Abstract
The impact of the local inhibition of soluble epoxide hydrolase, which metabolizes vasodilator and anti-inflammatory epoxyeicosanoids, on diabetic skin microvascular dysfunction was assessed. In diabetic db/db mice, basal skin blood flow assessed using laser Doppler imaging was similar to that of control mice, but thermal hyperemia was markedly reduced. At 2 h after the topical administration of an aqueous gel containing the soluble epoxide hydrolase inhibitor trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid (t-AUCB: 400 mg/L), the peak concentration of t-AUCB was detected in the skin of diabetic mice, which quickly decreased thereafter. In parallel, 2 h after application of t-AUCB treatment, thermal hyperemia was increased compared to the control gel. Quantification of t-AUCB in plasma of treated animals showed no or low systemic diffusion. Furthermore, haematoxylin and eosin histological staining of skin biopsies showed that skin integrity was preserved in t-AUCB-treated mice. Finally, for pig ear skin, a surrogate for human skin, using Franz diffusion cells, we observed a continuous diffusion of t-AUCB from 2 h after application to beyond 24 h. A single topical administration of a soluble epoxide hydrolase inhibitor improves microcirculatory function in the skin of db/db mice and might represent a new therapeutic approach for preventing the development of skin complications in diabetic patients.
Background
Diabetic foot ulcers (DFUs) are a common and serious complication of diabetes mellitus and are associated with major morbidity. Indeed, diabetes is the primary cause of non-traumatic lower limb amputation. 1 The skin’s microcirculation, by maintaining perfusion and delivering oxygen and nutrients, plays a key role in tissue survival. Skin microvascular dysfunction occurs early in the pathophysiology of diabetes and contributes to poor wound healing and the development of foot complications in diabetic patients.2–4 Endothelial dysfunction, as well as sensory and autonomic neuropathies, is thought to contribute to a reduction in the functional capacity of the microvasculature,2–4 and topical treatments targeting this microvascular dysfunction may help to improve wound healing while minimizing potential systemic side effects. 5
Increasing evidence suggests that alterations in the endothelium-derived epoxyeicosatrienoic acids (EETs) pathway are involved in the pathophysiology of the endothelial dysfunction associated with type 2 diabetes.6,7 One interesting approach would thus be to increase the bioavailability of EETs, which are formed by the action of cytochrome P450 and display powerful vasodilating, anti-inflammatory and angiogenic properties.6,7 In vivo, EETs are rapidly converted to the less active dihydroxyeicosatrienoic acids by soluble epoxide hydrolase (sEH), which is the target of a new class of pharmacological inhibitors.8,9 Interestingly, exogenous EET administration as well as genetic or pharmacological inhibition of sEH has been shown to accelerate wound epithelialization and neovascularization in ob/ob mice and in the hairless mouse ear wound model.8–10 However, to date, no study has evaluated their impact on skin microcirculatory function. Since in animals the systemic administration of sEH inhibitors has been successfully used to improve endothelial function of aorta and coronary arteries,11,12 we hypothesized that local cutaneous sEH inhibition may improve endothelium-dependent microvascular reactivity.
In this context, the aim of this study was to assess the effect of a topical formulation containing a sEH inhibitor on skin microcirculation in diabetic mice.
Methods
Animals and treatments
The protocol was approved by the local institutional review committee (decision number: C 38 516 10 006, n°2017011312598602-V5#8531) and conducted in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. Nine-week-old male wild-type C57BL/6J and db/db [BKS(D)-Leprdb/JOrlRj] mice, a genetic model of type 2 diabetes, were acquired from Janvier Labs (Le Genest-Saint-Isle, France). These mice were allowed to acclimate to the photoperiod (12 h of light/12 h of darkness) and temperature conditions (22 ± 1°C) for 1 week prior to the start of the study. A 2-h topical administration (20 µL) of a newly developed gel-like, aqueous pharmaceutical preparation containing the sEH inhibitor trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid (t-AUCB: 400 mg/L) dissolved in dimethyl sulfoxide (DMSO) or a vehicle control gel was applied to the dorsal skin of db/db mice, depilated 2 days before the experiments. Assessment of microvascular function, skin biopsies (50 mm2) and intra-cardiac blood sampling was performed at 2 and 24 h after gel application. Animals were anaesthetized with isoflurane (induction at 3% during 3 min and then maintained at 2%) and placed on a heated pad to maintain stable core body temperature (37.5 ± 0.5°C).
Local and systemic quantification of t-AUCB
Plasma and skin levels of t-AUCB were quantified by liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS). 13 Briefly, skin tissues were mixed with 1 mL of methanol–water (50:50 v/v) and ultrasonicated for 10 min, or 100 µL of plasma were mixed with 300 µL of methanol, allowing protein precipitation. Then, skin and plasma samples were thoroughly vortexed for 10 s and centrifuged at 16,100g for 5 min. The resulting supernatants were collected and analysed by LC-MS/MS. Chromatographic separation was performed on a Kinetex C18 column (2.6-μm particle size, 50-mm length × 3-mm inner diameter). The autosampler temperature was set at 8°C, the column oven at 30°C, the injected volume was 20 μL and the flow rate was 400 μL/min. The mobile phase was 0.2% formic acid in methanol (solvent A) and 2-mM ammonium formate with 0.2% formic acid in water (solvent B). The elution started with 95% B (0–2 min), 95–5% B (2–5 min), 5% B (5–10 min), 5–95% B (10–11 min) and 95% B (11–12 min). The following multiple reaction monitoring (MRM) transitions, m/z 412.9 to m/z 135.1 and m/z 412.9 to m/z 93.0 in positive ion mode, were used to detect t-AUCB (quantification and confirmation transitions, respectively). Skin levels were normalized to tissue weight.
Assessment of skin microvascular function
Skin microvascular reactivity to local heating was used as an index of endothelium-dependent function. 14 Dorsal skin blood flow was measured by laser Doppler imaging (LDI; PeriScan PIM, Perimed, Järfälla, Sweden) over 10 min before heating (baseline flow). The skin was then heated at 41°C for 20 min using a 0.5-cm2 heating probe regulated with an internal thermometer. Skin blood flow was then recorded during the following 15 min.
Data were digitized, stored on a computer and analysed offline with signal processing software (PimSoft v1.5.4.8078, Perimed). Baseline and peak hyperemia were expressed as arbitrary perfusion units (APU), averaged over 3 min immediately before and 1 min immediately after heating, respectively. Thermal hyperemia was subsequently calculated as the difference between peak hyperemia and baseline skin blood flow.
Skin integrity
Skin biopsies were carefully sampled and immediately fixed in a 4% formalin solution for 24 h. After proper fixation, tissue samples were embedded in paraffin and stored at room temperature until analysis, when 4-µm sections were deparaffinized and stained with standard haematoxylin–eosin (H&E) stains. Slides were analysed by an experienced pathologist (J.-M.P.).
Transdermal passage of t-AUCB across pig ear skin
The percutaneous absorption of t-AUCB was studied using Franz diffusion cells. 15 The skin from pig’s ears was chosen for the experiments as it is very similar to that of human skin and closer than mouse skin.16,17 The Franz’s cells had a contact area of 2 cm2, and the experiments were conducted at 32°C. The donor compartment was filled with 20 µL of the t-AUCB-containing gel (400 mg/L). The receptor compartment contained 4.5 mL of phosphate-buffered saline (PBS) and was under magnetic stirring. Samples from the receptor compartment were collected at different time points over 24 h to determine the percutaneous flow of t-AUCB. The t-AUCB quantification was performed by LC-MS/MS.
Statistical analysis
All values are expressed as mean ± standard error of mean (SEM). The Shapiro–Wilk test was used to assess normality. Analyses of the differences between diabetic and control mice for basal skin blood flow and thermal hyperemia were performed using an unpaired t-test or the nonparametric Mann–Whitney rank-sum test. Analyses of the variation in basal skin blood flow and thermal hyperemia induced by the t-AUCB-containing gel were performed using mixed effects models with time as fixed effect and the mouse as a random effect followed, in case of significance, by Bonferroni post hoc tests to compare the baseline value to other time points after application. Analyses of the differences in the effect of the t-AUCB-containing gel and the vehicle gel on basal skin blood flow and thermal hyperemia were performed by repeated measures analysis of variance (ANOVA), and we assessed the influence of the group, of time and the time × group interaction. Statistical analyses were performed with NCSS software (version 07.1.14). A two-sided p < 0.05 was considered as statistically significant.
Results
At untreated skin sites, cutaneous blood flow was slightly, but significantly, lower in db/db mice compared to control mice (Figure 1(a)). In contrast, there was a more marked reduction in thermal hyperemia in db/db mice compared to controls (Figure 1(b)), showing the presence of diabetic skin microcirculatory dysfunction.
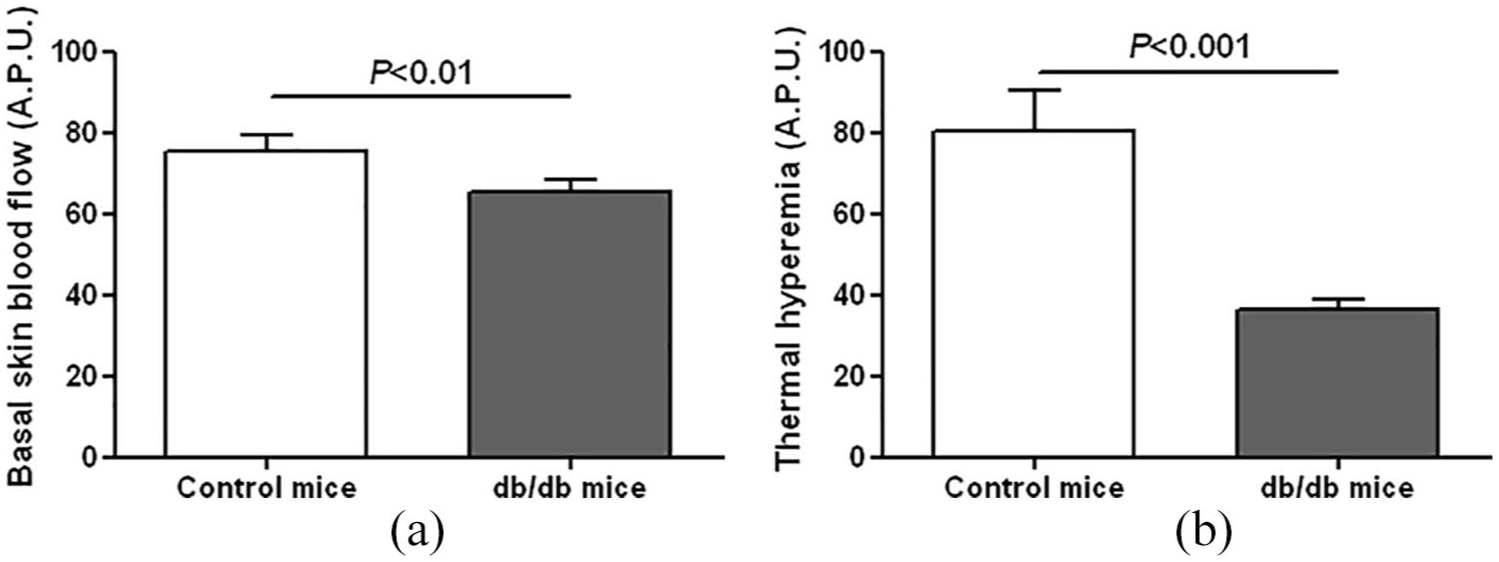
(a) Basal skin blood flow and (b) thermal hyperemia measured by laser Doppler imaging in control (n = 7) and db/db mice (n = 31).
We carefully compared the effect of the t-AUCB-containing gel to that of a vehicle control gel in db/db mice. Both gels were applied on the same animal, with a minimal distance of 1 cm between the two application sites. Both gels increased basal skin blood flow after a 2-h long application, but with no significant difference between groups (Figure 2(a)). However, the t-AUCB-containing gel significantly increased thermal hyperemia compared to the vehicle control gel (Figure 2(b)).
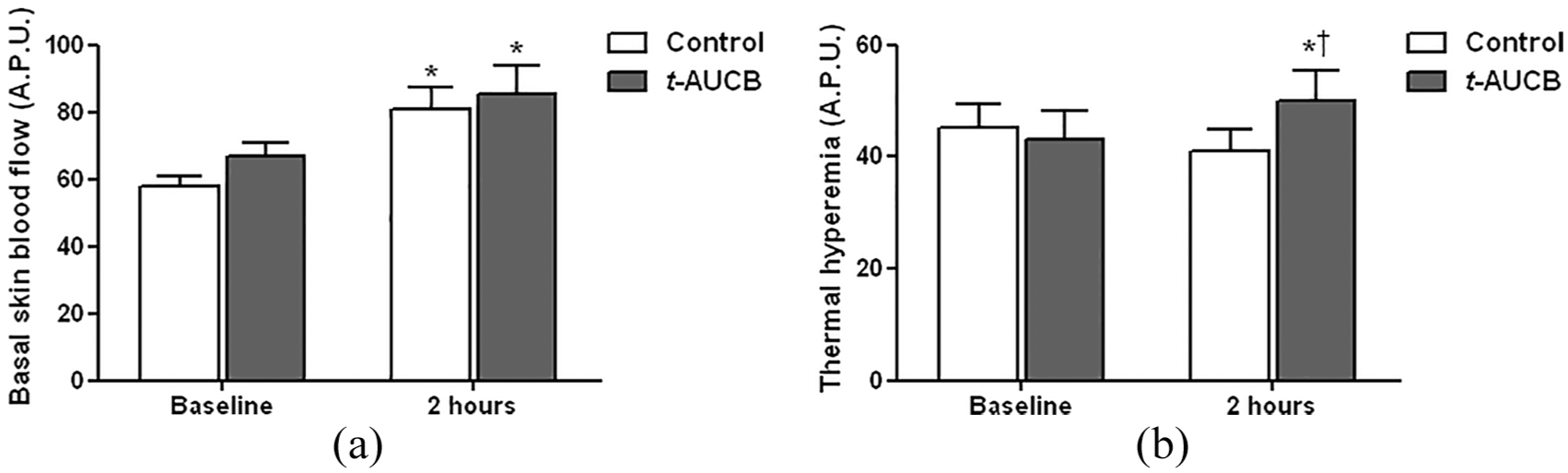
(a) Basal skin blood flow and (b) thermal hyperemia measured by laser Doppler imaging before and after a 2-hour topical application of the t-AUCB-containing gel (20 µL at 400 mg/L) and the vehicle control gel on the dorsal skin of db/db mice (n = 13).
After the 2-h gel application, t-AUCB was detectable in skin biopsies, and these levels had drastically decreased 24 h after application, demonstrating transdermal permeation of t-AUCB across db/db mouse skin (Figure 3(a)). Consistently, thermal hyperemia returned to baseline values 24 h after gel application (Figure 3(b)).
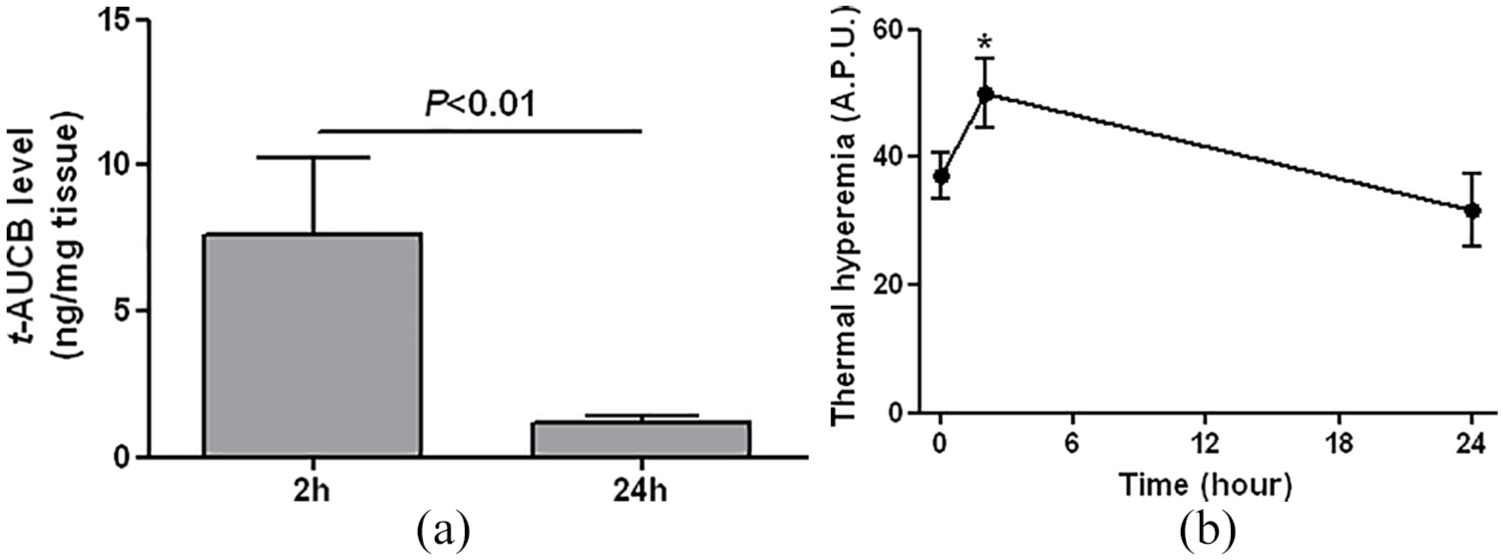
(a) Skin levels of t-AUCB, quantified by liquid chromatography coupled to tandem mass spectrometry, 2 (n = 5) and 24 hours (n = 6) after topical application of the t-AUCB-containing gel (20 µL at 400 mg/L) on the dorsal skin of db/db mice. (b) Thermal hyperemia measured by laser Doppler imaging before (n = 19), 2 (n = 13) and 24 hours (n = 6) after the topical application of the t-AUCB-containing gel on the dorsal skin of db/db mice.
Analysis of plasma samples showed no systemic diffusion of t-AUCB, assessed at 2 and 24 h after application of the t-AUCB-containing gel, except for one animal (Table 1). In addition, no significant inflammatory infiltration was observed in mouse skin at either 2 or 24 h after gel application (Figure 4).
Quantification of t-AUCB in plasma.

t-AUCB: trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid; LOQ: limit of quantification.
Data are represented as n (%).
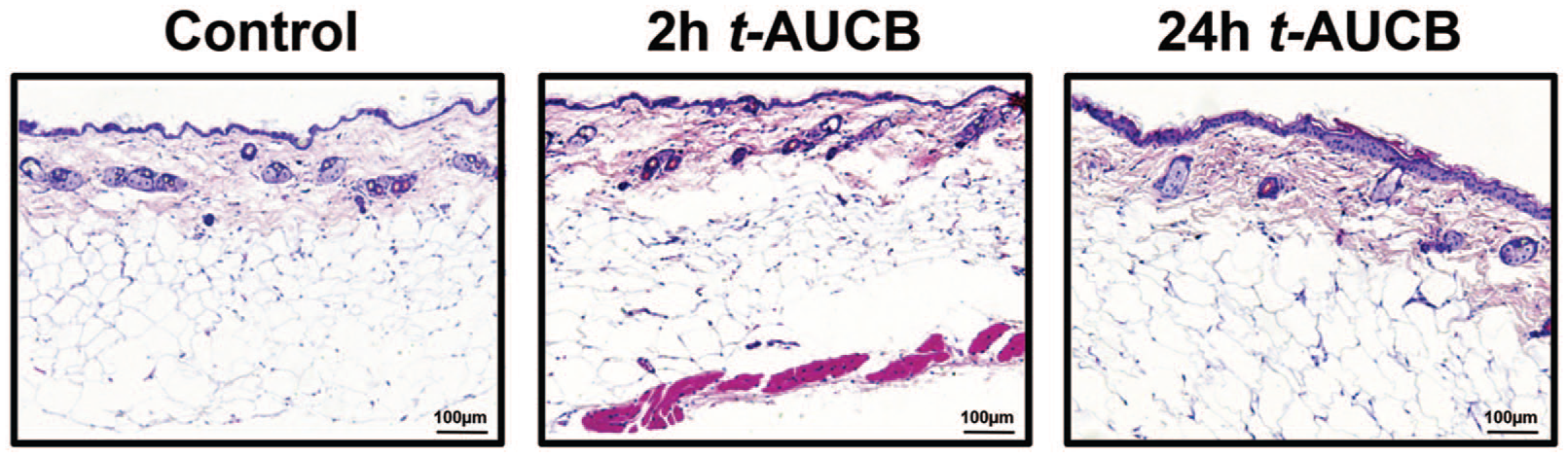
Representative images of haematoxylin and eosin staining of mouse skin 8 and 24 hours after topical application of t-AUCB-containing gel (20 µL at 400 mg/L) on the dorsal skin of db/db mice.
Finally, at the same dosage, a continuous diffusion of t-AUCB was observed across pig ear skin from 2 h up to 24 h after application (Figure 5).
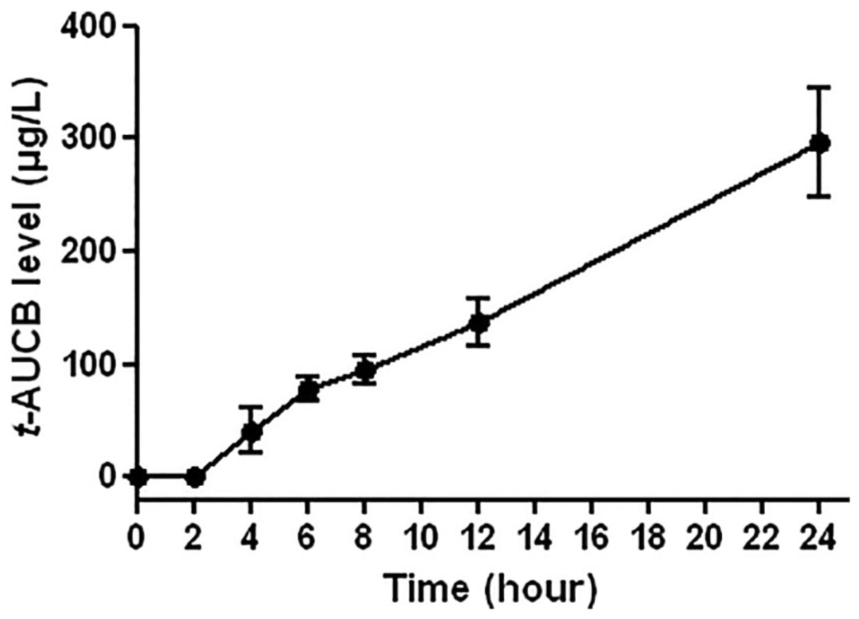
Evolution of t-AUCB level, quantified by liquid chromatography coupled to tandem mass spectrometry, in the receptor compartment of Franz cells from 0 to 24 hours after gel application (20 µL at 400 mg/L) to pig ear skin (n = 3 per time point).
Discussion
The major finding of this preliminary study is that sEH inhibition by t-AUCB using a topical formulation increases thermal hyperemia in the skin, an index of endothelium-dependent microvascular reactivity, in a murine model of diabetes. As microvascular endothelial dysfunction is a hallmark of diabetes, and considering the role of impaired cutaneous microcirculation in poor wound healing in diabetics, such a strategy may be an interesting therapeutic approach for DFUs.
EETs are endothelium-derived vasodilating factors with powerful anti-inflammatory and pro-angiogenic properties that could be useful in the treatment of the cardiovascular complications of type 2 diabetes.6,7 Despite increasing evidence suggesting a possible role for EETs in diabetes-related endothelial dysfunction, no study had previously focused on diabetic skin microvascular dysfunction. The use of thermal hyperemia as a reactivity test was motivated by the involvement of EETs, together with NO, in the response to local heating in humans. 18
We observed a reduction in basal skin blood flow in diabetic db/db mice compared to wild-type mice, which is probably mainly related to lower vascular density. 19 In addition, although no data were available in animal models of diabetes when we designed the study, we demonstrated altered microvascular reactivity to thermal hyperemia in diabetic mice. Thus, as shown in humans, 20 measuring blood flow response to a standardized local heat stimulus provides a suitable model for the study of the skin microvascular dysfunction associated with diabetes in mice.
In this context, we tested the impact of a topical formulation containing t-AUCB, an inhibitor of EET degradation by sEH,11,13 on microvascular dysfunction in the skin of db/db mice. Quantification of t-AUCB in skin biopsies revealed significant transdermal permeation of the molecule 2 h after gel application, associated with increased basal skin blood flow, compared to baseline, and thermal hyperemia. However, the vehicle control gel increased basal skin blood flow in a similar manner. This result supports previous data showing a direct vasodilating effect of the vehicle DMSO. 21 In fact, the topical administration of DMSO has even been proposed for use in humans to treat the skin complications of systemic scleroderma, which is also characterized by microvascular dysfunction and a risk of ulcers, but the results of randomized controlled trials were disappointing.22,23 While the DMSO vehicle had no effect on reactivity, in contrast, the t-AUCB-containing gel improved thermal hyperemia compared to the vehicle control gel, demonstrating an improvement in skin microvascular reactivity. This result shows that, as previously demonstrated in coronary and peripheral arteries,11,12 sEH plays a major role in the vascular dysfunction of the skin associated with type 2 diabetes. Although the objective of this preliminary study was not to assess the effect of sEH inhibition on wound healing, it provides a first proof of principle in an animal model with prolonged wound healing. 24
Importantly for potential human use, histological analysis revealed no signs of skin toxicity with the t-AUCB-containing gel. In addition, quantification of t-AUCB in plasma from exposed animals showed low systemic diffusion of the drug, except in only one animal out of five. This may be important because, although the first results obtained in the initial phases of clinical development suggest that sEH inhibitors were safe,25,26 some data show that increasing EET bioavailability might be associated with adverse effects and in particular might potentiate tumour development.6,7,27,28 Moreover, because mouse skin is thin and the animals were shaved for the experiments, which could have led to an underestimation of the time needed for the transdermal passage of t-AUCB compared to that in humans, the pharmacokinetic study was performed on isolated, more human-like, pig ear skin. A progressive and continuous diffusion of t-AUCB was observed, suggesting that topical sEH inhibitors could be particularly useful in the prevention and/or treatment of skin complications in patients with type 2 diabetes.
Conclusion
These results show that the acute topical administration of a sEH inhibitor improves skin microvascular reactivity in a model of type 2 diabetes. The absence of skin toxicity, the limited systemic diffusion and the demonstration of the progressive passage of the sEH inhibitor across a more human-like animal source of skin support the use of this therapeutic strategy in patients with type 2 diabetes, with potential for the prevention of skin complications and in particular DFUs development. The next steps will be to show, using repeated dosing experiments, that this improvement in skin microcirculatory function translates into effective prevention of diabetes-associated dermatological complications and to confirm the limited systemic diffusion of the molecule so as to avoid potential long-term side effects.
Footnotes
Acknowledgements
The authors thank Mr Tony Pereira (Laboratory of Pharmacokinetics, Toxicology and Pharmacogenetics, Rouen University Hospital, France) for his technical assistance in the quantification of t-AUCB and Dr Alison Foote (Grenoble Alpes University Hospital, France) for editing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was co-supported by grants from the Fondation de France (2011-20459), the French National Research Agency (ANR-16-CE17-0012) and the National Institute of Health (NIEHS/R01 ES002710).
