Abstract
Patients with chronic pulmonary disease often suffer from breathlessness or dyspnea. Traditional evidence generation techniques to expand upon current treatment paradigms are limited by the significant delay between study initiation and clinical implementation of findings. Rapid learning health care is a novel approach to health care delivery that relies on intelligent and continuous integration of clinical and research data sets to deliver personalized medicine using the most current evidence available. Results of important studies in the management of chronic respiratory disease are presented in brief; however, the focus of this review is on evidence supporting the implementation of a rapid learning model for symptom management. Recent findings suggest that a rapid learning system is feasible and acceptable to patients with advanced illness, helps monitor symptoms overtime, facilitates study of the impact of novel interventions, and can identify unrecognized needs and concerns. A rapid learning model improves comprehensive assessment, timeliness of intervention, and accrual of contemporaneous data to support best practice that tailors care specific to the needs of patients as their disease and lifestyle change overtime. Using the rapid learning health care model, data collected in the process of routine care can simultaneously function both as clinical information and as a resource for research on patient-centered experiences and outcomes.
Introduction
Dyspnea is a major cause of morbidity and distress in patients with chronic respiratory disease. Comprehensive, personalized management strategies for dyspnea are limited by the lack of robust methods to continuously integrate disparate clinical, laboratory, radiologic, biologic, and research data to intelligently, cohesively, and constantly develop refined predictive treatment algorithms, prognostic models, and strategies for care. This is timely because of the growing understanding that the dyspnea experience stems more from than just anatomic or pathophysiologic derangement; in order to care for people with dyspnea, data reflecting diverse biopsychosocial domains must also be integrated into the model.
As a larger population of people will be living with the chronic dyspnea due to improved disease-directed therapies, better strategies will be needed to tailor interventions to optimize independence relieving the symptomatic burden of dyspnea. Managing dyspnea among patients who have diverse pulmonary diseases and clinical presentations is not a simple task. Management must be aided by data collection, integration, and analyses of continuously collecting databases embedded within common clinical, quality improvement, and research efforts without causing significant burden to patients or their providers. This ability, which complements knowledge produced by phase III clinical trials, requires rigorous systems that can conduct ‘rapid learning health care.’ In this review, we discuss the role of rapid learning in chronic dyspnea management.
The burden of dyspnea in chronic respiratory diseases
Chronic respiratory disease is a growing global epidemic. As of 2009, respiratory diseases such as chronic obstructive pulmonary disease (COPD), lung cancer, bronchiectasis, asthma, cystic fibrosis, and interstitial lung disease were the third leading cause of death in the United States 1 and the European Union. 2 Furthermore, a recent report estimates that the prevalence of chronic respiratory conditions is around 10% in the general population worldwide. 3,4 In the United States, up to 24 million adults suffer from COPD or asthma with millions more who suffer from other obstructive and restrictive respiratory conditions. 5 This number is likely an underestimation as many patients live without a formal diagnosis 6 or have less-common conditions that are not included in prevalence estimates. In the United States, COPD is the only common cause of death that has increased over the last 40 years, in sharp contrast to the reductions seen in cardiovascular and infectious diseases and many cancers. 7 Within the coming decade, chronic respiratory diseases like COPD are predicted to become the fifth most common cause of chronic disability in patients worldwide. 8 These highlight the growing population of patients who will be living with chronic respiratory illness and who will require intensive symptom management.
Dyspnea is one of the most common, debilitating symptoms experienced by patients with chronic respiratory diseases. Its prevalence ranges from 56% in patients with severe COPD 9 to 90% in the end-of-life setting. 10 Dyspnea is experienced at some point by the majority of patients with chronic diseases including those that are not primary respiratory or cardiac conditions. For example, for almost half of all patients at the end of life, dyspnea severity increases significantly between 3 months and 1 month before death 11 ; half of these patients report that the symptom is severe in nature. 12,10 Effect of dyspnea on health-related quality of life (QoL) is well documented in large studies such as the National Emphysema Treatment Trial. This study of over 1600 patients with severe COPD demonstrated a strong negative relationship between dyspnea severity and QoL, which was remarkably independent of physiologic changes in lung function such as forced expiratory volume in 1 second. 13
Defining dyspnea
The American Thoracic Society defines dyspnea as ‘a subjective experience of breathing discomfort that consists of qualitatively distinct sensations that vary in intensity. The experience derives from interactions among multiple physiological, psychological, social, and environmental factors, and may induce secondary physiological and behavioral responses. 5 The definition of dyspnea is inherently dependent upon the words and descriptors of the patient, irrespective of any physiologic or anatomical derangements observed through physical examination, laboratory measurements, or radiologic assessment.
At its core, dyspnea is truly ‘a subjective experience’ that is shaped by the patient’s background, experiences, and perspectives. Similar to the groundbreaking description of ‘total pain’ by Saunders, 14 Abernethy and Wheeler 15 describe a biopsychosocial model of dyspnea that described the multifaceted nature of its etiologies, distracters, and exacerbators. The ‘total’ concept was recently applied to dyspnea because of its many parallels with the experiences of the patient with chronic pain. For example, the four domains of chronic pain (physical, psychological, interpersonal, and other distress) similarly contribute to the experiences of patient with dyspnea in both positive and negative ways. For example, depression, fatigue, and overall health perception associate with dyspnea in patients with heart failure 16 ; spiritual distress has also been shown to correlate with dyspnea severity. 17 Following the lead of Saunders, Kamal et al. 18 recently presented a mnemonic specific for dyspnea assessment and management whereby the biopsychosocial components can be recalled using ‘dyspnea’ as the acronym: D (depression); Y (yearning for peace, forgiveness, etc.); S (social issues); P (physical problems); N (nonacceptance or spiritual/existential distress); E (economic or financial distress); and A (anxiety and anger).
Assessing dyspnea
Several assessment tools are available for determining dyspnea prevalence and severity. 18 These tools range from those validated and used in the research setting to those that are user-friendly for usual clinical environments. Patient report continues to be the primary method for dyspnea assessment at the bedside; a number of validated tools are used. The tools most easily implementable in community settings are those that involve numerical or categorical responses and do not require functional assessment. These include standard single-item ordinal scales such as the Visual Analogue Scale, Numerical Rating Scale (e.g. 0 [no breathlessness] to 10 [worst possible breathlessness]), and the modified Borg Scale.
Research studies typically employ more complex measurements of physiologic parameters, such as the 6-min walking time, and surveys eliciting a report of the patient’s symptom experience and severity (e.g. ‘I feel out of breath,’ ‘My chest feels tight,’ etc.). 19 Summary tools include the Memorial Symptom Assessment Scale, 20 the Patient Care Monitor (PCM), 21 and the Edmonton Symptom Assessment System. 22 While many of the available scales overwhelmingly focus on physiological parameters or are too long to be practically applicable in the clinic, 18 the 12-item Cancer Dyspnea Scale 23 efficiently details the patient’s experience. The Cancer Dyspnea Scale was originally developed in Japanese and has been recently translated and validated in English. 24
In community practice, dyspnea assessments are either performed as part of paper-based systems review prior to the provider interview or during the history and physical examination itself. As a reflection of the limitations of data capture that ‘stand alone,’ these dyspnea assessments often disappear into the clinical records maintained by separate providers. Neither can these data points be readily integrated into the health records or decision making of other providers who also participate in the patient’s care nor can this information be put into the perspective of the patient’s psychosocial state (e.g. as assessed by a mental health professional), spiritual distress (from the interview of a hospital chaplain), or caregiver distress (as asked by the patient’s primary care physician) without adequate technologic systems in place to incorporate all these data into useful, intervenable, action points. As dyspnea is a symptom assessed and managed by many types of providers often concurrently (e.g. primary care physician and oncologist in lung cancer as well as pulmonologist and infectious disease in chronic bronchiectasis), combining data obtained through these usual clinical encounters naturally becomes a solution for integrated clinical management of dyspnea. This also prefaces how further research into optimized, personalized treatment strategies can be aided by such efforts, of significant importance, because of the major gaps in knowledge regarding dyspnea management.
Dyspnea management
As the biopsychosocial model of dyspnea proposes that dyspnea causes and exacerbators stem from diverse anatomic, physiologic, psychosocial, and emotional origins, and clinical management of this symptom remains particularly challenging. This is uniquely difficult because measures of ‘success’ are largely subjective and rely on patient report rather than measurable laboratory or radiologic changes. 25
When a patient’s dyspnea becomes refractory to the usual anatomic-directed interventions (e.g. thoracenteses for pleural effusions or airway dilation with beta agonists for bronchoconstriction), the focus of care shifts from disease-specific treatments to global or systemic approaches. This represents a shift in treatment goals from altering the disease trajectory to improvement in qualities of life and function. There remains good support for global palliative treatments to improve symptoms and QoL for patients with intractable dyspnea. 26
Opioids remain the pharmacologic mainstay of the global management of dyspnea. Morphine, the most commonly studied opioid, has been part of two important clinical trials to establish it as the gold standard. The largest double-blind, placebo-controlled trial versus 20 mg daily morphine was conducted in 2003 by Abernethy et al. in 48 opioid-naive patients, mostly with COPD. 27 In the morphine arm, significant benefits for both dyspnea and insomnia control were reported. More recently, Currow et al. 28 completed a phase II dose increment study to determine a minimum effective daily dose and evaluated potential longitudinal clinical benefit. A total of 85 participants were given escalating doses of sustained-release oral morphine, starting at 10 mg/day to a maximum 30 mg/day. Remarkably, for 70% of patients, the beneficial dose was 10 mg/day, and sustained benefit for 3 months was observed in 53% of patients at any dose. This was the first study to demonstrate that low doses of sustained opioids have persistent therapeutic effect. Thus, the usual recommended treatment dose is morphine either in divided doses or sustained release, totaling 10–20 mg/day with active evaluation and gradual titration to the desired effect.
Another mainstay of global management of dyspnea is supplemental medical oxygen, often supplied by tank through nasal cannulae. Supplemental oxygen is one of the interventions most frequently requested by patients 29 and implemented by hospitals to relieve dyspnea. 30 Although evidence in COPD clearly demonstrated a survival advantage with oxygen use in hypoxemic patients, 31, 32 other systematic reviews in cancer dyspnea have suggested that oxygen benefit may only be seen in patients with more severe hypoxemia. 33,34
To clarify the role of palliative oxygen for refractory dyspnea, Abernethy and colleagues recently conducted a large, international, randomized, controlled, double-blind study of palliative oxygen versus medical air (i.e. room air with ambient partial pressure of oxygen) for nonhypoxemic patients (PaO2 >55 mm Hg). 33 Neither gas demonstrated superiority in improving quality of life or relieving the sensation of breathlessness. Interestingly, both dyspnea and QoL improved over the study period in both arms, suggesting that patients may experience benefit derived from the sensation of moving air alone, rather than from the properties of a specific gas such as oxygen. Because some patients who might benefit from oxygen therapy may not want to receive it 34 and because the data on treatment preferences of patient with dyspnea are not conclusive, palliative oxygen should be delivered only with careful consideration of the intervention’s potential benefit versus patient burden and costs. 35
Importantly, certain patient demographics (minorities, young persons, and frail elderly), diseases (cancer and heart failure), and settings (opioid-tolerant and long-term efficacy) have not been subjected to robust clinical trials. The limitations of the applicability of available clinical trial data coupled with the unlikelihood that large, controlled trials for dyspnea control will occur frequently and rapidly require that new methods of evidence generation are needed to achieve true personalized health care delivery.
Rapid learning health care
The translational gap between the continual flood of new data generation and the clinical practice of medicine remains large. The inability to practice true personalized medicine stems from the health care system’s inefficiencies in matching—in real time and at the point of care—each patient with the most detailed evidence available. 36,37 The ability of health information technology to amass information from routine clinical care, laboratory and radiologic data, clinical trials, comparative effectiveness research, and molecular and genomic data for ready use during clinical encounters is the foundation of the rapid learning concept. In a rapid learning environment, newly available data become accessible so that the rapid learning system can continue to ‘learn’ in an iterative manner (Figure 1). This is because pertinent information: (a) is gathered in a planned and systematic manner; (b) is continually analyzed with each new use; (c) is reviewed within the context of available (retrospective analysis) and evolving (prospective) data; (d) can be implemented into current clinical care; (e) will evaluate current clinical outcomes; and (f) will generate new hypotheses and investigations. 38

Moving from patient-reported assessments to iterative decision support.
Current information technology has advanced to a level where it is now possible to move forward from concept and design to developing, initiating, conducting, and cultivating the data resources for the rapid learning model to aid in clinical decision making and drive the discovery process. Data from a variety of sources (e.g. genomics, proteomics, clinical research, clinical and patient-reported outcomes [PROs], clinical care, and administrative) are integrated to provide a diverse clinical resource that allows clinicians to balance their personal judgment with relevant evidence to achieve optimal outcomes while minimizing adverse events. 39 Accelerated information acquisition and processing support the clinician’s treatment decisions at the point of care because these decisions are based on the experiences and outcomes within very specific patient populations. The entry point, however, is the patient’s report of their experience.
Throughout the trajectory of chronic respiratory conditions, from diagnosis through death, a rich but underappreciated and underutilized source of information is the patient’s personal impression of the illness experience, including its associated symptoms and effects on outcomes like QoL. The importance of incorporating PROs into routine patient care has been recognized by the Institute of Medicine as a critical direction to generate patient-relevant evidence, encourage patient care to be the driving force for scientific discovery, and maximize health care. 39
The importance of electronic PROs in rapid learning systems
PROs are a valuable metric for evaluating a patient’s symptoms, experience, and QoL. Historically, PROs were collected during the clinician’s interview with the patient or by using paper-and-pencil surveys. Getting the information from a survey or a chart into a usable resource is both labor- and time-intensive. The advent of electronic systems has greatly improved the efficiency of data collection and incorporation. Electronic processing (e.g. tablet computers, smart phones, voice response systems, and secure Internet connections) also affords a level of privacy for sensitive topics as well as ensuring that patient concerns come to the attention of the health care provider and are not forgotten once the clinical encounter begins, thereby facilitating patient–physician communication. 40
Patients prefer electronic capture of PROs to paper and find the method easy and acceptable, 33,41 a conclusion confirmed by meta-analysis. 42 While the incorporation of PROs into a rapid learning system is gaining acceptance in the oncology community, widespread adoption in other subspecialties has not been as aggressive. Multiple health information technology platforms can be employed to join patient-reported data to data entered by patient, caregiver, or clinician. These platforms include the Internet, local clinic-based kiosks, tablet computers, digital pens, smart phones, and other handheld devices. The actual choice will depend on the local setting and patient population but needs to be flexible and sensitive to selected patient populations (e.g. a telephone-based platform may be best for the vision-impaired patient). Information technology-based platforms allow a common approach to data collection and management to be accessible to numerous care settings.
While the goals of rapid learning health care systems can be broadly defined, they often focus on relatively short timeframes—seeking to improve processes and interventions on a continuous basis. Thus, symptom prevalence, burden, and chronicity are ideally suited for measurement and intervention within a dynamic, continuously improving learning system. The impact of an intervention for symptomatic improvement made at one clinic visit can be followed over subsequent time points (with any follow-up interval) to assess the efficacy of that intervention. Such outcomes ideally align with the outcomes amenable to direct patient reports, laying the foundation for PROs as the critical element in rapid learning health care systems. Since dyspnea is a subjective experience that is best evaluated by direct patient reports, PROs will be the backbone of any rapid learning dyspnea management model.
Our experience with rapid learning
We have implemented rapid learning systems to support symptom management and monitor QoL in patients with advanced cancer and in palliative care settings. Using electronic user interfaces (e.g. tablet computers) and digital pens to collect PROs at the point of care, data are securely transferred to a central server to be aggregated with clinical results and maintained for longitudinal storage and analysis. We have shown that data collected electronically is both feasible and acceptable to patients 33 and is comparable with data collected by traditional paper-based questionnaires. 43 Patient’s symptoms can be tracked longitudinally, novel interventions can be evaluated, 44,45 and unmet patient needs can be identified and addressed. 46
The Duke Cancer Care Research Program and Duke Cancer Institute began longitudinal collection of PRO data in 2006 as part of a portfolio of clinical research studies that included pilot studies assessing feasibility and usability of specific interventions, such as the Pathfinders program, 44 to decrease psychosocial distress among cancer patients. The warm reception by patients and clinicians encouraged an expansion of the program medical oncology clinics; PRO data collection soon became a routine part of clinical care. The current rapid learning model, called the PCM version 2.0 (PCM 2.0), has patients with cancer complete an 80-item (86 for women) review of symptoms instrument while in the waiting room prior to each clinic visit with the treating medical oncologist. The psychometric properties (i.e. validity and reliability) of the PCM 2.0 have been demonstrated using personal tablet computers and a consistent information technology infrastructure. 21 Once the patient completes the instrument, the data are uploaded to a secure repository and a summary report is generated for the clinician. The report highlights symptoms concerning to the patient as well the direction and magnitude of symptom change since the previous assessment. The report also presents current symptom scores alongside scores from the previous three visits, so that the symptom trajectories and potential impact of interventions can be quickly evaluated. Research efforts are currently underway to characterize symptom burden in a disease-specific manner (and ultimately, in a treatment-specific manner) so that interventions can be designed and tested. The results of such characterization may allow identification of signals within PROs that can aide in discriminating between disease progression and treatment toxicities to support clinical decision making.
Dyspnea management in community settings has rarely been studied. In 2008, we formed a novel partnership, called the Carolinas Palliative Care Consortium. In this group, four North Carolina community palliative care organizations and Duke University collectively agreed to collect point-of-care data on patient distress within domains that range from symptoms to advance care planning. 47 This system uses patient responses recorded in a provider-entered, information technology-based program to aid in point-of-care observations being recorded, analyzed, and grouped for efficient incorporation into reports. This effort has been largely fruitful; data on over 6000 unique patients across over 20,000 patient visits have been recorded in a central database. Through data analyses, we have been able to define dyspnea prevalence, the efficacy of usual interventions, and how practice improvement can stem from the knowledge gained. 48
Seeing that this first iteration of the rapid learning concept could yield important clues about how to improve the delivery of personalized medicine, we aimed to make our rapid learning approach even more rapid. This required leaning more heavily on information technology to increase efficiency of data collection and provide useful feedback in real time. Thus, the new version, the Quality Data Collection Tool (QDACT) is a completely Web-based instrument that is accessible from any Internet-connected device and across diverse settings (e.g. hospital, patient’s home, long-term care centers, and outpatient clinics). This required an interdisciplinary team of software programmers, database analysts, project managers, physicians, public policy researchers, and the expertise of community palliative care providers to build a point-of-care, provider-entered clinical assessment tool that targets the important constructs related to dyspnea and its management (Figure 2). For example, it uses the Edmonton Symptom Assessment Tool 22 to document dyspnea severity during the visit on a scale from 0 to 10 and the Memorial Symptom Assessment Scale 20 to explore dyspnea severity, burden, and frequency. The QDACT also records specific pharmacologic and nonpharmacologic interventions for dyspnea management.
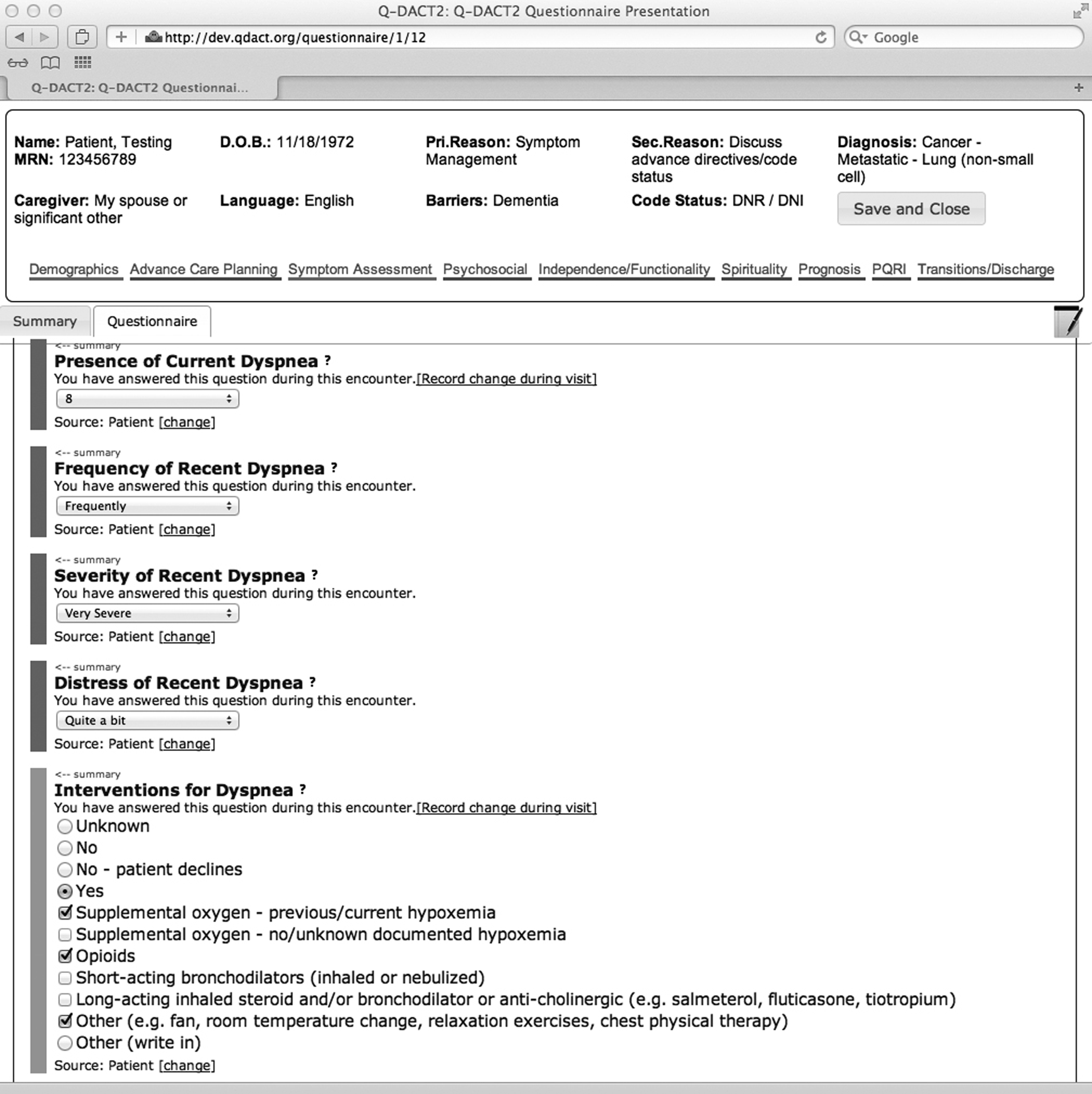
Screenshot of Quality Data Collection Tool (QDACT) for dyspnea assessment.
To support local quality improvement programs, we are also building a feedback framework within QDACT that grades the management of palliative care providers in dyspnea care against accepted quality measures. For example, the QDACT will be able to provide rapid feedback to providers on their conformance to a set of dyspnea quality measures and how this matches up with other providers within their practice, among providers with similar training background or specialty, or even across the entire consortium. Ultimately, in addition to being clinically useful, the QDACT becomes a rapid learning tool to help providers understand how data can drive quality improvement efforts and eventually patient care and outcomes.
Implementing rapid learning and PROs in community dyspnea management
Dyspnea is an ideal symptom where seamless integration of the biopsychosocial understanding of its causes with rapid learning health care systems can produce just-in-time solutions for clinical management. Figure 3 depicts the process integrating the biopsychosocial model of ‘total dyspnea’ into a technology-aided rapid learning system for dyspnea management. In this model, the typical components of the ‘total’ model are continuously integrated with other data ‘layers’ collected as part of clinical care and research. All this information can then generate a treatment plan for the individual patient, while simultaneously informing care strategies for the whole population. This is especially valuable for individuals who are rarely included in clinical trials (e.g. the frail and elderly, patients with complex comorbidities and associated medications, and those geographically far from academic medical centers), 38 who are large and important components of the community population.

Rapid learning integrated into biopsychosocial model of dyspnea.
For community implementation of PRO-based rapid learning systems for dyspnea assessment and management, four key steps are required (Table 1). First, a cultural buy-in from administrators, clinicians, and the health system as a whole is required to justify the commitment of resources to develop, manage, and maintain the necessary components. Second, the clinical team needs to choose among a variety of goal-based methods to ensure standardized, systematic collection of PRO dyspnea data of interest. This includes identifying the software and hardware platforms to conduct the system that are usable within the health care environment. These platforms, coupled with a chosen PRO method, can be used to: screen for known problems (i.e. setting thresholds), identify underappreciated problems, or monitor interventions. An example of implementation of PRO data for rapid learning at the Duke Cancer Institute focused on psychosocial distress management in patients with metastatic breast cancer that used the PCM as a method for screening patients for a social work–guided intervention and for responses to the intervention. 44 Within chronic dyspnea, one approach may involve setting thresholds for alerting symptoms (e.g. an absolute dyspnea score of 4 of 10 or an increase of 2 points from prior visit). The rapid learning system could then track dyspnea trajectory among those patients whose scores exceeded the threshold to determine whether alerting clinicians to problematic dyspnea resulted in improvement in symptom severity, even if varied interventions were implemented.
Characteristics of implementable PRO systems
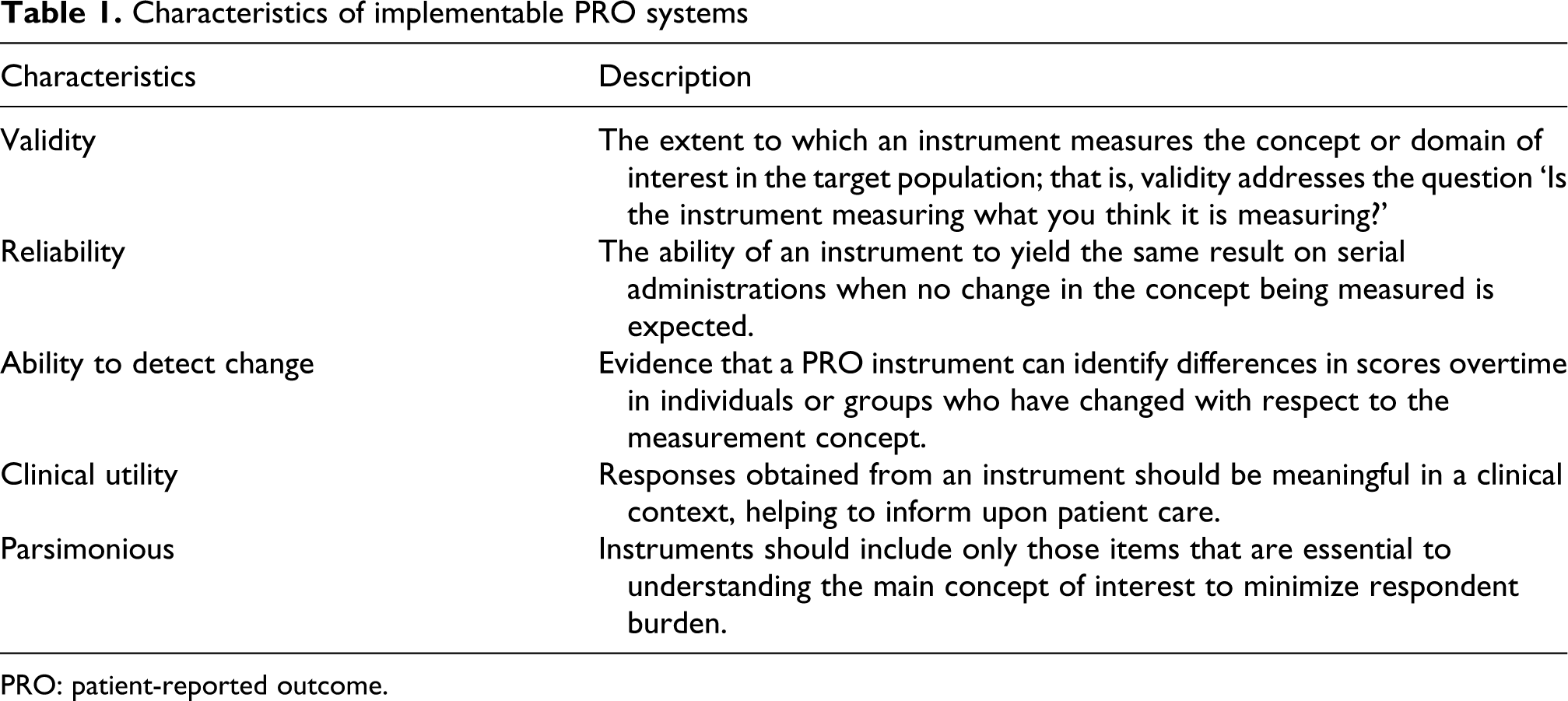
PRO: patient-reported outcome.
Next, the information must be turned around ‘rapidly’ to iteratively update the delivery of dyspnea care. This is the critical step that involves interpreting data, comparing it to previous knowledge, and reconciling this new data for near-term changes in clinical practice. Often, this is realized as clinical decision support measures where local evidence generation coupled with knowledge from external sources (i.e. pivotal clinical trials) modifies treatment pathways. For instance, consider the patient with a dyspnea score greater than 5 and anxiety subscales greater than 5. This person has refractory symptoms; the clinical decision support system informs the physician that a trial of morphine may be beneficial. If the initial pharmacologic management with morphine advised via the clinical pathway provides inadequate symptomatic relief, the next step in the pathway is recommended—perhaps the addition of psychological support. As the system learns about the relationship between the addition of psychological support and relief of dyspnea, the timing of the recommendation is shifted accordingly (e.g. if the adaptive learning analysis suggests that patients with combined anxiety and dyspnea have more effective and durable symptom relief with earlier psychological interventions, then an updated pathway may result, with oversight of the clinical pathway development process). Thus, the rapid learning system would provide the opportunity to deliver care truly tailored to local patient populations and practice patterns without causing undue burden to the provider or the patient. A system like that described also allows for rapid integration of new interventions from outside research, and more importantly, the opportunity to further refine implementation at the local level.
Conclusion
Chronic respiratory disease management is complex and the available evidence limits our ability to fully account for the multidimensional etiologies and the extensive number of factors that can aggravate the condition. Most assessment protocols and interventions are unidimensional that fail to value the power of concurrently collected clinical, laboratory, and radiologic data sets. An individually oriented, patient-centered approach is most likely to achieve the desired effect that begins with the routine incorporation of PROs into an ever-expanding record of the patient’s illness and experience over the trajectory of the disease. Rapid learning health care leverages existing evidence with the patient’s experience in both community and academic settings to optimize and tailor care specific to the individual. This approach shows promise for improving symptom management while reducing patient burden and disease-related suffering to enhance the QoL for patients with serious, life-limiting respiratory disease. Although conceptually grand in its vision, rapid learning dyspnea management can be easily implemented in the community through four critical steps.
