Abstract
Introduction:
Patients with diabetes mellitus are known to carry an increased risk for surgical site infections and perioperative complications. The subcutaneous implantable cardioverter defibrillator is an established treatment option in patients at risk for sudden cardiac death especially with an increased risk for infection over time.
Methods and Results:
Forty-eight patients (mean age = 55.0 ± 21.3 years, 31.3% patients with diabetes mellitus, 75% male) who underwent consecutive subcutaneous implantable cardioverter defibrillator surgery between February 2016 and May 2019 were retrospectively analysed. Overall adverse events including relevant bleeding complications, any surgical wound problems and infections requiring reoperation or device malfunction were evaluated as primary combined safety endpoint. Patients with diabetes mellitus tended to be older with a higher body mass index compared to non-diabetes mellitus. Procedure duration and postsurgery hospital days were not different in diabetes mellitus versus non-diabetes mellitus patients. Analysis of the primary combined endpoint showed no significant difference but a trend towards higher event rates in the diabetes mellitus group (diabetes mellitus vs non-diabetes mellitus: 20% vs 12.1%, p = 0.119).
Conclusion:
Diabetes mellitus is a frequent and relevant variable in patients undergoing subcutaneous implantable cardioverter defibrillator implantation represented by 31.3% in this consecutive cohort. Our results suggest that diabetes mellitus is not associated with a prolonged hospital stay or increased rate of periprocedural adverse events.
Introduction
Totally subcutaneous implantable cardioverter defibrillator (S-ICD) has recently been established in clinical practice and represents a valid alternative to the transvenous ICD (TV-ICD), while being beneficial in avoiding complications associated with transvenous endocardial leads. The pulse generator of the S-ICD has a large volume in comparison to a TV-ICD, which requires the preparation of a larger device pocket and distinct surgical technique compared to the traditional TV-ICD. Initial data of real subcutaneous implantation technique showed high rates of complications, for example, infections or skin erosions.1–4 Although S-ICD has a lower risk of lead-related complications than TV-ICD, the incidence of non-lead-related complications was higher for S-ICD than for TV-ICD. Especially the rate of pocket infections in S-ICD procedures in the earlier studies appeared to be up to four-fold higher than reported for conventional TV-ICDs (0.7%–1.2%).5–8 Altered immune function in patients with diabetes mellitus (DM) is suspected to lead to an increased risk of infections. 9 In general, wound healing problems occur more frequently in patients with DM.10,11 Due to higher riks for long-term lead infection rates in DM patients, S-ICD implantation is favorable. It remains unclear if these patients are going to suffer more often from the previously reported pocket infection problems in short-term. The objective of this study was to further evaluate the role of DM in a consecutive group of patients undergoing S-ICD implantation surgery in our single-centre study with respect to the procedural short-term outcome, including peri- and postoperative complications.
Methods
Study design
From February 2016 to May 2019, patients undergoing first S-ICD implantation and S-ICD generator replacements at our centre were included in this retrospective observational study. The study was conducted in compliance to the Declaration of Helsinki, and the research protocol was approved by the Ethics Committee of the Medical Faculty of the University of Essen (Protocol Number 19-8716-BO).
Implantation procedures
S-ICD surgery was performed using the intermuscular two-incision technique under conscious sedation with local anaesthesia, creating an intermuscular pocket between the anterior surface of the serratus anterior and the posterior surface of the latissimus dorsi muscles instead of a subcutaneous pocket (Figure 1) as previously described.12,13
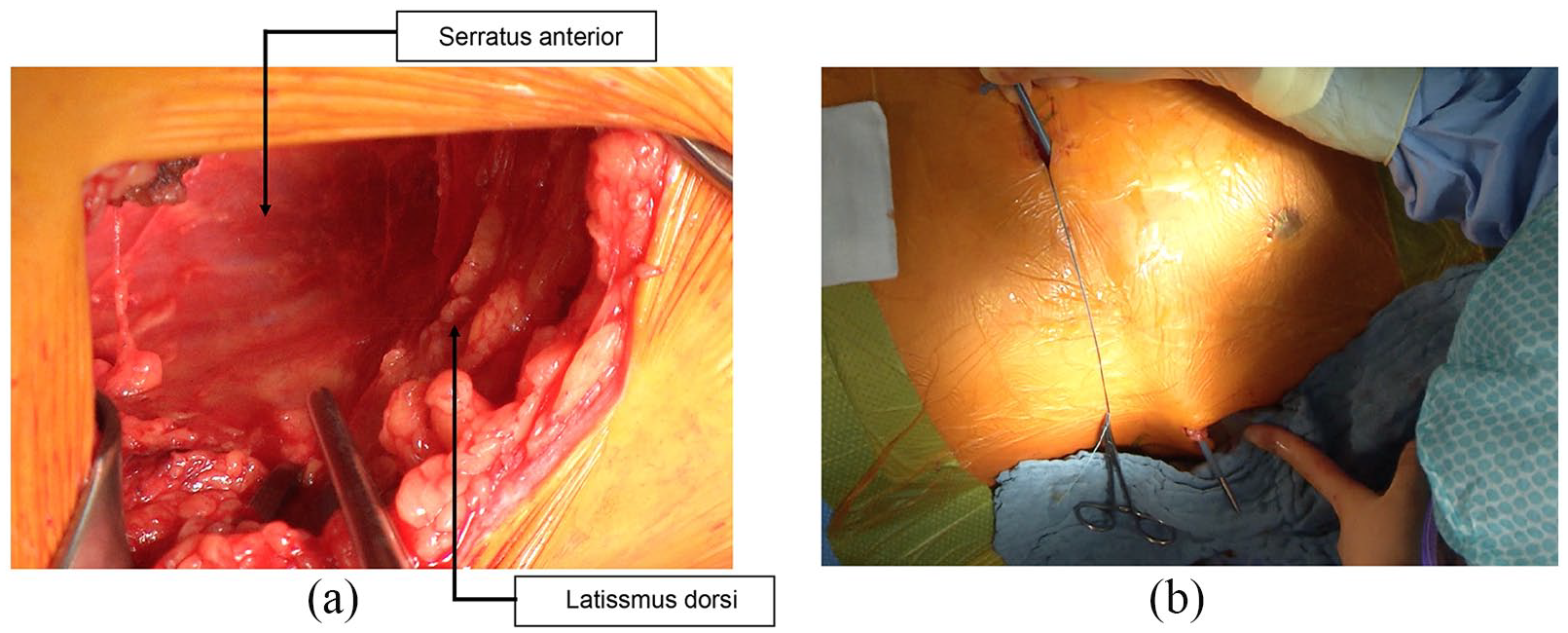
Representative pictures illustrating the implantation technique of the S-ICD: (a) superior view on the intermuscular generator pocket between the anterior surface of the serratus anterior muscle and the posterior surface of the latissimus dorsi muscle for the pulse generator and (b) tunnelling from xiphoid incision to the generator pocket with the tunnelling tool pre-loaded with an introducer sheet.
Five patients underwent generator replacement. In four out of five cases, a surgical intervention with creation of a new intermuscular device pocket was necessary according to current recommendations to improve patient comfort, cosmetic results as well as defibrillation efficacy. All patients received routine perioperative antibiotic prophylaxis (intravenous administration of 1,5 g cefazolin 15–30 min prior to the skin incision). All procedures were performed in a hybrid operating room without interruption of antiplatelet therapy. Novel oral anticoagulants (NOACs) were ceased at least 48 h before and restarted 24 to 48 h after the procedure independent of the presence of DM. In case of oral anticoagulation by vitamin K antagonist (VKA) use, oral anticoagulant (OAC) was continued with a target international normalized ratio (INR) between 2.0 and 3.0 on the day of surgery. Antisepsis procedures were performed immediately before surgery, with antiseptic solution successively applied to the skin. No local antibiotic pocket wash was performed. Skin closure was carried out by either polypropylene (Prolene) or polydioxanone (PDS) by Donati suture or intracutaneous suture technique. Regarding device programming, all patients had dual-zone programming as previously described. 2
Endpoints
We defined a primary combined safety endpoint including any bleeding complications according to International Society on Thrombosis and Haemostasis (ISTH) bleeding scale, any surgical wound problems, infections, adverse events requiring reoperation or device malfunction.
Secondary safety endpoints were divided into single endpoints: infectious complications (superficial infections, pocket infections and severe infections requiring complete device extraction), surgical wound problems (surgical wound dehiscence), device malfunction (lead or generator dislodgement, inappropriate shock therapies) requiring hospitalization, bleeding complications defined as major device-pocket haematomas requiring blood transfusion or device-pocket haematomas that required evacuation and resulting in prolongation (at least 24 h) of hospitalization, bleeding complications defined as minor pocket haematomas not requiring any treatment or prolonging hospitalization or subcutaneous haematomas with local ecchymosis, and death from any cause within 90 days.
Procedure-related endpoints
Procedure-related endpoints included the following: procedure duration (defined from the time of skin incision to the time of completion of skin closure) and length of postoperative hospital stay.
Secondary efficacy endpoints were defined with following points: successful defibrillation threshold testing, where a successful conversion of 50 Hz stimulation-induced ventricular fibrillation (VF) was defined as the delivery of 65 Joules (J) with any shock vector with a maximum output of 80 J, and appropriate shock therapy.
Postoperative evaluation and follow-up
Daily assessment of the device pocket and wound examination were performed until the patient was discharged from the hospital. Surgical wound infection was diagnosed by clinical assessment of usual infections signs like erythema, oedema, increased pain and heat. Measurements of inflammatory markers such as C-reactive protein (CRP) and white blood cell (WBC) were recorded. Leukocytosis was defined as an elevated WBC 10/nanolitre (nl). Temperature was daily taken with an ear thermometer. Follow-up visits were scheduled 4 weeks and 3 months after the procedure in our outpatient clinic. Patient follow-up comprised physical examination. During follow-up, adverse events and all relevant device parameters (battery, impedance, vector recognition, arrhythmia episodes and shock therapies) were recorded and documented.
Statistical analysis
Baseline characteristics, procedure-related data and procedure-related complications were extracted from a specifically designed database. Normal distribution was tested using a Shapiro–Wilk test. Continuous variables are expressed as mean ± standard deviation in case of normal distribution (compared with unpaired t test), and as median and interquartile range in the cases of other types of distribution (compared by using a Mann–Whitney U test).
Categorical variables are summarized as counts and percentages and compared using the chi-square test. For all analyses, a p value of <0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 24.0 (IBM SPSS, Chicago, IL, USA).
Results
Study population: overall patients and procedural characteristics
In our cohort, 15 (31.3%) of 48 patients had the diagnosis of DM. The diagnosis of DM was based on the initial questionnaire (use of antidiabetic medication or insulin treatment). The baseline clinical characteristics, prevalence and distribution of clinical data are provided in Table 1, illustrating the overall cohort and the subgroups of patients with DM versus non-DM. The overall mean age at implantation was 55.0 ± 21.3 years, while the majority of patients were male (75%). The mean ejection fraction (EF) was 29.0% ± 12.0%. Of the 48 patients, 46 (95.8%) had an underlying structural heart disease, while 2 (4.2%) patients were implanted after survived sudden cardiac death (SCD) due to idiopathic VF with no overt heart disease. Indications for S-ICD implantation were mainly for primary prevention in 38 patients (79.1%). Seven patients had a history of atrial fibrillation (AF; 14.6 %). Overall, the majority of patients either were receiving antiplatelet (85.4%) therapy or were on oral anticoagulation (31.3%) (Table 2).
Baseline demographics: clinical data and characteristics.
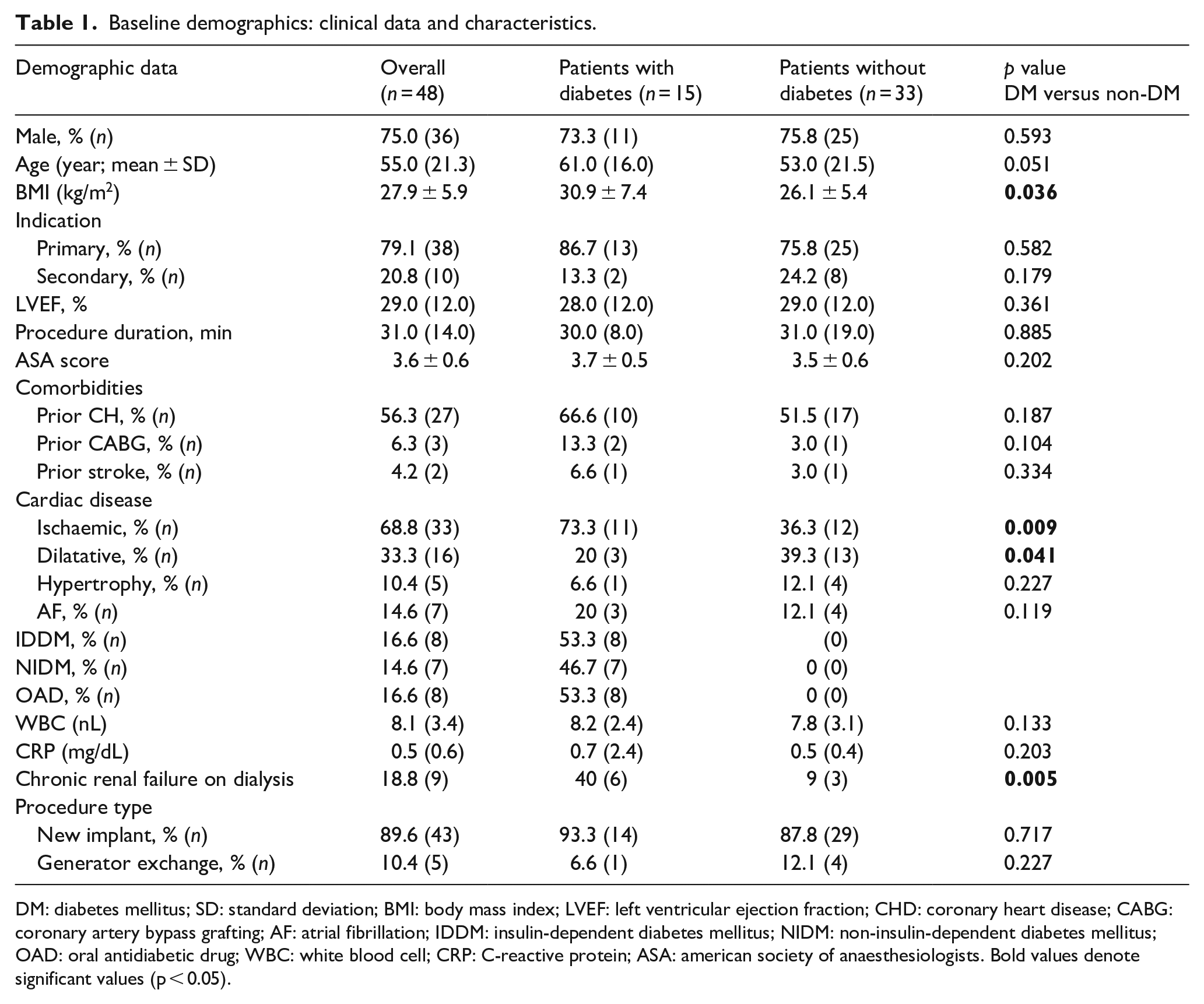
DM: diabetes mellitus; SD: standard deviation; BMI: body mass index; LVEF: left ventricular ejection fraction; CHD: coronary heart disease; CABG: coronary artery bypass grafting; AF: atrial fibrillation; IDDM: insulin-dependent diabetes mellitus; NIDM: non-insulin-dependent diabetes mellitus; OAD: oral antidiabetic drug; WBC: white blood cell; CRP: C-reactive protein; ASA: american society of anaesthesiologists. Bold values denote significant values (p < 0.05).
Peri-interventional anticoagulation.
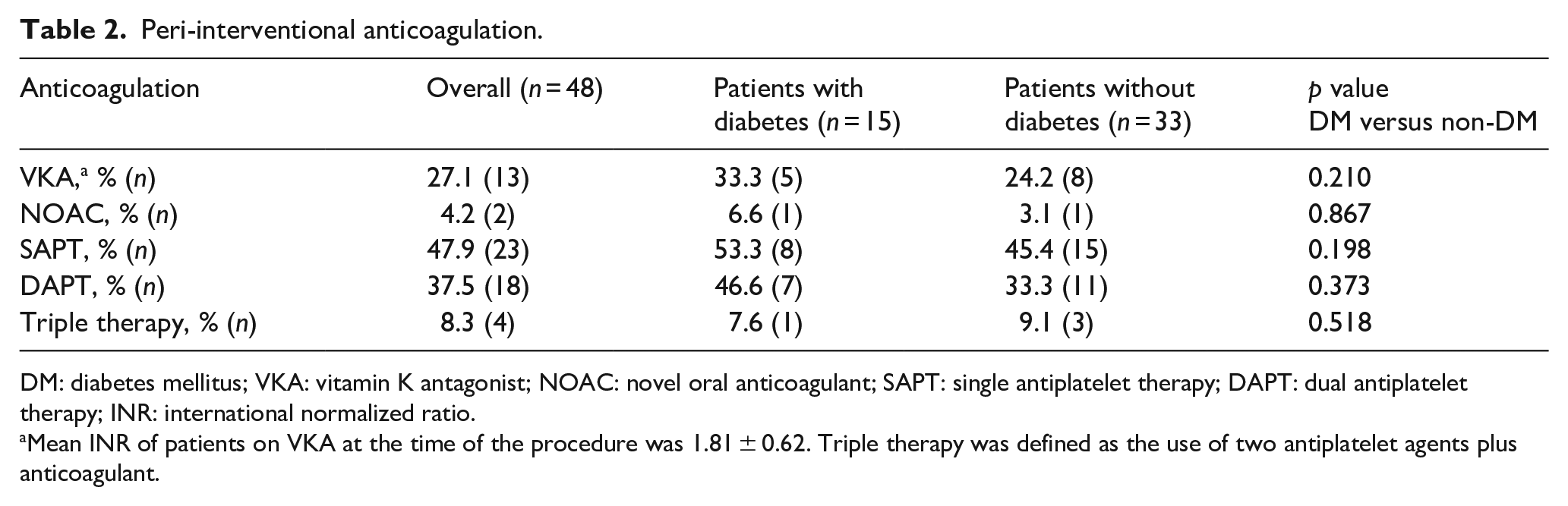
DM: diabetes mellitus; VKA: vitamin K antagonist; NOAC: novel oral anticoagulant; SAPT: single antiplatelet therapy; DAPT: dual antiplatelet therapy; INR: international normalized ratio.
Mean INR of patients on VKA at the time of the procedure was 1.81 ± 0.62. Triple therapy was defined as the use of two antiplatelet agents plus anticoagulant.
Diabetes versus non-diabetes patients
Patient characteristics comparing patients with DM versus without DM are listed in Table 1. There were no significant differences regarding gender and age; 73.3% of patients with DM and 75.8% of patients without DM were male, respectively. Indications for S-ICD therapy were 86.7% primary prevention in DM patients, whereas in the non-DM group, 75.8% of cases were primary prevention and 24.2% secondary prevention. Mean left ventricular ejection fraction (LVEF) was comparable between both groups (DM vs non-DM: 28.0% ± 12.0% vs 29.0% ± 12.0%; p = 0.361). Prevalence of comorbidities such as AF, history of coronary artery bypass grafting (CABG), coronary heart disease (CHD) or stroke was equally distributed between both groups. As expected, patients with diabetes had higher body mass index (BMI; DM vs non-DM: 30.9 ± 7.4 vs 26.1 ± 5.4 kg/m2; p = 0.036). Patients with DM showed a higher prevalence of ischaemic cardiomyopathy compared to non-DM patients (DM vs non-DM: 73.3% vs 36.3%; p = 0.009), while non-DM patients showed higher rates of dilated cardiomyopathy versus non-DM (DM vs non-DM: 20% vs 39.3%; p = 0.041).
Procedural characteristics
Regarding the procedural endpoints, the mean overall procedure duration was 31.0 ± 14.0 min. The mean postoperative hospital stay of the overall patients was 2.5 ± 2.0 days. There was no significant difference of procedure duration (DM vs non-DM 30.0 ± 8.0 min vs 31.0 ± 19.0 min; p = 0.885) and length of postoperative hospital stay with respect to DM versus non-DM (DM vs non-DM 3.0 ± 5.0 days vs 2.0 ± 2.0 days; p = 0.790).
Peri- and postprocedural outcome
Differences in risk of any complication with respect to DM and non-DM were not statistically significant (Table 3). Comparing DM and non-DM, there was no significant difference in the primary combined safety endpoint (DM vs non-DM: 20% vs 12.1%, p = 0.119), yet there was a trend towards higher complication risk in the DM group (Figure 2). Bleeding complications were the most common single component of the combined endpoint (8.7%), with respect to this bleeding complication numerically, but, not statistically significant, higher risks were observed in patient with DM (DM vs non-DM: 13.3% vs 6.1%, p = 0.178). The analysis of bleeding events revealed that all bleeding complications were detected as minor, defined as small superficial pocket haematomas requiring no clinical intervention. No patient developed a major device-pocket haematoma requiring surgical intervention, subsequent evacuation or blood transfusion.
In-hospital events and follow-up at 12 months.
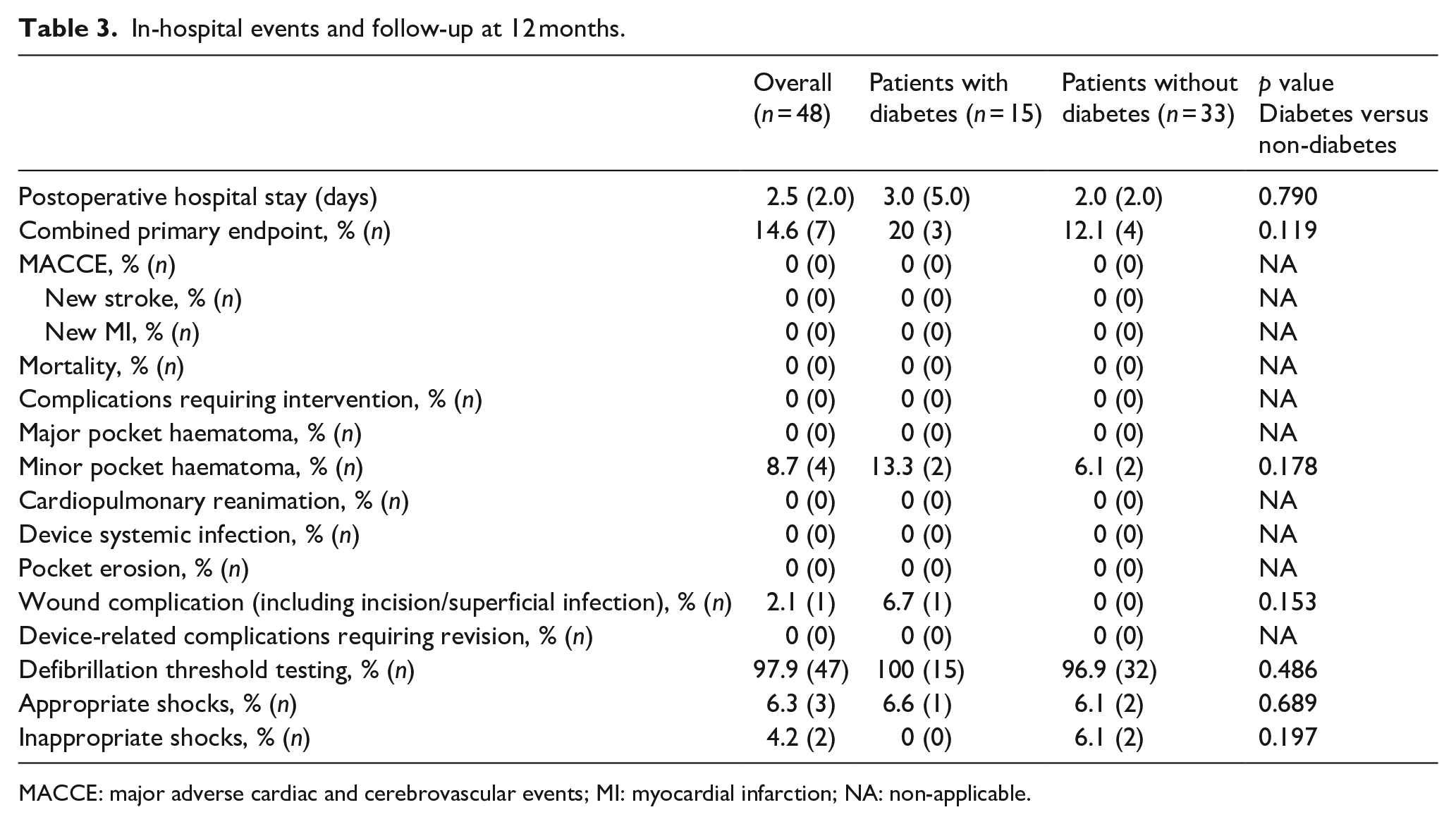
MACCE: major adverse cardiac and cerebrovascular events; MI: myocardial infarction; NA: non-applicable.
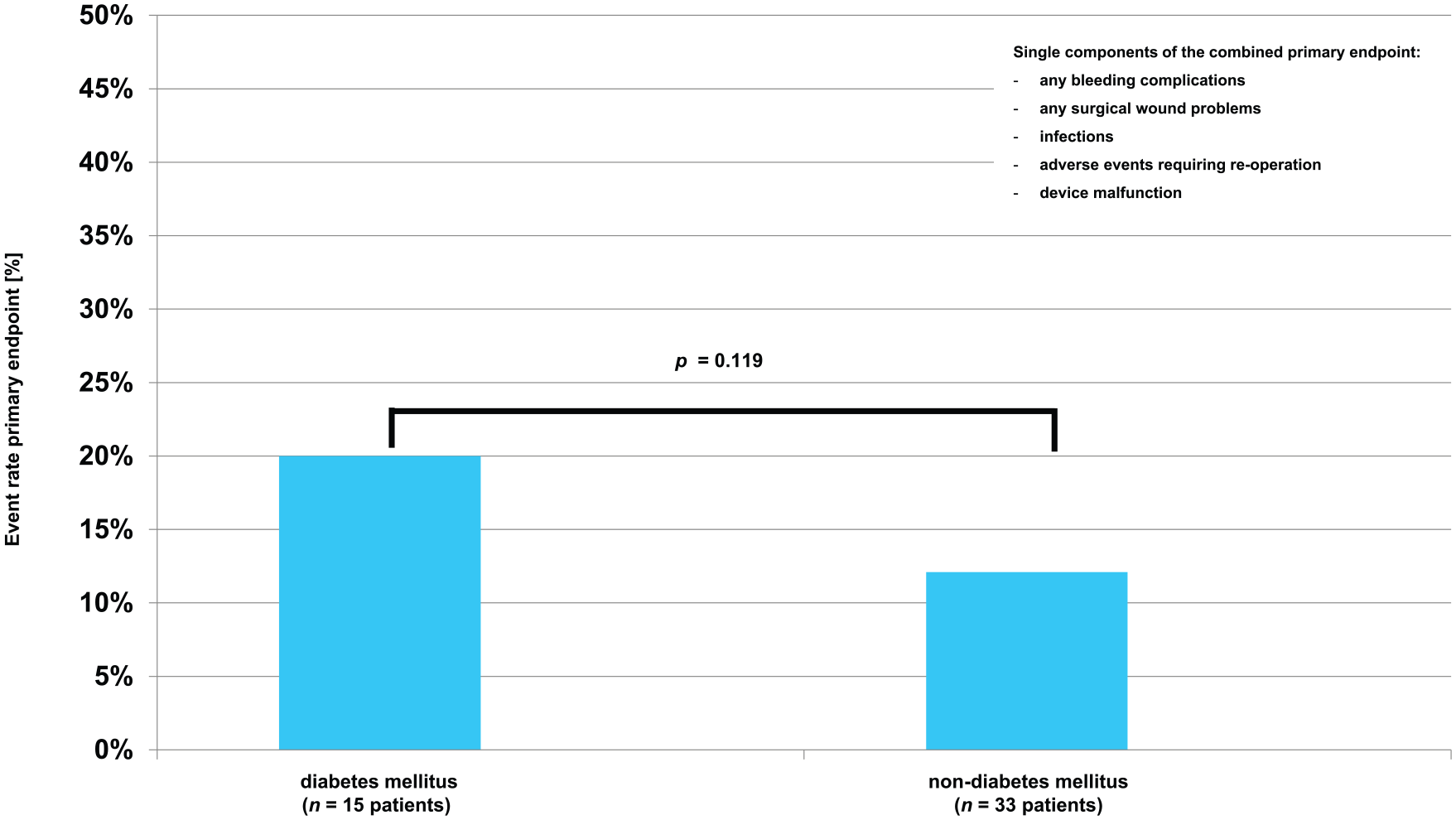
Outcome analysis: percentage of primary combined endpoint in the diabetes subgroup (left) and in the non-diabetes subgroup (right).
With respect to the secondary endpoint of infectious complications, we could not observe any pocket infection or systemic signs such as fever and leukocytosis in both groups. There was no case of wound infection requiring surgical revision or intravenous antibiotics nor an S-ICD-related bacteraemia. With respect to the secondary endpoint of wound infection, there was no difference. Regarding surgical wound problems, there were no case of wound dehiscence. Furthermore, we observed no electrode or pulse generator migrations.
During the initial implantation, the first defibrillation test was successful in about 98% of the patients. All patients but one underwent successful VF-induced defibrillation at 65 J shock. The patient with a defibrillation failure (non-DM group) underwent external defibrillation with 200 J biphasic shock, and, after repositioning of the pulse generator more dorsally, an effective internal shock was obtained in standard polarity.
During 3 months follow-up, there were two appropriate shock deliveries in the DM group, while one shock was delivered in non-DM group (DM vs non-DM: 6.6% vs 6.1%, p = 0.698). All true ventricular fibrillation/ventricular tachycardia (VF/VT) episodes (n = 3, 6.3%) were successfully terminated with appropriate ICD shock delivery; there was no case of non-successful VF/VT therapy in both groups. All of the shocks could terminate VF/VT with the first attempt.
Regarding shock-related hospitalizations due to inappropriate shocks, two events could be detected, which were patients of both DM and non-DM groups (DM vs non-DM: 0% vs 6.1%, p = 0.197). One non-DM patient suffered from multiple inappropriate shocks due to oversensing caused by residual air surrounding the proximal electrode 5 days after the implantation. The device was re-programmed to a different vector which prevented any further inappropriate shocks. The inappropriate treated episodes of the other patient of the non-DM group were due to rapidly conducted AF.
In the overall cohort, there was no case of perioperative (<24 h) mortality, and no patient died within first 3 months.
Discussion
The objective of this study was to evaluate whether DM might be a risk factor for peri- and postinterventional complications in patients undergoing S-ICD implantation. Therefore, we evaluated periprocedural outcome, including complication rates during 3 months follow-up. Our study cohort showed a relatively high prevalence of DM in 30.9% of patients. This rate is higher than in the first large prospective S-ICD study EFFORTLESS 14 (11%) but consistent with recently published S-ICD studies showing rates of DM up to 33%.15,16
Primary endpoint
Despite other studies of ICD patients who have found DM to be an independent risk factor for complications, 17 we observed that DM patients implanted with an S-ICD defibrillator did not show an association with an increased risk for the predefined combined primary safety endpoint of any bleeding complication according to ISTH criteria, infection or device malfunction compared to non-DM patients. Still, there was a clear trend towards higher numbers of complications in the DM group, mainly driven by minor complications.
The most common reported complication associated with S-ICD implantation is device infection. The pooled analysis of the investigational device exemption (IDE) and EFFORTLESS studies reported a 2% rate of device infection, with 1.7% requiring revision. 2 The results of the midterm analysis of the EFFORTLESS showed an infection rate of 2.9% of patients, with 2.4% requiring device explant during a 3.1-year average follow-up. 14 In our cohort, there was no case of a severe infection with necessity of a revision, including the DM group. Looking at haematoma complications after S-ICD surgery, the incidence of relevant device-pocket haematoma was consistently reported <1% across different registries in the literature.2,14,15 Our results are in line with these results confirming relevant haematoma as a rare complication after S-ICD implantation. We experienced no clinically significant pocket haematoma complication in our cohort. However, our results point towards higher periprocedural minor bleeding complications in DM patients undergoing an S-ICD surgical procedure without the need for revision or further intervention.
Secondary safety endpoints
Analysis of secondary safety endpoints showed no significant difference for DM compared to non-DM patients.
Regarding the secondary safety endpoint of bleeding complications, the literature reports high rates of S-ICD pocket complications in conventional subcutaneous implantation technique ranging from 7.6% to 11.9%. 4 Anticoagulation and/or antiplatelet therapy with clopidogrel appeared to increase the risk for haematoma in this context.4,18 As reported above in our study, no case of a significant device-pocket haematoma requiring surgical intervention was observed, despite the fact that the majority of patients were on oral anticoagulation and/or antithrombotic therapy.
In our overall cohort, the periprocedural safety was high: there were no operative revisions required or needed for explanation of S-ICD and no perioperative mortality. Previous published data showed a lower rate of lead-related complications in S-ICDs compared to TV-ICDs, whereas S-ICD patients had more non-lead-related complications, which mostly consisted of inappropriate sensing and infections. 19 In contrast to these results, there were no wound infections requiring surgical revision within our cohort independent to the presence of DM.
Procedure duration and length of stay were not different in patients with DM when compared to non-DM patients. This finding is in line with the results of post hoc subgroup analyses of TV-ICD studies and other ICD trials, in which the length of stay was not different in DM when compared to non-DM patients.20,21
Efficacy
Recently reported intraoperative shock conversion rates were consistently very high so that in >99% of patients, induced ventricular arrhythmia was terminated.5,22 In our cohort, only one patient in the DM group, induced ventricular arrhythmia was terminated within the first shock therapy. In this single case, a repositioning with a more dorsal position of the pulse generator created an improved shock field and resulted in an effective internal shock test.
One of the limitations inherent to the S-ICD device is the presence of inappropriate shocks. Earlier studies, for example, from the EFFORTLESS S-ICD registry, showed a quite high rate of inappropriate shocks occurring in up to 7% per year. 6 The majority of inappropriate shocks were resulting of T-wave oversensing. A significant overall reduction in inappropriate therapy was seen in dual-zone programming2,22 as well as the new SMART-PASS 23 algorithm of advanced technology with second-generation S-ICD. All our S-ICD patients were implanted with a second-generation Emblem S-ICD with new SMART-PASS algorithm and had dual-zone programming, resulting in complete absence of inappropriate therapies due to T-wave oversensing.
Nevertheless, in our cohort, inappropriate shocks occurred in two patients of the non-DM group (n = 2, overall 4.2%; DM vs non-DM: 0.0% vs 6.1%) and could effectively be addressed by re-programming of the S-ICD. In general, AF is the most common cause of inappropriate therapy. In our cohort, we observed inappropriate therapies due to AF episodes in one patient of the non-DM group.
In general, in our study, DM was not associated with an increased rate of ICD therapies of any type, including appropriate or inappropriate shocks during 3 months of follow-up. This is in concordance with subgroup analyses of the Comparison of Medical Therapy, Pacing, and Defibrillation in Heart Failure (COMPANION) and MADIT II (Multicenter Automatic Defibrillator Implantation Trial–II) trials that did not find an increased rate of appropriate therapies in the DM cohort.20,21 A sub-study of the MADIT–Reduce Inappropriate Therapy (MADIT RIT) did find that DM patients had a 58% increased risk of appropriate therapy, but a 46% decreased risk of inappropriate therapy, compared to no DM patients during a mean follow-up of 17 months. 24 Impact of DM in shock therapies of S-ICD recipients has not been adequately investigated yet.
Potential relevance and clinical implications
Our data suggest that S-ICD implantation in DM patients is not associated with a poor procedural outcome or a statistically significant higher risk of wound infections and other procedure-related complications within a follow-up of 3 months.
However, considering the retrospective analysis of a small patient cohort, it is hard to conclude that S-ICD implantation in DM patients is as safe – especially since there was a numerical difference towards more adverse events in the DM group driven by higher number of minor bleeding complications. It might be speculated that patients with DM might have a worse outcome when evaluated in larger cohorts. In view of the relatively high percentage of DM patients in an S-ICD cohort, the need of future larger trials can be postulated – especially taken the fact that S-ICD is already recommended as a first-line therapy in patients with an increased infection risk by the AHA guidelines, 25 and DM is known to go along with higher infection risks.
Furthermore, DM is associated with chronic kidney disease (CKD). End-stage renal dysfunction is a frequent comorbidity in patients with diabetes in our cohort - represented by 6 out of 15 patients (40%) with diabetes being on haemodialysis. Therefore, this subgroup seems to be overrepresented in our study but probably reflects a relevant pre-selected subgroup of patients, which is one main target population for this device according to the current guidelines. 25
Limitations
The present study has several limitations. It is limited by its retrospective design investigating a small patient cohort with a limited follow-up time of 3 months. Another limitation of our study is the designation of patients as DM based on self-reporting rather than regular determination of laboratory results such as serum glucose or glycosylated haemoglobin (HbA1c) levels that constitute the criteria for the diagnosis of diabetes. Furthermore, detailed data regarding the length of time since diagnosis and information on diabetic medication are missing. Considering the retrospective analysis of only a small patient cohort group and the lack of a powered analysis, it is not possible to conclude that S-ICD procedures in DM patients are equally safe. Our results can only be considered as hypothesis-generating and has to be further evaluated in larger cohorts.
Conclusion
Our retrospective, single-centre experience primarily assessed the safety of S-ICD procedures in patients with DM within an unselected all-comers consecutive S-ICD cohort. DM patients represented one-third of the cohort underlying the relevance of this variable taken as the S-ICD recommendation in patients with an elevated infection risk. Procedural parameters, efficacy and perioperative complication rate were not increased in patients with DM compared to non-DM patients. These results suggest that S-ICD implantation seems to be a valid option in DM patients requiring ICD implantation and might be even considered as a first-line option considering that patients with type 2 DM might bear an elevated infection risk.
Still, results of prospective randomized trials with more patients and longer follow-up will be required for assessing the outcome of S-ICDs in DM and the potential to reduce the long-term risks of serious systemic infection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
