Abstract
This study investigated the effects of diaphragmatic breathing (DB) on ventilation and breathing pattern, seeking to identify predictors of its efficacy in patients with chronic obstructive pulmonary disease (COPD). Twenty-nine patients with moderate and severe COPD were monitored using respiratory inductance plethysmography and metabolic gas analysis. After 4 minutes of natural breathing, subjects completed 2 minutes of DB followed by 4 minutes of natural breathing. Dyspnea was measured using a visual analogue scale. Diaphragmatic mobility was assessed using chest radiography. DB was associated with a significant increase in tidal volume and reduction in breathing frequency, leading to higher ventilation and oxygen saturation, with a reduction in dead space ventilation and ventilatory equivalent for carbon dioxide. A total of 10 subjects with moderate (5) and severe (5) COPD performed DB with asynchronous thoracoabdominal motion, worsening the dyspnea, and decreasing the gain of tidal volume. Diaphragmatic mobility, inspiratory muscular strength, lower scores for dyspnea and hypoxemia as well as coordinated thoracoabdominal motion are associated with effective DB. In patients with COPD, DB can improve breathing pattern and ventilatory efficiency without causing dyspnea in patients whose respiratory muscular system is preserved.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) has a significant impact on the performance of activities of daily living. 1 Diaphragmatic breathing (DB) is a breathing strategy frequently taught as a component of self-management for this population. Its objective is to minimize the respiratory demands of the disease and in turn reduce the impact of the disease upon daily life. 2
During DB, patients should predominantly move the abdominal wall during inspiration and reduce upper rib cage motion. It is suggested that this manoeuvre improves chest wall motion and the distribution of ventilation, decreasing the energy cost of breathing, the contribution made by the rib cage muscles, and dyspnea, while improving exercise performance. 3 During DB, patients with COPD are able to increase the excursion of the abdominal wall 4 and reduce accessory muscle activity, 5 leading to an improvement in breathing pattern 6 and arterial gases. 7 However, there have also been reports of reduced mechanical efficiency of the respiratory muscles, 5 abnormal chest wall motion, 5 a tendency toward increased work of breathing and dyspnea, 7 and absence of changes in the distribution of pulmonary ventilation. 8 The differences between the results could be related to the manner of application of the technique or to the capacity of the respiratory musculature to increase the pulmonary volume. Our hypothesis is that the effects of DB in patients with COPD depend upon the preexisting condition of the musculature. Thus, our objective was to study (a) the breathing pattern of subjects with COPD with various degrees of airflow obstruction during DB and (b) what physiological variables can identify patients as responders or nonresponders in terms of increasing inspiratory volume.
Methods
The study was approved by the Research Committee on Human Investigation at the authors' institution, and a written informed consent was obtained after the purposes and potential risks of the investigation were fully explained and understood by each patient. The study population consisted of outpatients of the Pulmonology Department, Heart Institute, University of São Paulo School of Medicine, recruited over a 1-year period. Diagnosis of COPD was made according to American Thoracic Society (ATS) guidelines 9 : history of smoking (>10 pack/year), a post-bronchodilator forced expiratory volume in 1 s (FEV1) <80% of predicted, and a forced expiratory volume at first second (FEV1)/forced vital capacity (FVC) ratio <0.7. The moderate group was defined 9 as having FEV1 among 35–50% predicted and the severe group FEV1 <35%. All patients had stopped smoking for 1–38 years, were in a stable condition, as assessed by stability in blood gas values, and were free from exacerbations in the 4 weeks prior to study. Patients with a post-bronchodilator (200 mcg of salbutamol) change in FEV1 >15% or >200 ml were excluded from the study, as were patients with other organ failure, cancer, or with an inability to cooperate. All patients received regular treatment with inhaled bronchodilators and inhaled steroids according to current guidelines for their disease stage. 10 The control group consisted of healthy individuals without respiratory symptoms or impairment in activities of daily living. They had normal spirometry, FEV1/FVC > 0.7 and FEV1 > 80% predicted, and were recruited during the study period.
Study design
During a period of 2 weeks, patients followed the protocol on four nonconsecutive days. On the first day after providing informed consent, patients underwent a physical examination; on the second day, conventional pulmonary function tests were performed; on the third day, assessment of the diaphragmatic movement was undertaken; and on the fourth day, breathing pattern, thoracoabdominal motion, and metabolic gas analyses were conducted. Subjects participated in DB practice each day of the protocol. In the semi-recumbent position (45°), the subjects were instructed to inspire predominantly with abdominal motion, while reducing upper rib cage motion. Respiratory control was achieved by encouraging deep, slow inspiration followed by gentle expiration without pursed lips. Tactile feedback was given with one of the therapist’s hands on the patient’s abdomen. In each session, three sets of 20 DB were performed and the physiotherapist was satisfied with the DB pattern if this pattern was associated with a significant increase in abdominal tidal excursion, while rib cage excursion was greatly reduced. 5 Patients were encouraged to practice DB at home. Individuals in the control group underwent the same procedures as those of patients with COPD.
Lung function
Spirometry and lung volumes were assessed according to ATS and European Respiratory Society (ERS) guidelines. 11 Dynamic lung volumes were calculated with a flow spirometer (Puritan-Bennett PB 100, Pleasanton, CA, USA). Predicted values were used according to Knudson et al. 12
Respiratory muscle function
Maximal inspiratory (MIP) and maximal expiratory (MEP) pressures were measured at the level of functional residual capacity, 11 using a respiratory module system (Marshall Town—Instrumentation Industries, Bethel Park, PA, USA).
Diaphragmatic movement
Diaphragmatic movement (DM) was measured using chest radiography under full inspiration and expiration, with the patient in the orthostatic position. 13 We performed two radiographic exposures of the same film in posterior–anterior and lateral view. Images were digitalized by a computerized system. We used software (Image Tool for Windows, version 1.28) to calculate the area between the right dome of the diaphragm in full inspiration and expiration using a posterior–anterior view (DMPA) and the area between the two domes in lateral view (DML) expressed in square centimeters. 14 Calibration of the computer software was made based on a 5-cm line.
Breathing pattern, thoracoabdominal motion, and exhaled gas analysis
Breathing pattern and thoracoabdominal motion were recorded using a respiratory inductive plethysmography system (Respitrace®, Noninvasive Monitoring Systems, Miami, FL, USA). The least squares method calibration technique was used. 15 During the study, tidal volume (V T) in milliliters (ml), respiratory frequency (f) in breaths per minute (bpm), inspiratory and expiratory times (T I,T E) in seconds (s), fractional inspiratory time (T I/T TOT), mean inspiratory flow (V T/T I) in milliliters per second (ml/s), percentage of abdominal contribution to tidal volume (%Ab), and ratio of the maximum compartmental amplitude to tidal volume (MCA/V T) were calculated continuously. After obtaining the curves corresponding to V T, we drew a line at the level of the end expiration. The volumes measured above this line were termed as volume of expansion (V Exp; Figure 1 ). Ventilatory variables were obtained breath-by-breath using a gas metabolic analysis system with a pneumotachograph (Cardiopulmonary Exercise System CPX, Medical Graphics Corporation, St Paul, MN, USA) by means of a mouthpiece and nose clip. The system was calibrated before measurements using the manufacturer’s recommended procedures. Minute ventilation (VE) in liters per minute (L/m), ventilatory equivalents (VE/VCO2, VE/VO), dead space ventilation (VD/VT), and oxygen saturation (SpO2) were measured. The percentage of ventilatory reserve (%RVE) was calculated using VE and maximum voluntary ventilation (MVV), according to RVE% = (1 − (VE/MVV) × 100.

Respitrace® record—patient responder to diaphragmatic breathing. Black line represents expiratory rest level.
An A-B-A design was adopted for the study. The subjects were comfortably placed in a semi-recumbent position, and following stabilization, breathing at rest was measured for 4 minutes (phase A), 2 min of DB (phase B) and 4 min of quiet breathing (phase A). The modified Borg scale (0–10) 16 was used to assess the feeling of breathlessness in the first minute, after the DB period and at the last minute of the study.
Statistical analysis
Data are presented as means and standard deviations (SDs). One-way analysis of variance (ANOVA) and Kruskal-Wallis one-way analysis of variance were utilized to determine the differences between the groups. A Tukey test and Dunn’s method were used for multiple comparisons. Two-way repeated measures analysis of variance was used to test the differences between the phases. A post hoc test was added when indicated by ANOVA interaction. All the results of multiple comparisons were corrected using the Bonferroni test. Pearson’s coefficient was obtained to assess the correlation between variables; p < 0.05 was considered statistically significant.
Results
The study included 29 COPD patients divided into two groups: a moderate COPD group (n = 14) and a severe COPD group (n = 15), as well as a control group with 15 healthy subjects. Anthropometric data, lung, and respiratory muscle function are illustrated in Table 1 . A significant difference between the groups was found in body mass index, which was significantly lower in severe COPD patients. Mean functional pulmonary values between groups were different, whereas respiratory muscle function was similar in COPD and control groups. DM was significantly lower in both COPD groups than in the control group.
Anthropometric data, lung and respiratory muscle functions a

M/F: male/female, BMI: body mass index, COPD: chronic obstructive pulmonary disease, FEV1: forced expiratory volume in 1 s, FVC: forced vital capacity, MIP: maximal inspiratory pressure, MEP: maximal expiratory pressure, DMPA: area between the right dome of the diaphragm in full inspiration and expiration using radiographic exposures in posterior–anterior view, DML: area between diaphragmatic domes using radiographic exposures in lateral view.
a Values are mean ± SD.
b p < 0.05 versus control group.
c p < 0.05 versus moderate group.
DB significantly increased the tidal volume, mean inspiratory flow, and reduced the respiratory frequency of all groups, resulting in elevated V E and SpO2 with reduced V D/V T and VE/VCO2. Patients were able to perform DB from the longest movement of the abdominal wall with a tendency toward thoracoabdominal asynchrony and the presence of dyspnea (Table 2 ).
Breathing pattern, thoracoabdominal motion, ventilation and dyspnea during quiet (phase A) and diaphragmatic breathing (DB) a
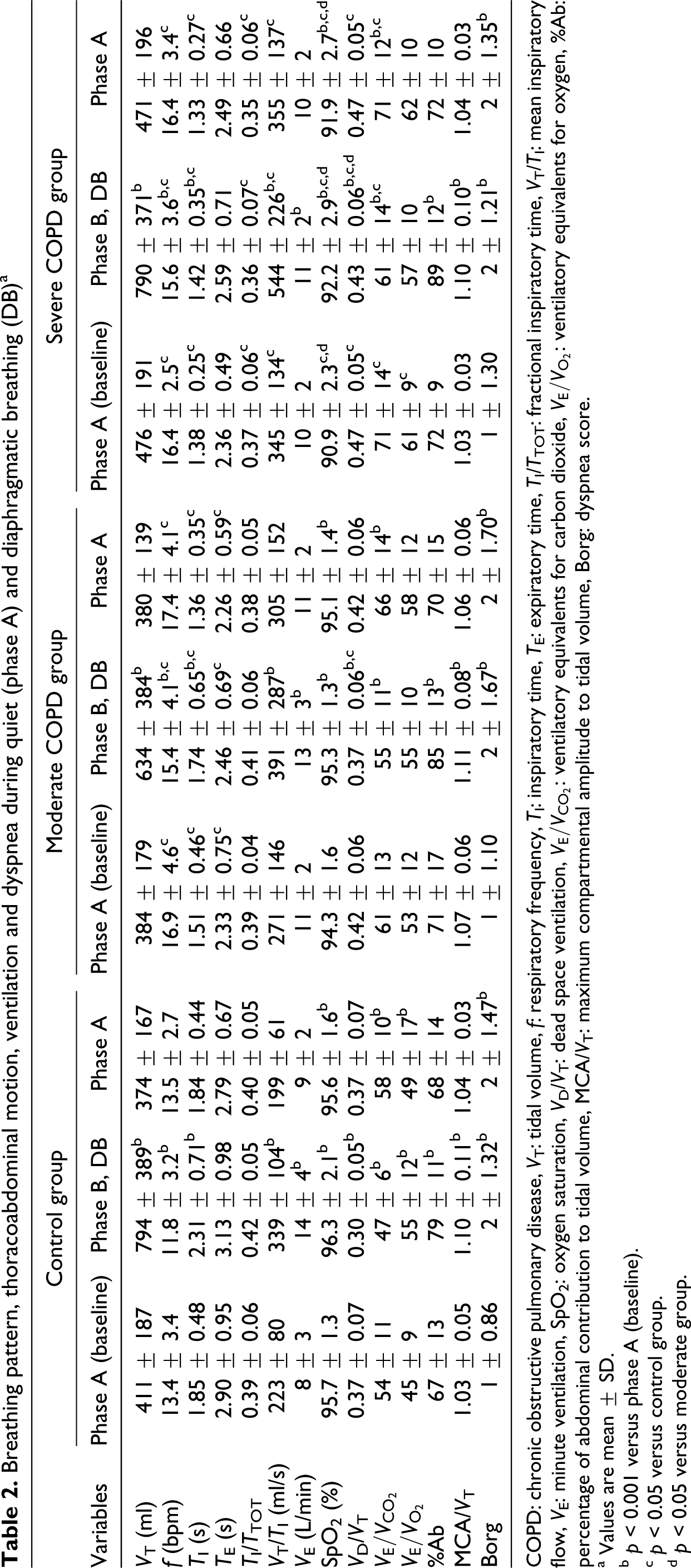
COPD: chronic obstructive pulmonary disease, V T: tidal volume, f: respiratory frequency, T I: inspiratory time, T E: expiratory time, T I/T TOT: fractional inspiratory time, V T/T I: mean inspiratory flow, V E: minute ventilation, SpO2: oxygen saturation, V D/V T: dead space ventilation, VE/VCO2: ventilatory equivalents for carbon dioxide, VE/VO2: ventilatory equivalents for oxygen, %Ab: percentage of abdominal contribution to tidal volume, MCA/V T: maximum compartmental amplitude to tidal volume, Borg: dyspnea score.
a Values are mean ± SD.
b p < 0.001 versus phase A (baseline).
c p < 0.05 versus control group.
d p < 0.05 versus moderate group.
A total of 19 subjects with COPD (9 moderate and 10 severe) performed DB with coordinated thoracoabdominal motion (MCA/V T < 1.10), 17 and no statistically significant differences were observed between V Exp and V T during DB. However, 10 patients (5 moderate and 5 severe) had asynchronous thoracoabdominal motion during DB (MCA/V T > 1.10). 17 The curves referring to DB for those patients showed oscillations of end-expiratory level (Figure 2 ) and the V Exp was statistically lower compared with V T. The increase of V T induced by DB above the resting expiratory level had a positive correlation with diaphragmatic mobility, SpO2 and %RVE, and a negative correlation with dyspnea index and MCA/V T (Table 3 ).
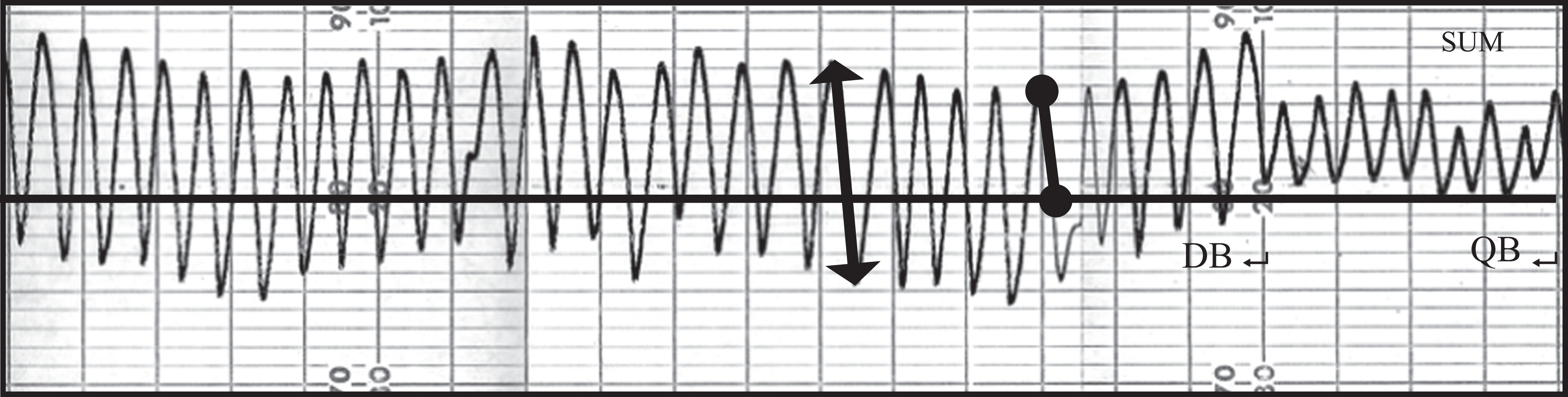
Respitrace® record–patient nonresponder to diaphragmatic breathing (DB). Black line represents expiratory rest level. Note. V Exp (black circle) and V T (line arrow). QB: quiet breathing.
Efficiency of diaphragmatic breathing and characteristics of patients with COPD at rest

▵V T: variation of volume between V T phase A (baseline) and V Exp DB, DMPA: area between the right dome of the diaphragm in full inspiration and expiration using radiographic exposures in posterior–anterior view, DML: area between diaphragmatic domes using radiographic exposures in lateral view, Borg: dyspnea index, SpO2: oxygen saturation, MCA/V T: maximum compartmental amplitude to tidal volume, %RVE: percentage of ventilatory reserve, r: correlation coefficient.
Following this analysis, we defined individuals who showed DB with higher V Exp and thoracoabdominal coordination as responders (Figure 3 ). The characteristics of the responder (R) and nonresponder (NR) groups showed statistical differences in inspiratory muscle strength and dyspnea scores. The NR group showed lower diaphragmatic mobility and lower muscular expiratory strength, but not at statistically significant levels. During DB the NR group showed a significant increase in the f, dyspnea scores, V D/V T and VE/VCO2 (Table 4 ).

Tidal volume (V T) and expansion volume (V Exp) in patients responders (continuous line) and nonresponders (chain line) to diaphragmatic breathing (DB).
Characteristics of patients in the responder and nonresponder groups a

BMI: body mass index, FVE1: forced expiratory volume in 1 s, DMPA: area between the right dome of the diaphragm in full inspiration and expiration using radiographic exposures, DML: area between diaphragmatic domes using radiographic exposures in lateral view, MIP: maximal inspiratory pressure, MEP: maximal expiratory pressure, Borg: dyspnea score, f: respiratory frequency, MCA/V T: maximum compartmental amplitude to tidal volume, V D/VT: dead space ventilation, VE/VCO2: ventilatory equivalents for carbon dioxide.
a Values are mean ± SD.
b p < 0.05 versus responder group.
Discussion
Our study shows that DB altered the respiratory pattern, increasing the tidal volume and reducing the respiratory frequency. This technique resulted in increases in V E and in gas exchange, as shown in the reduction in V D/V T and VE/VCO2 and the increase in SpO2. These data suggest that the increase in V E occurred in the alveolar zone.
Previous studies showed the capacity of patients with COPD to adopt a breathing pattern with increased V T and lower f, increasing ventilatory efficiency. 6,7
During the exercise, the displacement was predominantly abdominal with some patients showing asynchronous thoracoabdominal movement and a greater sensation of perceived exertion. The mechanisms associated with a greater sensation of perceived exertion 7 and mechanical inefficiency 5 during DB seem to be associated with an increase in tension–time index and pulmonary hyperinflation with consequent diaphragmatic flattening. 9 In the present study, the technique was used with low dyspnea ratings and thoracoabdominal coordination in several patients. We believe that preserved respiratory muscle strength and adaptive mechanisms in the diaphragm, such as muscle remodeling, 18 change in the fiber composition, 19 reduction in the number of sarcomeres in series, 20 and maintenance of diaphragmatic geometry 21 contributed to the maintenance of diaphragmatic excursion efficiency, contributing to a suitable balance between developed work and reserve of strength.
The capacity for increase in V T has been considered a marker of DB efficiency in patients with COPD, 5,22 and it seems to have little relation to the degree of airway obstruction. 22 Our data support this hypothesis, because the increase of V T in moderate and severe groups was similar during the technique in relation to phase A (baseline). Thus, FEV1 may not be the best parameter for identification and stratification of patients as candidates for performing DB.
We identified an R group who showed increased tidal volume with synchronous thoracoabdominal movement during DB. These patients were less hyperinflated and had a higher inspiratory strength than those in the NR group. The absence of differences between V T and V Exp suggests the capacity of these patients to use their inspiratory reserve volume. On the other hand, some patients performed the technique with asynchronous thoracoabdominal motion. These patients needed to recruit the expiratory reserve volume to try to raise V T, which means an incapacity for ventilatory increase above functional residual capacity. In these subjects, V Exp was significantly lower and had higher scores for dyspnea than responders during the exercise. This kind of DB breathing execution pattern can explain the results by Pasto et al. 23 who showed that although DB corresponds, in general, to a greater use of the diaphragm, it does not lead to greater ventilatory effectiveness. Probably the relaxation of the abdominal muscle prior to the onset of diaphragmatic contraction could be responsible for further expansion of abdomen during the technique in these patients.
Chest wall movement seems to be closely linked to DB efficiency, which is in accordance with what Cahalin et al. 22 have pointed out. Abnormalities in the coordination between the thorax and abdomen are related to an increase in the respiratory muscle load and are considered precursors of muscle fatigue. 24 Gosselink et al. 5 reported that DB may not be the best choice as a ventilatory strategy for COPD patients with hyperinflation and mechanical inefficiency of the diaphragm. Our data confirm that patients with preserved respiratory muscular strength and diaphragmatic mobility, lower levels of dyspnea, thoracoabdominal coordination within normal limits, oxygenation, and adequate ventilatory reserve may be considered the best candidates to perform DB.
The presence of these factors may indicate the likelihood of benefits from the technique for these patients, despite the demand inherent in its execution. In conclusion DB is a ventilatory strategy able to promote an increase in pulmonary ventilation in COPD patients with preserved respiratory mechanics. Thus, the inclusion of DB as a ventilatory strategy in a self-management program may produce positive effects in these patients.
Limitations and comments
The method of choice for evaluating the diaphragmatic mobility was based on the acquisition of images of anatomical structures and then on the calculation of its area. Some factors may interfere with this kind of investigation, such as technical variations during the radiographic exposure, the posture of the subject, the degree of hyperinflation and active contraction of inspiratory and expiratory muscles during MIP and MEP maneuver, and also the intensity of stimulus applied to the patient by the examiner during the maneuver. 25 To minimize such problems, the radiographic technique was standardized, and examinations were always performed by the same examiner. This evaluation was chosen because it is easy to perform and understand and provides an indirect measure of the degree of hyperinflation; it is also noninvasive and inexpensive.
The high values of V E observed in the groups might be attributable to the use of a mouthpiece and nasal clip which promotes an increase in V T and a decrease in f, 26 which may influence ventilatory response. This effect, however, was common to all subjects who participated in the study. Several patients reported that perceived exertion during the phases of the study was mainly related to these devices rather than solely to DB itself. Thus, these data must be considered with caution.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declared no conflicts of interest.
