Abstract
Obstructive sleep apnoea (OSA) may increase the risk of hyperuricaemia and predispose to gout. The evidence for the effects of OSA on serum urate in severe obesity is limited. This study investigated whether OSA was associated with serum urate in severe obesity and whether continuous positive airway pressure (CPAP) treatment was associated with a fall in urate. Severely obese subjects without known OSA or gout were recruited. Baseline assessments included urate, metabolic parameters, spirometry and overnight polysomnography. OSA patients were initially naive to treatment and were offered CPAP. At follow-up, change in urate was compared between CPAP-treated and non-CPAP-treated subjects. A high urate was defined as greater than the median. Logistic regression was performed to identify associations between (1) OSA and high urate at baseline and (2) use of CPAP and change in urate at follow-up. In total, 92 subjects were recruited (61 (66%) OSA and 31 (34%) non-OSA). Median urate was 345 μmol/L. OSA was associated with high urate in females at baseline after adjusting for confounders (adjusted odds ratio ORadj = 10.2; 95% CI: 1.1, 93.5). At follow-up (14 months), 58 subjects (28 on CPAP and 30 not on CPAP) were reassessed. CPAP was significantly associated with a fall to a low urate category at follow-up ( = 0.017). Regression revealed a trend for a fall in urate category in the CPAP-treated group (ORadj = 9.3; 95% CI: 0.8, 97). Serum urate is associated with OSA in severely obese females and CPAP may reduce levels in patients with OSA. There may be a need to consider and assess for OSA in obese patients with hyperuricaemia and recurrent attacks of gout.
Introduction
Obstructive sleep apnoea (OSA) is a common condition associated with metabolic dysregulation and cardiovascular disease. It is characterized by repeated upper airway occlusion during sleep, leading to arterial oxygen desaturations resulting in repeated arousals from sleep. Patients typically report symptoms of excessive somnolence, poor cognition and unrefreshed sleep.
The prevalence of OSA has been increasing and estimates indicate 13% of men and 6% of women between the ages of 30 years and 70 years have moderate to severe forms of OSA. 1 It is well established that obesity is a major risk factor for OSA, and the prevalence of OSA in severe obesity has been estimated to be between 40% and 90%. 2
OSA is diagnosed by overnight polysomnography (PSG), and the apnoea–hypopnoea index (AHI) is used to assess the severity of OSA. In addition to the AHI, the frequency of oxygen desaturation episodes and severity of somnolence symptoms are also used. 3 There is effective treatment for OSA in the form of continuous positive airway pressure (CPAP).
Epidemiological studies have suggested an association between gout and OSA. 4,5 Urate levels may be influenced by cell apoptosis secondary to apnoea-induced episodes of hypoxia. 6 Furthermore, increased adenosine triphosphate degradation in recurrent hypoxia may increase uric acid levels. 7 Given that these events occur intermittently throughout the sleep cycle, urate levels may fluctuate during sleep and may contribute to an increased risk of gout attacks. This may explain the increased predilection for the nocturnal onset of many gout attacks.
Elevated levels of serum urate have been associated with OSA and increased cardiovascular risk. 8 This may reflect shared risk factors of increased body weight associated with gout and OSA. 9 Although several studies have shown that urate levels are raised in OSA, most studies were cross-sectional in design and did not control for obesity or gender that may confound this relationship. 10,11 Additionally, the association between urate and OSA has not been investigated in patients with severe obesity (body mass index (BMI) >35 kg/m2). Therefore, we aimed to explore, in a severely obese population, whether the presence of OSA was associated with urate. In addition, we were keen to identify whether the use of CPAP treatment in OSA was associated with a fall in the serum urate.
Methods
Ethics statement
Research approval was granted by the local research ethics committee (NRES 12/NW/123). The research was performed in accordance with the Declaration of Helsinki 2008. All patients gave written informed consent.
Participants
Subjects were recruited from weight management and sleep clinics at University Hospital Aintree from March 2012 to January 2013 and follow-up visits were from September 2013 to February 2014.
Patients were eligible if they were ≥21 years old and had BMI ≥ 35kg/m2. Exclusion criteria were patients who were being treated or had prior treatment for OSA, those with known cardiorespiratory disease, current smokers or those with more than 10 pack years smoking history, kidney and liver disease and acute illnesses and pregnancy. No patients had a history of gout or were on urate-lowering medications. At follow-up assessment, subjects with OSA who were non-compliant with CPAP (usage <4 hours/night) and those who had bariatric surgery were excluded from the study.
Protocol
At baseline, subjects attended between 08:00 and 10:00 hours and underwent a detailed history and physical examination. Body composition measurements, anthropometry, serum urate and biochemical tests were performed. An overnight sleep study was performed and patients were then grouped according to their sleep status.
Blood pressure
Blood pressure was measured at the arm in the sitting position after resting for at least 5 minutes at 1 minute intervals between each measurement using an oscillometric digital blood pressure monitor (HEM-705CP, Omron, Japan). The mean of three measurements was calculated.
Body measurements
All measurements were done in triplicate. Weight and height were measured without shoes and with light clothing. Other measurements included neck circumference at the level of the laryngeal prominence, waist circumference midway between the lower rib and iliac crest and hip circumference was measured horizontally over the widest part of the gluteal region. The tape measure was ensured to be snug and not compressing the skin, parallel to the floor with measurement at the end of a normal expiration.
Spirometry assessment
Spirometry was performed at baseline with a Spiro Air LT system (Medisoft, Sorinnes, Belgium) supervised by an experienced technician. This was performed to assess for coexisting lung pathology.
Sleep diagnostic assessment
Diagnosis was confirmed by overnight multichannel respiratory limited PSG (Somnoscreen Digital PSG acquisition system, version 2.0, SomnoMedics, Germany). Sleep studies were independently assessed by experienced sleep physiologists using software (Domino PSG analysis software version 2.5.0; SomnoMedics). Apnoea was defined as a cessation of airflow for >10 seconds. Hypopnoea was defined as a 50% reduction in airflow accompanied by a >4% desaturation and a reduction in chest wall movement. OSA was diagnosed if the AHI was ≥5. The oxygen desaturation index, which is a measure of the hourly average number of desaturation episodes during sleep, was also recorded for each patient.
Biochemical measurements
Biochemical tests including serum urate were measured using standard laboratory assays (Roche, UK). Blood gases were analysed with a Cobas blood gas analyser (Roche).
Assessment of urate
The category ‘high urate’ was defined as a value above the baseline median urate measure. This was necessary, given that all subjects did not have a history of gout, and it was expected that their urate levels were unlikely to exceed the upper reference range.
To compare the change in urate with treatment, a binomial variable of ‘change in urate’ category was created. This represented a change from a high urate category at baseline to a low urate category at follow-up.
Metabolic syndrome
Subjects were assessed for metabolic syndrome according to the National Cholesterol Education Program guidelines. 12 Patients had metabolic syndrome if three or more risk factors were present: waist circumference (males >102 cm; females >88 cm), triglycerides ≥1.7 mmol/L, high-density lipoprotein cholesterol (males <1.04 mmol/L; females <1.3 mmol/L), blood pressure ≥130/≥85 mmHg and fasting glucose ≥6.1 mmol/L.
CPAP therapy
The decision to initiate CPAP was based on a positive sleep study, OSA with evidence of daytime sleepiness and Epworth sleepiness questionnaire scores. OSA patients received standard therapy with S8/S9 Escape machines (ResMed, Abingdon, UK). CPAP compliance was based upon usage in hours per night at the prescribed pressure. Compliance data were recorded by the CPAP machines and were downloaded using ResScan software (version 4.2, ResMed). This was assessed at each patient’s most recent routine CPAP compliance clinic. Adequate compliance was defined as the usage time of >4 hours/night on >70% of nights in the treatment group. 13
Follow-up
Patients were reassessed after 12–14 months with repeat urate measurement. Change in urate levels compared to baseline measures was recorded. Study participants with OSA who were compliant with CPAP treatment (CPAP-treated group) were compared to non-OSA participants and OSA participants who chose not to undergo CPAP treatment (non-CPAP group).
Statistical analysis
Statistical analyses were performed using Stata 13 (StataCorp LP, College Station, Texas, USA). Normal distribution of data was confirmed using the Shapiro–Wilks test. Patient demographics were summarized by OSA group as means (SD) (or median (interquartile range [IQR] if non-normal) for continuous variables and frequencies (%) for categorical variables. Comparisons of demographics between groups were performed using a t-test for continuous variables that were normally distributed and a Mann–Whitney test for those that were not. Categorical variables were tested using a χ 2 or Fisher’s exact test depending on the frequencies.
Univariate analysis was performed and variables with significance level <0.1 were entered into the multivariate model. The candidate variables that were included in the model selection were based on parameters that were identified as potentially having a confounding influence on OSA and those that were associated with an increased risk of gout. The final multivariate models were generated using negative stepwise logistic regression.
Multivariate logistic regression analysis was performed at baseline exploring associations between high urate and OSA adjusting for age, previous smoking status, highly sensitive C-reactive protein (hsCRP) and BMI. As urate levels are higher in males, gender-stratified analysis was conducted.
In addition, multivariate logistic models were generated to explore whether CPAP was associated with a fall in urate category.
Results
Ninety-two consecutive patients with severe obesity were recruited at baseline (61 with OSA (mild OSA n = 25, moderate OSA n = 18 and severe OSA n = 18) and 31 without OSA; Figure 1). All subjects were of White European ethnicity. Baseline descriptors are summarized in Table 1 and in Supplementary Table 1. The ODI and AHI were significantly different between groups. The median urate for all 92 subjects was 345 μmol/L (IQR: 288–414). At baseline, the presence of OSA was significantly associated with high urate (35 (57%) of the OSA group had a high urate compared with 11 (35%) in non-OSA group). However, when urate was analysed as a continuous variable, no significant association was observed with OSA. In addition, subjects with OSA had significantly increased systolic blood pressure (BP), hsCRP and metabolic syndrome compared with the non-OSA group. BMI was higher in the OSA group but did not reach significance. Therefore, this covariate was adjusted for in the multivariate analysis.

Study profiles at baseline and follow-up. OSA: subjects with obstructive sleep apnoea; non-OSA: subjects without obstructive sleep apnoea; CPAP: continuous positive airway pressure treatment.
Patient demographics at baseline are summarized by OSA prior to CPAP versus non-OSA groups.a

AHI: apnoea–hypopnoea index; BMI: body mass index; BP: blood pressure; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; hsCRP: highly sensitive C-reactive protein; MDRD-GFR: glomerular filtration rate using modification of diet in renal disease equation; ODI: oxygen desaturation index; OSA: obstructive sleep apnoea; non-OSA: subjects without obstructive sleep apnoea; IQR: interquartile range; SD: standard deviation.
aValues are expressed as means (SD) (or median [IQR] if non-normal) for continuous variables and frequencies (%) for categorical variables.Significant p values are highlighted in boldface. A high urate was defined as a urate greater than the median urate (345 μmol/L). Metabolic syndrome was assessed according to National Cholesterol Education programme guidelines.
Gender stratification revealed that more females with OSA had a high urate level (13 (42%)) compared with females without OSA (1(7%; p = 0.015; Table 1). Systolic BP in females with OSA (p = 0.025) and hsCRP in males with OSA (p = 0.047) were significantly different compared with the controls. Metabolic syndrome was significantly increased in male (p = 0.012) and female (p = 0.001) OSA subjects compared with non-OSA subjects.
Logistic regression showed a significant association between OSA and high urate (OR = 10.1; 95% CI: 1.2, 86.8) only in female participants (Table 2). This remained significant after adjusting for confounding variables age, previous smoking status, hsCRP and BMI (ORadj = 10.2; 95% CI: 1.1, 93.5). However, this association was not significant in males (ORadj = 0.8; 95% CI: 0.1, 4.1) and when both genders were combined (ORadj = 2.3; 95% CI: 0.8, 6.7; Table 2).
Baseline regression analysis – association between OSA and high urate.a
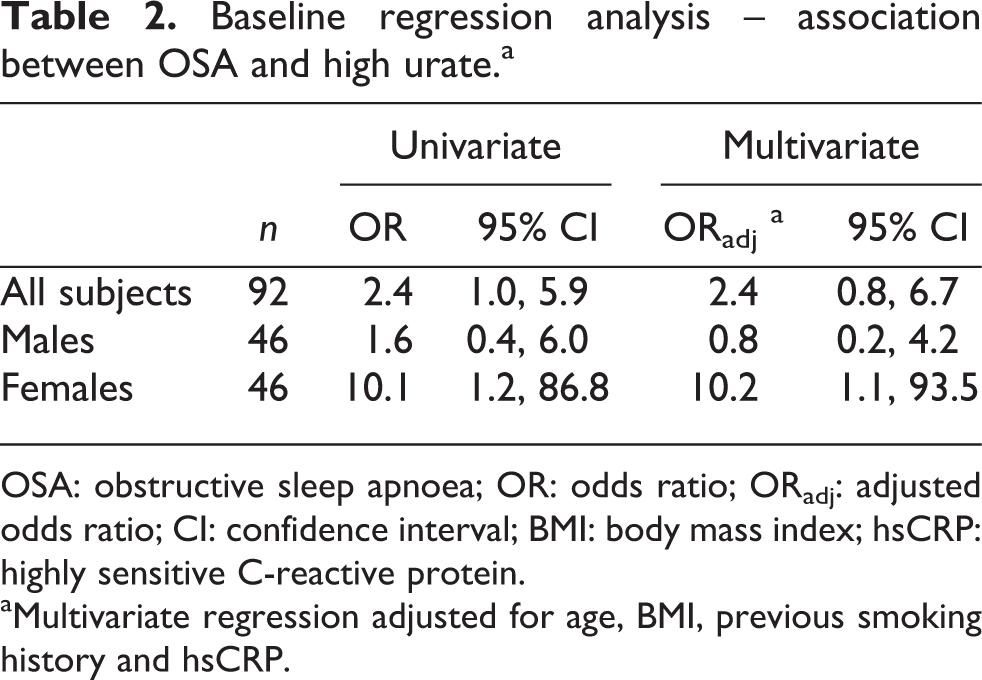
OSA: obstructive sleep apnoea; OR: odds ratio; ORadj: adjusted odds ratio; CI: confidence interval; BMI: body mass index; hsCRP: highly sensitive C-reactive protein.
aMultivariate regression adjusted for age, BMI, previous smoking history and hsCRP.
The median follow-up period was 14 months (IQR = 13–15). At follow-up, 58 subjects were reassessed (28 on CPAP (mild OSA n = 6, moderate OSA n = 12 and severe OSA n = 10) and 30 not on CPAP). The characteristics of the follow-up cohort are presented in Table 3 and in Supplementary Table 2. Subjects were grouped into those who received CPAP and those who were not treated with CPAP. Mean CPAP use was 14 months (SD 1.5). Mean CPAP duration per night was 4.7 hours/night (SD 0.6). Mean CPAP pressures were 10 cm H2O (SD 1.0). Metabolic syndrome (p = 0.036) was more prevalent in CPAP users.
Patient demographics at follow-up were summarized by non-CPAP versus CPAP groups.a

BMI: body mass index; BP: blood pressure; hsCRP: highly sensitive C-reactive protein; MDRD-GFR: glomerular filtration rate using modification of diet in renal disease equation; CPAP: continuous positive airway pressure; pCO2: partial pressure of carbon dioxide; IQR: interquartile range; SD: standard deviation.
aValues are expressed as means (SD) (or median [IQR] if non-normal) for continuous variables and frequencies (%) for categorical variables. Significant p values are highlighted in boldface. A high urate was defined as a urate greater than the median urate (345 μmol/L). Metabolic syndrome was assessed according to National Cholesterol Education programme guidelines.
At follow-up, CPAP-treated patients had similar mean urate (mean 345 μmol/L (SD 61) to non-CPAP users (mean 342 μmol/L (SD 81). However, in CPAP-treated subjects, 7 (25%) had a change from a high urate at baseline to a low urate level and this was significant compared with 1 (3%) for non-CPAP subjects (p = 0.017; Table 3).
For CPAP-treated subjects, the mean urate significantly fell between baseline 357 μmol/L (95% CI: 339, 392) and follow-up 345 μmol/L (95% CI: 321, 368; p = 0.016). This was non-significant in non-CPAP subjects (baseline 340 μmol/L (95% CI: 307, 376) and follow-up 342 μmol/L (95% CI: 311, 372; p = 0.980). In the CPAP-treated group, there was no correlation between change in urate and hours of CPAP use per night (r = 0.035, p = 0.860) and between change in urate and follow-up duration (r = 0.012, p = 0.952). Regression analyses stratified by gender did not demonstrate significant differences for change in urate.
Using the category of high urate (above 345 μmol/L). Seven subjects (25%) of CPAP users had urate levels that fell from a high to a low category; 20 (72%) CPAP-treated subjects remained in the same category; and 1 (3%) subject had a change in urate from a low to a high category. Effective CPAP was significantly associated with a fall from high urate into a low urate category at follow-up (p = 0.017). Multivariate logistic regression analysis revealed a trend for CPAP use with the fall in urate category, but the result did not reach significance (ORadj = 9.3; 95% CI: 0.8, 97).
Discussion
We have found evidence of an association between OSA and urate in severely obese females. We also observed a trend towards a fall in urate levels in OSA patients treated with CPAP. The evidence from the current study may suggest an explanation for the onset of nocturnal gout flares in some patients. It may also be argued that the treatment of OSA with CPAP may potentially influence urate responses to gout treatment for patients with hyperuricaemia and OSA. Thus there may be a need to consider OSA in severely obese subjects who have hyperuricaemia or recurrent gout, as there may be a possible role for CPAP in influencing urate levels.
Our findings are in agreement with the previous evidence of an association between the severity of OSA and increased urate levels. 14,15 However, at baseline, we did not observe a significant association between urate and OSA in male participants. This may be explained by the lower BMI in this group compared with the female subjects, and it is conceivable that a difference in adiposity may explain the disparity in findings in the analysis stratified by gender, given that obesity is linked with uric acid levels. 10
At follow-up, the mean CPAP use was 4.7 hours/night, which is comparable to a previous study with compliance of 4.6 hours/night. 16 We observed a significant fall in mean urate in OSA patients at follow-up with CPAP therapy compared with baseline and a trend for a fall in urate category with effective CPAP. However, multivariate regression analysis did not show a significant association. A randomized controlled trial (RCT) in males with type 2 diabetes and OSA did not demonstrate a significant change in serum urate with either 3 months of therapeutic or placebo CPAP. 17 Conversely, two studies have previously reported reduced levels of urate following CPAP treatment. 18,16 However, these studies were not placebo controlled.
There were differences in several characteristics between the groups at baseline and between the groups at follow-up. At baseline, OSA patients had higher systolic BP and hsCRP and many had metabolic syndrome compared with the non-OSA group. At follow-up, more patients on CPAP had metabolic syndrome. Previous work in our group has demonstrated an association between OSA and metabolic syndrome in an RCT. 19 There is also evidence that OSA is associated with hypertension. 20 It has been suggested that hyperuricaemia is associated with metabolic syndrome. 8 Thus it is conceivable that there may be an interaction between these factors that may potentiate changes in serum urate. It is unclear whether OSA is independently associated with increased CRP, as it is known that obesity induces chronic low-grade inflammation and may be associated with CRP. 21
In the CPAP-treated group, there was no correlation between change in urate and hours of CPAP use per night. Although the link between CPAP treatment and decrease in serum urate may appear less likely, it is possible that this may be a true reflection of the trend with significance not being apparent due to a larger effect size.
The median BMI in the CPAP-treated group was lower at follow-up compared to baseline. It should be noted that the majority of patients recruited were attending a multidisciplinary weight management service during this time and would have received weight management care during their CPAP treatment, and it is possible that weight loss may have occurred during the intervening time between baseline and follow-up. However, a proportion of the patients were also recruited from the sleep service who were not attending weight management clinic. It remains plausible that combination treatment with weight loss and CPAP may have potential effects on the change in urate levels.
It has been postulated that intermittent hypoxia in OSA leads to increased adenosine triphosphate degradation and associated purine catabolism that may lead to increased levels of urate. 7,22 Additionally, it has been suggested that elevated oxidative stress in OSA may be linked with uric acid production, and there is evidence implicating serum urate as an oxidative stress marker. 23,24 It is known that OSA is associated with cardiovascular disease and hypertension through mechanisms including intermittent hypoxia, leading to oxidative stress and inflammation. 20 Given our findings of an association between serum urate and OSA in females with severe obesity, there may be a possible role of uric acid in cardiovascular risk. 8
There are several limitations in this study. Without a placebo CPAP group, it was not possible to categorically make inferences on cause–effect and to demonstrate that our observations at follow-up were indeed CPAP effects. However, it would have been ethically questionable to conduct a placebo-controlled randomized trial over such a prolonged duration (>1 year) as in our study, and our aim was to investigate the changes in real-life settings. We did not deliberately attempt to match patients in the groups as this would have restricted eligible patients who were willing to participate, potentially introducing a selection bias, and pragmatically, it was important to try to recruit as many patients as possible for both groups.
In this study, urate was measured early morning and not at repeated times throughout the night. Therefore, this may potentially reflect a random urate level. It is possible that the time of sampling may not directly reflect the fluctuations in urate during nocturnal hypoxic episodes. Although multiple sampling of urate at the time of sleep apnoea episodes may have provided further precise information, this would have made sleep difficult and therefore a pragmatic approach for blood sampling was adopted.
The respiratory multichannel studies that were used in this study were in line with local protocols and current practice. Electroencephalographic activity was not assessed, however, and it may have been possible for the AHI to have been underestimated as this measurement would have enabled the detection of respiratory arousals.
Although we found a trend that approached significance for an association between CPAP and fall in urate, this may have been attributable to the small patient numbers at follow-up. Thus further studies are needed to investigate these observations with larger sample sizes. It is also conceivable that a combination of several modalities of treatment may be needed for patients with hyperuricaemia including lifestyle and weight loss, where appropriate, in combination with CPAP, may have a synergistic effect on serum urate levels in severely obese populations or patients with recurrent gout. These will also needto be explored in future work.
Conclusion
To conclude, serum urate levels are associated with OSA in severely obese females, and CPAP treatment may influence serum urate. Further study is necessary to investigate the effects of a combination of several modalities of treatment including lifestyle and weight loss, where appropriate, in combination with CPAP on serum urate levels in severely obese cohorts and also to ascertain whether the duration of CPAP therapy has a role in influencing urate outcomes. Intervention to treat nocturnal hypoxia in OSA may be a consideration for patients with hyperuricaemia and recurrent attacks of gout.
Footnotes
Acknowledgements
We would like to thank Dr Stephen Embego and Ms Tessa McKown (Sleep Lab), Mr Stuart Wragg (Lung function Lab) and Mr Peter Thompson (Biochemistry Lab).
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was funded by the University of Liverpool, University Hospital Aintree and St Helens and Knowsley Teaching Hospitals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
