Abstract
Background:
Peripheral arterial disease in patients with type 2 diabetes mellitus is an important risk factor for vascular events. Recommendations about whether ankle-brachial index should be performed differ depending on the source; therefore, it is necessary to re-evaluate the most important risk factors associated with peripheral arterial disease and whether it is useful to perform ankle-brachial index in newly diagnosed and drug-naïve patients with diabetes, independent of age or peripheral arterial disease symptoms.
Methods:
A total of 711 subjects were divided into groups: group 1, 600 type 2 diabetes mellitus patients, symptomatic or not for peripheral arterial disease; group 2, 61 type 2 diabetes mellitus patients newly diagnosed and drug naïve; and group 3, 50 subjects without diabetes. Ankle-brachial index, medical records and physical examination were performed in all patients, accessing cardiovascular risk factors.
Results:
Analysing group 1 asymptomatic patient to peripheral arterial disease, we found abnormal ankle-brachial index in 49% (77/156) ⩾50 years and 42% (16/38) <50 years (p = not significant). Considering drug-naïve patients, a peripheral arterial disease prevalence of 39% (24/61) was found; among these, 48% (13/27) were <50 years and 32% (11/34) were ⩾50 years (p = not significant). A forward stepwise regression model was developed, with type 2 diabetes mellitus duration (r2 = 0.12) and sedentary lifestyle (r2 = 0.14) found as independent variable predictors of severity of peripheral arterial disease, related to ankle-brachial index.
Conclusion:
We suggest that, in type 2 diabetes mellitus, ankle-brachial index should be measured at diagnosis. In addition, sedentary lifestyle was strongly associated with presence and severity of peripheral arterial disease.
Background
Atherosclerotic cardiovascular diseases are the main cause of mortality in individuals with diabetes mellitus (DM) and are responsible for most of the public health costs related to this disease. 1 In addition, peripheral arterial disease (PAD) in patients with DM is an important risk factor for lower limb amputation, being associated with other vascular events (stroke and myocardial infarction) 1 and increasing the probability of cardiovascular diseases up to one-third in patients with diabetes at a more advanced stage. 2
Despite the fact that risk factors related to PAD in patients with DM are still not well established, it is well known that the ankle-brachial index (ABI) is the procedure of choice for the diagnosis of this pathology, 3 since it is non-invasive, low cost, easy to perform 4 and precedes PAD symptoms. The American College of Cardiology/American Heart Association (AHA) has recommended screening asymptomatic patients with diabetes older than 50 years or who are younger than 50 years with additional risk factors for cardiovascular disease for PAD. 5 Incongruously, American Diabetes Association (ADA) recommended, in 2018, that ABI test should only be performed in type 2 diabetes mellitus (T2DM) patients with symptoms or signs of PAD. 2 However, the increase in DM prevalence makes it necessary to re-evaluate the most important risk factors associated with PAD and whether it is useful to perform ABI in newly diagnosed and drug-naïve patients with diabetes, independent of age or PAD symptoms.
Methods
Study design and patients
We performed a cross-sectional and prospective study to evaluate PAD in patients with T2DM and controls. This study was developed according to the Declaration of Helsinki and the Nuremberg Code and was approved by the University Hospital João de Barros Barreto ethics committee. Signed consent was obtained from all patients.
A total of 711 subjects were divided into three groups: group 1, 600 T2DM patients, symptomatic or not for PAD; group 2, 61 patients with T2DM newly diagnosed (<3 months) and drug naïve; and group 3, 50 subjects without diabetes. Drug-naïve patients with diabetes are a rare cohort, which explains the small number of patients in this group. Moreover, control group was used only to compare ABI to the drug-naïve group and showed that ABI was lower even in recently diagnosed patients when compared to subjects without diabetes. All participants were between 30 and 85 years and patients with T2DM must have been followed at the endocrinology outpatient clinic and must not have undergone peripheral revascularization surgery.
Clinical and laboratory data
Analysis of medical records and physical examination were performed. Information on demographics, physical measures, cardiovascular risk factors, pre-existing clinical conditions, duration of T2DM (in years), medications in use, laboratory tests, ABI and body mass index (BMI) was analysed.
The cardiovascular risk factors accessed were as follows: obesity and overweight, dyslipidaemia, systemic arterial hypertension, alcohol intake, smoking, diabetes duration and sedentary lifestyle. All parameters were based on ADA’s recommendations for individuals with T2DM. 2 Sedentary lifestyle was defined as performing physical activity less than 150 min per week according to American College of Sports Medicine and the AHA. 6 Acute myocardial infarction, stroke, cardiac catheterization, coronary revascularization [percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG)], heart failure, angina pectoris and atrial fibrillation were the clinical conditions evaluated. After first evaluation, patients were followed for 3 years with standard care visits each 3 months to register the occurrence of cardiovascular events. Only cardiovascular events adjudicated in medical records, and evaluated by the study team, were considered. This search for cardiovascular events 3 years after the measurement of ABI is important to analyse risk factors. The study was carried out between January 2013 and February 2018.
ABI was performed using a vascular Doppler, based on the American College of Cardiology/AHA recommendations. 7 As a parameter of normality, the lowest ABI value between right and left was considered. Patients with an ABI ⩽0.9 were diagnosed as having PAD. PAD symptoms and signs included claudication, ischaemic rest pain and tissue loss (ulcer/gangrene).
Values between 0.71 and 0.9; 0.41 and 0.7; and <0.41 were considered as mild, moderate and severe obstruction, respectively. Values >1.40 indicate that the arteries were not able to be compressed, which is more common among individuals with DM and/or advanced chronic kidney disease. According to AHA guidelines for management of PAD, in the setting of non-compressible ABI values, additional imaging is highly recommended to diagnose PAD if the clinical suspicion is significant. 7 During our recruitment, we have found only five patients with ABI >1.4 and those cases were excluded from the study.
As we are a tertiary hospital for PAD and diabetes, we expected to find a high prevalence of PAD in our sample. The control group was considered as a parameter for normal decrease in ABI that occurs with ageing.
HbA1c and fasting glycaemia were measured by the high-performance liquid chromatography (HPLC) method. In addition, serum triglycerides, total cholesterol, low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) were measured, as well as serum creatinine, which was used to calculate the glomerular filtration rate (GFR) using the simplified Modification of Diet in Renal Disease study equation. 8 Ultrasensitive C-reactive protein (PCR-US) was measured by the nephelometry method, with a detection limit of 0.01 mg/dL. To evaluate the presence and degree of nephropathy, the patients were submitted to albuminuria/creatinine (three isolated urinary samples) by immunoturbidimetry.
Statistical analysis
An ABI score was established to evaluate the severity of the arterial obstruction, in which normal individuals were assigned a value of 0 - normal, 1 - mild, 2 - moderate and 3 - severe obstruction. To establish the relationship between risk factors, linear and logistic regression models were created. Drug-naïve patients were excluded from linear and logistic regression, considering that their duration of diabetes equals 0. To establish correlations between variables, Pearson’s or Spearman’s test was used. Value of p less than 0.05 was considered statistically significant. All calculations were performed with SPSS Statistics 22.0 (IBM, USA).
Results
In our study, in group 1 (N = 600), 490 (82%) presented PAD and 110 (18%) had normal ABI. Considering only PAD patients, 296 (60%) were symptomatic [claudication, ischaemic rest pain and tissue loss (ulcer/gangrene)] and 194 (40%) presented no symptoms. Clinical and laboratory characteristics of the groups are described in Tables 1 and 2, respectively.
Clinical features and cardiovascular risk factors.
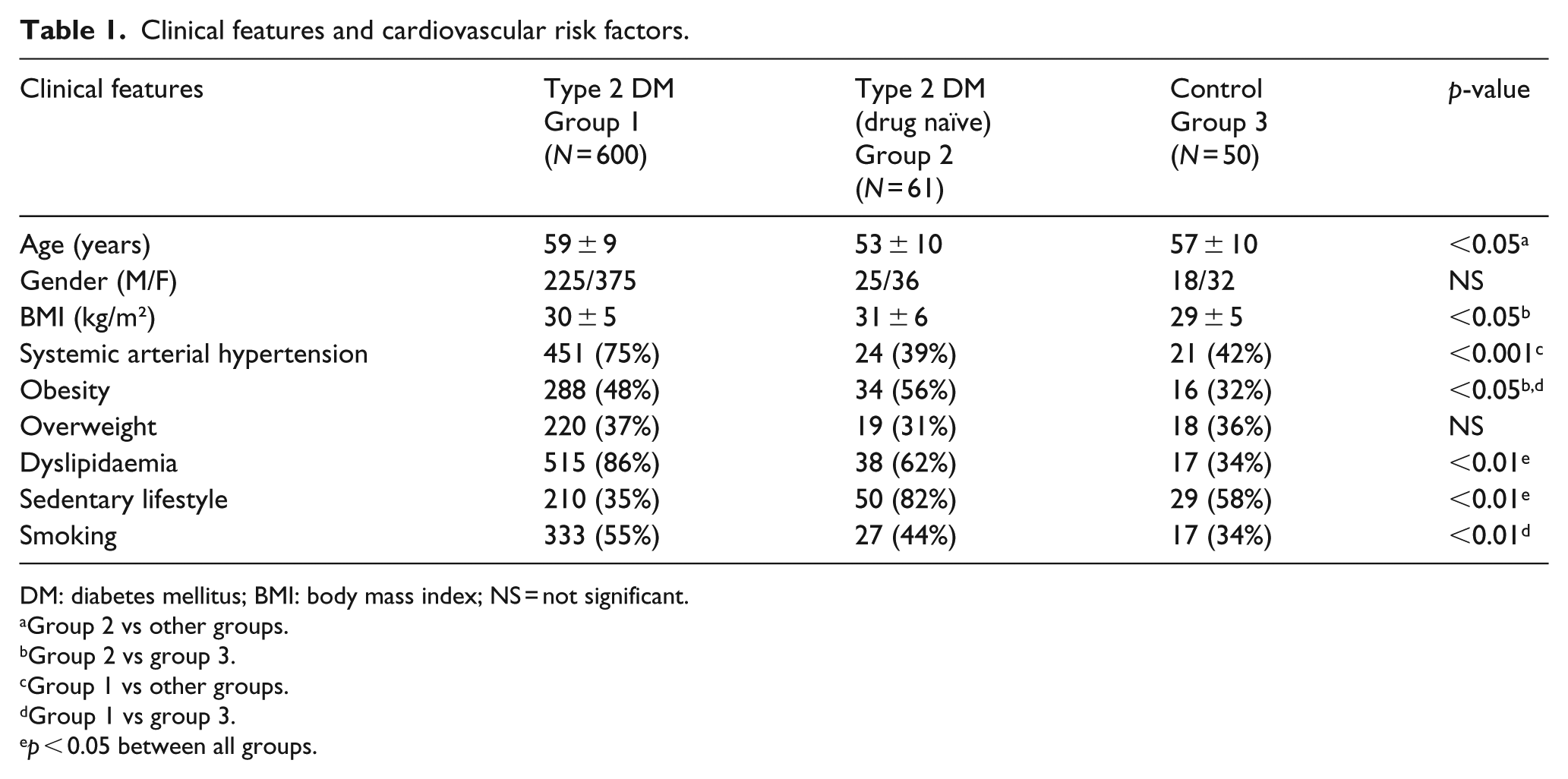
DM: diabetes mellitus; BMI: body mass index; NS = not significant.
Group 2 vs other groups.
Group 2 vs group 3.
Group 1 vs other groups.
Group 1 vs group 3.
p < 0.05 between all groups.
Laboratory features of T2DM individuals.
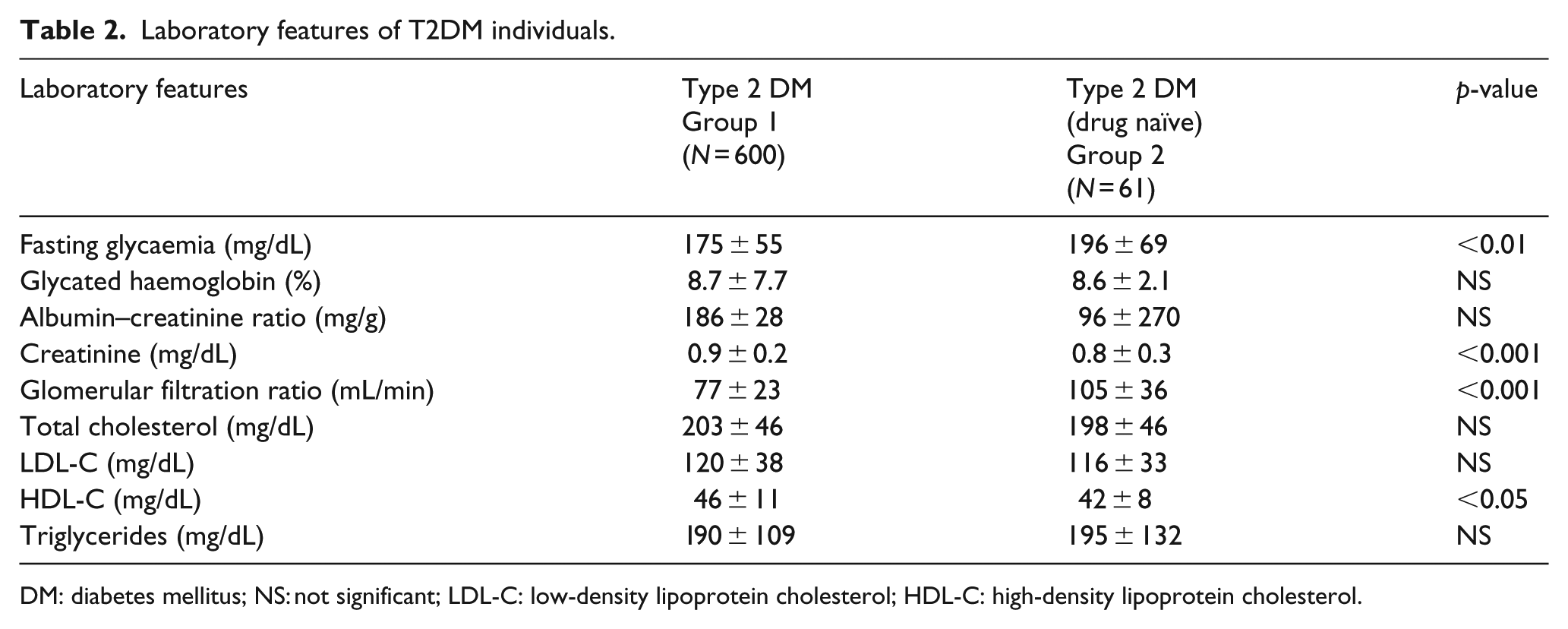
DM: diabetes mellitus; NS: not significant; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol.
Prevalence of cardiovascular events and comparison of ABI and ABI index between groups are shown in Tables 3 and 4, respectively. Table 3 observations were collected by medical records during the 3-year follow-up. When PAD severity was evaluated, group 1 showed worse ABI score compared to group 2 (0.98 ± 0.6 vs 0.51 ± 0.70; p < 0.01).
Prevalence of cardiovascular events in T2DM.
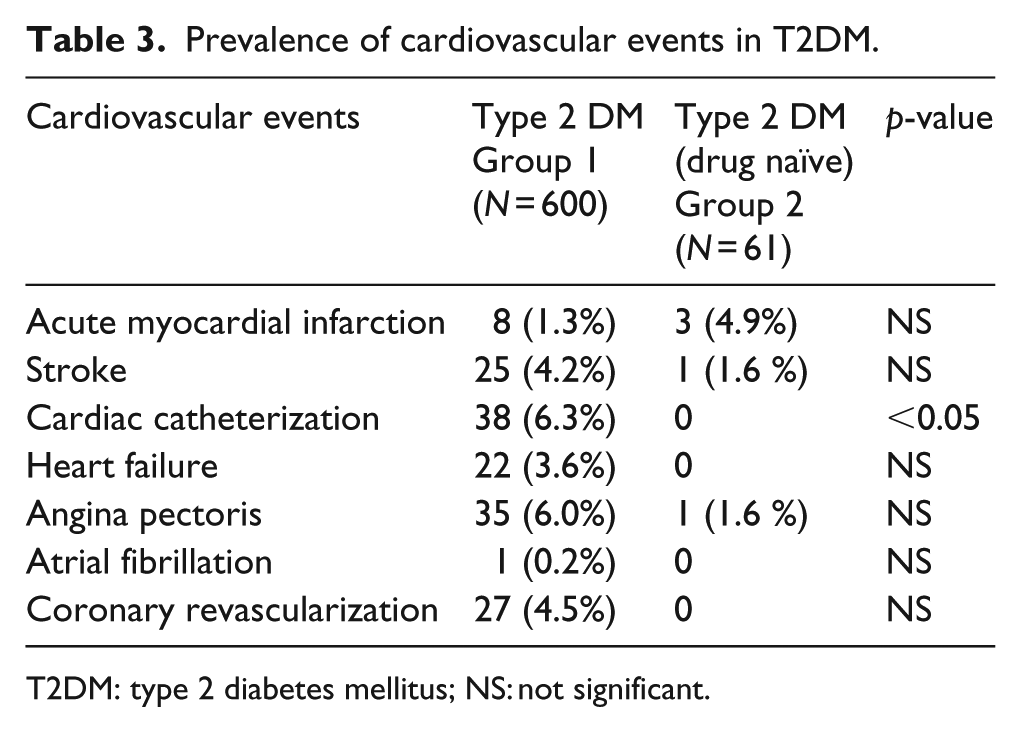
T2DM: type 2 diabetes mellitus; NS: not significant.
ABI, PAD prevalence and severity.
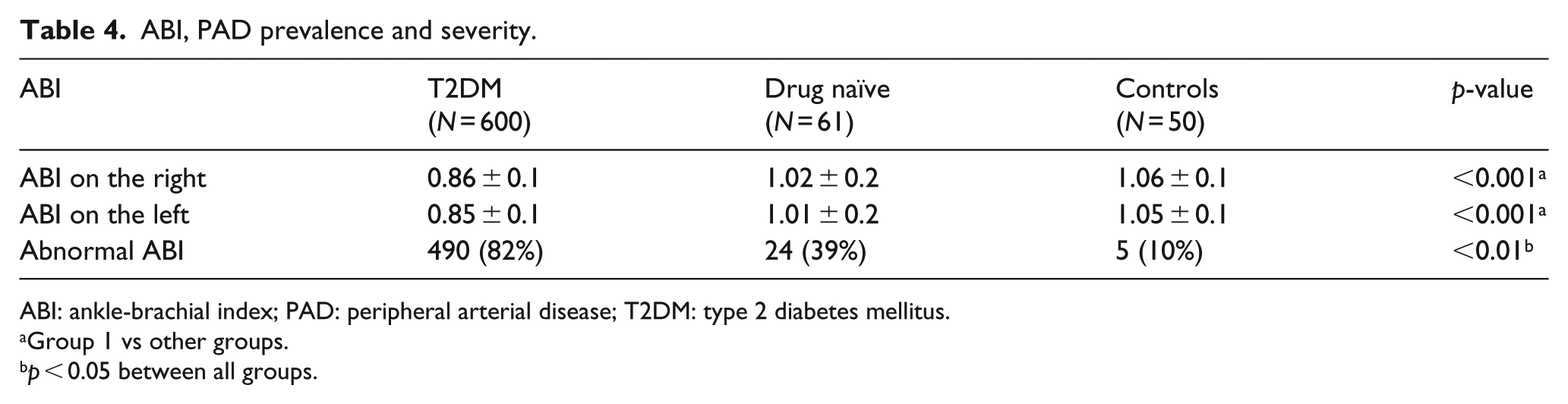
ABI: ankle-brachial index; PAD: peripheral arterial disease; T2DM: type 2 diabetes mellitus.
Group 1 vs other groups.
p < 0.05 between all groups.
The occurrence of cardiovascular events showed correlations with ABI levels and ABI score (r = −0.2, p < 0.01; r = 0.2, p < 0.01), respectively. The correlations between presence of PAD, ABI levels, ABI score and their risk factors are shown in Tables 5 and 6, respectively.
Correlations between presence of PAD and their risk factors.
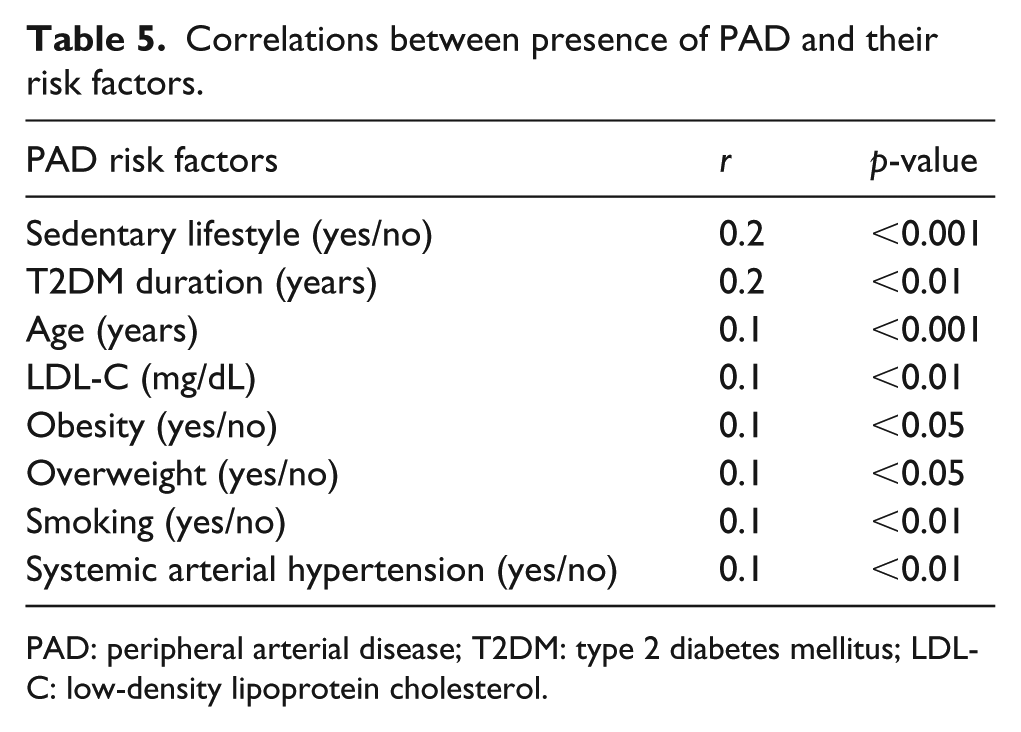
PAD: peripheral arterial disease; T2DM: type 2 diabetes mellitus; LDL-C: low-density lipoprotein cholesterol.
Correlations between ABI values, ABI score and PAD risk factors.
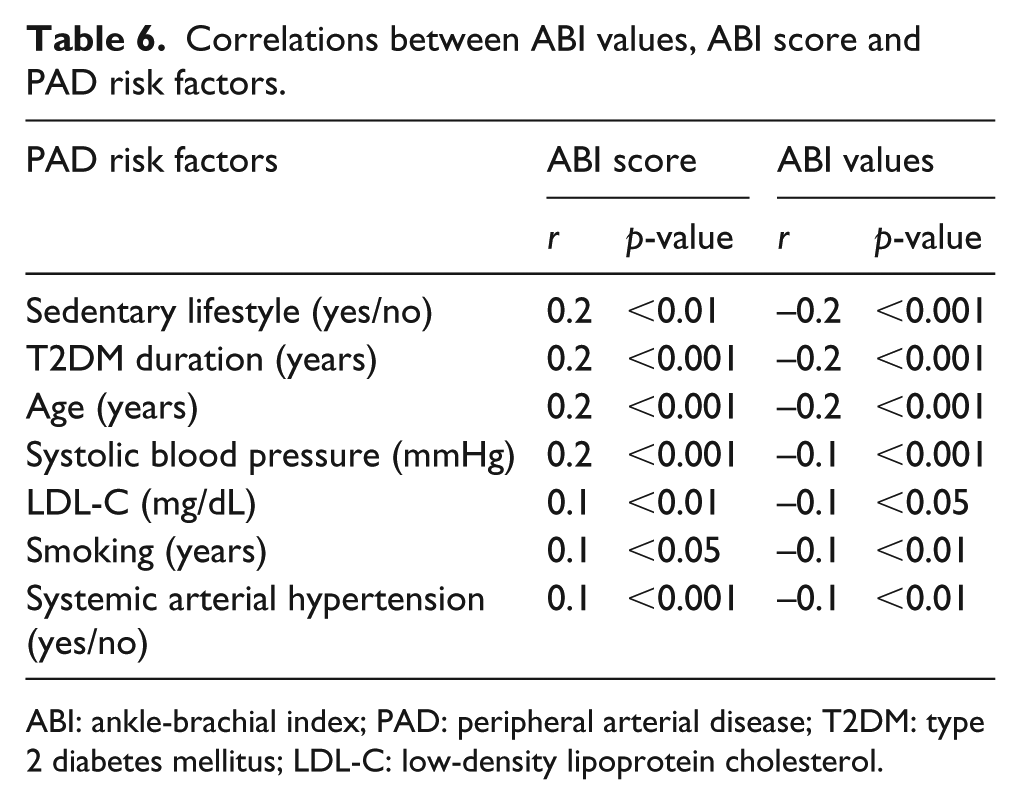
ABI: ankle-brachial index; PAD: peripheral arterial disease; T2DM: type 2 diabetes mellitus; LDL-C: low-density lipoprotein cholesterol.
Glycated haemoglobin and fasting glycaemia did not correlate with ABI value or presence and severity of PAD in T2DM group.
A forward stepwise regression model was developed using ABI score (PAD severity) as dependent variable and age, systolic blood pressure, BMI, sedentary lifestyle and T2DM duration as independent variables. Only T2DM duration (r2 = 0.12) and sedentary lifestyle (r2 = 0.14) were found as independent variable predictors of severity of PAD, related to both ABI level and ABI score.
An inclination coefficient of −0.003 was obtained using ABI and T2DM duration as variables in a simple linear regression model according to the formula ‘ABI = 0.847 – (0.00327 × T2DM duration)’, which suggests that, for each year of T2DM evolution, there would be a decrease of 0.003 in ABI.
Using a binary logistic regression model, patients with diabetes duration of 10 years or more were at a higher risk of PAD [odds ratio = 3.3 (confidence interval (CI) = l.7–6.2; Wald statistic = 14.1; p < 0.001)] compared to patients with less than 10 years of diagnosis. Also, through a binary logistic regression model, sedentary individuals presented a higher prevalence of PAD [odds ratio = 2.5 (CI = l.4–4.5; Wald statistic = 9.2; p < 0.01)] compared to those who practised regular physical activities according to ADA recommendations.
When only group 1 asymptomatic patients to PAD were analysed, we found abnormal ABI in 49% (77/156) ⩾50 years and 42% (16/38) <50 years (p = NS). Considering just drug-naïve patients, a PAD prevalence of 39% (24/61) was found; among these, 48% (13/27) were <50 years and 32% (11/34) were ⩾50 years (p = NS). In addition, 28% (17/61) of all drug-naïve patients had PAD at the time of diagnosis without any symptoms.
Discussion
Our study found a high prevalence of abnormal ABI values in early diagnosed drug-naïve patients with diabetes. In addition, in our 600 diabetic cohort group, sedentary lifestyle and diabetes duration were the risk factors most intimately related to presence and severity of PAD.
The drug-naïve group showed a PAD prevalence of 28%, in asymptomatic patients, including younger than 50 years. Currently, based on ADA 2018, it is not recommended to perform ABI in T2DM patients without PAD symptoms. 2 Our data suggest that this recommendation should be expanded, but a larger cohort group study would be necessary to confirm our results.
In addition, our data indicated sedentary lifestyle and diabetes duration as important risk factors related to presence and severity of PAD. Recently, it has been shown that muscle strength is related to flat ground walking ability in PAD.9,10 In a recent meta-analysis, Parmenter et al. 11 have also found a strong relation between regular exercising and PAD. Their findings suggested that despite significant improvements in cardiorespiratory fitness and walking distances, diagnostic and prognostic measures such as ankle-brachial, blood pressure index and flow-mediated dilatation would remain unchanged following exercise training. It suggested that changes in blood flow and/or pressure should not be the mechanism to explain why exercise improves walking ability in this cohort. Nevertheless, they have not assessed T2DM cohort groups. It has been suggested that ABI would not improve with exercise in patients with diabetes and PAD, but it remains controversial.11,12 Barone Gibbs et al., 13 in a randomized study, evaluating T2DM without PAD, showed an improvement in ABI, suggesting a possible preventive effect of physical activity. If this is true, performing ABI as a screening method for PAD in T2DM at the time of diagnosis becomes even more important.
Diabetes duration was also an important risk factor for PAD according to our results. A few previous researches suggested that time of disease should remain a risk factor even for patients who were still young when diagnosed.14,15 The importance of this risk factor, associated with the high prevalence of PAD in early diagnosed drug-naïve T2DM patients, just confirms the existence of a delay to detect that disease. 16 In addition, Faglia et al., in a recent cross-sectional study with a larger number of newly diagnosed type 2 diabetes patients in Italy, showed that 21% of these already had ABI ⩽0.9, reinforcing our findings. Therefore, increasing screening actions to T2DM is imperative to promote prevention of PAD.
Joosten et al., 17 analysing the association between smoking, hypertension, hypercholesterolaemia and T2DM as PAD predictors in men, have concluded that patients who did not present one or more of these risk factors had 77% lower risk of developing this disease. Smoking was not strongly associated as a risk factor for PAD in our study. Given our use of ADA 2018 criteria to classify smokers, those who smoked only one cigarette per day were also taken into account. However, a cross-sectional study with 403 patients showed that a minimum quantity of 20 cigarettes per day was necessary to increase the risk of PAD development. 18 Moreover, Malta et al. 19 found a trend in smoking reduction (<20 cigarettes/day) in the northern region of Brazil, from 3.2% in 2007 to 1.7% in 2014. Therefore, the broad ADA criteria and a decrease in our population smoking load could be the reason why we have not found this habit as a relevant risk. In addition, another possible explanation could be that diabetic patients often have distal arterial disease (such as tibial) rather than proximal disease (such as iliac), the one associated with smoking.
Finally, Weragoda et al. 15 pointed not only DM, hypertension, dyslipidaemia and smoking as principal risk factors for PAD but also its lack of awareness, which leads to delayed diagnosis, low compliance and unfavourable outcomes. Therefore, measuring ABI as early as possible in all T2DM patients becomes more imperative. Our data reinforce that approach.
Our main finding conveys that patients with diabetes should perform ABI at the time of diagnosis, which is justified by the need of early PAD detection. Diabetes duration is a well-described risk factor for PAD in the literature and was also found in our study. T2DM patients already have delayed diagnosis – estimated to be around 5 years – thus being exposed to an important risk factor since disease detection. In our study, drug-naïve patients were examples of newly diagnosed patients who had received no influence of diabetes medications whatsoever, and yet 39% had PAD – regarding drug-naïve patients with PAD, 28% were asymptomatic. Early recognition would help prevent and treat PAD adequately, regarding especially asymptomatic patients (40% in PAD patients) whose course of illness, according to current screening recommendations, would not be avoided due to lack of prompt diagnosis and treatment.
The main limitation of our study was not stratifying exercise practice in mild, moderate and intense obstruction, which could have led to more adequate analysis of the real influence of physical activity in PAD. It occurred because we have used ADA 2018 statements to classify physical activity. Other weakness is the small number of patients in group 2 (drug-naïve group). Control group was used only as a parameter for ABI values in our population and drug-naïve group was limited because of narrow inclusion criteria. The small difference between age and BMI between those two groups does not really interfere in our results, since drug-naïve patients presented worse ABI values but were younger, and this index decreases with age.
Conclusion
Our study suggests that, in T2DM, due to high prevalence of abnormal ABI in newly diagnosed drug-naïve patients, this measurement should be performed independently of age and PAD symptoms. In addition, sedentary lifestyle was strongly associated with the presence and severity of PAD.
Footnotes
Acknowledgements
The authors thank Pró-Reitoria de Pesquisa (PROPESP) of Federal University of Pará and Programa de Pós-Graduação em Oncologia e Ciências Médicas of University Hospital João de Barros Barreto-Endocrinology Division, Federal University of Pará, for the assistance. The authors thank Dr J.F. from the University Hospital João de Barros Barreto – Endocrinology Division, Federal University of Pará, for the technical assistance. All persons who met authorship criteria are listed as authors, and all authors certify that they have participated sufficiently in the work to take public responsibility for the content, including participation in the concept, design, analysis, writing or revision of the manuscript: C.C.K. and J.S.F. took part in conception and design of study; K.M.F., N.N.M.Q., A.L.A., M.C.S., D.D.S., A.C.C.B.S. and F.T.C.M. were responsible for acquisition of data, while N.A.Z., F.S.R., R.J.M.C.P. and J.F.A.N. have done the analysis and interpretation of data; and M.N.L., L.M.C.F., N.J.K.S.N., I.I.F.F. and M.C.N.I.O. have drafted the manuscript together. All authors have revised the manuscript critically and approved the version to be published.
Availability of data and materials
The datasets during and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. This study was approved by the University Hospital Joao de Barros Barreto ethics committee.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Informed consent was obtained from all patients for being included in the study.
