Abstract
Background:
This cross-sectional study was designed to examine the association between home blood pressure and cognitive impairment in elderly patients with type 2 diabetes.
Methods:
Home blood pressure was measured in the morning and evening for 14 consecutive days in 749 patients with type 2 diabetes. A total of 231 patients were included in the study population. Cognitive function was evaluated using the Mini-Cog test, which is used as a screening for cognitive impairment in elderly patients. We performed a logistic regression analysis and measured the area under the receiver operating characteristic curve and estimated home blood pressure as a marker of cognitive impairment.
Results:
The adjusted odds ratio (95% confidence interval) of evening systolic blood pressure for cognitive impairment was 1.24 (1.02–1.53). Evening systolic blood pressure showed the highest area under the receiver operating characteristic curve for cognitive impairment in both unadjusted and adjusted models. In all subgroup analyses except gender, home blood pressure showed higher area under the receiver operating characteristic curve than clinic blood pressure.
Conclusion:
Home blood pressure was associated with cognitive impairment in elderly patients with type 2 diabetes.
Introduction
Both type 2 diabetes and cognitive impairment are disorders with high morbidity in the elderly and are major social problems. Several large population-based studies have suggested an association between type 2 diabetes and cognitive impairment,1,2 and it has been reported that diabetes almost doubles the risk of cognitive impairment. 2 The morbidity of hypertensive patients with type 2 diabetes is also higher than that in the general population. 3 Home blood pressure (HBP) has been found to have a stronger relation with target organ damage than clinic blood pressure (BP) in several population-based studies and prospective clinical trials.4,5 However, there have been no studies to investigate the relationship between HBP and cognitive impairment in patients with diabetes. The aim of this study was to evaluate whether HBP is associated with cognitive impairment in elderly patients with type 2 diabetes.
Methods
Study design
This cross-sectional study is based on data from the HBP cohort of type 2 diabetes (KAMOGAWA-HBP study). The KAMOGAWA-HBP cohort included patients who had regularly attended the diabetes outpatient clinic at the Kyoto Prefectural University of Medicine or one of four other hospitals. In this study, we evaluated the association between cognitive impairment and HBP in patients who had attended the Kyoto Prefectural University of Medicine. All procedures were approved by the local Research Ethics Committee and were conducted in accordance with the Declaration of Helsinki; informed consent was obtained from all patients.
Study population
HBP measurements were performed in 749 patients with type 2 diabetes who had regularly visited the diabetes outpatient clinic at the Hospital of Kyoto Prefectural University of Medicine between 2008 and 2012. Of the 749 patients, we excluded those with inappropriate BP recordings (HBP measured by patient’s family, measured after breakfast or no measurement) (n = 10), who had progressive renal dysfunction (Cr ⩾2.0 mg/dL) 6 (n = 4), who were younger than 65 years (n = 323), who declined to have cognitive function evaluated and whom the physician recognized as inappropriate patients for this research (patients who cannot understand the intention of the study due to serious dementia, psychiatric disorders or underlying diseases, those with visual impairment, with hearing impairment or had cardiovascular events within half a year) (n = 181). Finally, we included a total of 231 patients for the study population (122 males and 109 females; Figure 1). The diagnosis of type 2 diabetes was based on the American Diabetes Association criteria. 7

Flow diagram for the KAMOGAWA-HBP cohort.
BP measurements
HBP was self-measured using a validated, automatic device (HEM-7080IC; Omron Healthcare Co. Ltd, Kyoto, Japan) 8 equipped with memory to store the date, time and readings. We used the electronic readouts from the device for our study analyses. Patients were instructed to perform triplicate morning and evening BP with at least 1 min between recordings for 14 consecutive days. We calculated the mean of three measurements per morning and per evening each day, and the level of HBP was computed from those 14 mean values. Morning BP measurements were made within 1 h of waking, before breakfast or taking any medicines, with the patient seated and rested for at least 5 min. 9 Evening BP measurements were obtained in a similar manner just prior to bedtime. The cuff was placed directly around the non-dominant arm, and the position of the cuff was maintained at the level of the heart. Clinic BP was calculated as the mean value of triplicate measurements using the HEM-70801C device, at the time of study entry.
Assessment of cognitive function outcome
Cognitive function was evaluated by the physician using the Mini-Cog test, which is a practical screening tool for cognitive impairment in primary care.10,11 The Mini-Cog can be used effectively after brief training in medical settings. It consists of two components, a three-item recall test for memory (0–3 points; each correctly recalled word = 1 point) and a simply scored clock drawing test (abnormal clock = 0 points and normal clock = 2 points). 12 In this study, cognitive impairment was defined as Mini-Cog score equal to or less than 2 points, which is the cut-off value to screen for cognitive impairment.
Physical and biochemical examinations
Information, including age, duration of diabetes, microvascular complications, macrovascular complications, smoking habit and alcohol intake, hypoglycemic medication and antihypertensive medication, were obtained at the time of study entry. Retinopathy was graded as follows: no diabetic retinopathy (NDR), simple diabetic retinopathy (SDR) and proliferative diabetic retinopathy (PDR). Neuropathy was defined by the diagnostic criteria for diabetic neuropathy proposed by the Diagnostic Neuropathy Study Group. 13 Nephropathy was defined as urinary albumin excretion (UAE) equal to or more than 30 mg/g Cr. A macrovascular complication was defined as the presence of previous cerebrovascular disease, cardiovascular disease and/or arteriosclerosis obliterans based on the clinical history or physical examination. Smoking status (current, past or never smokers) and alcohol drinking status (everyday, social and never) were assessed by interview. Blood samples were collected for biochemical measurements. Haemoglobin A1c, triglyceride, cholesterol and other biochemical data were determined using standard laboratory assays. UAE was measured with an immunoturbidimetric assay. A mean value for UAE was determined from triplicate urine collections.
Statistical analysis
Baseline characteristics were summarized by median with interquartile range (IQR) or numbers. We performed a logistic regression to analyse the relationship between HBP and presence of cognitive impairment. We established a multivariate logistic model adjusted by age, sex, body mass index, haemoglobin A1c, cholesterol, use of antihypertensive medications, use of insulin, history of cerebral infarction and alcohol consumption status. Unadjusted and adjusted odds ratios and 95% confidence intervals (CIs) were estimated. We computed day-by-day variability of BP as within-patient coefficient of variation (CV) of measurements. CV was determined as within-patient standard deviation divided by within-patient mean. In addition, we compared the area under the receiver operating characteristic curve (AUC) of HBP and clinic BP for cognitive impairment as Mini-Cog score ⩽ 2.
We performed subgroup analyses by gender, duration of diabetes (10 years as a cut-off value), insulin use, history of cerebral infarction and age (equal to or more than 75 years and less than 75 years).
The JMP version 10.0.2 software (SAS Institute Inc., Cary, NC, USA) was used for statistical analyses, and p-values < 0.05 (two-sided) were considered statistically significant.
Results
The clinical characteristics of the patients are shown in Table 1. Median (IQR) age, duration of diabetes, body mass index and haemoglobin A1c were 72.5 (69.0–76.0) years, 14.5 (7.0–20.0) years, 23.2 (21.1–24.9) kg/m2 and 6.9 (6.2–7.4)%, respectively. Of the 231 patients, 67 (29.0%) had a Mini-Cog score equal to or less than 2 points and 126 (54.5%) were treated with antihypertensive drugs.
Clinical characteristics of patients.
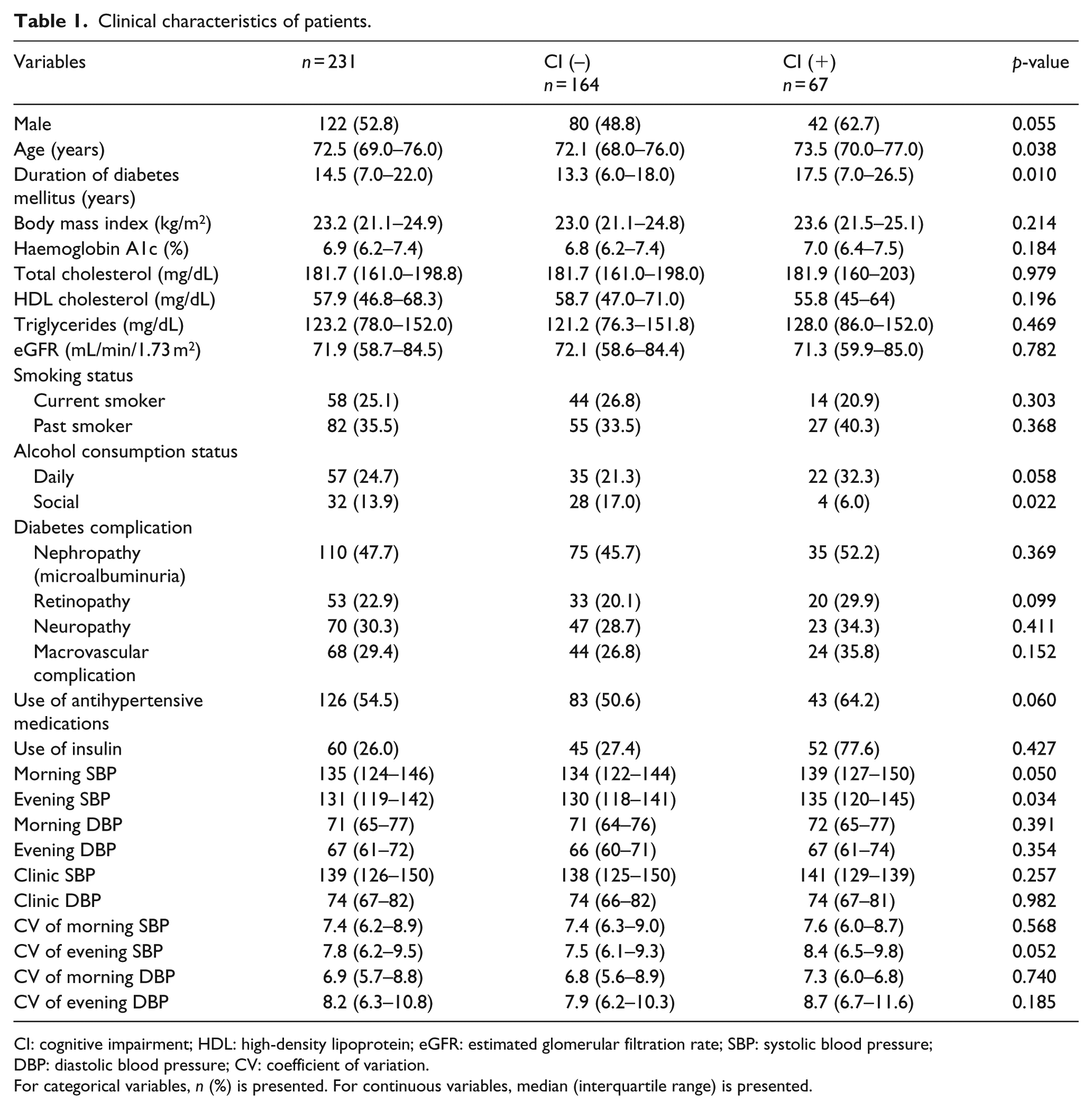
CI: cognitive impairment; HDL: high-density lipoprotein; eGFR: estimated glomerular filtration rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; CV: coefficient of variation.
For categorical variables, n (%) is presented. For continuous variables, median (interquartile range) is presented.
The unadjusted odds ratio (95% CI) of morning, evening and clinic systolic blood pressure (SBP) for cognitive impairment were 1.18 (1.00–1.40; p = 0.051), 1.20 (1.01–1.43; p = 0.036) and 1.09 (0.94–1.26; p = 0.257), respectively. The adjusted odds ratio (95% CI) of morning, evening and clinic SBP for cognitive impairment were 1.19 (0.98–1.45; p = 0.080), 1.24 (1.02–1.53; p = 0.037) and 1.12 (0.94–1.34; p = 0.192), respectively. Evening SBP showed the highest AUC in both unadjusted and adjusted models (Table 2). The unadjusted odds ratio (95% CI) of CV of morning SBP and evening SBP were 1.03 (0.92–1.16; p = 0.566) and 1.09 (1.00–1.19; p = 0.057), respectively. The adjusted odds ratio (95% CI) of CV of morning SBP and evening SBP were 1.04 (0.90–1.19; p = 0.590) and 1.11 (1.00–1.22; p = 0.049), respectively. We have evaluated the relation between cognitive impairment and the transition of BP as a graph of percentage of cognitive impairment and BP distribution (Figure 2). In all subgroup analyses except gender, morning or evening SBP showed the highest AUC, while in female, clinic SBP showed the highest AUC (Supplementary Tables 1–5).
Unadjusted and adjusted odds ratios (ORs) for cognitive impairment.
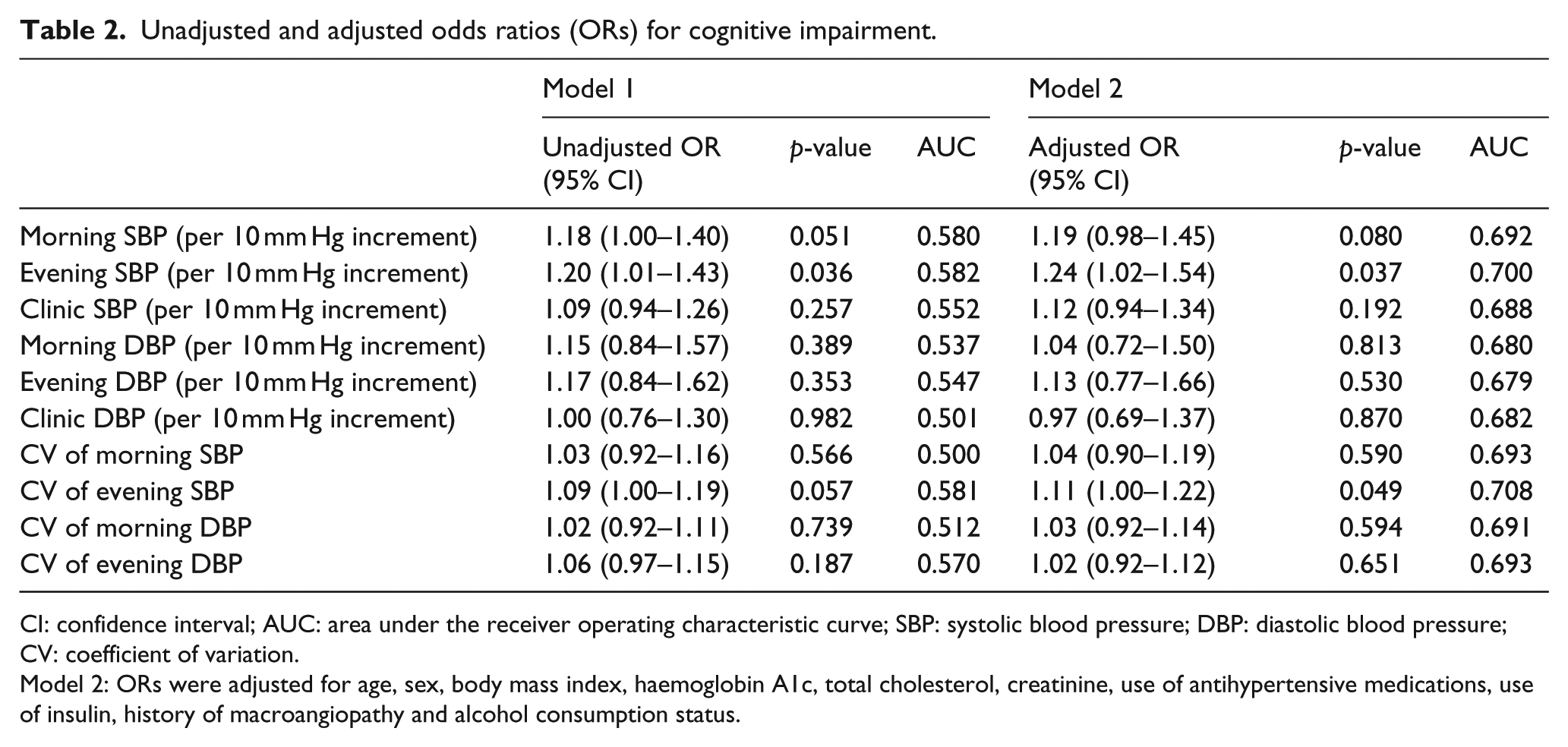
CI: confidence interval; AUC: area under the receiver operating characteristic curve; SBP: systolic blood pressure; DBP: diastolic blood pressure; CV: coefficient of variation.
Model 2: ORs were adjusted for age, sex, body mass index, haemoglobin A1c, total cholesterol, creatinine, use of antihypertensive medications, use of insulin, history of macroangiopathy and alcohol consumption status.
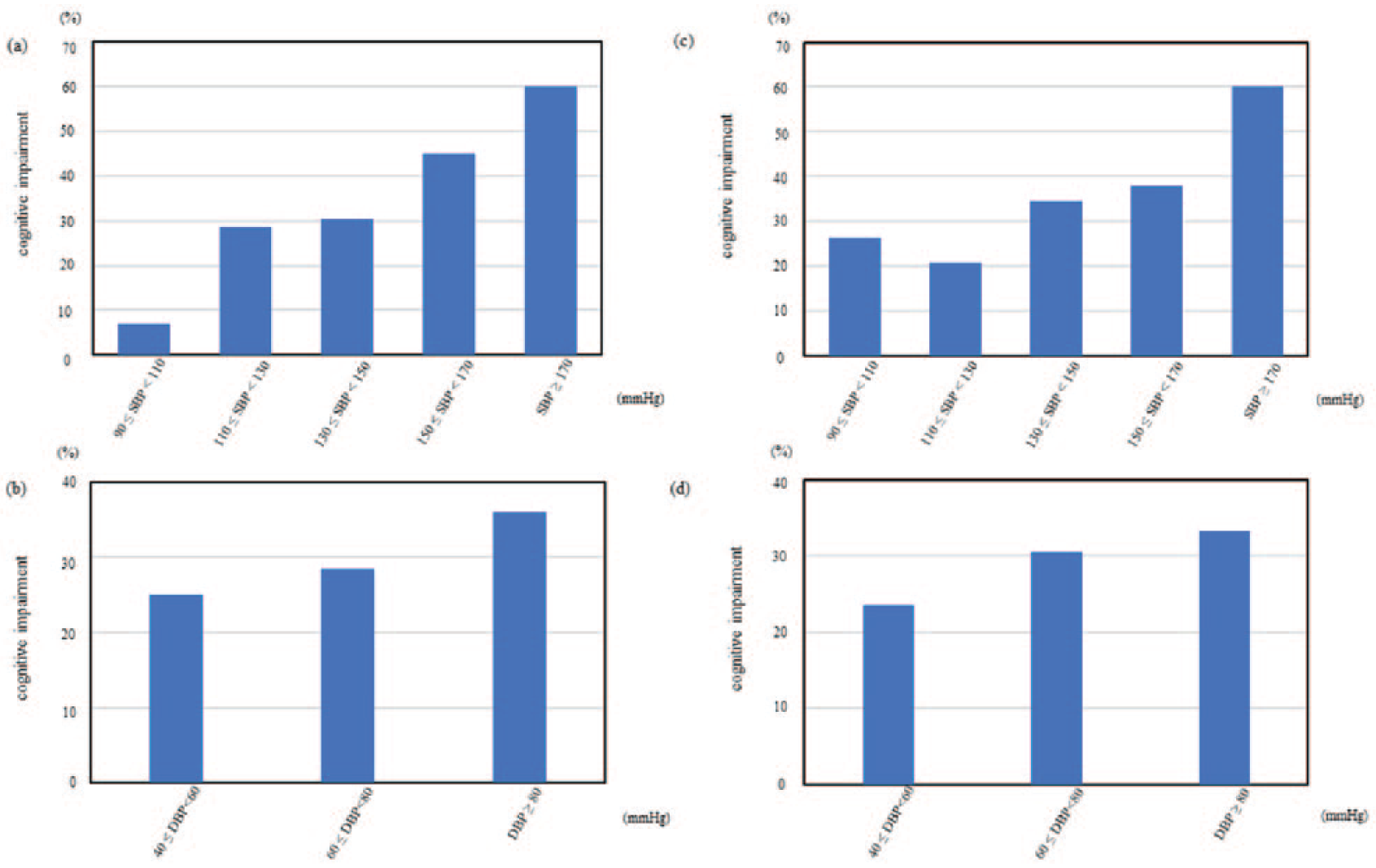
Relationship between cognitive impairment and (a) morning SBP, (b) morning DBP, (c) evening SBP and (d) evening DBP.
Discussion
Principal findings
This study indicated that increased HBP is associated with poorer cognitive performance than clinic BP in patients with diabetes. Evening SBP showed a positive relationship with cognitive impairment independent of age, sex, body mass index, haemoglobin A1c, total cholesterol, creatinine, use of antihypertensive medications, use of insulin, history of macrovascular complications and alcohol consumption status. The results of this study are consistent with those in the previous report, 14 in which Wu et al. have showed in a cross-sectional study in elderlies with hypertension that clinic BP control correlated with cognitive function evaluated by Mini-Mental State Examination.
Interpretations
In regard to associations of HBP and cognitive function, Yeung and Loken Thornton 15 reported HBP as a predictor of neuropsychological function in older adults beyond cognitive screening. They showed that higher home systolic BP predicted worse processing speed, executive function and everyday cognitive function. The possible reasons can be explained by the fact that HBP measurements provide more reproducible information on BP, exclude the white coat effect and have more prognostic significance 16 than conventional BP measurements.
Another study reported that increased day-to-day BP variability is, independent of average HBP, a significant risk factor for the development of dementia in the general elderly Japanese population. 17 Day-to-day BP variability and short-term BP variability can be related to autonomic dysfunction, 18 and it has been reported that orthostatic hypotension, one of the symptoms of autonomic dysfunction, can cause cerebral ischaemia and associate with dementia. The results of this study, which showed the association of day-by-day variability and cognitive impairment, were also consistent with the previous reports.18,19 In diabetes patients, autonomic dysfunction frequently occurs which may influence BP variability and may be associated with the risk of cognitive impairment, equal to or higher than that of the general elderly Japanese population.
In this study, evening SBP showed the highest AUC for cognitive impairment. Ribeiro et al. 20 reported that the BP circadian profiles showed a lower variability, with the absence of a nocturnal fall, especially in diabetes patients with chronic complications such as autonomic neuropathy and macroangiopathy. In diabetes patients with cardiovascular autonomic dysfunction, the relative risk of mortality is high due to the high incidence of painless myocardial infarction and severe arrhythmia. 21 In this study, we did not measure the nocturnal BP, but in patients with high evening BP, there might be an absence of a nocturnal fall 22 related to the onset of cardiovascular events.
Lv et al. 23 reported a U-shaped association between BP values and prevalence of cognitive impairment, but there was no such relationship found in this study.
In all subgroup analyses except gender, morning or evening SBP showed the highest AUC for cognitive impairment, while in female, clinic SBP showed the highest AUC of all (Supplementary Tables 1–5).
A previous study has demonstrated that female gender is positively associated with HBP variability. 24 As a mechanism for increasing female BP variability, intrinsic factors such as instability of the autonomic nervous system and more obvious emotional fluctuation during female BP measurement can be considered. These findings may well explain why females have a higher incidence of white coat hypertension and, in this study, why clinic SBP showed the highest AUC.
This study has several limitations. First, since this is a cross-sectional study, the causal relationship is not clear. For a useful evaluation of the relationship between HBP measurements and the cognitive impairment among patients with type 2 diabetes, a randomized controlled trial with larger sample sizes and a long-term follow-up period should be conducted. Second, the evaluation of cognitive impairment is performed only for a single Mini-Cog test, and the evaluation (Alzheimer’s type dementia, vascular dementia, Lewy body disease, etc.) or classification of dementia such as image evaluation is insufficient. Third, it has been reported that there was not enough evidence to support the routine use of the Mini-Cog test as a screening test for dementia in primary care. 11 However, we prioritized and selected the benefits of the Mini-Cog test, which can be done in a short time during the busy outpatient clinic in this study. Finally, this study lacks data for daytime BP and nocturnal BP measurements.
Conclusion
Cognitive impairment, including mild cognitive impairment, significantly reduces quality of life and shortens life expectancy. Considering both the social burden of cognitive impairment and the higher risk for cognitive impairment in diabetic patients, it is as important to prevent cognitive impairment as to treat diabetes. Our findings implicate the possibility that measuring not only clinic BP but also HBP, especially evening SBP, may lead to prediction of progress of cognitive impairment. Appropriate control for HBP and blood glucose level might be important to prevent cognitive impairment as well as cardiovascular diseases in clinical practice.
Supplemental Material
R_Supplementaries – Supplemental material for Home blood pressure is associated with cognitive impairment among elderly patients with type 2 diabetes: KAMOGAWA-HBP study
Supplemental material, R_Supplementaries for Home blood pressure is associated with cognitive impairment among elderly patients with type 2 diabetes: KAMOGAWA-HBP study by Keiko Iwai, Emi Ushigome, Shinobu Matsumoto, Nobuko Kitagawa, Hidetaka Ushigome, Isao Yokota, Mai Asano, Masahide Hamaguchi, Masahiro Yamazaki and Michiaki Fukui in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors acknowledge Naoko Higo R.N., Machiko Hasegawa R.N. and Terumi Kaneko R.N. of the Kyoto Prefectural University of Medicine for teaching patients how to measure their blood pressure and also Sayoko Horibe of the Kyoto Prefectural University of Medicine for their secretarial assistance.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.I., E.U., S.M., N.K., M.A., M.H., M.Y. and M.F. have received grants, honoraria and research supports from AstraZeneca plc., Astellas Pharma Inc., Nippon Boehringer Ingelheim Co., Ltd., Daiichi Sankyo Co., Ltd., Eli Lilly Japan K.K., Kyowa Hakko Kirin Company Ltd., Kissei Pharmaceutical Co., Ltd., MSD K.K., Mitsubishi Tanabe Pharma Corporation, Novo Nordisk Pharma Ltd., Sanwa Kagaku Kenkyusho Co., Ltd., Sanofi K.K., Ono Pharmaceutical Co., Ltd. and Takeda Pharmaceutical Co., Ltd. The sponsors were not involved in the study design; in the collection, analysis and interpretation of data; in the writing of this manuscript; or in the decision to submit the article for publication. The authors, their immediate families and any research foundations with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article. The authors declare that although they are affiliated with a department that is supported financially by pharmaceutical company, the authors received no current funding for this study and this does not alter their adherence to all the journal policies on sharing data and materials. The other authors have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japanese Study Group for Physiology and Management of Blood Pressure and the Astellas Foundation for Research on Metabolic Disorders (grant number 4024).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
