Abstract
The aim of pharmacological therapy for asthma is to improve symptoms and lung function and minimize the risk of asthma attacks. The intensity of treatment is based on the level of asthma control and the potential risk of future deterioration. In the British asthma guidelines, treatments are divided into steps 1 to 5, with each step signifying a need for an increase in therapy in response to symptoms or to prevent exacerbations. Treatments comprise of inhaled or systemic medications. Inhaled therapy includes short-acting and long-acting medication to improve symptoms and inhaled corticosteroids that reduce airway inflammation. Systemic treatments include medications that act on specific biological pathways, such as the leukotriene or immunoglobulin E pathways, or systemic corticosteroids. In choosing a particular therapy, treatment benefits are balanced by the potential risks of medication-related adverse effects. This review will provide a practical guide to the key pharmacological therapies for adult asthma at steps 1 to 5 based on British guidelines and consider future options for new treatments.
Introduction
Pharmacological therapy is a key component of the management of people with asthma. 1 Current established treatments can be classified into two groups: medication taken via the inhaled route and treatment taken via the systemic route (e.g. oral, intravenous, subcutaneous or intramuscular; Table 1). 2 –4 Inhaled therapy includes short-acting β-agonists (SABA), which are used to provide short-term improvement in symptoms, and inhaled corticosteroids (ICS), which suppress airway inflammation. In addition, inhaled long-acting β-agonists (LABA) are symptom-controller medications that are used in combination with ICS. Systemic therapy for asthma includes systemic corticosteroids, 5 leukotriene receptor antagonists and more recently, ‘targeted’ therapy towards specific underlying biological processes, such as anti-IgE (immunoglobulin E), anti-IL-5(interleukin-5) and anti-IL-13 (Table 2).
Currently approved pharmacological therapy for adult asthma.

SABA: short-acting β-agonists; ICS: inhaled corticosteroids; LABA: long-acting β-agonist; IgE, immunoglobulin E; BDP: beclometasone dipropionate.
aTerbutaline is available in oral and parenteral preparations.
bLow-to-medium adult doses are: 200–800 μg/day of BDP or budesonide; 100–500 μg/day of fluticasone propionate; 400 μg/day of mometasone furoate. High adult doses are: >800–2000 μg/day of BDP or budesonide; >500–1000 μg/day of fluticasone propionate; >400–800 μg/day of mometasone furoate.
cOnly used in combination with ICS, preferably in a single combination ICS/LABA inhaler.
dLicensed for use with 100/6 μg or 200/6 μg budesonide/formoterol dry powder inhaler or 100/6 μg beclometasone/formoterol metered dose inhaler.
Emerging therapies for adult asthma.

ICS: inhaled corticosteroids; LABA: long-acting β-agonist; IgE, immunoglobulin E; IL: interleukin.
Treatment is generally tailored to the severity of underlying disease activity, usually measured by a combination of symptoms, lung function and assessment of the risk of adverse outcomes such as exacerbations, lung function decline or treatment-related adverse effects. 6 The intensity of the treatment, or treatment ‘step’, reflects the perceived level of current impairment or future risk of an adverse event such as a severe exacerbation or diminished lung function. People are prescribed medications that range from step 1 (‘low’ intensity treatment) to step 5 (‘high’ intensity treatment), with an increase or decrease in step based on reviewing the adherence and response to current therapy, generally over a period of 2–6 months (Figure 1). 4 There should, therefore, be a close relationship between the assessment of current asthma control, identification of the risk of future adverse outcomes and the prescribed pharmacological treatment step.
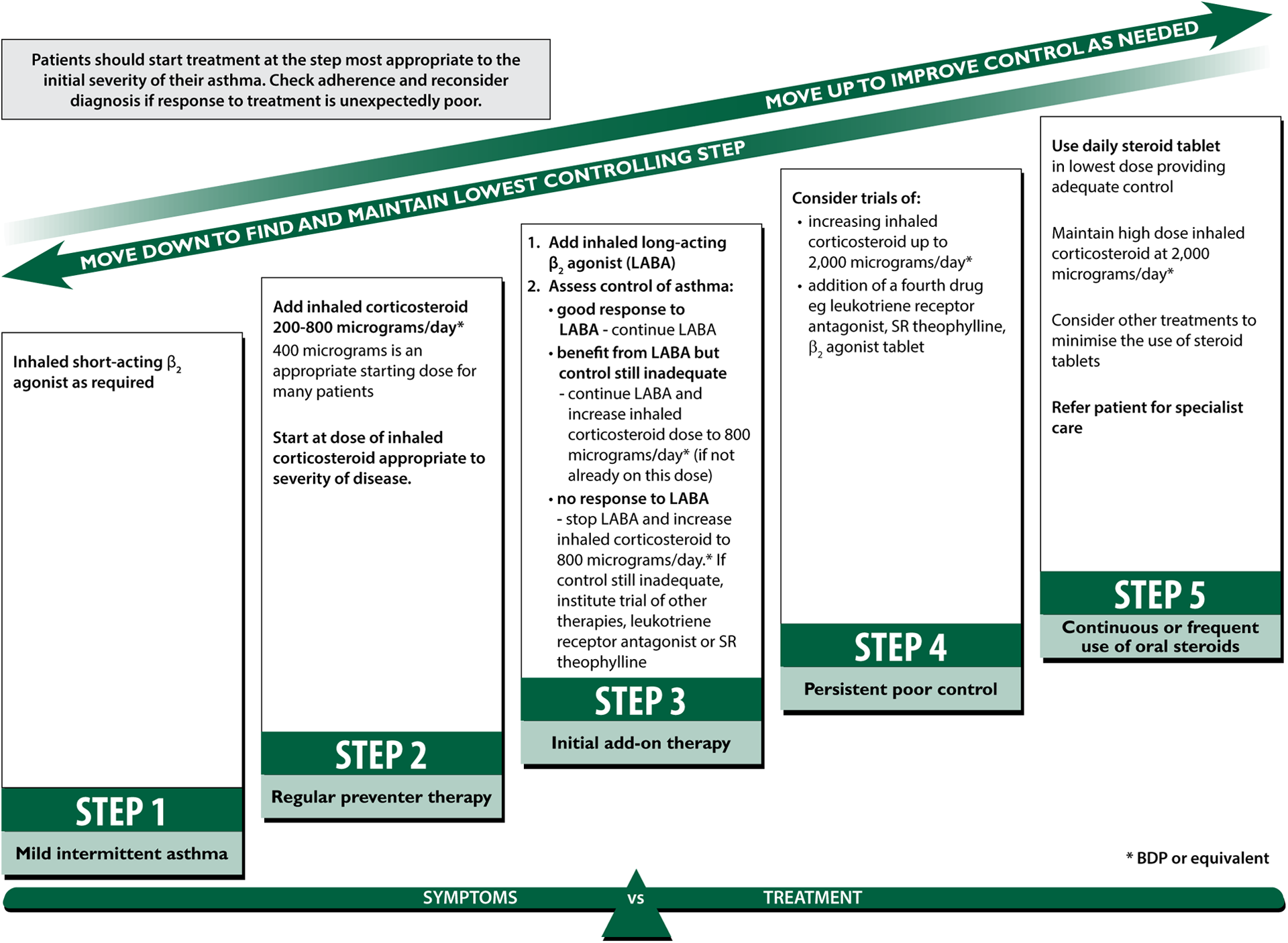
Treatment steps in adult asthma. Source: Reproduced from the 2014 BTS/SIGN British guideline on the management of asthma, by kind permission of the Scottish Intercollegiate Guidelines Network. 4
The aim of this review is to provide a concise summary of the key pharmacological therapy for adult asthma and to highlight emerging treatments that may be recommended in the future. We will base our review on British asthma guidelines, 4 supported by both the American 3 and international 2 asthma guidelines. We will focus on the practical use of these treatments in the clinical setting rather than on detailed pharmacological mechanisms and biological modes of action, which are discussed elsewhere. 7
Step 1: As-required SABA
Step 1 therapy is generally recommended for people with very mild ‘intermittent’ asthma. Guidelines suggest that these people are characterized by intermittent, mild symptoms, infrequent nocturnal awakening, normal lung function and a low risk of future adverse events. 2 –4 Common triggers for symptoms are exercise, allergens or infections. 8
People are offered symptomatic treatment in the form of inhaled SABAs, such as salbutamol or terbutaline. These drugs have a rapid onset but short duration of action, and act as bronchodilators. As-required SABA treatment is comparable to regular SABA treatment but with lower drug exposure and is generally the preferred option. 9
Future directions
The commencement of step 1 therapy relies on establishing a diagnosis of probable asthma together with an assessment of an adequate level of current control and low risk of future exacerbations. In clinical practice, barriers exist to all of these processes. The majority of people with mild intermittent asthma are seen in primary care, where access to diagnostic aids, such as tests of atopy, airway inflammation and bronchial hyper-responsiveness, may be poorly, if at all available. There is also variable use of available tools, such as questionnaires used to assess asthma control 10 and lung function testing. In addition, it remains difficult to predict which individuals are at risk of future poor control or exacerbations. 11 This can cause uncertainty in selecting those people who could benefit from early commencement of regular preventer therapy.
Future work is likely to focus on more comprehensive initial evaluation of people presenting with possible asthma in order to confirm the diagnosis and help define individual characteristics that can help to predict an increased risk of future adverse outcomes. For example, early detection of uncontrolled eosinophilic airway inflammation could help to identify individuals with an increased risk of severe exacerbation. 12 This information will then help to inform initial treatment choices, such as the early use of ICS. Additionally, novel treatment strategies, such as the on-demand use of combination ICS/fast-onset LABA inhalers in response to asthma symptoms, 13 will provide further options for earlier ICS treatment in people with intermittent symptoms. The focus of therapy at step 1 will therefore move towards improved identification of individuals at risk of poor control and earlier exposure to ICS.
Step 2: Regular ICS treatment
In people with persistent symptoms, despite step 1 therapy (e.g. nocturnal wakening or regular need for SABA therapy), or in whom initial assessment suggests a greater risk of future adverse outcomes (e.g. prior severe exacerbation or impaired lung function), the standard approach is regular maintenance treatment with low-dose ICS and SABA for relief of breakthrough symptoms. 14 Corticosteroids act by inhibiting pro-inflammatory cellular processes as well as having topical effects that may reduce airway inflammation. 15,16 Treatment with ICS is associated with an improvement in symptoms and lung function 17 and a reduction in exacerbations 17,18 and risk of death. 19 ICS treatment is generally prescribed twice daily, and 400 μg/day of beclometasone dipropionate (BDP) (or equivalent) is a usual starting dose for mild-to-moderate asthma severity. 14 Most of the therapeutic benefit from ICS is attained at low-to-moderate doses, 20,21 though people with more severe disease (e.g. frequent exacerbations) may benefit from higher doses. 22 In corticosteroid-naive individuals with mild asthma, initial use of combination ICS/LABA therapy offers no advantages compared with ICS alone. 18,23
ICS is available in a range of formulations, with variability in pharmacological properties and drug delivery devices. 24 Common adverse effects include dysphonia and oropharyngeal candidiasis, and use of spacers together with advice on mouth rinsing after dosing may help reduce this effect. 3 Systemic side effects have been associated with ICS therapy, 25 usually with high-dose/long-term use, though the risk/benefit profile generally favours their use.
Alternative preventer therapy at step 2 includes the use of oral leukotriene receptor antagonists. These drugs have both bronchodilator and anti-inflammatory properties 26 and may offer an alternative for some people with mild asthma. 27 However, current recommendations are that ICS remain the preferred option for use as sole maintenance therapy. 28
Future directions
Poor adherence to maintenance in ICS therapy is a key factor contributing to the risk of future severe exacerbations. 29,30 Thus, people with asthma at step 2 may remain poorly controlled and ‘at risk’ because they perceive little symptomatic benefit day-to-day from their prescribed ICS therapy and so can underuse their maintenance inhaler and over-rely on their reliever SABA inhaler. Two strategies have been studied that aim to promote exposure to ICS, namely, intermittent ‘as-needed’ ICS treatment and as-needed combination ICS plus SABA treatment.
In adults, as-needed ICS used in response to worsening symptoms provided comparable improvements in morning peak expiratory flow rate compared with regular ICS therapy, at markedly reduced overall ICS exposure. 31 However, participants in the regular ICS group had significantly greater improvements in asthma symptoms, pre-bronchodilator forced expiratory volume in 1 second and airway inflammatory measures than the as-needed group. 31 Asthma quality of life scores and exacerbation rates were similar between these two groups. 31 These findings have led to differing opinions regarding the role of as-needed ICS therapy in mild asthma. 32,33 At present, there is insufficient evidence to recommend the routine use of the as-needed ICS regimen 34 and further study in real-world trials is required. 35
An alternative strategy uses symptom-driven as-required ICS/SABA (in a combination inhaler) 36 or ICS + SABA (taken via separate inhalers). 37 The advantage of this strategy is that people are exposed to ICS at the time of SABA use, thus providing both immediate bronchodilation and anti-inflammatory treatment during worsening asthma. These studies demonstrated that the symptom-driven approach provided similar improvements in asthma symptoms, lung function and exacerbations at reduced ICS exposure compared with regular ICS therapy. 36,37 However, combination ICS/SABA inhalers are currently not available in the United Kingdom and the use of separate ICS + SABA inhalers is likely to be impractical in the clinical setting.
Trials using the as-needed approach, but using combination ICS/fast-onset LABA inhalers, will provide further information on the utility of this approach using inhalers that are available to prescribe in the clinical setting. 38 Results from real-world trials, in particular, and utilizing electronic monitoring to describe patterns of medication use, 39 will help guide clinicians as to the risks versus benefits of this approach in adults with mild persistent asthma.
Step 3: Add-on treatments
International guidelines recommend the addition of a LABA to low-dose ICS as the preferred option for step 3 maintenance treatment, with SABA for relief of symptoms. 2,4 There are, however, a number of alternative strategies available at step 3.
A major asthma study showed that the combination of low-dose ICS (200 μg budesonide/day) and LABA (formoterol) significantly reduced severe exacerbations compared with low-dose ICS alone. 22 Low-dose ICS and LABA in combination were also superior to a fourfold higher dose of ICS (800 μg budesonide/day) in improving asthma symptoms. 22
However, in this study, higher dose budesonide treatment resulted in significantly greater reductions in asthma exacerbations compared with combination low-dose ICS and LABA treatment. 22 Therefore, an alternative option at step 3 is to increase the dose of ICS to 800 μg/day BDP (or equivalent) instead of using a LABA with low-dose ICS. 2,3 Although this is not the preferred strategy in the United Kingdom, 4 this option remains available in people with asthma who have no improvement following the initial add-in of LABA therapy.
Greatest improvements (reductions) in exacerbations in this study were achieved with the higher dose ICS and LABA in combination. 22 Individuals who are considered at higher risk for poor control and asthma exacerbations can benefit from the use of higher dose (up to 800 μg/day of BDP or equivalent) combination ICS/LABA therapy. 22,40
In clinical practice, use of a combination ICS/LABA inhaler is likely to encourage adherence to ICS 41,42 and avoid the risk of LABA monotherapy. 43 British guidelines recommend this practice as the preferred option when people are prescribed these two classes of drugs. 4 In recent years, there have been concerns raised regarding the safety of LABAs 44 and large-scale studies are underway to further investigate this issue. 45
Another option at step 3 is the use of a single combination ICS/fast-onset LABA inhaler as regular maintenance therapy, but with additional doses of the same inhaler also taken for relief of symptoms. 46 – 49 This treatment plan, called single combination ICS/LABA inhaler as maintenance and reliever therapy (SMART), has the advantage of providing individuals with a dose of ICS every time their inhaler is used for relief of symptoms. This has the dual effect of reducing non-adherence to ICS and allowing higher doses of ICS to be delivered at the time of worsening symptoms. 48 This regimen significantly reduces severe exacerbations of asthma by around 50% compared with the same fixed dose of maintenance ICS/LABA therapy with SABA for relief. 46 – 49 SMART can be considered the preferred option in adults at risk of future severe exacerbations. 2,50
An alternative to the addition of LABA is the addition of a leukotriene receptor antagonist to ICS treatment. A recent study has suggested that the add-on of leukotriene receptor antagonists may promote adherence to treatment compared with the addition of a LABA in a real-world setting. 27 However, the most recent Cochrane review suggests that LABAs provide greater advantages in terms of reducing severe exacerbations, 51 and so they remain the preferred add-on treatment at step 3. 4
Future directions
Future research at step 3 and above is likely to focus on the use of therapy guided by underlying disease biological processes. 52 –54 In this regard, the application of findings from cluster analyses can help to delineate phenotypic characteristics. 55,56 For instance, recognition of the eosinophilic/allergic endotype will help to predict improved ICS responsiveness, 57 whereas a favourable response to leukotriene receptor antagonist therapy could be suggested by an eosinophilic endotype characterized by altered eicosanoid metabolism. 52,58,59 Study of genetic variations in the β-adrenoceptor protein and risk of adverse outcomes may help to inform clinicians as to particular individuals in whom LABA therapy may be of diminished value. 60 The application of pharmacogenetics to help predict response to clinical treatments could also be valuable for other classes of asthma medication. 61
A recent trial has investigated patient use of electronic monitors that measure ICS/LABA inhaler use and provide medication reminders for missed doses as a method to improve medication adherence. 62 This trial was conducted in primary care and 32% of participants had received at least one course of oral corticosteroids in the prior year for asthma. Participants randomized to receive medication reminders had significantly greater ICS/LABA adherence at 6 months (60% of daily prescribed doses taken) compared with participants who were electronically monitored without receiving reminders (29% of daily prescribed doses taken). However, there were no significant differences between groups in asthma control or severe exacerbations. Future work will focus on the utility of electronic monitoring technology to improve inhaler adherence and clinical outcomes in selected cases, 63 such as individuals at risk of asthma exacerbations or people with unrecognized poor adherence under consideration for novel biological treatments for severe asthma. 64
Finally, novel inhaled LABA and ICS drugs are emerging that will provide greater choice for use with adults at step 3 and above. These include newer ICS/LABA combinations, 65,66 as well as ICS in combination with 24-hour acting LABA, which can allow once-daily inhaled treatment. 67
Step 4: High-dose ICS with other controller medications
There are few data on the optimal strategies at steps 4 and 5. Options are therefore based on individual discussions around the risks versus benefits of a trial of additional treatments. In people with asthma in whom add-on therapy provides no benefit, or results in side effects, then the usual action is to return to the previous regimen and consider an alternative add-on option. At steps 4 and 5, it remains important to consider adherence to treatment, avoidance of asthma triggers, assessment for coexisting disease and psychological factors, all of which may affect asthma control.
An initial option is to increase the ICS dose to up to 2000 μg/day (BDP or equivalent, in addition to LABA treatment), recognizing that the risk of potential side effects of ICS may be greater than the benefits at these doses. 68 Alternative options include the addition of a leukotriene receptor antagonist, oral theophylline or oral β-agonist tablets. 4
In selected people at step 4 with allergic asthma who require frequent courses of oral corticosteroids (four or more in the prior year), the National Institute for Health and Care Excellence has approved the use of omalizumab, a monoclonal antibody against IgE. 69 This treatment is also approved at step 5 and there are specific eligibility criteria regarding IgE levels and lung function. Omalizumab is administered in specialist centres by subcutaneous injection every 2–4 weeks, usually for a period of 4–6 months, at which point reassessment of the clinical benefit is required. For some adults, this treatment is effective in reducing severe asthma exacerbations. 70,71
Future directions
A trial in people poorly controlled on moderate-to-high dose ICS plus LABA therapy has suggested that the addition of the long-acting muscarinic antagonist tiotropium (via soft mist inhaler) reduces the risk of a severe exacerbation by 21% compared with placebo. 72 Participants in this trial had persistent airflow limitation, though potentially eligible participants using maintenance oral corticosteroids at more than 5 mg/day were excluded. 72 Tiotropium has recently been approved for maintenance asthma treatment in Europe. In the United Kingdom, a therapeutic trial can be considered in people on medium-to-high dose (≥800 μg/day of budesonide or equivalent) ICS/LABA with at least one severe exacerbation in the previous year. Additionally, a recent meta-analysis has indicated that tiotropium may be useful as an alternative to salmeterol add-on therapy in poorly controlled individuals on medium-to-high dose ICS, 73 although this indication remains unlicensed currently.
Macrolide antibiotics, such as azithromycin or clarithromycin, have also been used for their antibiotic and anti-inflammatory properties in asthma. 74 Their use may be of particular benefit in reducing exacerbations in severe neutrophilic asthma. 75 However, further long-term study of the impact of treatment on airway microbiota, 76 cardiovascular risk and mechanisms of action 74 is required.
Step 5: Maintenance oral corticosteroids
Step 5 is defined by the requirement for maintenance oral corticosteroid treatment. This is usually reserved for people requiring frequent short courses of oral corticosteroids for persistently uncontrolled asthma despite other treatments above. 77 The aim is to titrate treatment to the lowest maintenance dose that achieves asthma stability and to monitor or treat for steroid-related adverse effects. 78
Methotrexate, azathioprine, ciclosporin and gold have been used as corticosteroid-sparing agents, but these drugs are associated with significant side effects and their routine use is not supported by the findings from Cochrane reviews. 79 – 82
Future directions
Recent work has focussed on the use of biological agents targeted at T-helper type-2-driven cytokines, such as IL-4, 83 IL-5 84 –86 and IL-13. 87 These drugs have generally been tested in rigorously conducted clinical trials in carefully selected participants, and have shown promising results. Most recently, two studies have demonstrated the efficacy of subcutaneous mepolizumab (a monoclonal antibody against IL-5) in reducing asthma exacerbations in severe eosinophilic asthma, 84,85 and this treatment will likely become available for use in clinical practice in the near future. Further work is underway to identify biological and clinical characteristics that may help to predict favourable treatment response with other novel agents. 88,89
Step up and step down of therapy
Step up of therapy is generally considered if there is evidence of persistent symptoms, diminished lung function or ongoing airway inflammation after a period of 2–3 months on the current treatment regimen. 90 Prior to step up, it is important to review the validity of the original diagnosis of asthma, because misdiagnosis, or the presence of coexisting conditions requiring their own specific treatments, may help to explain the incomplete response to current therapy. Review of adherence to treatment, triggers for worsening asthma and inhaler technique 2 are other factors that should be considered with every change in therapy.
Step down of treatment may be considered after 3–6 months of clinical stability, with the intention of maintaining asthma control at a reduced medication burden. There are a variety of options available, 2 including ICS dose reduction, 91 use of a lower dose combination ICS/LABA preparation 92 and change from twice daily to once daily dosing. Recently, the US Food and Drug Administration has recommended that once asthma control is achieved, LABAs should be withdrawn and treatment maintained with ‘controller medication such as ICS’. 45 However, this approach can lead to loss of asthma control, compared with continuation of ICS/LABA therapy, 93 and so may not be appropriate for individuals at risk of future asthma exacerbations. 94
In general, a 25–50% ICS dose reduction is considered safe for most well-controlled people with asthma. 2 One caveat is that in adults who are stable on a low maintenance dose of ICS, there is an increased risk of future asthma exacerbation after ICS cessation compared with continuation of ICS. 95 In these individuals, complete cessation of ICS therapy is therefore not advised. 2
Factors determining choice of inhaler
The key factors that determine the choice of inhaler device are individual preference, acceptable inhaler technique for the chosen device and local availability and cost. In general, metered-dose inhalers are considered first-line for both preventer 96 and reliever 97 inhaler prescription. Inhaler use training helps to improve technique 98 and is an important factor in helping to optimize drug delivery to the lungs. 99 However, three situations when specific formulations may be selected are for improving drug deposition in the small (<2 mm) airways, reduced exposure to the systemic effect of ICS and for reduction in risk of ICS-associated pneumonia and lower respiratory tract infection.
Inflammation occurring in the distal small airways of the bronchial tree may contribute to the clinical features of asthma, such as poor control and airway hyper-responsiveness. 100 Newer inhaler devices, which generate smaller particle aerosols, can allow improved drug deposition in the small airways 15 and there is increasing evidence to suggest that this may help to improve asthma control. 101 However, challenges remain in identifying small airways dysfunction in a clinical setting, 15,100 limiting the widespread use of these inhalers. One approach is to switch to a small particle formulation in the setting of persistent poor control despite adequate inhaler technique (and adherence) with a standard large particle inhaler.
Systemic effects of ICS may be reduced by specific pharmacological properties of the drug. 24 For example, ciclesonide is a prodrug, which is converted to an active metabolite in the lung and which has limited oral bioavailability after inhalation. 102 Mometasone is another ICS with limited systemic bioavailability, because most of the swallowed dose is excreted unchanged and any absorbed drug undergoes liver metabolism into less active compounds. 103 However, the superiority of these drugs over other ICS formulations prescribed at low-to-medium daily doses has not been established, and in clinical practice, they remain an option for selected cases only.
Finally, there is emerging evidence to suggest that high-dose ICS use is associated with an increased risk of pneumonia or lower respiratory tract infection and that this risk is enhanced by use of certain ICS formulations, particularly those containing high-dose fluticasone, in comparison to budesonide or beclomethasone. 104 In people with asthma with recurrent respiratory infections, consideration should be given to reviewing the choice and dose of the prescribed ICS treatment.
Influenza vaccination and smoking cessation
Viral infections, including those caused by influenza, are associated with asthma exacerbations in observational studies. 105,106 Although there is insufficient evidence to demonstrate a reduction in exacerbations as a result of influenza vaccination, 107 current UK guidelines recommend influenza immunization in people with chronic asthma in order to help reduce the potential morbidity associated with deteriorating asthma control. 108
Current smoking is associated with increased odds of asthma hospitalization, greater asthma severity and worse quality of life compared with never smoking. 109 Smokers may also have a diminished response to ICS compared with non-smokers. 110 Support to access smoking cessation services should be considered for all people with asthma. 2
Conclusion
Pharmacological therapy for asthma is prescribed based on the level of current symptoms and lung function together with an assessment of the risk of future adverse events such as exacerbations. The intensity of the treatment ranges from as-needed use of short-acting reliever therapy to the use of systemic anti-inflammatory therapy. At each treatment step, adherence to medication, occurrence of side effects and assessment of coexisting conditions and triggers should be undertaken. In this review, we have provided a practical summary of the current standard pharmacological management of adult asthma based on British guidelines and suggested emerging treatments that may be recommended for use in the future.
Footnotes
Conflict of interests
MP is a member of the guideline development group for the Adolescent Asthma section of the BTS/SIGN Asthma Guideline. We have no other relevant conflicts of interest.
Funding
MP is funded by a National Institute for Health Research (NIHR) Clinical Lectureship. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
