Abstract
Patients with chronic obstructive pulmonary disease (COPD) and chronic heart failure (CHF) commonly suffer from exertional symptoms of breathlessness and fatigue. The similar systemic manifestations of the conditions, including skeletal muscle dysfunction, are a major contributing factor to the limitation in exercise capacity. A period of exercise training has been shown to improve exercise performance and health-related quality of life for both conditions. Exercise training is a key component of pulmonary rehabilitation (PR) which is now a standard of care for patients with COPD and is symptom based. Although it may be assumed that patients with CHF could be incorporated into cardiac rehabilitation, this is predominantly a secondary prevention programme for patients who are largely asymptomatic. It has been shown that patients with CHF can be successfully trained together with patients with COPD by the same therapists within PR. There are comparable outcome measures that can be used for both COPD and CHF. Many patients with CHF still do not have access to an exercise rehabilitation programme and incorporating them into the PR model of care could be one solution. This article reviews the (1) similar symptoms, mechanisms and consequences between COPD and CHF, (2) rationale and evidence for exercise training in CHF, (3) model of PR, (4) safety of exercise training in CHF, (5) evidence for combined exercise rehabilitation for CHF and COPD, (6) adaptations necessary to include patients with CHF into PR, (7) the chronic care model and (8) summary.
Introduction
Chronic diseases are the leading cause of death worldwide, representing 60% of all deaths according to the World Health Organization. 1 Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death worldwide, and chronic heart failure (CHF) has a 5-year mortality similar to common cancers. 2 Both COPD and CHF exert a large and similar burden on health care resources. 3,4 In summary, they are common diseases, with a high mortality, morbidity and socioeconomic burden, making an important target for any health care provider. This article describes how one aspect of their management namely exercise rehabilitation could potentially be combined.
Similar symptoms and mechanisms between chronic obstructive pulmonary disease and chronic heart failure
Exertional breathlessness and fatigue are common problems in patients with chronic obstructive pulmonary disease (COPD) and chronic heart failure (CHF). 5,6 Typically patients become less active to avoid these symptoms leading to deconditioning which further worsens the symptoms. This reduction in activity can lead to loss of confidence, depression, loss of work or inability to perform hobbies and social isolation, resulting in substantial disability.
In both conditions, the degree of primary organ impairment was found to correlate poorly with exercise capacity, particularly in severe disease, 7–9 and investigation began into other contributing factors. In addition to shortness of breath, leg fatigue was also found to be a common limiting symptom in patients with cardiopulmonary diseases. 10 It is now recognized that there are similar and important secondary pathological alterations in COPD and CHF including skeletal muscle dysfunction, mood disturbances, osteoporosis, hormonal alterations and anaemia, many of which contribute to exercise limitation and poor prognosis. 11,12 There is debate over the mechanisms behind these alterations, but inactivity, systemic inflammation, oxidative stress, poor nutrition, hypoxia and neurohormonal activation are all likely to contribute. 11,13,14
The skeletal muscles of locomotion are affected in performance, morphology and metabolism. 15,16 There is a reduction in muscle mass, strength and endurance in both diseases. 17–19 Muscle fibre type proportion is similarly altered with a relative decrease in type I oxidative fibres (less fatigable), with a reduction in oxidative enzymes and mitochondria. 20–24 Energy metabolism in the muscle is unfavourably altered in both conditions. 25,26 There has been one study directly comparing COPD and CHF skeletal muscle performance which confirmed a similar reduction in skeletal muscle strength, endurance and contribution to exercise intolerance. 27 The skeletal muscle abnormalities are thought to be a major contributor to the early lactate rise during exercise in COPD and CHF. 26,28 In addition to the negative effects on function, quadriceps weakness is also associated with a poor prognosis. 29,30 The skeletal muscle alterations are at least partially reversible with exercise training, making them an area of interest for both conditions. 20,22
Exercise training as a strategy for CHF
Historically, bed rest was recommended for patients with CHF as a result of small observational studies 31 and concerns over safety probably slowed the development of exercise training as an available therapy. Studies of exercise training in CHF occurred later (1980s) 32 than in COPD (1960s). 33 One of the earliest and an influential study was a home-based training study with a crossover design. 34 Patients performed 8 weeks of cycling 3 times a week or 8 weeks of activity restriction in a random order. There were no adverse events with both peak exercise capacity and symptoms improved after exercise training. The first randomized controlled trial involving long-term training (over 1 year) confirmed these improvements in peak exercise capacity and health status with exercise training and also showed a reduction in hospitalisation and mortality. 35
There is now a body of evidence demonstrating that a period of exercise training is beneficial for patients with CHF improving exercise tolerance and health status 36–39 and may reduce mortality. 37,40 A further trial to highlight was a large (n = 2331) multicentre, randomized controlled trial of 3 months of exercise training followed by home-based training versus usual care with 3-year follow-up (HF-ACTION). 37 The results reported were a trend towards a reduction in mortality, but the improvements in walking distance and exercise capacity were small. It should be noted that interdisciplinary education and self-management were not included in the protocol.
Few studies in CHF have involved a practical service involving both exercise training and multidisciplinary education. Austin et al. conducted a randomized controlled trial of exercise rehabilitation and enhanced medical management versus enhanced medical management only and showed small improvements in walking distance and health status in the exercise limb. 41 Hospital admissions and days spent in hospital were also significantly lower in the exercise group. Pharmacological therapy was optimized during the study in both the limbs.
Exercise therapy for CHF is recommended in national and international guidelines, 42 but currently they are not as detailed as for COPD. 43 The updated American Heart Association guidelines for CHF state that ‘Exercise training is beneficial as an adjunctive approach to improve clinical status in ambulatory patients with current or prior symptoms of HF and reduced LVEF.’ 44 More comprehensive guidance on cardiac rehabilitation (CR) is provided by the recent position article by the European Association of Cardiovascular Prevention and Rehabilitation Committee (EACPRC) and heart failure is included in one of the seven groups targeted. 45 The components of CR include patient assessment, physical activity counselling, exercise training, diet/nutritional counselling, weight control management, lipid management, blood pressure monitoring, smoking cessation and psychosocial management. Despite the supportive evidence for exercise training in CHF, many patients may not have access to this as part of a comprehensive service. 46,47 Reimbursement is currently not provided for patients with CHF to undergo structured exercise training programmes in Northern America.
The model and patient population of pulmonary rehabilitation
Pulmonary rehabilitation (PR) is a therapy targeted at the secondary alterations (systemic manifestations) of COPD, aiming to improve the functional and psychosocial aspects of an individual. The key components of a rehabilitation programme are symptom directed (i.e. exertional dyspnoea) and include individually prescribed exercise training, multidisciplinary patient education, psychological support and self-management. 48 PR is an integral part of the clinical management of patients with COPD and has a strong, supportive evidence base demonstrating a reduction in dyspnoea, with improvements in exercise tolerance and health-related quality of life (HRQOL). 49,50 There is increasing evidence that the benefit extends to other chronic respiratory diseases, for example interstitial lung disease, bronchiectasis, asthma, with a similar rationale at targeting exertional dyspnoea and activity limitation. 48,51,52 Patients with pulmonary hypertension may also benefit from exercise training 53 and are starting to be referred for PR. Hence, the PR population is increasingly diverse.
It may seem logical to include patients with CHF to the existing service of CR. However, this service is primarily focused on secondary prevention. The traditional CR patients with post myocardial infarction, post coronary artery bypass surgery or percutaneous angioplasty, or post valvular surgery often have little limitation in functional status and patients with CHF share more in common with the PR population. 54 Currently many CR programmes exclude patients with CHF 47,55 in part because they are recognised as having different requirements and being complex.
So far COPD and CHF have been discussed as separate diseases, but the evidence shows that they often coexist (over 30% in some series). 56,57 The combination of COPD and CHF is associated with increased symptoms, further exercise intolerance and worse prognosis. 58,59 Patients with combined disease are currently undertaking CR and PR; CHF was known to be present in 15% of patients with COPD undergoing PR 60 and 19% patients with post coronary artery bypass graft surgery (CABG) had COPD. 61 Guidance for PR is starting to focus on the cardiovascular disease associated with COPD. 62 This furthers the rationale for a combined service that has the necessary expertise for both conditions (occurring separately or in the same patient). A potential strategy is to organize exercise rehabilitation services around a common disability (symptom based) rather than around the primary organ disease.
Safety and practical considerations for exercise training in CHF
There is now a body of evidence supporting exercise training as a safe therapy in CHF for stable disease. Fears of worsening left ventricular function have not been founded and beneficial left ventricular remodelling is more likely to occur, particularly with aerobic training. 63,64 A randomized controlled trial reported no increase in adverse events with exercise training (CHANGE) 65 supported by the findings of a Cochrane meta-analysis. 66 The HF-ACTION trial included New York Heart Association (NYHA) IV for the first time and patients with implantable cardioverter defibrillators (ICDs) with no increase in adverse events reported. 37
Similar to COPD, patients with CHF should be optimally medically managed prior to enrolment to exercise rehabilitation. The underlying cause of the heart failure should be addressed (e.g. ischaemia, valvular lesions, arrhythmias and hypertension) and pharmacological management optimized; typically a combination of an angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB), β-blockade (e.g. metoprolol, bisoprolol or carvedilol), spironolactone and loop diuretics. There should be specific investigation and management of the secondary alterations and comorbidities such as COPD, diabetes and renal dysfunction. Electrolyte imbalances occur commonly with diuretic therapy, particularly hypokalaemia, and should be corrected. There are cardiac-specific contraindications to exercise training including moderate-to-severe aortic stenosis, a recent myocardial infarction (<4 weeks), unstable angina, uncontrolled blood pressure, uncontrolled atrial or ventricular dysrhythmias. 67 At least one member of the rehabilitation team should probably be trained in advanced life support if a cardiopulmonary resuscitation team is not readily available at the location. Defibrillation equipment should be available.
A maximal exercise test should be performed with an exercise ECG. Commonly this is undertaken by a laboratory cardiopulmonary exercise test, but an incremental shuttle walk test (ISWT) with telemetry could be used. The American College of Sports Medicine (ACSM)
68
recommend exercise discontinuation for diastolic BP ≥110 mm Hg decrease in systolic BP >10 mm Hg significant ventricular or atrial dysrhythmias 2nd or 3rd degree heart (atrioventricular [AV]) block signs/symptoms of exercise intolerance, including angina, marked dyspnoea, and ECG changes suggestive of ischaemia
Is combined exercise rehabilitation for COPD and CHF feasible and effective?
In order to implement combined programmes, the complete process of PR (assessment, outcome measures and the programme components) need to be applied to CHF. An assessment of dyspnoea, functional exercise performance and health status is standard and can inform both individual progress and program quality.
The Medical Research Council (MRC) dyspnoea scale is commonly used to screen for referral to PR typically included with grades III–V. The scale was successfully applied to patients with CHF, demonstrating construct validity with decreasing exercise performance and health status with increasing (worsening) grade comparable to COPD. 69
Outcome measures used for the assessment of pulmonary rehabilitation
Exercise tests
The gold standard measurement of exercise capacity is peak oxygen consumption. A maximal, incremental, symptom-limited cardiopulmonary test with expiratory gas analysis can provide this measurement, as well as useful information about the precise limitation to exercise. Typically tests are performed using a cycle ergometer or a treadmill. Both incremental and constant power/speed tests can be performed using the same equipment.
Field testing has been used in COPD and CHF with the benefit of not requiring expensive laboratory equipment or the necessary expertise. The 6-minute walk test (6MWT) 70 is completed over a 30-m flat course. Patients are asked to walk as far as they can for 6 min and the result is presented as the distance walked which can be reported against normal reference values. 71 Encouragement during the test is standardized to minimize its influence on test results.
The ISWT is a symptom limited, externally paced test, conducted along a 10-m course. 72 The walking speed increases every minute until the patient is too breathless or fatigued to continue or can no longer maintain the required speed. Patients are given standard instructions on how to complete the test. The ISWT performance has been shown to have a graded cardiovascular response throughout. 73 In patients with heart failure, there is a high correlation between the peak oxygen consumption (peak VO2) derived from the ISWT performance and a maximal, incremental cardiopulmonary exercise test on a treadmill, also shown in patients with COPD. 72,73 However, there was no correlation between the peak VO2 from the maximal treadmill test and that achieved on the 6MWT in CHF. The ISWT also has a higher correlation with mortality than the 6MWT distance in patients with CHF. 74
Both the 6MWT and the ISWT are repeatable, valid and responsive after a familiarisation test 70,75,76 and can be used for the exercise assessment and for setting the exercise prescription for PR.
The endurance shuttle walk test (ESWT) is a constant speed endurance test which is externally paced and uses the same course as the ISWT. It has been validated in COPD 77 and is highly responsive to the effects of rehabilitation. 78
Health-related quality of life
HRQOL is recognized as an important outcome measure in both diseases. 79 For chronic heart failure, both generic questionnaires such as the Medical Outcomes Short Form 36 (SF-36) 80,81 and disease-specific questionnaires such as the Minnesota Living with Heart Failure questionnaire (MLWHFQ) 82 or Kansas City Cardiomyopathy Questionnarie (KCCQ) 83 have been reliably used. The disease-specific questionnaires tend to be more responsive than generic questionnaires. 84 The Chronic Heart Questionnaire (CHQ) 85 was developed by the same group as the Chronic Respiratory Questionnaire (CRQ) 86 and is identical except for one question. It has been found to be reliable, valid and responsive in stable heart failure. 85,87,88 A self-reported version of the CHQ has recently been found to be comparable with the interview-led version and also to be valid, repeatable and responsive. 89 It is therefore possible to compare disease-specific HRQOL between these two populations.
Are the recommendations for the exercise training programme similar between COPD and CHF?
The current evidence for the exercise training component is very similar between the two groups of patients (Table 1 ) including high-intensity aerobic lower limb training, frequency and duration of programmes. The addition of strength training has been examined in both groups. 91,92 Muscle strength was increased in the combined training groups compared to aerobic training alone but so far has not translated into further improvements in exercise performance or health status than aerobic training alone. A meta-analysis reported that high-resistance training is likely to be safe in CHF, 93 whereas caution above moderate resistance training has always been advised. In CHF, aerobic training alone appears to result in beneficial left ventricular remodelling, whereas this was not seen with combined training. 64
Comparison between content of exercise training programmes between COPD and CHF
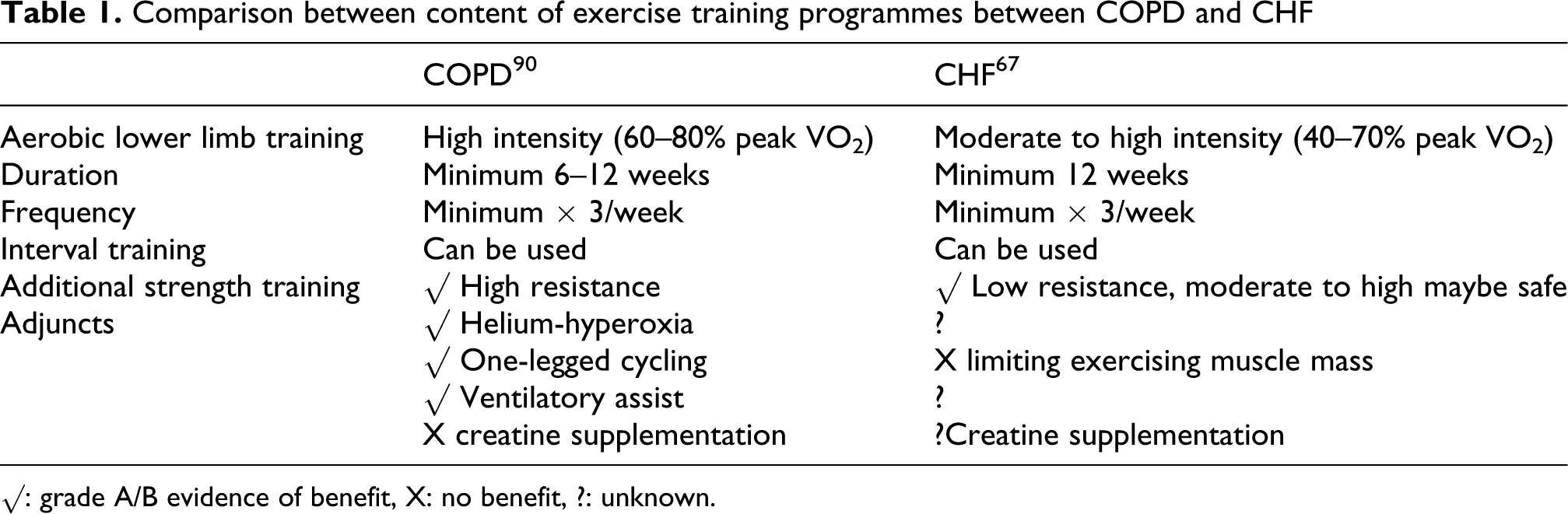
√: grade A/B evidence of benefit, X: no benefit, ?: unknown.
Interval training has been compared to continuous training and is tolerated and safe in both groups. A recent meta-analysis has shown no overall benefit with interval training compared to continuous training in patients with COPD. 94 There has been a positive randomized controlled trial for interval training in CHF, 95 but to date there is not enough data for a meta-analysis to be performed.
Creatine supplementation with exercise training has been found to have no effect on exercise performance in COPD and CHF. 96,97 The exception for the similarities in training programmes is in the use of training adjuncts for patients with COPD designed to relieve the ventilatory limitation to exercise particularly dynamic hyperinflation such as helium hyperoxia, 98 proportional assist ventilation 99 and single leg cycling. 100 The rationale for the use of these adjuncts in COPD is unlikely to apply in chronic heart failure. 101 However, few of these adjuncts are currently used in practical PR programmes.
Evidence for combined exercise rehabilitation for patients with COPD and CHF
Preliminary and anecdotal data supported the hypothesis that patients with CHF could gain comparable benefit from PR. Subsequently, the model of PR was successfully applied to patients with CHF in a randomized controlled trial of PR versus usual care. 102 Patients undergoing PR made significant improvements in exercise tolerance and HRQOL. A parallel, comparative observational study of PR was conducted between COPD and CHF. 102 The significant improvements with PR, in exercise tolerance and HRQOL, were not statistically different between the groups. However, there was a trend for the HRQOL improvements to be greater for COPD. Both groups could be successfully trained together, at the same time and location, by the same therapists.
The management of patients with COPD and CHF has also been successfully combined in a pilot study assessing a joint community-based exercise programme. 103
Potential adaptations of PR for the inclusion of patients with CHF
The exercise assessment with an exercise ECG is recommended for entry to exercise rehabilitation for patients with CHF. The PR team have considerable and valuable skills in dealing with the severely dyspnoeic patients, but some additional education may be necessary. Therapists should understand the symptoms and signs of an episode of decompensated heart failure similar to an exacerbation of COPD. Much of the education in PR is symptom based and generic. However, thought should be given to how to provide disease-specific education, either during the programme or by another health care professional (UK patients commonly have access to a heart failure or COPD specialist nurse). The delivery of disease-specific education is also relevant for the increasing number of patients with respiratory disease other than COPD referred for PR.
The benefit of self-management education is established in both COPD and CHF
104,105
with the use of action plans forming part of this.
106
PR aims to continue self-management learning. Some of the general principles apply to all chronic lung diseases and chronic heart failure, but the more detailed disease-specific information is unlikely to be able to be tackled in a mixed, group environment. Further evidence and guidance is necessary to understand the optimal delivery.
Chronic care model
Over a decade ago, a model of chronic care was described aiming to improve recognized deficiencies in health care for patients with chronic conditions. Delivery, system and design are just one component of the health system. Exercise rehabilitation forms part of the management for many chronic conditions with the broad aims of improving function and quality of life and/or mortality via secondary prevention. It may be that the overall design and delivery of these programmes would be more efficient and cost effective if they were categorized by symptoms or disability rather than being disease based. Instead of multiple individual disease-specific programmes, if exercise rehabilitation could be categorized broadly into two programmes, resources could be focused on delivery of these programmes widening patient choice, for example geography: hospital, community or home based and type: supervised or tele-health care, enabling access and compliance. A theoretical model is shown in Table 2 . Work is currently in progress integrating a chronic disease health care model between four different conditions including COPD and chronic heart failure. 107
A theoretical symptom-based model of care for providing exercise rehabilitation programmes
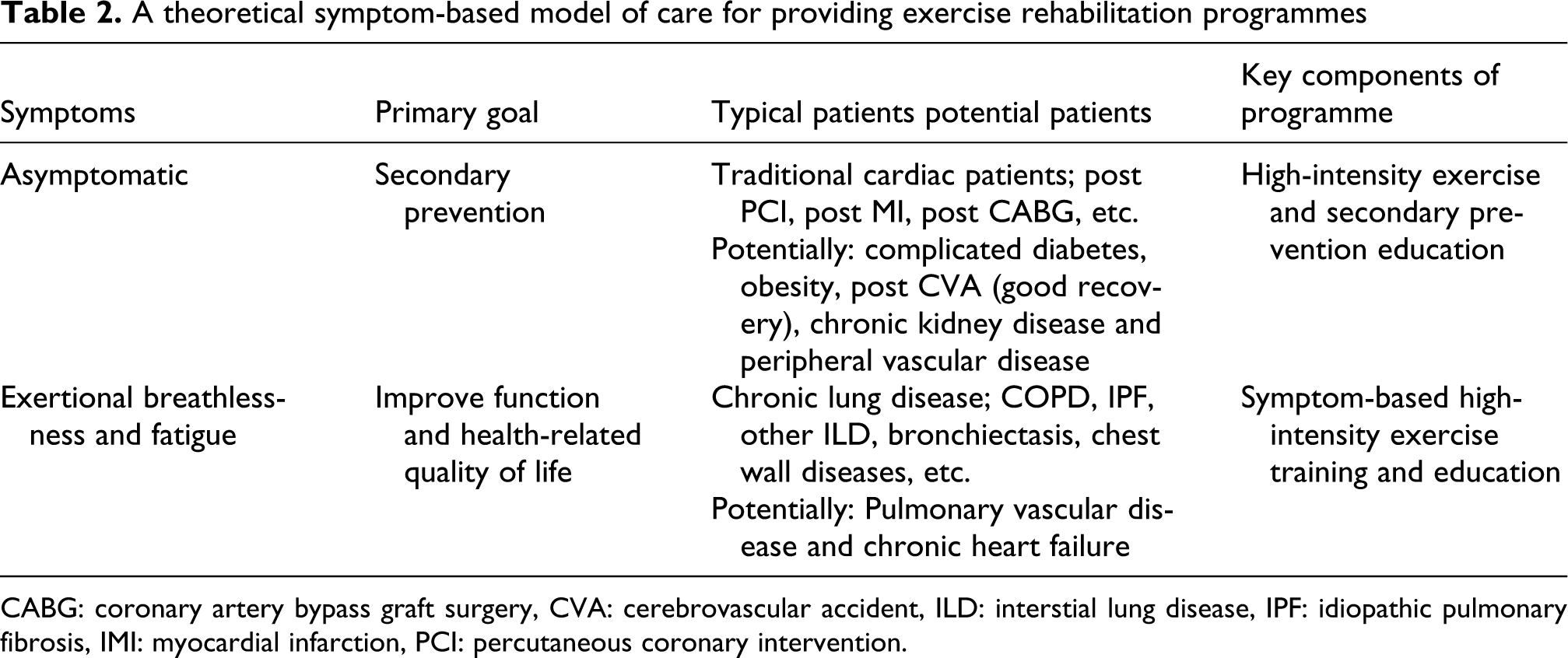
CABG: coronary artery bypass graft surgery, CVA: cerebrovascular accident, ILD: interstial lung disease, IPF: idiopathic pulmonary fibrosis, IMI: myocardial infarction, PCI: percutaneous coronary intervention.
Summary
The model of PR can be successfully applied to patients with CHF. Combined symptom-directed exercise rehabilitation is feasible and effective for patients with COPD and CHF. Further work is needed to assess sustainability and to assess whether the strategy offers economies of scale to both populations. Chronic disease models of care are under development and perhaps the strategies could focus on common symptoms and disabilities rather than being disease specific.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
