Abstract

Liraglutide, a glucagon-like peptide-1 receptor agonist (GLP-1 RA), has been used to treat patients with type 2 diabetes. Previous study showed that using liraglutide to treat type 2 diabetes patients with established ASCVD decreased adverse cardiovascular events and mortality compared with placebo. 1 However, the mechanism whereby liraglutide decreases atherosclerosis and mortality remains unclear.
MicroRNAs (miRNAs) are small non-coding RNA molecules that regulate post-transcriptional gene expression.2,3 Recently, miRNAs have been proposed as potential biomarkers or targets for treatment of cardiovascular diseases.4,5 To date, in vitro studies have demonstrated that liraglutide could prevent atherosclerosis through miRNA pathway.6–8 We hypothesized that patients treated with liraglutide may have alterations in serum miRNA expression that are associated with atherosclerosis via the AMPK signaling pathway. This suggests that miRNAs could be developed as a novel biomarker-guided therapy or as a targeted treatment for cardiovascular disease in the future.
This observational study was performed at the Faculty of Medicine Ramathibodi Hospital Mahidol university. The study enrolled patients with type 2 diabetes mellitus. Inclusion criteria for the liraglutide group were patients with established ASCVD or who were at high risk of atherosclerosis.
A 5-mL venous blood sample was collected from each participant before prescription of liraglutide (day 0). After liraglutide prescription, the participant was scheduled for follow-up at weeks four and 8. Drug compliance was monitored by telephone at weeks 2 and 6 after prescription.
During follow-up, patients were asked about drug compliance, and a venous blood sample was collected. The samples were analyzed for hs-CRP and NT-proBNP, IL-6 and miRNA analysis. We hypothesized that liraglutide improved the atherosclerosis risk in the patient though the AMPK signaling pathway. Thus, we selected miRNA, which was considered to be associated with the AMPK signaling pathway, for in vitro studies.9–12 We focused on miRNA144/145, miRNA-451a, miRNA33, and miRNA195. 13 Informed consent was provided by all participants after the study was explained to all of them. This study was approved by Instructional Review Board Faculty of Medicine Ramathibodi Hospital, Mahidol university. All methods were carried out accordance to Declaration of Helsinki.
Serum RNA extraction was performed using a Monarch Total RNA Miniprep kit (New England Biolabs, USA), in accordance with the manufacturer’s instructions. The miRNA-451a primer and the miRCURY LNA RT kit were purchased from Qiagen and were used for cDNA synthesis. Serum miRNA expression was determined using ddPCR, and expression levels were quantified using QuantaSoft software (Bio-Rad, USA).
Sixteen participants with type 2 diabetes mellitus who received liraglutide for 8 weeks were evaluated. The mean age of the study population was 62.3 ± 6.3 years, and 10 participants (62.5%) were men. The mean baseline hemoglobin A1c (HbA1c) was 8.5 ± 1.1% (range, 7.05–10.92 %). All the participants had established ASCVD.
Compared with pretreatment, mean serum high-sensitive C-reactive protein (hs-CRP) levels were not significantly different after treatment for 8 weeks (3.3 ± 4.4 mg/L vs 5.8 ± 17.3 mg/L, p = .3) (Figure 1). N terminal-pro B-type natriuretic peptide (NT-proBNP) tended to decrease from baseline to week eight in participants who received liraglutide, although the difference failed to reach statistical significance (410.13 ± 602.32 pg/mL vs 305.81 ± 485.35 pg/mL, p = .11). Serum interleukin (IL)-6 levels were measured, but there was no significant difference at week 8 compared with the baseline (4.83 ± 5.6 pg/mL vs 13.55 ± 21.82 pg/mL, p = .14). Serum miRNA451a level at baseline and 8 weeks (copy/µL).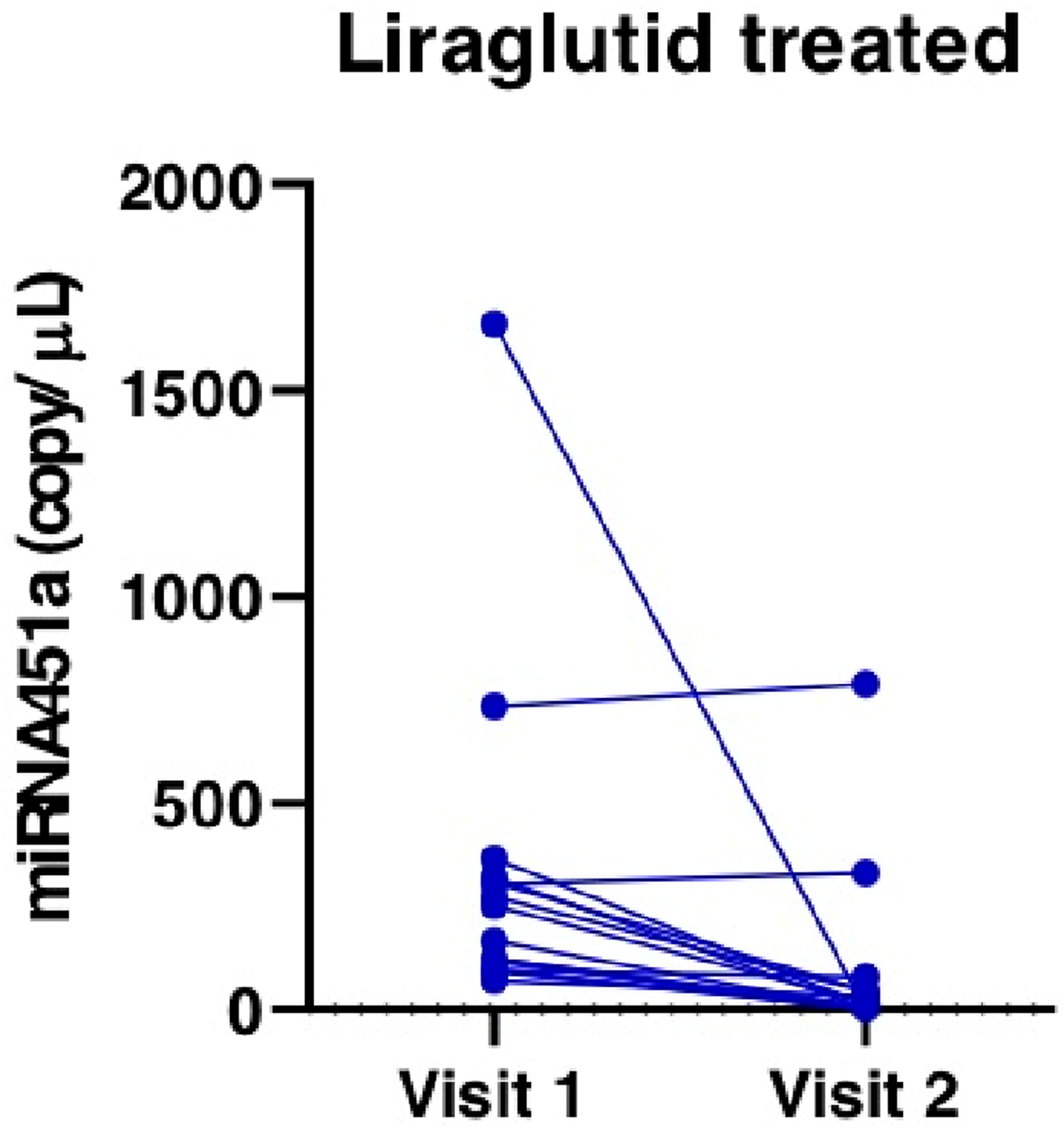
Of miRNA tested, only miRNA-451a was detected in patient sera. In most participants who were treated with liraglutide, serum miRNA-451a showed prominent expression (mean value, 318.59 ± 394.92 copy/µL). Serum miRNA-451a was reduced after treatment to 85.92 ± 203.97 copy/uL (p < .001). Only two participants had stable miRNA-451a expression compared with the baseline level.
Our work demonstrates that patients with type 2 diabetes mellitus and established ASCVD have significant miRNA-451a expression. Studies have shown that some agents may down-regulate miRNA-451a in diabetes, such as vitamin C supplementation in those with inadequate glycemic control. 14 Our study showed, for the first time, that liraglutide significantly down-regulated miRNA-451a, which could be one mechanism by which cardiovascular events are decreased in liraglutide-treated patients.
In conclusion, liraglutide treatment appears to down-regulate miRNA-451a in individuals with type 2 diabetes and future research is needed to understand whether this is linked to improved cardiovascular outcomes. If a link with cardiovascular events is established, miRNA-451a may become a helpful biomarker for the cardiovascular protective effects of liraglutide and other agents in this class.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NT-proBNP measurement equipment was supported by Roche Diagnostics (Bangkok, Thailand). The study was funded by a grant from Institutional Income, Ramathibodi Hospital, and The Heart Association of Thailand under the Royal Patronage of H.M. the King.
