Abstract
Carboxymethyl-lysine is an advanced glycation end product that is detectable in the serum. Higher carboxymethyl-lysine levels have been associated with increased risk of coronary heart disease, stroke and cardiovascular mortality. We determined whether high carboxymethyl-lysine levels are also associated with the risk of peripheral artery disease in Cardiovascular Health Study participants who were all aged 65 years and older at baseline. Multivariate Cox proportional hazards models were used to determine the association of baseline carboxymethyl-lysine levels with incident peripheral artery disease in 3267 individuals followed for a median length of 10.0 years. A total of 157 cases of incident peripheral artery disease occurred during follow-up. No significant relationship between carboxymethyl-lysine and risk of peripheral artery disease was found (hazard ratio per standard deviation increment = 1.03; 95% confidence interval = 0.87, 1.23).
Advanced glycation end products (AGEs) are implicated in atherogenesis through both non-receptor-mediated and receptor-mediated pathways, which lead to macrophage activation, pro-inflammatory cytokine release, smooth muscle cell proliferation and impaired endothelial function. 1 While various AGEs are known to exist in the circulation, carboxymethyl-lysine (CML) is the most well-studied. Higher serum CML levels have already been associated with coronary heart disease and stroke in older adults. 2 Given known differences in risk factors between vascular beds, we determined whether high CML levels are also associated with the risk of peripheral artery disease (PAD).
The Cardiovascular Health Study (CHS) is a community-based, longitudinal study of 5888 individuals aged 65 years and older. Eligible participants were those without PAD and who had CML measured at Year 9 (1996–1997), which served as the baseline for this study. CML measurement was performed in 2011 by an immunoassay [AGE-CML enzyme-linked immunosorbent assay (ELISA); Microcoat, Penzberg, Germany] in serum specimens stored at −70°C since collection in 1996–1997.
The ankle–brachial index (ABI) was assessed at Year 11 (1998–1999), and the measurement protocol has been described previously. 3 A low ABI was defined as a value of 0.9 or less. An ABI of >1.4 was defined as a non-compressible vessel. Clinical PAD was identified by self-report, review of medical records for other events or as review of Center for Medicaid Services (CMS) records for the ICD-9 codes 440.2 and 443.9 and adjudicated by the cardiac subgroup of the CHS Clinical Events Subcommittee. Detailed criteria for the diagnosis of clinical PAD have already been described. 3 Follow-up data for clinical PAD were available by June 2015.
Individuals who did not attend the Year 9 visit (n = 1475), had prevalent clinical PAD at Year 9 (n = 106) or were missing CML measurement (n = 1040) were excluded from the analysis. In addition, those without ABI measurement or with non-compressible vessels were excluded from the low ABI analysis (n = 984). Logistic regression modelling was performed to investigate the associations of baseline CML levels with low ABI at Year 11. Cox proportional hazards models were used to determine the association of baseline CML levels with incident PAD. In these analyses, CML was modelled continuously [per 1 standard deviation (SD) increment] and also categorized into quartiles. Models were adjusted for age, sex, race, clinic site, total cholesterol, myocardial infarction, stroke, heart failure, anti-hypertension medication use, systolic blood pressure, cigarette smoking status and pack-years, diabetes medication use, glucose, physical activity, statin use, estimated glomerular filtration rate, albumin and C-reactive protein.
Mean age for the included participants was 78.0 years, 39% were male and 16% were black. There were 417 (17.2%) participants found to have a low ABI at Year 11. Over a median follow-up of 10.0 years, 157 (4.8%) participants developed clinical PAD. No significant relationship between CML levels and risk of either prevalent low ABI or incident clinical PAD was found (Table 1).
Associations between carboxymethyl-lysine (CML) levels (pmol/L) and peripheral artery disease (PAD). a
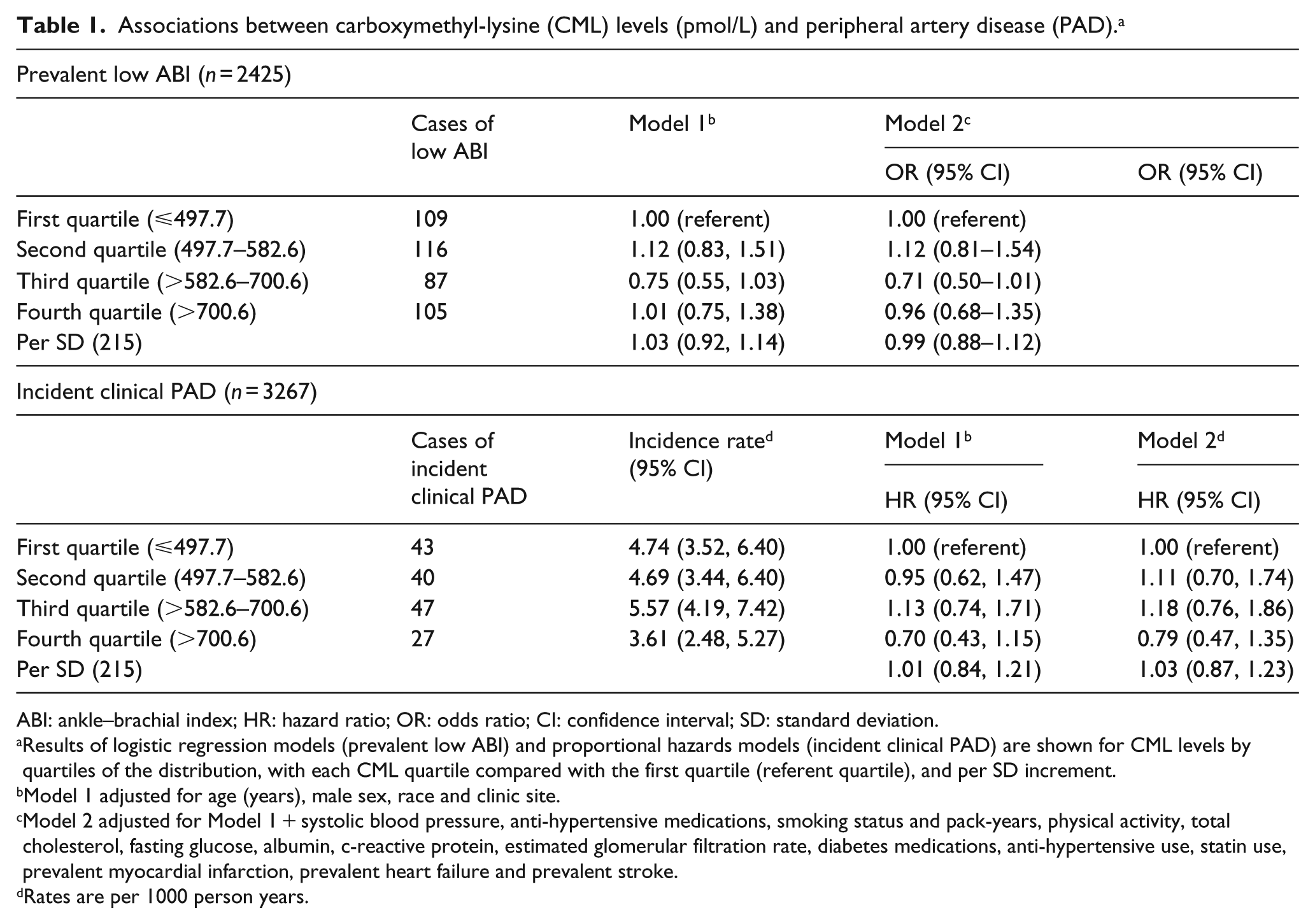
ABI: ankle–brachial index; HR: hazard ratio; OR: odds ratio; CI: confidence interval; SD: standard deviation.
Results of logistic regression models (prevalent low ABI) and proportional hazards models (incident clinical PAD) are shown for CML levels by quartiles of the distribution, with each CML quartile compared with the first quartile (referent quartile), and per SD increment.
Model 1 adjusted for age (years), male sex, race and clinic site.
Model 2 adjusted for Model 1 + systolic blood pressure, anti-hypertensive medications, smoking status and pack-years, physical activity, total cholesterol, fasting glucose, albumin, c-reactive protein, estimated glomerular filtration rate, diabetes medications, anti-hypertensive use, statin use, prevalent myocardial infarction, prevalent heart failure and prevalent stroke.
Rates are per 1000 person years.
In a large community-based cohort of older adults, we found no association of CML levels with either an increased risk of low ABI or incident clinical PAD in multivariate adjusted models. Current literature regarding associations of CML and PAD were limited to small case–control studies that measured tissue AGE with skin autofluorescence, showing higher levels in those with PAD compared to those without. 4 Atherosclerosis is a complex process that demonstrates heterogeneity in different-sized vascular beds. Significant differences in the strength of the association of traditional cardiovascular risk factors such as diabetes or cigarette smoking with PAD versus other forms of atherosclerotic disease have already been established. 5 It is possible, therefore, that the relationship of CML with the larger vessels of the lower extremities may differ from its relationship with other smaller arterial vessels located in the coronary or cerebral vasculature. In addition, it is also possible that the relationship of CML in the development of PAD may also differ with age and that a role for this AGE may exist in younger individuals. Participants in this cohort have had over 70 years to develop PAD through multiple pathways and elevated CML levels found later in life may not be significant enough to make a difference.
This study has limitations. Eligibility for this analysis required that participants should be healthy enough to attend the Year 9 visit and have CML measured at this time. ABI examination was not performed at the same time of CML measurement.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by contract numbers HHSN268201200036C, HHSN268200800007C, HHSN268201800001C, N01HC55222, N01HC85079, N01HC85080, N01HC85081, N01HC85082, N01HC85083, N01HC85086 and grant numbers U01HL080295, U01HL130114 and R01HL094555 from the National Heart, Lung, and Blood Institute (NHLBI), with additional contribution from the National Institute of Neurological Disorders and Stroke (NINDS). Additional support was provided by R01AG023629 from the National Institute on Aging (NIA). A full list of principal CHS investigators and institutions can be found at ![]() .
.
