Abstract
Besides being a metabolic disease, diabetes is considered a vascular disease as many of the complications relate to vascular pathologies. The aim of this study was to investigate how vascular tone and reactivity and vascular cell metabolism were affected in type 2 diabetes mellitus and whether β-hydroxybutyrate could have a positive effect as alternative energy substrate. Isolated mesenteric arteries of db/db and control mice were incubated in media containing [U-13C]glucose or [U-13C]β-hydroxybutyrate, and tissue extracts were analysed by mass spectrometry. Functional characterization was performed by wire myography to assess vasodilation and vasocontraction. Hypermetabolism of glucose and β-hydroxybutyrate was observed for mesenteric arteries of db/db mice; however, hypermetabolism was significant only with β-hydroxybutyrate as energy substrate. The functional characterization showed impaired endothelial-dependent vasodilation in mesenteric arteries of the db/db mice, whereas the contractility was unaffected. This study provides evidence that the endothelial cells are impaired, whereas the vascular smooth muscle cells are more robust and seemed less affected in the db/db mouse. Furthermore, the results indicate that hypermetabolism of energy substrates may be due to adaptive changes in the mesenteric arteries.
Introduction
Type 2 diabetes mellitus (T2DM) is a metabolic disease characterized by a defect in both insulin secretion and insulin sensitivity, leading to increased plasma glucose levels. 1 T2DM has by now reached a pandemic level 2 and is associated with a host of comorbidities, 3 which imposes a substantial socioeconomic burden and negatively affects patient’s quality of life. 4 Interestingly, many diabetic comorbidities are related to vascular complications, such as nephropathy and neuropathy,5,6 and that is why diabetes is also considered a vascular disease in addition to being a metabolic disease. Vascular smooth muscle cells (VSMCs) make up the vascular wall and undergo contraction and dilation to regulate vessel tone, blood flow and pressure.7,8 The vascular lumen is covered by a monolayer, the vascular endothelium, which has multiple functions, including regulation of vasodilatation and vasoconstriction, growth and metabolism. 5 The structure of blood vessels can be remodelled and adapt to new environments. 9 In relation to this, evidence suggests that prolonged energy surplus, as presented to vascular and endothelial tissue during T2DM, in the form of glucose or free fatty acids, may lead to generation of reactive oxygen species (ROS) causing cellular oxidative stress and cell damage. 10 Vascular smooth muscle energy metabolism is compartmentalized as the ATP production arriving from glycolysis is primarily associated with membrane pumps, such as the Na+/K+ pump activity, and ATP derived from oxidative phosphorylation fuels contraction,11,12 whereas the energy source for active vasodilation seems unclear. Besides this, little is known about vascular metabolism and how it is affected in T2DM. If treated in time, the disease progression of T2DM can potentially be attenuated, but in order to successfully treat patients, thorough investigation of the mechanisms and causalities of the disease is needed. The aim of this study was to assess how vascular tone and reactivity, and vascular cell metabolism are affected in T2DM. Furthermore, whether the ketone body, β-hydroxybutyrate, which is often found to be elevated in plasma of T2DM patients, 13 could have a positive effect as alternative energy substrate needs clarification. To investigate this, isolated mesenteric arteries from the db/db mouse, a commonly applied mouse model of T2DM, were incubated in media containing energy substrates enriched with stable carbon isotope 13C to assess vascular metabolism. Vasomotor response was measured by a wire myograph to assess vasodilation and vasocontraction. The experiments were performed with glucose either as lone energy substrate or in combination with β-hydroxybutyrate.
Materials and methods
Materials and solutions
Physiological salt solution (PSS) had the following composition (in mM): NaCl 119, NaHCO3 25, KCl 4.7, CaCl2 1.5, KH2PO4 1.18, MgSO4·7H2O 1.17, ethylenediaminetetraacetic acid (EDTA) 0.027 and glucose 10, with pH adjusted to 7.4. Potassium-enriched physiological salt solution (KPSS) was prepared by replacing all sodium with an equimolar amount of potassium resulting in a total K+ concentration of 125 mM. Physiological buffer solution (PBS) had the following composition (in mM): NaCl 58.4, KCl 74.6, KH2PO4 136.1 and Na2HPO4·2H2O 178.0, with pH adjusted to 7.4. PSS and KPSS buffer were prepared with 10-mM glucose, 5-mM glucose + 5-mM β-hydroxybutyrate or 10-mM sucrose as a glucose-free preparation.
N,N-Dimethylformamide (DMF), Bradford reagent, N-tert-butyldimethylsilyl-N-methyltrifluoroacetamide (MTBSTFA), prostaglandin F2α(PGF2α), serotonin [5-hydroxytryptamine (5-HT)], U46619, norepinephrine (NE) and acetylcholine (ACh) chloride were purchased from Sigma-Aldrich (St. Louis, USA). Methanol (⩾99.8%, filtered at 0.2 µm) and acetonitrile (⩾99.9% filtered at 0.2 µm), both gradient grades, for high-performance liquid chromatography (HPLC) were purchased from VWR (Fontenay-sous-Bois, France). Ampoules with o-phthalaldehyde (OPA) and 3-mercaptopropionic acid in 0.4-M borate buffer and borate buffer were purchased from Agilent Technologies (Santa Clara, USA). [U-13C]glucose (99%) was purchased from ISOTEC (Miamisburg, USA), and [U-13C]β-hydroxybutyrate (99%) was from Sigma-Aldrich. All other chemicals used were of the purest grade available from regular commercial sources.
Animals
Diabetic and obese homozygote (db/db) and control mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA) and bred in the animal facilities at the Department of Drug Design and Pharmacology, University of Copenhagen, Denmark. The animals were kept in a humidity- and temperature-controlled environment with 12 h/12 h of light/dark cycle with free access to water and standard chow. Sex- and age-matched animals were used at 16–18 weeks of age, and both female (n = 19) and male (n = 25) mice were included in the study. Heterozygote (db/+) littermates were used as controls. 14 Blood glucose levels were assessed, using a BAYER Breeze 2 instrument, in neck blood immediately after decapitation. The experiments were approved by the Danish National Ethics Committee and performed in accordance with the European Community Council Directive on ‘The Protection of Animals Used for Scientific Purposes’ (2010/63/EU).
Vascular tissue preparation and viability test
One db/db mouse and one littermate control mouse were used for each experimental round, that is, animals were analysed in tandem. The animals were sacrificed by cervical dislocation, and the mesenteric arcades were carefully removed. The mesenteric arcades were pinned to Petri dishes in glucose-free PSS, and all surrounding fatty tissue was removed from the mesenteric arteries. Three segments, approximately 2-mm long, from each animal were used for the functional characterisation. The rest of the isolated arteries were incubated for the metabolic characterisation. The vessel segments were mounted and normalized as previously described 15 using a multi-channel wire myograph system 610M and glucose-free PSS.
Vascular tone measurement
The vascular tone was measured three times using the same blood vessel segments, initially with a starvation period in glucose-free medium. Subsequently, vascular tone was measured in presence of 5-mM β-hydroxybutyrate + 5-mM glucose, then in 10-mM glucose, and finally in glucose-free medium for comparison with the initial starvation period to ensure viability of the vessels during the assay. Depolarized forced contraction was induced by KPSS followed by wash with PSS to reach baseline. This was repeated three times to ensure reproducible contractions. Receptor-mediated contraction was induced by 100-µM NE and subsequently, after contraction plateau, 100-µM ACh was added to measure endothelial-dependent vasodilation. Finally, the vessel segments were allowed to reach baseline before maximal contractile response was measured by a ‘cocktail’ solution containing 5-HT (10 µM) and thromboxane receptor agonist U46619 (10 µM) in KPSS. 16 The contractile response was calculated as the difference between vessel wall tension (N/m) during the resting baseline level in PSS buffer and maximal contraction with KPSS, NE or cocktail. The values were normalized to maximal contractile capacity by ‘cocktail’ solution in 10-mM glucose medium. Endothelial-dependent vasodilatation response was determined as percentage relaxation relative to the precontraction tone (N/m) induced by NE (100 µM).
Metabolic mapping using 13C-labelled substrates
The mesenteric arcade was divided into two equally sized segments. One was incubated in PSS containing 10-mM [U-13C]glucose and one in PSS containing 5-mM [U-13C]β-hydroxybutyrate + 5-mM glucose for 1 h at 37°C and oxygenation (95% O2/5% CO2). NE (final concentration 0.1 mM) was added to the medium after half an hour to stimulate metabolism. The incubation was terminated by rinsing the tissue with ice-cold PBS and freezing in liquid nitrogen before being placed in 1 mL of 70% EtOH at −20°C. The tissue samples were subsequently homogenized in a precooled glass douncer and sonicated. Samples were centrifuged (20,000g × 20 min), and the supernatant, that is, blood vessel extract, was lyophilized prior to analysis by gas chromatography–mass spectrometry (GC-MS) and HPLC. The pellets were kept for protein determination.
Metabolic mapping by GC-MS
Metabolic mapping was used to study metabolism in the mesenteric arteries. The percentage of 13C labelling in metabolites of interest was determined using a method described in detail by Walls et al. 17 Briefly, lyophilized extracts were dissolved in water, and an aliquot was acidified with HCl to pH 1–2 and evaporated under nitrogen. Organic extraction was performed using 96% ethanol and benzene followed by evaporation under nitrogen. The samples were derivatized using MTBSTFA in presence of 15% DMF and analysed by GC (Agilent Technologies 7820A, J&W GC column HP-5MS) coupled to an MS (Agilent Technologies 5977E). Isotopic labelling was corrected for natural abundance of 13C determined from an unlabelled standard solution containing the metabolites of interests. The percentage of M + X, where M correlates to the molecular mass of the unlabelled metabolite and X is the number of 13C labelled atoms, was calculated as described by Walls et al. 17
Labelling from 13C glucose and 13C β-hydroxybutyrate
[U-13C]Glucose metabolized via glycolysis leads to [U-13C]pyruvate (M + 3), which may be converted to alanine M + 3 by alanine aminotransferase (ALAT) or lactate M + 3 by lactate dehydrogenase (LDH). Alternatively, pyruvate M + 3 can enter the tricarboxylic acid (TCA) cycle as acetyl coenzyme A (CoA) M + 2 units and condense with unlabelled oxaloacetate giving rise to citrate M + 2. α-Ketoglutarate M + 2, formed by oxidative decarboxylation catalysed by isocitrate dehydrogenase, can be either metabolized in the TCA cycle into succinate M + 2 or converted to glutamate M + 2 via transamination or glutamate decarboxylase (GDH). Glutamate M + 2 is the precursor of glutamine M + 2. In the TCA cycle, succinate M + 2 is converted into fumarate M + 2, malate M + 2 and oxaloacetate M + 2. Labelled aspartate can be formed by transamination of oxaloacetate M + 2. Oxaloacetate M + 2 additionally serves as the substrate for the second turn in the TCA cycle following condensation with acetyl CoA.
[U-13C] β-hydroxybutyrate enters the TCA cycle as acetyl CoA M + 2 condenses with unlabelled oxaloacetate. This will not give rise to 13C labelling in alanine or lactate, but the labelling pattern in the TCA cycle is otherwise similar to the pattern of [U-13C]glucose.
Measurement of total amounts of amino acids using HPLC
Quantitative amounts of amino acid metabolites from mesenteric artery extracts were measured by reversed-phase HPLC (Agilent Technologies 1260 Infinity). The lyophilized extracts were dissolved in water, and an aliquot was online derivatized with OPA and detected using fluorescent detection (excitation 1: 338 nm and emission 1: 390 nm). The amino acids were separated on a Zorbax Eclipse Plus C18 column (4.6 × 150 mm2, 3.5 µm) and a mobile phase gradient of mobile phase A (10-mM Na2HPO4, 10-mM Na2B4O7 · 10 H2O and 0.5-mM NaN3, pH 8.2) and B [acetonitrile 45%: methanol 45%: H2O 10% (v:v:v)]. The percentage of the mobile phase B increased linearly from 2% to 57% in 30 min, from 57% to 100% in 0.1 min and returned to 2% in the last period. The total run time was 35 min, and the flow was maintained at 1.5 mL/min throughout the run. The amounts of amino acids in the samples were calculated from standards containing the amino acids of interest and related to protein content in the samples.
Protein determination
The protein content was determined by the Bradford protein assay. The pellet was dissolved in 150-µL ddH2O, and 15 µL of the suspension was transferred to 40 µL of 1-M KOH and left overnight at room temperature. Five-microlitre samples and bovine serum albumin (BSA) standards (0–1 mg/mL) were added to a 96-well plate. The Bradford reagent of 250 µL was added to each well, and the plate was read at 595 nm.
Statistical analysis
Statistical analyses were performed with GraphPad Prism version 7. Grubb’s test was used to identify significant outliers (alpha = 0.05) prior to analysis. Unpaired Student’s t test was used when comparing two groups of data (i.e. control vs db/db). One-way analysis of variance (ANOVA) followed by Sidak’s multiple comparison test was used to identify significant differences, when three or more groups of data were compared. The values of p < 0.05 were considered statistically significant and indicated by a single asterisk. The data presented are mean values ± standard error of the mean (SEM).
Results
Physiological parameters
The db/db mice are a model of both diabetes and obesity. 14 Indeed, the body weight (control 28.4 ± 0.9 g, db/db 53.1 ± 1.0 g) and blood glucose (control 9.6 ± 0.3 mmol/L, db/db 24.5 ± 2.0 mmol/L) of the db/db mice were significantly higher than those of control mice (Figure 1(a) and (b)). Interestingly, it was also observed that the lumen diameter (µm) of the mesenteric artery used for wire myography was significantly increased in the db/db mice (Figure 1(c)).
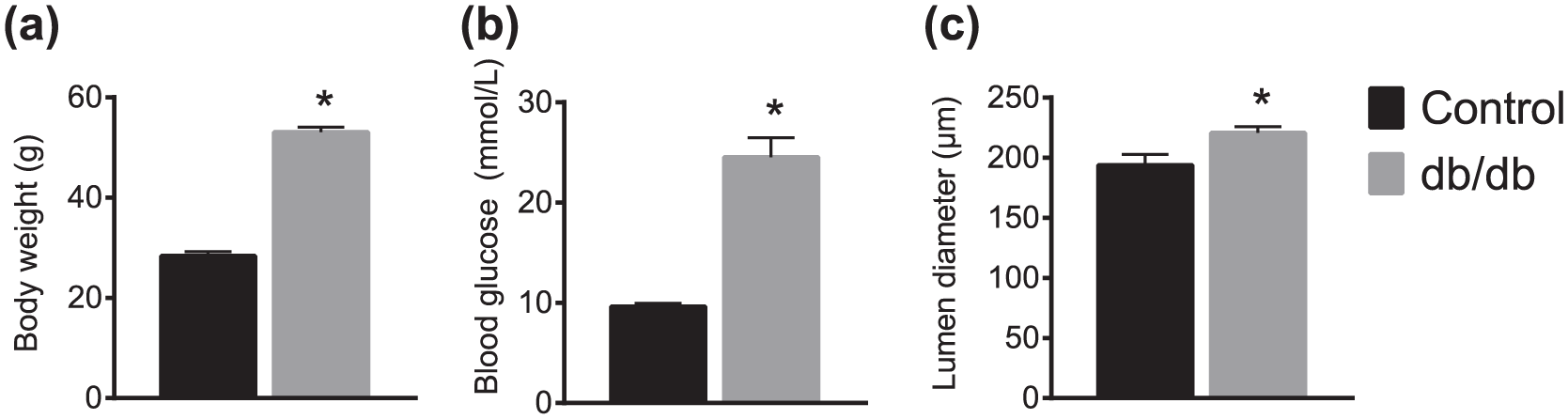
Physiological parameters of control and db/db mice. (a) Body weight (g), n = 22; (b) blood glucose (mmol/L), n = 20; and (c) mesenteric artery lumen diameter (µm), n = 8–9. Each n in lumen diameter represents the average of two to three technical replicates. Results are presented as mean ± SEM. Student’s t test, *p< 0.05.
Vasomotor response
The contractile response of isolated mesenteric arteries of db/db and control mice was tested by eliciting contraction with KPSS and NE. A cocktail (KPSS, 5-HT and U46619) was used to determine the maximal contractile capacity. This was performed with three different buffer conditions: glucose free, glucose and glucose supplemented with β-hydroxybutyrate. No differences in contractile response were observed between db/db and control mice, regardless of the mode of constriction, KPSS (Figure 2(a)), NE (Figure 2(b)) or cocktail (Figure 2(c)). This was observed for all three buffer conditions. However, a lower contractile response, elicited by NE (Figure 2(b)) and cocktail (Figure 2(c)), was observed in the blood vessels of db/db and control mice during the starvation period in the absence of glucose. The starvation period did not affect the vasoconstriction elicited by depolarization with KPSS (Figure 2(a)). Overall, these results show similar contractile response in the mesenteric arteries of db/db and control mice, indicating that the contractile functions of the VSMCs are unaffected in the db/db mice.
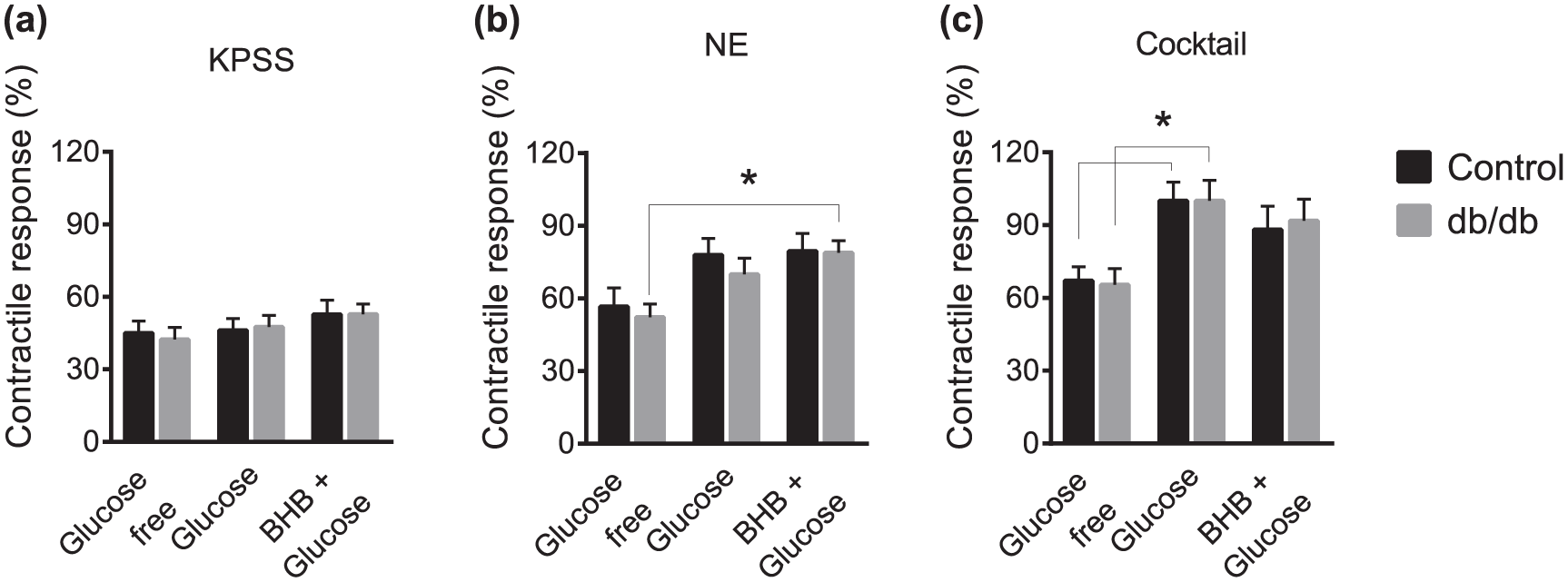
Contraction of small mesenteric arteries from control and db/db mice reveals no significant changes in contractile response. (a) Depolarization induced by KPSS, (b) receptor-mediated contraction induced by 100-µM NE and (c) maximal contraction induced by a cocktail of KPSS, 10-µM 5-HT and 10-µM U46619 in either glucose-free buffer or buffer with 5-mM BHB + 5-mM glucose or 10 mM glucose alone as substrate. Results are presented as mean ± SEM, n = 8–9, one-way ANOVA, *p< 0.05. Each n represents the average of two to three technical replicates.
Endothelial-dependent vasodilation
The functionality of the vascular endothelium was tested by adding ACh (i.e. endothelial-dependent vasodilation) to NE-precontracted mesenteric arteries. Mesenteric arteries from db/db mice showed an attenuated dilatory response to ACh (Figure 3), which was observed in all three buffer conditions (buffer containing glucose alone did not reach statistical significance, p = 0.053). These results illustrate a diminished endothelial-dependent vasodilation in the mesenteric arteries of db/db mice, indicating impaired function of vascular endothelium, regardless of the available energy substrates.
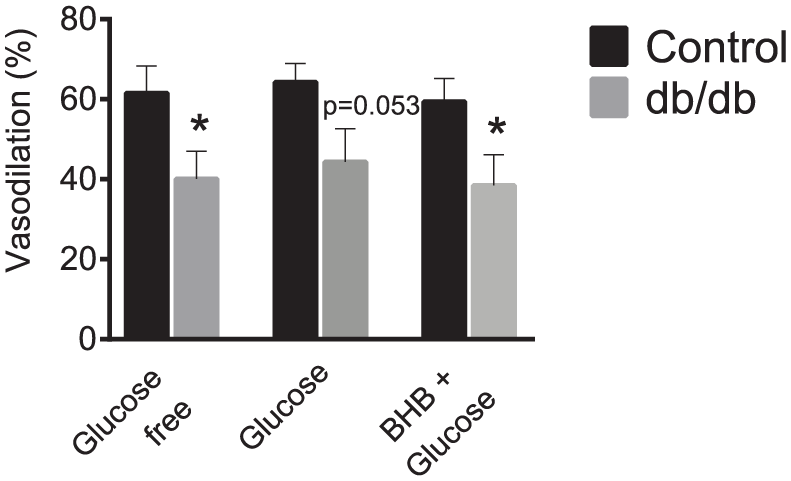
Impaired endothelial-dependent vasodilation in db/db mice. Vasodilation induced by 100-µM ACh after NE-mediated contraction in mesenteric arteries from control and db/db mice either in glucose-free PSS or with 5-mM BHB + 5-mM glucose or 10-mM glucose alone as substrate. Results are presented as mean ± SEM, n = 9, Student’s t-test, *p< 0.05. Each n represents the average of two to three technical replicates.
Glucose metabolism in mesenteric arteries
The 13C labelling patterns of TCA cycle intermediates and amino acids obtained from incubation of isolated mesenteric arties from control and db/db mice in medium containing [U-13C]glucose are presented in Figure 4. The 13C enrichment in alanine was significantly higher in the arteries of the db/db mice, whereas the 13C lactate labelling was similar in control and db/db mice, likely indicating unchanged glycolytic activity but increased ALAT activity. 13C Enrichment in first-turn TCA cycle intermediates from metabolism of [U-13C]glucose showed a tendency towards elevated 13C labelling in the arteries of the db/db mice when compared to controls. This tendency was observed in citrate, fumarate and malate and was statistically significant in succinate. In the second turn of TCA cycle metabolism, the tendency was observed in fumarate and was significant in malate. No difference was observed between the db/db and control mice in the 13C glutamate labelling in the first turn of the TCA cycle, whereas the second turn showed significantly lower labelling in glutamate of db/db mice. The 13C labelling in glutamine originating from glutamate was very low and similar in mesenteric arteries of db/db and control mice. Collectively, these results indicate elevated metabolism of glucose in db/db blood vessels when compared to controls.
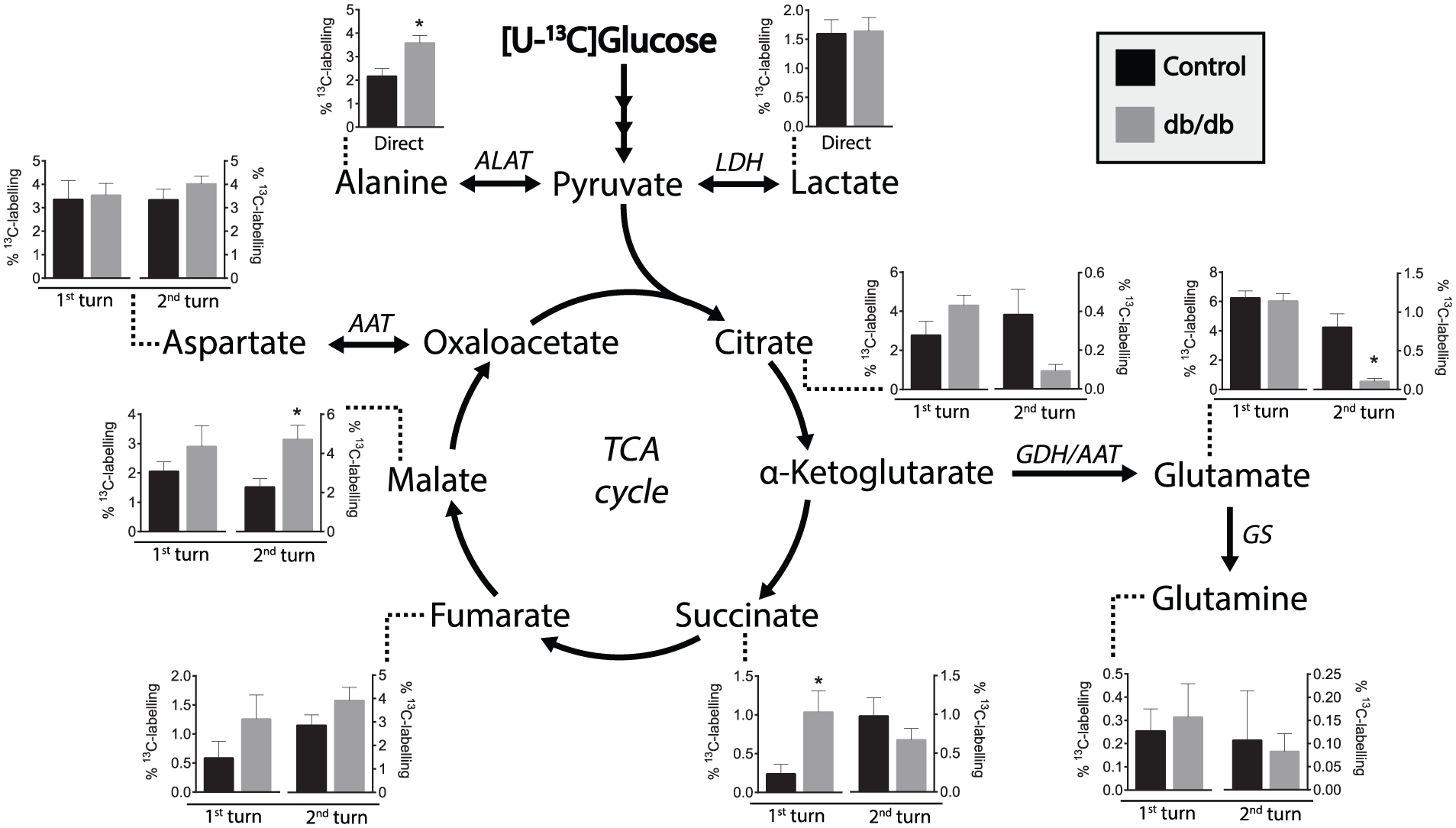
13C Labelling from [U-13C]glucose metabolism indicates elevated glucose metabolism in mesenteric arteries of db/db mice. % 13C Labelling of TCA cycle intermediates and amino acids from metabolism of [U-13C]glucose during incubation of isolated mesenteric arteries from control and db/db mice. Results are presented as mean ± SEM, n = 10–13, Student’s t-test, *p< 0.05.
β-Hydroxybutyrate metabolism in mesenteric arteries
The 13C labelling patterns of TCA cycle intermediates and amino acids obtained from incubations of isolated mesenteric arteries from control and db/db mice in medium containing [U-13C]β-hydroxybutyrate are presented in Figure 5. All TCA cycle metabolites reflecting both the first and the second turn in the TCA cycle showed significantly elevated 13C labelling in the arteries from db/db mice when compared to controls. The 13C labelling in aspartate and glutamate from both the first and second turn was also significantly elevated in the db/db mice, whereas the 13C labelling in glutamine originating from glutamate was similar in mesenteric arteries of db/db and control mice. Interestingly, the labelling percent was much higher using [U-13C]β-hydroxybutyrate (Figure 5) compared to [U-13C]glucose (Figure 4), even though the amount of added 13C labelled carbon was halved when applying β-hydroxybutyrate and despite dilution from metabolism of unlabelled glucose. These results demonstrate that the utilization of β-hydroxybutyrate clearly is augmented in the mesenteric arteries from the db/db mice when compared to the control mice.
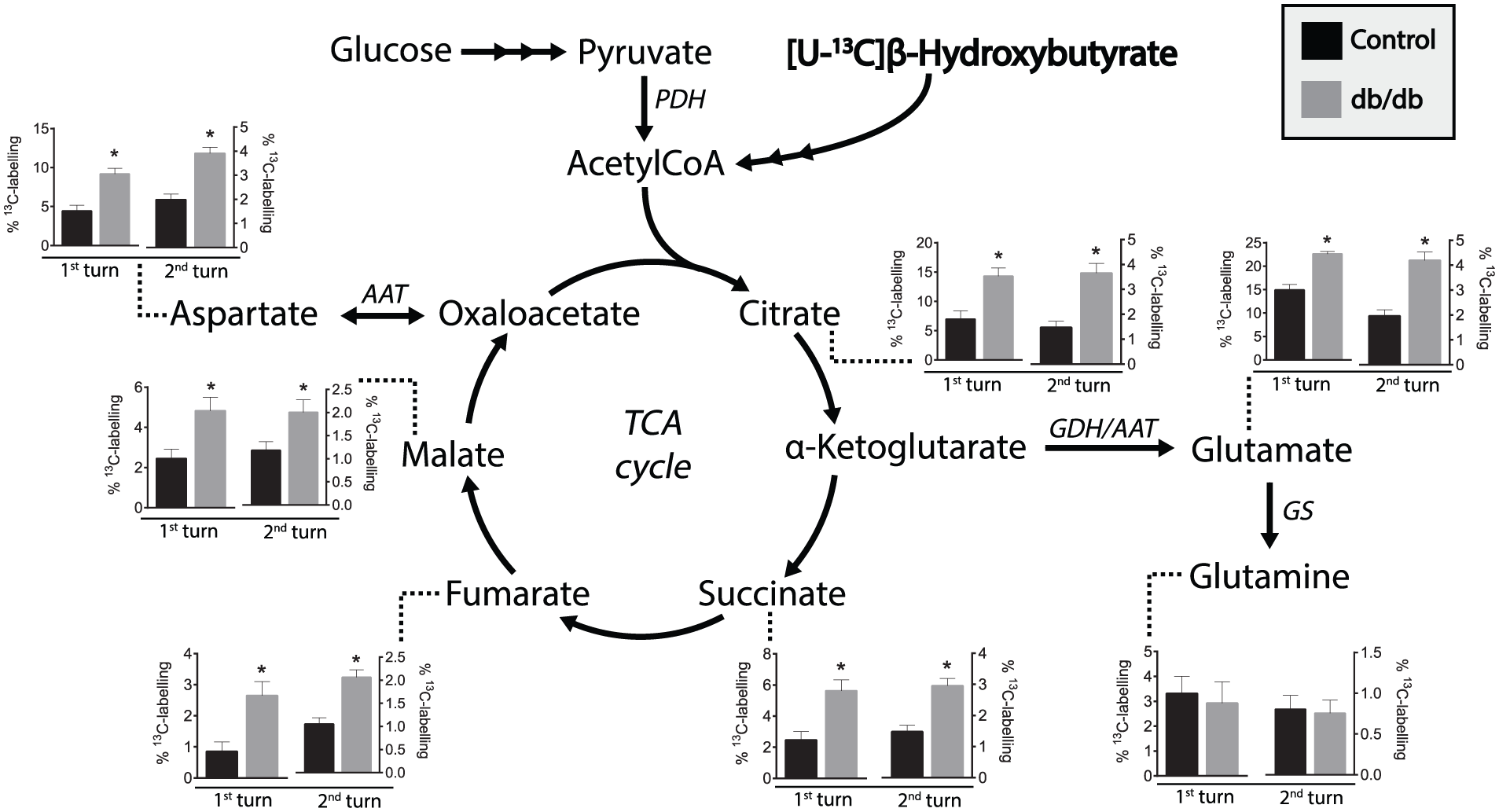
13C Labelling from [U-13C]β-hydroxybutyrate metabolism demonstrates significant elevated β-hydroxybutyrate metabolism in db/db mesenteric arteries. % 13C Labelling of TCA cycle intermediates and amino acids from metabolism of [U-13C]β-hydroxybutyrate during incubation of isolated mesenteric arteries from control and db/db mice. Results are presented as mean ± SEM, n = 10–13, Student’s t-test, *p< 0.05.
Amino acids
The intracellular amounts of amino acids were determined in the incubated mesenteric arteries from the db/db and control mice (Table 1). No significant differences were observed in the amounts of amino acids between db/db and control mice incubated with neither [U-13C]glucose nor [U-13C]β-hydroxybutyrate.
Amounts (nmol/mg of protein) of amino acids in mesenteric artery extracts.
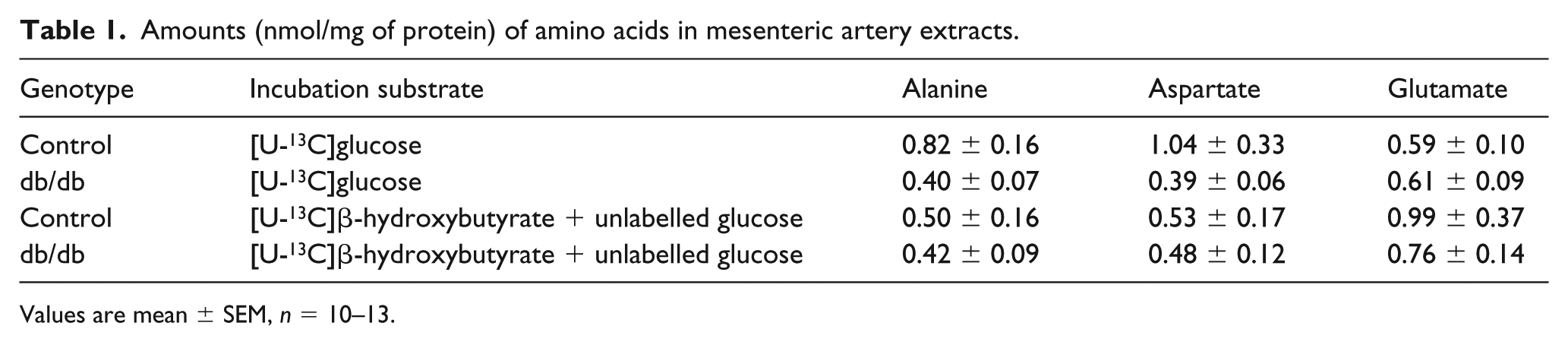
Values are mean ± SEM, n = 10–13.
Discussion
This study sought to assess how vascular functionality (contractile and dilatory) and vascular metabolism are affected in T2DM. The functional data provide evidence of impaired endothelial cell response in mesenteric arteries from the db/db mice leading to impaired endothelial-dependent vasodilation, while the contractility appeared to be unaffected. These findings were accompanied by hypermetabolism of 13C-glucose and 13C-β-hydroxybutyrate in the db/db mesenteric arteries.
Impaired vasodilation but unchanged contractile response in db/db mice
Endothelial dysfunction is well known to be associated with diabetes mellitus and perturbed glucose metabolism.18,19 The results of the present study confirm this association by demonstrating attenuated dilatory response to ACh in mesenteric arteries of db/db mice independent of the available energy substrates: glucose alone, glucose in presence of β-hydroxybutyrate or during starvation (absence of any exogenously provided substrates). These results are in agreement with those of previous studies reporting similar findings in mesenteric arteries isolated from Zucker diabetic fatty (ZDF) rats and db/db mice.20,21 Supporting the clinical relevance of this finding, similar observations have been made in both diabetic patients 22 and non-diabetic, healthy humans during acute hyperglycaemia. 23 Based on the results from non-diabetic healthy humans, it was suggested that the endothelial dysfunction may occur as a result of oxidative stress induced by hyperglycaemia, which is supported by other studies.24–26 The hampered endothelial-dependent vasodilation could be explained by compromised contribution of one or more of the three key endothelium-derived relaxing mediators: (a) nitrogen oxide, (b) prostacyclines and (c) endothelium-derived hyperpolarizing factor. 27 However, further investigation is needed to explore the possible mechanisms. Diminished glycogen in skeletal and cardiac muscles of ZDF rats has previously been described, 28 and to ensure that any observed differences would not be due to difference in glycogen depots, blood vessels of both db/db and control mice were starved prior to measurement of vascular tone. Similar contractile responses were observed in db/db and control mice, regardless of how contraction was induced. In line with this finding, a study performed on mesenteric arteries from ZDF and lean rats turned out similar results 20 and that vasocontraction appears unaffected in the db/db mesenteric arteries. While no differences were observed between db/db and control mice when exposed to different energy substrates, there was, however, a tendency towards decreased contraction during starvation in both phenotypes. This is in contrast to the endothelial-dependent vasodilation, which was unaffected by the starvation period. The tendency to a decrease in vasoconstriction seemed to primarily affect the receptor-mediated contraction, as the potassium-induced contraction (i.e. via depolarization) appeared unaffected. This is in accordance with findings in rabbit aortas where potassium-induced contraction was less vulnerable to glucose-free treatment compared to contraction stimulated by catecholamines, 29 suggesting that contraction induced by depolarization of the blood vessels is less energy demanding.
β-Hydroxybutyrate and glucose metabolism in db/db mesenteric arteries
VSMC energy metabolism is highly compartmentalized, and ATP production from glycolysis is primarily used to fuel membrane pumps, such as the Na+/K+-ATPase, whereas ATP derived from oxidative phosphorylation is used to fuel contraction.11,12,30 The ATP production providing Na+/K+-ATPase activity is oxygen independent, and most of it results in lactate formation. 22 No difference was seen in lactate labelling (%) between the db/db and control mice, suggesting unchanged glycolytic activity and likely equal amounts of ATP to drive Na+/K+-ATPase activity. Metabolism of 13C labelled glucose showed a general tendency towards an elevated labelling (%) in TCA cycle metabolites in db/db blood vessels. These results could indicate that the membrane pumps work equally in db/db and control mice, but more energy is needed to drive contraction in mesenteric arteries of db/db mice. Alanine labelling (%) from 13C glucose metabolism was found to be significantly elevated in mesenteric arteries of db/db mice, which could indicate increased ALAT activity. ALAT can introduce the carbon skeleton from glutamate into the TCA cycle as α-ketoglutarate, which can subsequently be consumed as an energy substrate. Even though previous results indicate that amino acids only have a minor role as energy substrates in blood vessels, 31 this increased formation of α-ketoglutarate could serve as an anaplerotic substrate. This would increase the TCA cycle capacity for oxidative metabolism 32 of two-carbon units, that is, acetyl CoA, thereby also increasing oxidative metabolism of glucose. In T2DM, insulin resistance results in increased gluconeogenesis, 33 thereby depleting TCA cycle intermediates and reducing capacity for oxidative metabolism of glucose. The increased ALAT activity could, thus, serve as an anaplerotic mechanism to support oxidative metabolism and maintain energy homoeostasis. Significantly elevated 13C labelling was found in all metabolites, except glutamine, originating from metabolism of [U-13C]β-hydroxybutyrate in mesenteric arteries of the db/db mice. This correlates with previous findings, 34 where increased utilization of ketone bodies was observed in the db/db brain. The levels of ketone bodies have been found to be increased in both db/db mice35,36 and T2DM patients, 13 and thus, it could be speculated that diabetic vascular cells might have adapted towards using ketone bodies instead of or in addition to glucose in response to a perturbed glucose metabolism. Supporting this, increased plasma levels of free fatty acids37,38 and increased fatty acid oxidation in the heart muscle 38 have been found in db/db mice, and this could indicate that blood vessels exhibit the same increased β-oxidation. As seen from the glucose incubations, increased ALAT activity in the db/db mesenteric arteries could possibly allow glucose to function as an anaplerotic substrate, thereby further increasing the oxidative metabolism of β-hydroxybutyrate to maintain energy homoeostasis. This speculation is supported by the large increase in relative labelling of the TCA cycle intermediates in the db/db mice in the co-presence of glucose and β-hydroxybutyrate compared to glucose alone. In absence of β-hydroxybutyrate, glucose serves both as a mediator of anaplerosis and as the source of acetyl CoA, thereby reducing the input of 13C in the TCA cycle. However, when β-hydroxybutyrate is present, glucose serves only as a mediator of anaplerosis while β-hydroxybutyrate is the major source of acetyl CoA.
Hypermetabolism and structural remodelling in db/db mesenteric arteries
Cells need energy for normal function, and oxidative phosphorylation is responsible for generating the majority of cellular energy in the form of ATP. 39 Hyperglycaemia, as seen in diabetes, leads to an oversupply of electrons in the electron transport chain, which results in formation of ROS. 10 Interestingly, elevated levels of uncoupling proteins have been found in cardiac mitochondria of db/db mice, 37 and mild uncoupling has been suggested as a possible way to control and limit formation of free radicals and ROS production.40,41 If uncoupling occurs in blood vessels from the db/db mice, it will result in decreased ATP production. Diminished ATP production through oxidative phosphorylation could result in increased TCA cycle activity to maintain the level of ATP production. This would be in line with our observations of increased 13C labelling (%) in metabolites from both 13C labelled β-hydroxybutyrate and glucose, but whether uncoupling occurs in the mesenteric arteries in the db/db mice needs to be verified experimentally. In support of the present findings, a previous study, utilizing mesenteric arteries from 16-week-old db/db mice, also found increased lumen diameter as well as increased external diameter and medial cross-sectional area, indicating hypertrophic outward remodelling of the mesenteric arteries. 42 Interestingly, no mesenteric artery structural remodelling was observed in 12-week-old db/db mice, suggesting that the artery remodelling changes occur between the age of 12 and 16 weeks. During hypertrophy, the number of mitochondria increases, and it is often associated with an increased oxidative metabolism. 30 Thus, it might be speculated that the vascular remodelling induces an increased energy demand, suggesting that the hypermetabolism observed in the mesenteric arteries from the db/db mice could be due to vascular remodelling. In case of uncoupling or remodelling, the blood vessels might need additional energy substrates, and the increased ALAT activity in mesenteric arteries of the db/db, indicated by elevated 13C alanine labelling, might, therefore, have a supportive role in introducing energy substrates into the TCA cycle as outlined above.
Conclusion
Our findings suggest that changes occur in the mesenteric arteries of db/db mice, leading to hypermetabolism and impaired endothelial-dependent vasodilation, which could be linked to the comorbidities of T2DM with vascular complications. As the contractile response seemed to be less affected, these results might indicate that the VSMCs are more robust and therefore less affected in the db/db mouse model, while the endothelial cells are more vulnerable. Ideally, these findings should be followed up by investigation of the endothelial cell metabolism alone to experimentally verify this.
Footnotes
Acknowledgements
The excellent laboratory assistance of Bahareh Abdolalizadeh is cordially acknowledged.
Author contributions
L.A. performed all experiments with help from J.D.N and drafted the initial manuscript. All authors have contributed with critical intellectual content and have approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project is financially supported by Novo Nordisk Foundation
