Abstract
Some patients suffer from clinical symptoms of chronic obstructive pulmonary disease (COPD) but their pulmonary function tests are in the normal range (at risk group). The objective of this study was to discover a practical test to distinguish these patients from non-COPD subjects. A total of 77 subjects including 40 COPD patients, 37 subjects at risk for developing COPD, and 32 control subjects were entered in this study. The accuracy of maximal-mid expiratory flow (MMEF)/forced vital capacity (FVC) for the diagnosis of COPD in at risk patients and its capability to differentiate from early COPD and normal patients were evaluated. Body plethysmography was used for measurement of lung volume as the Global Initiative for Obstructive Lung Disease standard. MMEF/FVC in the at risk group of COPD (0.73 ± 0.19) was significantly lower than the normal control group (0.9 ± 0.24, respectively), and also, it was significantly higher than the COPD group (0.31 ± 0.17). There was significant correlation between the MMEF/FVC and amount of smoking measured by pack year (r 2 = 0.112, p = 0.005) and stages of COPD (Spearman’s ρ = 0.82, p = 0.0001). Early stage COPD (smoker subjects without spirometry derangement) can be diagnosed by MMEF/FVC. Using this tool we may be able to detect this highly preventable disease at an earlier stage.
Introduction
Chronic obstructive pulmonary disease (COPD) is a slow progressive disease and is valuable for early detection physiologic evaluations such as the spirometry. 1 According to the last Global Initiative for Obstructive Lung Disease (GOLD) criteria for staging COPD, the mild stage is a forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) below 70%. 2 The at risk stage is the mildest form of this disease with the FEV1/FVC and FEV1 being in the normal range. 2 Recent revisions in the GOLD guideline do not persist with the significance of the at risk group. 3 According to a previous screening study in asymptomatic cigarette smokers, 27% of these subjects showed some degree of airway obstruction. 4 Diagnosis of airway obstruction in at risk stage can help in the treatment of patients with cough and sputum and prevent further damage from air pollution and cigarette smoking. In this regard, physiological assessment including spirometry may help earlier detection of COPD.
Maximal-mid expiratory flow (MMEF) is a physiological parameter that enables us to estimate the obstruction in small airways when larger airways are not involved. However, this parameter changes with elongation of the FVC maneuver; in this regard, MMEF corrected with FVC (MMEF/FVC) 5 was a more reliable test to evaluate airway obstruction in small airways.
Moreover, dysanapsis is an anatomical term that is used for subjects with a smaller airway caliber diameter relative to their lung volume. The MMEF/FVC ratio (forced expiratory flow at 25–75% (FEF 25–75)/FVC ratio) is a parameter that was previously introduced from the spirometry for its close relation with dysanapsis. 6 In COPD, reduction in airway caliber owing to inflammation, mucosal gland hyperplasia, an increase in lung volume due to air trapping, and decreased lung elasticity are common. 7 According to this similarity with dysanapsis, a role for the MMEF/FVC ratio for evaluating COPD is suggested. This study was conducted to evaluate the accuracy of the MMEF/FVC ratio to find early COPD without gross spirometric abnormalities.
Materials and methods
Study design
A total of 77 subjects in the COPD group (37 cases in at risk and 40 in the obstructive groups) and 32 control subjects were recruited for this prospective case-control study (Table 1). Inclusion criteria for COPD patients was a history of long time cigarette smoking (20 pack-year or more) or exposure to air pollution and the presence of one or two clinical findings: (1) cough and sputum for at least 3 months within the past 2 years and (2) dyspnea and airway obstruction demonstrated by FEV1/FVC < 0.7 with or without FEV1 < 80% predicted for obstructive group and FEV1 > 80% predicted and FEV1/FVC > 70% for at risk group.
Classification and staging of COPD according to GOLD guidelines which used in this study.
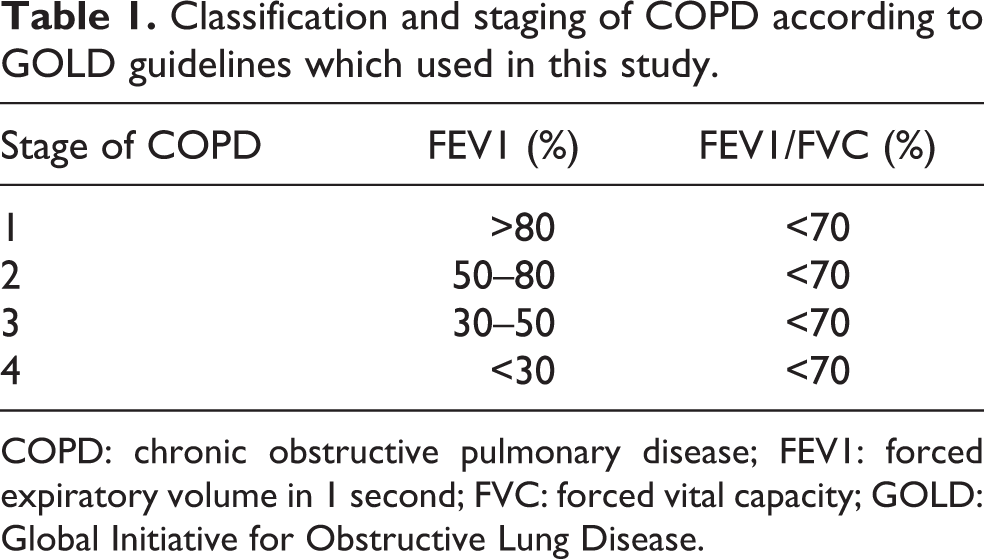
COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; GOLD: Global Initiative for Obstructive Lung Disease.
All the control subjects had no history of smoking, asthmatic symptoms, abnormal pulmonary function test results, lack of cooperation during the spirometry test, exposure to air pollution, and recent lung infections. Airway hyperresponsiveness was ruled out in the control group by the methacholine challenge test.
The Ethical Committee of our university approved the experiment and each subject gave his informed consent.
Methods
The study was performed in the pulmonary function laboratory of the Department of Pulmonary Medicine, Ghaem Hospital, Mashhad University of Medical Sciences, Mashhad, Republic of Iran.
Classification and staging of COPD was performed according to GOLD guidelines 3 (Table 1).
Emphysema was considered according to imaging (chest roentgenogram and/or computed tomography) or decrement of diffusion lung capacity for carbon monoxide (DLCO and DLCO/alveolar volume < 80% of predicted).
Techniques and protocol
The FVC maneuver and lung volumes were measured using a body plethysmograph, (Model Vmax 6200, SensorMedics, California Co. Ltd., Yorba Linda, California, USA) with acceptability standards outlined by the American Thoracic Society (ATS). 8 All COPD subjects were tested in a sitting position inside a box and were wearing nose clips. Standard spirometry was performed and the MMEF/FVC ratio (indicating dysanapsis of the lung) was calculated.
Control subjects were refrained from any drugs and caffeinated drinks for 2 hours before methacholine challenge test. Standard spirometry was performed as mentioned and then methacholine challenges with the cumulative concentration–response technique were used according to the recommendations of the ATS. 9
Analysis
Sample size was calculated according to the standard deviation of the MMEF/FVC ratio derived from a pilot study and α = 0.05.
Normal distribution of the data was checked using the Kolmogorov–Smirnov test. In comparing the quantitative values, the unpaired “t” test was used. Correlations between the MMEF/FVC ratio and spirometry parameters and lung volumes were tested using the least square regression. Correlation of the stage of COPD according to the GOLD classification and MMEF/FVC was measured by Spearman’s ρ. The receiver operator characteristic (ROC) curve was plotted for four diagnostic categories and the area under the curve was compared. Criteria mentioned above for the diagnosis of COPD were used as the GOLD standard for measuring the sensitivity, specificity, negative predicted value, and positive predicted value of the residual volume and MMEF/FVC ratio. EPI INFO 2003 (Centers for Disease Control and Prevention (CDC)) and SPSS 14 (SPSS INC.) were used for statistical analysis. Significance was accepted at p < 0.05.
Results
Baseline values
Sex distribution showed that the male to female ratio in the COPD group was 66 to 8, being significantly higher than the control group (Table 1; X 2 = 9.002, p = 0.002, Relative risk = 2.42).
Mean age of COPD subjects was 58 ± 12.15 years, being significantly higher than the at risk group (t = 3.9, p = 0.001; Table 2). Mean duration of smoking in COPD subjects was significantly longer than the at risk group (t = 3.06, p = 0.003; Table 2). The regression model revealed a significant correlation between the MMEF/FVC and amount of smoking measured by pack year (r = 0.336, p = 0.005) that was higher than correlation of smoking (pack year) with FEV1 and FEV1/FVC (r = 0.306 and r = 0.242, respectively).
Baseline demographic difference between groups tested.

Descriptive data of clinical symptoms and pulmonary function test
Frequencies of major clinical findings are shown in Table 3. Dyspnea was significantly more frequent in obstructive COPD than in at risk COPD patients. Frequencies of other symptoms were similar in both the at risk and COPD groups, for example, wheezing was seen in 60% of at risk COPD patients.
Comparison of clinical findings between at risk and obstructive stage of COPD.

COPD: chronic obstructive pulmonary disease; DLCO: diffusion lung capacity for carbon monoxide.
aEmphysema was approved according to chest roentgenogram and/or computed tomography and DLCO.
Progression of COPD according to its staging did not influence clinical symptoms (Table 4). Emphysema was diagnosed in 19% of at risk COPD patients which was significantly lower than the obstructive COPD group.
Clinical findings in different stage of COPD.

COPD: chronic obstructive pulmonary disease; DLCO: diffusion lung capacity for carbon monoxide.
aEmphysema was approved according to and chest roentgenogram and/or computed tomography and DLCO.
Pulmonary function tests revealed significant differences between obstructive and at risk COPD patients in routine spirometric parameters and lung volumes (except in total lung capacity (TLC); Table 5). However, in the at risk group, respiratory volume (RV) and functional residual capacity (FRC) more than 120% of predicted was detected in 35% (13 of 37) and 29% (11 of 37) of cases, respectively.
Comparison of spirometric evaluation between at risk and obstructive stage of COPD.a

COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; FEV: forced expiratory volume; VC: vital capacity; MMEF: maximal-mid expiratory flow; RV: respiratory volume; TLC: total lung capacity; FRC: final residual capacity.
aAll parameters except FEV1/FVC and MMEF/FVC are expressed in percentage of predicted value.
Evaluation of MMEF/FVC for diagnosis of at risk group
Mean MMEF/FVC in the at risk group of COPD was significantly higher than the obstructive group (0.73 ± 0.19 and 0.31 ± 0.17, respectively; t = 9.65, p = 0.0001). On the contrary, the mean MMEF/FVC in the at risk group of COPD was significantly lower than the normal control group (0.73 ± 0.19 and 0.9 ± 0.24, respectively; t = 3.2, p = 0.002). Figure 1 shows that the range of mean and 95% confidence level of MMEF/FVC in the three groups of obstructive, at risk, and normal subjects were distinct from each other.

Mean and standard error of MMEF/FVC in at risk and obstructive stages of COPD and normal subjects (error bars showed 95% CI of means). COPD: chronic obstructive lung disease; FVC: forced vital capacity; MMEF: maximal-mid expiratory flow; CI: confidence interval.
The stage of COPD according to GOLD classification also showed a significant correlation with MMEF/FVC (Spearman’s ρ = 0.82, p = 0.0001; Table 6).
Correlation of some important spirometric and lung volume parameters with different stage of COPD (FEV1 and RV are percentage of predicted value).

COPD: chronic obstructive lung disease; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; RV: respiratory volume; MMEF: maximal-mid expiratory flow.
Accuracy of MMEF/FVC for diagnosis of COPD in patients with normal FEV1 and FEV1/FVC
Figure 2 shows the ROC curve for four major pulmonary function tests used for the diagnosis of obstruction. MMEF/FVC showed the best results and the best cutoff point was 0.8. The accuracy of MMEF/FVC in three different cutoff points was shown in Table 7 and they were compared to the residual volume. The area under the curve for the MMEF/FVC was 0.849 (p = 0.0001), which was the largest followed by FEV1/FVC at 0.843 (p = 0.0001).

ROC curve in the form of diagram, specificity, and sensitivity of percentage predicted for FEV1, FEV1/FVC, MMEF/FVC and percent of predicted of RV. ROC: receiver operating characteristic; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; RV: respiratory volume; MMEF: maximal-mid expiratory flow.
Accuracy of MMEF/FVC for evaluation of patients suffered from COPD when FEV1 and FEV1/FVC are normal.

PPV: positive predicted value; NPV: negative predicted value; COPD: chronic obstructive lung disease; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; RV: respiratory volume; MMEF: maximal-mid expiratory flow.
Discussion
Today, the diagnosis of COPD requires physiological confirmation of bronchial obstruction. 1 The at risk subgroup referred to patients with cough and/or phlegm, with routine spirometry such as the evaluation of FEV1 and FEV1/FVC being in the normal range. 2,10 In a study of incident COPD cases, symptoms leading to seek medical advise were cough (84%), sputum (45%), and dyspnea (75%). 11 In this group, 37% had stage 0 (at risk).
The most recent revision of GOLD guidelines removed stage 0 disease from its classification. 3 However, there is still a group of patients that are at risk or suffer from COPD, but their obstruction cannot be confirmed with FEV1 or FEV1/FVC parameters.
In this study, the MMEF/FVC ratio for diagnosis of bronchial obstruction was examined. MMEF/FVC ratio reflects the relation of airway caliber to lung volume. Therefore, in COPD patients, although some lung volumes including FRC increase mainly due to air trapping, significant reduction of airway caliber due to inflammation, and submucosal gland hypertrophy 12 can lead to a significant reduction of this ratio. Since FEV1 and FEV1/FVC are not diagnostic in the at risk stage, accuracy of the MMEF/FVC ratio is higher than FEV1 and FEV1/FVC and it can detect patients who are in the at risk group. This parameter helps to diagnosis early stages of COPD. In comparison with some other sophisticated methods, it has an advantage, in that it can be evaluated with routine spirometry equipment. Depression of the middle part of expiratory flow volume curve in routine spirometry also has the capacity to detect early obstruction, but the shape of loop sometimes is not clear and it is interpreter dependent. MMEF/FVC can be calculated very easily and by the cutoff point defined in result section we can accurately separate the COPD and normal subjects.
COPD patients may present with a chronic cough and sputum that often precedes airflow limitation by many years. 12 For early diagnosis and to increase the chance of successful smoking cessation, it is better to repeat the physiologic testing every 3–5 years. 1 The use of the spirometry doubled the number of known patients with COPD 13 in comparison to a questionnaire used for diagnosing COPD by clinical symptoms. In a study in Poland using spirometric screening, it was able to diagnosis airway limitation in 24.3% of smokers aged more than 40 years with a history of smoking more than 10 pack-years. 14 In this group, anti-smoking intervention was more successful than in patients with normal spirometric parameters, which is promising for the prevention of the disease progression. A previous report showed a close correlation between the amount of smoking (pack-years) and decrement in FEV1. 15 In this study, a close correlation between the amount of smoking and the MMEF/FVC ratio was shown. The use of FEV1 and FEV1/FVC as the standard test for diagnosis of COPD was successful in 50% of our cases (37 of 74). Using the MMEF/FVC ratio, airflow limitation was detected in 86% (64 of 74).
Therefore, the results of the present study on COPD patients indicated that the MMEF/FVC ratio could be a useful test for the earlier diagnosis of this highly preventable disease and lead to better management of patients at-risk for developing COPD.
Footnotes
Authors’ Note
The present work was conducted in the Chronic obstructive pulmonary disease research center, Mashhad University of Medical Sciences, Islamic Republic of Iran.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
