs: asthma, COPD (including emphysema and chronic bronchitis), cystic fibrosis, lung cancer and mesothelioma.
The listed NCRDs were selected because of their high international prevalence 35 –37 and varying aetiologies (e.g. behavioral, occupational and genetic), perceived controllability (e.g. role of modifiable or non-modifiable factors) and severity. This allowed for potential stigma-related consequences to be compared and contrasted based on specific disease factors.
Articles were excluded if they: were duplicates; were not published in English; were a review, editorial, commentary or protocol; did not describe original research; described solely qualitative findings; exclusively reported aggregate findings across conditions not of interest to this review; or did not assess stigma-related experiences in relation to patient outcomes (e.g. examined inequities or discrimination based on gender, race, socioeconomic status, etc.).
As research in health-related stigma is an emerging field, the year of publication was not limited. The information extracted from articles included author, sample size, sample characteristics (age, gender, smoking status, disease severity and time of diagnosis), disease type, the stigma-related measure used and stigma-related findings.
Quality assessment
Appraisal of the inclusion and exclusion criteria for each article was conducted by a first coder (SR) and a random sample (10%) was independently checked by a second coder (DR). Of included articles, data was extracted by a first coder (SR) and a random sample (20%) was independently checked by a second coder (DR). Data extraction of all included articles was performed independently by two coders (SR and DR). Discrepancies were resolved through discussion. At all stages, more than 90% agreement between coders was achieved.
The methodological quality of all included studies was also assessed independently by two separate coders (SR, CP, AB and BK) using an amended version of the McMaster criteria form for quantitative studies (intervention criteria removed and replaced with sample representativeness criteria). Discrepancies were discussed until consensus was reached.
Results
Volume of articles
A total of 1431 articles were retrieved from the literature search (Figure 1). An additional 27 articles were identified via manual searches. Of these, 25 (peer-reviewed, n = 22; grey literature, n = 3) were included for this review.
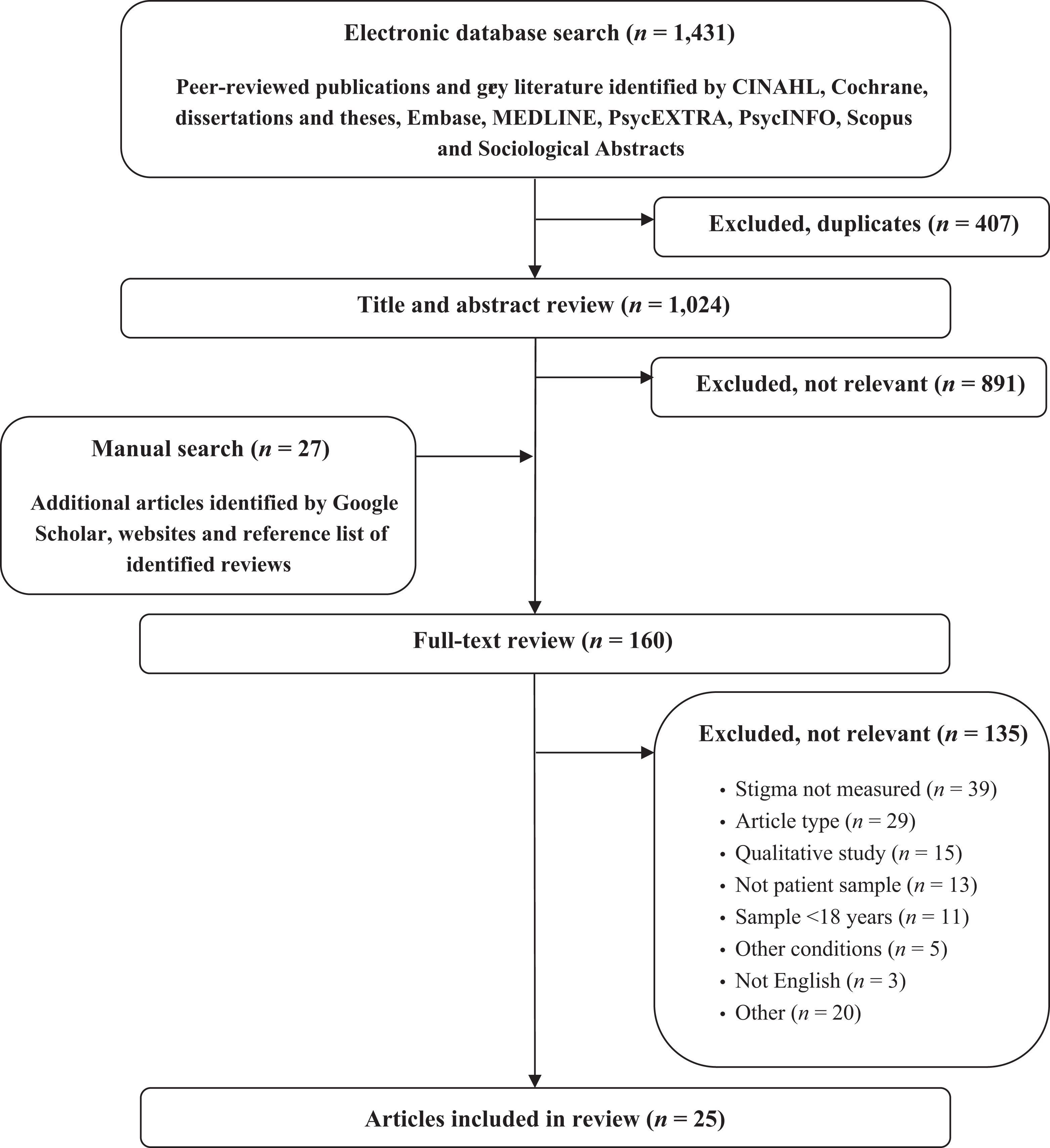
Flow chart of review process.
Most studies (n = 20) involved people with lung cancer. 39 –58 The next most common participant group was COPD (n = 4), 18,49,59,60 followed by asthma 27,59,61 and mesothelioma 43 (some articles examined more than one NCRD). No identified article examined stigma-related experiences in relation to cystic fibrosis. Table 2 summarizes the sample characteristics.
Summary of sample characteristics and description of stigma-related findings.

dx: diagnosis; mo: months; NR: not reported; NSCLC: non–small cell lung cancer; SCLC: small cell lung cancer; SD: standard deviation; tx: treatment; yr: years; COPD: chronic obstructive pulmonary disease.
Prevalence of stigma-related experiences
Articles examined NCRDs in relation to perceived stigma, 27,39 –42,44,45,47 –52,56 –59,61 shame or guilt 43 –46,52,54,60 and blame. 18,50,51,53,55 Generally, most samples within each NCRDs tended to report low (i.e. aggregate scores in the lowest third of the possible total) to moderate (i.e. aggregate scores in the middle third of the possible total) scores of stigma-related experiences (Table 2). However, higher aggregate stigma-related scores (i.e. aggregate scores in the highest third of the possible total) were reported in COPD 18 and lung cancer samples. 42,50,51 Overall, perceived stigma was measured via 10 different scales, blame via six separate scales and shame or guilt via five unique scales (Table 2).
Associations between stigma-related experiences and patient outcomes
Twenty of the 25 articles investigated stigma-related experiences with patient outcomes. 18,27,39 –45,47 –49,52,53,55 –60 The remaining studies either did not investigate patient outcomes 46,54,61 or reported combined findings with other disease groups (i.e. not NCRD) that were not relevant to this review. 50,51
Outcomes associated with stigma-related experiences by the study participants were identified in the following six domains: psychosocial, quality of life, behavioral, physical, treatment and employment. Broadly, stigma-related experiences were shown to have significant associations with all domains (Table 3). These relationships were investigated more widely in COPD and lung cancer; however, fewer domains were explored in asthma (psychosocial, physical and employment) and mesothelioma (behavioral only).
Frequency and summary of associations between stigma-related experiences and patient outcomes.
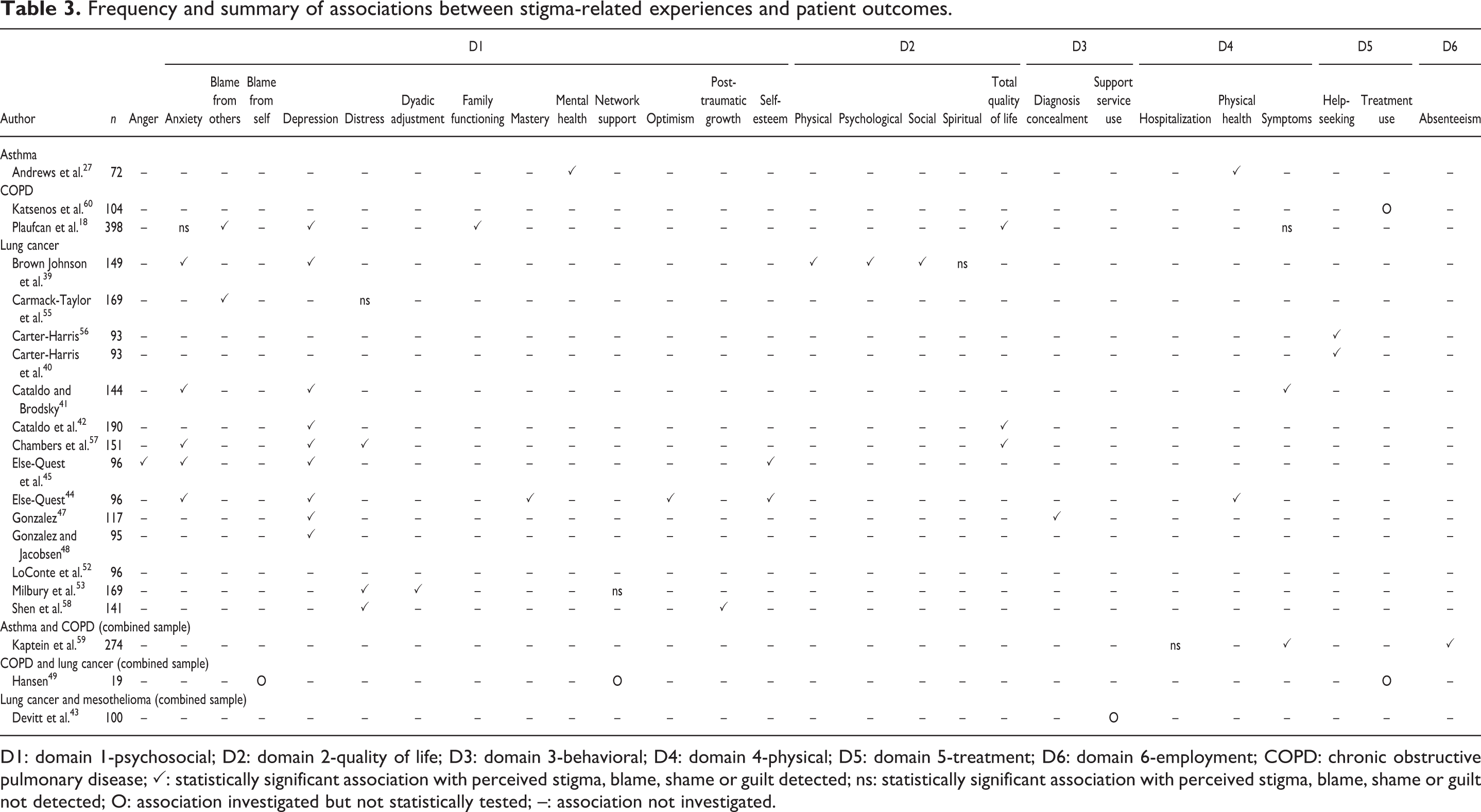
D1: domain 1-psychosocial; D2: domain 2-quality of life; D3: domain 3-behavioral; D4: domain 4-physical; D5: domain 5-treatment; D6: domain 6-employment; COPD: chronic obstructive pulmonary disease; ✓: statistically significant association with perceived stigma, blame, shame or guilt detected; ns: statistically significant association with perceived stigma, blame, shame or guilt not detected; O: association investigated but not statistically tested; –: association not investigated.
Stigma-related experiences and psychosocial outcomes
Fourteen articles investigated the possible associations between stigma-related experiences and psychosocial outcomes among patients with asthma, 27 COPD 18,49 and lung cancer. 39,41,42,44,45,47 –49,53,55,57,58 In particular, the association between perceived stigma and depression was most frequently investigated, with all studies indicating a significant positive relationship.
Studies in COPD and lung cancer examined similar patient psychosocial outcomes. Blame from others, family functioning and dyadic adjustment were found to significantly correlate with self-blame in both disease groups. 18,53,55 Prevalence data also indicated that COPD and lung cancer patients frequently experience perceived stigma, where disease onset can be attributed to personal responsibility. 49 However, anxiety was found to be significantly associated with perceived stigma in lung cancer 39,41,44,45 but not COPD. 18 The relationship between perceived stigma and mental health was investigated in asthma only and could not be compared to other NCRDs. In lung cancer, non-significant associations were reported when self-blame was measured in relation to distress 55 and social support 53 only.
Stigma-related experiences and quality of life
Four articles investigated stigma-related experiences with quality of life outcomes among people with COPD 18 and lung cancer. 39,42,57 Significant inverse relationships were reported in both disease groups when measured with perceived stigma 39,42,57 and self-blame. 18
Stigma-related experiences and behavior
Two studies investigated the associations between stigma-related experiences and behavior among people with lung cancer 43,47 and mesothelioma. 43 Diagnosis concealment was reported to be significantly associated with internalized shame within the last month and since diagnosis in patients with lung cancer. 47
Stigma-related experiences and physical health
Six studies investigated the associations between stigma-related experiences and physical outcomes among people with asthma, 27,59 COPD 18,59 and lung cancer. 41,44,52 Poorer health status scores were significantly linked to greater levels of perceived stigma in asthma patients, 27 and feelings of greater shame, guilt and self-blame in people with lung cancer. 44 No links between perceived stigma and hospitalizations were found in asthma and COPD patients. 27,59
Findings in relation to patient physical symptoms were mixed. Among people with lung cancer, perceived stigma was found to have a positive relationship with perceived symptom severity, as well as with all investigated symptoms including appetite loss, cough, dyspnea, fatigue, haemoptysis and pain. 41 Similarly, in asthma and COPD patients combined, perceived stigma was significantly associated with dyspnea and cough symptoms (only aggregate findings were reported). 59 However, self-blame was shown to have no association with breathlessness in COPD patients. 18
Stigma-related experiences and treatment experience
Four articles investigated the possible associations between stigma-related experiences and treatment experiences within COPD 49,60 and lung cancer. 40,49,56 Higher levels of perceived stigma were shown to predict longer delays between symptom onset and medical help-seeking in lung cancer patients. Descriptive data also suggested that perceived stigma may be connected to poor treatment compliance in both COPD and lung cancer patients. 49,60
Stigma-related experiences and employment
Only one article investigated the associations between stigma-related experiences and work factors within a combined sample of asthma and COPD patients. 59 Higher levels of perceived stigma were shown to be significantly linked to absenteeism.
Determinants of stigma-related experiences
Determinants of stigma-related experiences were explored in a small proportion of articles. Smoking history was a common factor explored in COPD and lung cancer samples. In COPD, it was found that people with greater duration and intensity of smoking were more likely to endorse the maximum score of behavioral self-blame. 18 Similarly, in lung cancer, smoking history was reported to be correlated with greater self-blame, 44,53,57,58 guilt and shame. 52 Time since diagnosis was explored in lung cancer samples, where relationships between higher perceived stigma and increased number of days to seek medical advice for symptoms were reported. 52,56 Younger age was also shown to be significantly linked to higher perceived stigma scores in COPD and lung cancer patients. 49
Quality of evidence (methodological rigor)
A summary of scores for methodological rigor is described in Table 4. Overall, no articles adequately met all criteria; however, three satisfactorily met six of the seven criteria 44,47,48 (all lung cancer samples). One article only met one criteria for methodological rigor. 54 The majority of articles (n = 20) included a sample with moderate to high risk of bias. Sample features (such as characteristics and justification of sample size) were also lacking sufficient detail, with a small number of articles (n = 5) adequately addressing this criteria.
Quality assessment of included articles.
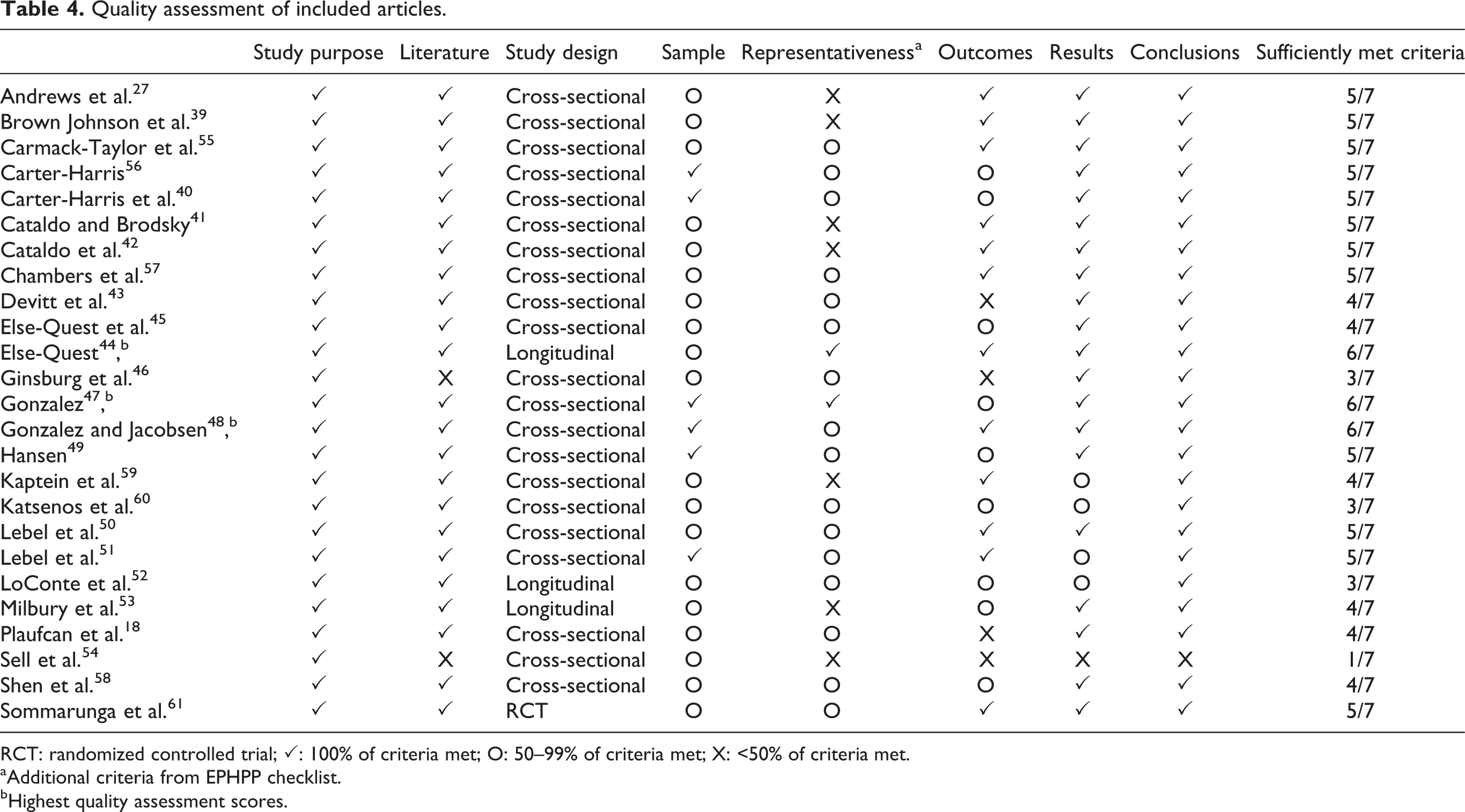
RCT: randomized controlled trial; ✓: 100% of criteria met; O: 50–99% of criteria met; X: <50% of criteria met.
aAdditional criteria from EPHPP checklist.
bHighest quality assessment scores.
Asthma samples were older than the general population of people with asthma. However, it should be noted that this review focused on studies conducted with in adult samples. Approximately, one-fifth of the lung cancer studies included samples younger than the average patient (mean sample age less than 60 years old). 39,41,42 Participants in the remaining lung cancer and COPD studies appropriately reflect the age typically seen within their respective disease groups (approximately 60 years and older).
Discussion
This review builds on previous work 30 by identifying correlates of stigma-related experiences across a larger number of patient domains within various NCRDs. Our findings were directed towards patient experiences and look more in depth at the potential stigma-related impacts overall, inclusive of day-to-day effects (e.g. work and social interactions). Common NCRDs of different prognoses and causative factors (such as behavioral, environmental or genetic) were targeted to determine whether such features can be attributable when comparing differences in patient outcomes. Understanding the possible factors that may contribute to or influence stigma-related experiences will identify relevant areas for delivering appropriate care and support to patients with NCRDs to promote improved patient outcomes.
Studies addressing stigma-related effects on patient outcomes were predominantly in relation to psychosocial domains. This may be because stigma is largely viewed as a social phenomenon that centres on public identity and group membership. 12 As such, the psychological effects may be seen as more pertinent by researchers. Despite this, in accordance with health-related stigma, 13 a broader range of patient outcomes can be impacted, which is worthy of further exploration. For example, only a small proportion of studies identified in this review investigated the relationship between perceived stigma and shame to either an individual’s behaviour or treatment experiences 18,40,43,47,49,56 with statistically significant relationships detected. Previous qualitative studies have demonstrated that factors such as fear or reluctance in discussing symptoms as a barrier to seeking medical advice. 21,62 However, in the instance of NCRDs (such as lung cancer), prompt symptom recognition and medical help-seeking is key to potentially extending survival. 62 As data are limited, it is of particular importance to examine whether stigma-related experiences can have detrimental implications later in the disease trajectory, particularly in relation to treatment decisions or adherence, and the role of nihilism on treatment choices.
Studies included in this review were primarily focused on COPD or lung cancer, indicating their vulnerability to stigmatization compared to other NCRD groups. Potentially contributing to this is the known role of tobacco smoking to the onset of COPD and lung cancer, 16 with the perception that the behavior is controllable and the disease therefore preventable. This has been reflected in a population-based study in which 70% of participants believed that lung cancer patients were at least partially to blame for their diagnosis. 63 Qualitative data suggests that anti-tobacco campaigns may contribute to additional distress in COPD and lung cancer samples. 21,28 However, further research is needed 30 as these data are anecdotal and the potential influence of media campaigns on stigma-related experiences is not clear. The bulk of identified studies were published within the last 10 years. This recent increase in stigma-related research, particularly in relation to COPD and lung cancer, may reflect the emergence of anti-tobacco campaigns that have come into prominence in recent years. For example, the United States’ ‘Truth’ campaign and Australia’s ‘Make Smoking History’ campaign were launched in 2000 and graphically depict the adverse effects of smoking.
A key issue is the ambiguity when interpreting the reported findings for this review. Across the 25 articles included in this review, 20 different scales were used to measure an element of stigma-related experience (i.e. perceived stigma, shame, blame or guilt). None of the measures used had specified thresholds by which to categorize the level of stigma-related experiences, and only two measures were specifically for NCRDs (the Cataldo Lung Cancer Stigma Scale and the Respiratory Illness Opinion Survey). Studies commonly used measures validated in mental health samples, 18,27,44,45,47 –52 or author-developed scales where validity and reliability were not assessed. 43 –46,49,52,54,60 Most measures comprised of one item, with only the Cataldo Lung Cancer Stigma Scale, the Social Impact Scale and the adapted version of the Shame and Stigma Scale providing a more comprehensive assessment of perceived stigma with 31, 24 and 21 items, respectively. While these findings provide an indication of stigma-related experiences across NCRDs, the vast differences between measures and the lack of clarity around interpretation of scores create challenges in objectively comparing experiences and effects on patient outcomes across (and within) disease groups.
The limitations of this review also need to be considered. As only quantitative English articles were included, some relevant articles may have been excluded. A broad set of search terms were used with the intent of adopting an inclusive approach to retrieving articles. Due to the scope covering multiple conditions, the authors considered it was appropriate to use a generalized set of search terms to capture a thorough pool of articles in an area that is limited. While it was considered unlikely that relevant articles would be missed, additional search strategies were employed, such as conducting searches via Google Scholar search and websites of key organizations, to ensure that this was minimized. While only a proportion of article titles, abstracts and full texts were screened by a second coder, there was a high level of agreement which suggests that potential errors were minimal. The aims of the included studies were not reported in this review. It may be that stigma-related experiences were not the primary focus of some of the included articles, which may account for the selection of less thorough or robust stigma-related measures. Finally, the authors’ categorization of the scale scores as low, moderate and high was a specific strategy for the purposes of this review and requires validation.
Stigma-related research in NCRDs is increasing. However, knowledge gaps are still evident with further investigation towards stigma warranted. Greater clarity about the various concepts (perceived stigma, shame, blame and guilt) commonly grouped together under the umbrella term ‘stigma’ is likely to be helpful. The authors also suggest that attention be directed towards the following:
Further development of stigma-related measures: The use of different tools across included articles contributed to the variability of findings. It may also be that the stigma-related scales used were not appropriate. As most scales originated and were developed for mental illness, they may not have the sensitivity to detect similar outcomes in patients with other chronic health conditions, such as NCRDs. In order to address this, future efforts need to be directed towards establishing stigma scales applicable to these disease groups. Further, development of generic scales for use in populations with chronic conditions will enable comparisons of stigma across different populations. The establishment of score thresholds would enable the identification of individuals experiencing high levels of perceived stigma who may benefit from intervention.
Ensuring studies are methodologically robust: Steps to minimize potential selection bias to improve methodological rigor need to be considered in the design of studies. Randomizing participants for intervention studies and adopting recruitment methods that facilitate representative sampling (i.e. limiting self-selection or volunteer strategies) in cross-sectional studies are strategies that can alleviate this risk. Complete descriptions of sample characteristics are needed to provide a more comprehensive picture. This will assist to determine whether the stigma impacts particular patient groups or whether particular patient subgroups are more vulnerable to stigmatization to target future interventions.
Broadening research to examine stigma-related experiences beyond psychosocial correlates: It is timely to explore stigma-related consequences that reflect patient outcomes more widely, integrating the multiple factors that are involved in their care processes (such as treatment decision-making) and day-to-day management (such as mobility). In conditions such as lung cancer or mesothelioma where early detection is particularly pertinent in providing greater treatment options, addressing stigma may assist in reducing barriers to seeking timely medical help. This in turn may improve clinical outcomes and reduce psychosocial distress.
Investigating temporal relationships of stigma-related experiences: Most studies utilized a cross-sectional design, where causality cannot be established. It would be opportune to identify whether stigma-related experiences are transitory or enduring to enable recommendations for future support services, as well as the appropriate timing of services to promote optimum patient outcomes.
Clinical implications
The stigma of NCRDs is an important issue to address, and clinicians can play a key role in effectively responding to these patient experiences. As found in this review, perceived stigma and shame were shown to be associated with delayed medical help-seeking, diagnosis concealment, symptom severity and poor treatment use for patients, 40,49,56,60 which can adversely impact patient health outcomes. Other studies have demonstrated that clinicians who communicate empathetically with their patients can lead to higher satisfaction and improved health outcomes (such as compliance with treatment) for patients. 64,65 However, the stigma that may manifest with NCRDs can impede opportunities in facilitating open dialogue between the patients and their clinician to promote best possible care. 62 Further, it may be that opportunities are missed by clinicians due to time constraints or focus towards making diagnoses or recommending appropriate treatment. It is therefore important for clinicians to be vigilant in recognizing and responding to these opportunities when they arise, but also to identify potential patient stigma or related experiences to understand and appropriately discuss concerns sensitively to alleviate the impact. Such practices can be instrumental in addressing the overt as well as underlying issues that may influence stigma-related experiences to support the patient self-esteem, disease management and treatment 66 to assist with improving both clinical and psychosocial outcomes for the patient.
Conclusion
The findings of this review indicate that patients with NCRDs report low to moderate scores of stigma-related experiences. However, more rigorous studies are needed in order to determine this more accurately and comprehensively. Within the large pool of literature resulting from this search, only 25 stigma-related studies were identified in relation to NCRDs. This literature gap is of concern, particularly given the association between stigma-related experiences and important patient outcomes. Building the evidence base will be critical in developing appropriate interventions and the delivery of best care within population groups vulnerable to stigma.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by infrastructure funding from the Hunter Medical Research Institute. Ms. Shiho Rose was supported by a University of Newcastle Research Postgraduate Scholarship. A/Prof. Christine Paul was supported by a Hunter Medical Research Institute Fellowship and a National Health and Medical Research Council Career Development Fellowship. Dr. Allison Boyes was supported by a National Health and Medical Research Council Early Career Research Fellowship Cancer Institute NSW Early Career Research Fellowship.
