Abstract
Gas exchange impairment is primarily caused by ventilation–perfusion mismatch in chronic obstructive pulmonary disease (COPD), where diffusing capacity of the lungs for carbon monoxide (DLCO) remains the clinical measure. This study investigates whether DLCO: (1) can predict respiratory impairment in COPD, that is, changes in oxygen and carbon dioxide (CO2); (2) is associated with combined risk assessment score for COPD (Global Initiative for Chronic Obstructive Lung Disease (GOLD) score); and (3) is associated with blood glucose and body mass index (BMI). Fifty patients were included retrospectively. DLCO; arterial blood gas at inspired oxygen (FiO2) = 0.21; oxygen saturation (SpO2) at FiO2 = 0.21 (SpO2 21) and FiO2 = 0.15 (SpO2 15) were registered. Difference between arterial and end-tidal CO2 (ΔCO2) was calculated. COPD severity was stratified according to GOLD score. The association between DLCO, SpO2, ΔCO2, GOLD score, blood glucose, and BMI was investigated. Multiple regression showed association between DLCO and GOLD score, BMI, and glucose level (R 2 = 0.6, p < 0.0001). Linear and multiple regression showed an association between DLCO and SpO2 21 (R 2 = 0.3, p = 0.001 and p = 0.03, respectively) without contribution from SpO2 15 or ΔCO2. A stronger association between DLCO and GOLD score than between DLCO and SpO2 could indicate that DLCO is more descriptive of systemic deconditioning than gas exchange in COPD patients. However, further larger studies are needed. A weaker association is seen between DLCO and SpO2 21 without contribution from SpO2 15 and ΔCO2. This could indicate that DLCO is more descriptive of systemic deconditioning than gas exchange in COPD patients. However, further larger studies are needed.
Keywords
Introduction
To evaluate the severity of chronic obstructive pulmonary disease (COPD), a number of measurements are required. These include spirometry, including forced expiratory volume during the first second (FEV1), forced vital capacity (FVC), and the ratio between these two measurements (FEV1/FVC). 1 Spirometry measures are supplemented by evaluation of dyspnea using, for example, the modified Medical Research Council (mMRC) score 2 and considerations of the number of exacerbations in the preceding year. These figures allow stratification of the patient using the combined risk assessment score for COPD (GOLD score) as recommended by the Global Initiative for Chronic Obstructive Lung Disease (GOLD). 1 The GOLD score does not hold any information about pulmonary gas exchange, even though this is an important pathophysiological finding in COPD. The main pathophysiological reason for gas exchange impairment is ventilation/perfusion (V/Q) mismatch with heterogeneous distribution of both areas of high and low V/Q ratios. 3,4 However, V/Q mismatch is not easily measured in routine clinical practice. The reference technique for the measurement of V/Q mismatch is the multiple inert gas elimination technique. 5 It involves the infusion of six inert gasses and, based on the measurement of output and mathematical processing of these measurements, evaluation of V/Q ratio in a model with 50 pulmonary compartments. Yet, the use of this method is limited in daily clinical care by its complexity of both procedure and interpretation. In the clinical setting, diffusing capacity is often used, as a surrogate measure, to estimate impaired gas exchange. In COPD, both areas of low V/Q ratios, that is, impaired ventilation, and high V/Q ratios, that is, areas with impaired perfusion, are seen in the lung. 4,6 As such, changes in arterial oxygen saturation (SpO2) and difference between arterial and end-tidal carbon dioxide (ΔCO2) can describe both areas of low and high V/Q ratios in the lung. 7 The question therefore arises as to what diffusing capacity of the lungs for carbon monoxide (DLCO) describes in COPD and whether it is a useful measurement. It has previously been shown that in COPD, DLCO is associated with dyspnea, evalated by the Borg scale, a dyspnea score used during exercise. 8 In addition, previous studies have shown that DLCO measurements are influenced by, and possibly describe, a number of systemic variations: an increase in body mass index (BMI) 9 –11 and lean body mass have been associated with an increase in DLCO 12 ; both acute 13 and long-term poor glycemic control 14 have been shown to correlate negatively with DLCO in diabetic patients, a finding that may be present even in early life. 15 Moreover, it is well known that even when DLCO is adjusted according to the hemoglobin level, 16 only anemic levels of hemoglobin are negatively associated with DLCO. 17,18
While these studies show that DLCO is a useful parameter to describe severity of perceived dyspnea and perhaps systemic variations, they do not confirm DLCO as a valid measurement of pulmonary gas exchange. Two factors are available in the clinical setting to understand whether DLCO is a useful approximation of gas exchange. The first is whether DLCO describes the relationship between inspired oxygen (FiO2) and SpO2 adequately. Small changes in oxygenation and the resulting change in arterial values have previously been shown to characterize V/Q mismatch in the lungs. 4,19,20 The second is whether it describes the relationship between end-tidal PCO2 and arterial PCO2 adequately. Indeed, the ratio between these two measurements (referred to here as ΔCO2) has previously been considered an appropriate measure for carbon dioxide (CO2) and dead space monitoring in COPD. 21 Taken together, this suggests that in COPD patients, where deoxygenation is primarily caused by impairment of pulmonary gas exchange, DLCO may provide a poor description of susceptibility to changes in oxygenation and differences in arterial and end-tidal PCO2. DLCO may instead describe the systemic deconditioning of the lung and body as a whole. This hypothesis was tested in a cohort of 50 COPD patients.
Therefore, the specific aims were to investigate the association between DLCO and (1) clinical measurements describing gas exchange, that is, SpO2 and its response to change in FiO2 and ΔCO2, (2) GOLD stratification score, and (3) systemic parameters, that is, BMI, blood glucose, and hemoglobin.
Methods
Patients
A cohort of 50 patients were included in this retrospective study. All patients suffered from COPD according to GOLD standards. 1 All were in a stable state, with none having had either COPD exacerbation 6 weeks prior to examination or major changes in the treatment of either COPD or other chronic diseases such as diabetes and heart disease. None of the patients had previously undergone lung surgery or were suspected of lung cancer.
Twenty-five of the above mentioned 50 COPD patients were enrolled from a study investigating physiological and radiological parameters in COPD. Data were collected from June 2011 till June 2012 (local ethical approval N-20090012; data regarding computed tomography (CT) scans have been published in the study by Thomsen et al. 22 ). The COPD patients were examined with a high-resolution CT scan, a body plethysmography and DLCO measurement, and measurement of pulmonary gas exchange via step changes in FiO2 mediated by the ALPE equipment (ALPE Essential®, Mermaid Care, Nørresundby, Denmark). 23,24 Arterial blood gases were obtained via puncture at FiO2 = 0.21, and partial pressure of oxygen (PaO2) and partial pressure of carbon dioxide (PaCO2) were registered for further analyses. In the background information in the “Results” section, this group is referred to as “data set 1.”
An additional 25 COPD patients were enrolled from a feasibility project carried out in the Department of Pulmonary Medicine at Aalborg University Hospital. Data were collected from April 2011 to June 2012 and registered and kept according to the legislation of the Danish Data Protection Agency. The feasibility project was presented to the ethical committee of Northern Jutland who found no need for ethical approval. The study elucidated the practical implications of using changes in FiO2 as a measure of gas exchange in a daily clinical setting. In this study, 147 patients with various pulmonary diseases, who were referred to a DLCO measurement, were offered a supplementary ALPE measurement, where SpO2 was monitored during an automatized stepwise reduction and final return to initial values of FiO2. Of the 147 included, 25 presented with COPD and therefore met the inclusion criteria for the present study. No data have previously been published from this study. Describing background information in the Results section, this group is referred to as data set 2.
Demographic data describing age, gender, smoking status, and BMI, and paraclinical data on hemoglobin and blood glucose were obtained from the patients' case files as well as patients' spirometry, information on dyspnea, using the mMRC score 2 , and the number of exacerbations in the preceding year to evaluate the GOLD score, judged by the GOLD stratification. 1
Pulmonary function
DLCO was measured prior to step changes in FiO2 in all 50 patients.
The DLCO procedure was performed according to international standards 25 using MasterScreen® Body (VIASYS Healthcare GmbH, San Diego, California, USA). DLCO expressed as the percent of the predicted value (DLCO%) was used for further analysis.
Changes in FiO2 were carried out in steps to a minimum of 0.15 and finally returned to initial or higher values, mediated by the ALPE equipment. At each step, SpO2 was registered when oxygen (O2) steady state was obtained. SpO2 at FiO2 = 0.21 (SpO2 21) and at FiO2 = 0.15 (SpO2 15) were used for further analysis. Furthermore, end-tidal PCO2 was measured, the value from the end of the first step at FiO2 = 0.21 was used for further analysis.
The difference between end-tidal PCO2 and PaCO2 (ΔCO2) was calculated.
Systemic parameters
Data on nonfasting blood glucose were included when available within 60 minutes of the DLCO measurement. Data on hemoglobin were included when measured on the same date when DLCO was measured.
Comorbidity
From patients’ case files, information on heart failure and ischemic heart disease was obtained.
Statistics
Demographic data were presented as median and interquartile range (non-normally distributed). A Kruskal–Wallis test was carried out to secure that data sets 1 and 2 were comparable. Further, to secure that SpO2 21 was a representative of the oxygenation a Kruskal–Wallis test was carried out between PaO2 and SpO2 21. Kruskal–Wallis test was also used to investigate possible differences in DLCO in patients with and without cardiac comorbidity.
Spearman’s correlation analyses were performed to investigate: (1) the association between SpO2 21 and SpO2 15; (2) the association between DLCO% versus SpO2 21, SpO2 15, and ΔCO2; (3) the association between DLCO% and GOLD score; and (4) the association between DLCO and systemic parameters: BMI, blood glucose, hemoglobin, and smoking status.
Multiple regression analyses were performed to investigate any interdependencies between variables and to evaluate the association between (1) DLCO% versus SpO2 21, SpO2 15, and ΔCO2 and (2) DLCO% versus GOLD score, mMRC, and systemic parameters: BMI, blood glucose, and hemoglobin. Variables were chosen by forward selection.
Analysis was performed using STATA 12 ® , and p values <0.05 were considered statistically significant.
Results
Demographics
Background information on the study population is presented in Table 1. There was no statistical difference between the parameters from data set 1 and data set 2.
Demographic data on the study population including age, gender, smoking status, pack years; lung function parameters, FEV1, FEV1 in percent of expected value (FEV1%), and DLCO%; BMI and paraclinical data on hemoglobin and glucose; data on blood gasses, PCO2−endtidal CO2 (ΔCO2) SpO2 at FiO2 = 0.21 (SpO2 21), and PaO2 from each of the two data sets and in the total study population.a

mMRC: modified Medical Research Council; GOLD: Global Initiative for Chronic Obstructive Lung Disease; CO2: Carbon dioxide; FEV1: forced expiratory volume in the first second; DLCO: diffusing capacity of the lung for carbon monoxide in percent of expected value; BMI: body mass index; ΔCO2: difference between arterial and end-tidal CO2; PaO2: partial pressure of oxygen.
aData are presented in percentages or medians and quartiles, and in the paraclinical data, ranges are included. Where N deviates from the total number in the data set and in the total study population, the observed number of cases is written in italic in the table.
b p > 0.3.
c p = 0.07.
Five patients had blood glucose above 8 mmol/L, and 5 patients had hemoglobin below 7 mmol/L.
Patients were classified according to the GOLD scores demonstrated in Figure 1.

The distribution (in total numbers) of the participants according to the GOLD score. GOLD: Global Initiative for Chronic Obstructive Lung Disease.
Twenty-six percent (13 of 50) of the patients had concomitant heart disease, 38% (5 of 13) of these suffered from ischemic heart disease, and the remaining heart failure.
DLCO and oxygenation
There was no significant difference in the oxygenation expressed in SaO2 and SpO2,21 respectively (p = 0.4). Figure 2 demonstrates relations between SpO2 21 and SpO2 15 in the individual patients. There was no significant correlation between these (R 2 = −0.2, p = 0.3).

Peripheral oxygen saturation for individual patients at the initial level of FiO2 (upward triangle, FiO2 = 0.21) and at the lowest level (downward triangle, FiO2 = 0.15). Patients are sorted according to saturation at the initial level in the descending order. The vertical gray lines connect pairs of measurement in the individual patients. There was no correlation between saturation at FiO2 = 0.21 and FiO2 = 0.15, p = 0.3.FiO2: inspired oxygen.
There was a significant association between DLCO% and SpO2 21 (R 2 = 0.2, p = 0.01) and between DLCO% and ΔCO2 (R 2 = 0.13, p = 0.04), but no significant association between DLCO% and SpO2 15 (R 2 = 0.1, p = 0.09).
DLCO and GOLD score
A significant association was found between DLCO% and the GOLD stratification score (R 2 = 0.4, p < 0.001), depicted in Figure 3, and a very minor but significant association between DLCO% and mMRC score (R 2 = 0.09, p = 0.02).

Plot of correlation between DLCO (percent of expected value) and the GOLD score for COPD. Open circles demonstrates median values, bars the 95% confidence intervals (R 2 = 0.4, p < 0.001). DLCO: diffusing capacity of the lungs for carbon monoxide; GOLD: Global Initiative for Chronic Obstructive Lung Disease; COPD: chronic obstructive pulmonary disease.
DLCO and systemic parameters
Blood glucose was measured a median of 10 minutes after the DLCO measurement (range 40 to 60 minutes). Hemoglobin was also measured a median of 10 minutes after the DLCO measurement (range 150 to 60 minutes).
No significant association was between DLCO% and BMI (R 2 = 0.04, p = 0.2), blood glucose (R 2 = 0.04, p = 0.2), hemoglobin (R 2 = 0.005, p = 0.6), and smoking status (R 2 = 0.002, p = 0.9).
DLCO and comorbidities
There was no significant difference in DLCO% in patients with and without cardiac comorbidity (p = 0.4).
Multiple regression analyses
The association found between DLCO% and the GOLD stratification score was confirmed when controlling for BMI, hemoglobin, and glucose (R 2 = 0.6, p < 0.0001) with the multiple regression equation describing the influence of the different components on the diffusion capacity being:
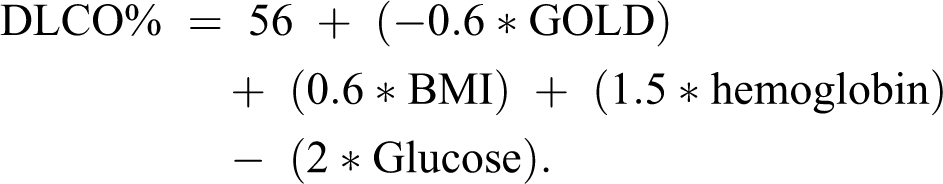
Multiple regression analysis showed an association between DLCO%, SpO2 21, SpO2 15, and ΔCO2 (R 2 = 0.3, p = 0.03) with the regression equation describing the influences of the different components as follows:

Discussion
This study indicates that in COPD patients at rest, DLCO% is significantly associated with SpO2 21 and ΔCO2 when breathing room air. No association was shown between DLCO% and SpO2,15 induced by deoxygenation. Furthermore, there was an association between DLCO% and GOLD score, which was stronger than the association between DLCO% and the respiratory parameters. This could indicate that DLCO is more descriptive of systemic deconditioning than gas exchange status in COPD patients.
The association of DLCO with the multifactorial GOLD score indicates the potential use of the method as part of the assessment of the general health status of the patient with COPD. Several studies have described COPD as a multifactorial systemic disease, 26 –28 with this recognition leading to the latest revision of the GOLD guidelines in 2011. 1 This study is, to these authors’ knowledge, the first to investigate a possible association between DLCO and the GOLD combined assessment score. The results indicate that DLCO is closely associated with the GOLD score. Even in patients with mild COPD, a modest reduction in DLCO% was seen. This may indicate that DLCO, as the GOLD score, describes systemic deconditioning.
The results are consistent with previously reported findings, which indicate that DLCO is sensitive to systemic changes. An increase in BMI or lean BMI over time leads to an increase in DLCO. 9 –12 Hyperglycemia in diabetics, both acute 13 and long-term poor glycemic control, 14 has been shown to decrease DLCO. In the data presented here, no linear association between DLCO% and BMI or between DLCO% and blood glucose was shown; however, both contributed significantly to the association between DLCO% and GOLD in the multiple regression analysis. This finding can most likely be explained due to BMI, which in this cohort was either normal (<25) or slightly overweight (<30) ranges. As such, neither obese nor underweight patients participated in the study. A similar pattern is existing for the blood glucose levels; in the cohort, the majority of blood glucose values were within the normal range, and only five patients had blood glucose values above 8 mmol/L. No association was found between DLCO and smoking status in this study. This is consistent with a previous study by Frans et al., investigating which parameters should be considered as reference values in DLCO measurements. 29
High-resolution CT scans were available in part of the patients, demonstrating both emphysema and airways disease and as such the diversity in the study population. However, the limited number did not allow inclusion of these data in statistical analysis. Nevertheless, the heterogeneity allows the assumption that different combinations of V/Q mismatch are represented in the study population. 4 In the present study DLCO% was, although significantly, only moderately associated with SpO2,21 and no association was found for SpO2.15 Furthermore, a weaker association was seen between DLCO% and ΔCO2. The difference in arterial and end-tidal CO2 has previously been shown to express the high V/Q component of V/Q mismatch in COPD patients. 21,30 It has previously been shown that even in patients with mild COPD, with limited airflow obstruction and no DLCO impairment, an extensively abnormal V/Q ratio can be found. 6 This poses the question what does DLCO really measure in COPD?
It is interesting to speculate whether DLCO might provide a good description of systemic deconditioning and a poor description of pulmonary gas exchange. The latter may seem controversial, given that DLCO is measured by inhalation. While DLCO is usually considered a diffusion measure, it is well understood that V/Q mismatch is the primary problem associated with gas exchange in COPD. 4 As such, if DLCO should provide an adequate representation of V/Q abnormalities, it would need to describe changes in ventilation and perfusion adequately. As the standard method for measuring DLCO involves breath holding at maximal inspiration, the amount of CO diffused is more likely to reflect maximal inspiratory volume than the individual’s normal ventilation. Indeed, it could be speculated that maximal inspiration may increase gas volume in all regions of the lung, which would potentially normalize low V/Q regions on the one hand and increase gas exchange abnormality in regions with high V/Q on the other.
It has previously been reported that the amount of CO diffusion may be highly affected by changes in perfusion and blood hemoglobin concentration. 6,17 Changes in hemoglobin and circulation are seen in other systemic diseases. 31,32 Recent studies indicate that this is also the case in COPD. 33,34 As such, it is perhaps not surprising that DLCO seems to reflect systematic changes well, without any association with O2 and CO2 levels.
The study has a number of limitations. As it was a retrospective study, only sparse information on concomitant systemic diseases was available. However, as DLCO has previously been associated with cardiac disease, 35,36 available data on cardiac comorbidity were included. No association was found. Results should be interpreted with care as data are provided retrospectively from the case files. Furthermore, values of blood glucose, BMI, and hemoglobin were primarily around normal ranges, and in some patients, values of blood glucose and ΔCO2 were not available. This may influence the generalizability of the model. Furthermore, (as 6 minute walking test would enable calculation of body mass index) to write the Body mass index; airflow obstruction; Dyspnea and Exercise Index (BODE-Index) which could have contributed to the characterization of the study population. Moreover, in a larger study population, it would have been interesting to investigate the association between DLCO and oxygenation in patients in GOLD strata A–D, to consider whether there is an association dependent on disease severity. In the same line, methodologically there is a risk of overfitting when applying multivariate regression analysis to a data set of this size. To avoid this, a limited number of variables, chosen by forward solution, were included in the analysis. As such, a larger prospective study is therefore needed to support the results shown here.
In conclusion, this study indicates that even though an association is seen between DLCO%, and SpO2,21 and ΔCO2 in COPD patients at rest, a stronger association was seen between DLCO% and GOLD score. This suggests that DLCO is more descriptive of systemic deconditioning than gas exchange status in COPD patients at rest. However, further study is required to explore this association in larger COPD cohorts.
Footnotes
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Professor Stephen Rees is a board member of Mermaid Care, the manufacturer of the ALPE system. None of the other authors have any conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
