Abstract
Aim:
The aim of this study was to assess the renal effects of the glucose-lowering SGLT2 inhibitors in Japanese type 2 diabetes mellitus patients with chronic kidney disease.
Methods:
The Kanagawa Physicians Association maintains a registry of patients who visit their 31 clinics. Clinical data of type 2 diabetes mellitus patients with chronic kidney disease, who were prescribed SGLT2 inhibitors in addition to other treatments, were collected and analysed.
Results:
SGLT2i was associated with a fall in HbA1c from 64.1 ± 16.7 mmol/mol (8.0 ± 1.5%) to 56.5 ± 12.9 mmol/mol (7.3 ± 1.2%) (p < 0.01) in 869 analysed cases, a decrease in urine albumin-creatinine ratio from a median of 47.1 to 41.1 mg/gCr, and decrease in estimated glomerular filtration rate from 77.7 ± 23.9 to 75.0 ± 23.9 mL/min/1.73 m2 (p < 0.01). The effect on albumin-creatinine ratio was independent of age or stage of estimated glomerular filtration; however, there was a significant negative correlation between albumin-creatinine ratio at the initiation of SGLT2 inhibitor and change in ACR. Multiple linear regression analysis identified use of empagliflozin, β-blockers, and sulphonylureas, Δsystolic blood pressure at office, serum Cr and albumin-creatinine ratio value at initiation of SGLT2 inhibitor as independent and significant determinants of change in ACR.
Conclusions:
This study confirmed that the beneficial renal effects of SGLT2 inhibitor in Japanese type 2 diabetes mellitus patients with chronic kidney disease, similar to those reported in large-scale clinical trials conducted in Western countries.
Keywords
Introduction
In addition to their direct effect on blood glucose level, the sodium-glucose co-transporter 2 inhibitors (SGLT2i) have other indirect effects, such as reduction of blood pressure (BP) and improvement of dyslipidemia and liver function. These pleiotropic effects have attracted attention and wide use of SGLT2i in the treatment of type 2 diabetes mellitus (T2DM). The results of several large-scale cardiovascular outcome trials on SGLT2i, such as the EMPA-REG OUTCOME trial 1 and CANVAS/CANVAS-R programme 2 showed significant improvement in cardiovascular events and diabetic nephropathy in T2DM patients. In Japan, six types of SGLT2i have already been approved. To our knowledge, only one large-scale clinical trial of Japanese patients has reported SGLT2i-related improvement in cardiovascular events. 3 The majority of patients in the above clinical trials of SGLT2i1–3 had past history or high-risk factors for cardiovascular events. In Japan, more than one million people are thought to suffer chronic kidney disease (CKD), 4 and we selected CKD to represent high-risk patients in this study. The aim of this retrospective study was to define the renal effects of SGLT2i in Japanese T2DM patients with CKD.
Methods
Study design and study population
The study subjects included all 935 T2DM patients who were registered and visited the clinics of members of the Kanagawa Physicians Association between November 2016 and March 2017. The inclusion criteria were as follows: T2DM patients (1) who commenced treatment with SGLT2i for the first time, (2) had CKD as defined by the K/DOQI clinical practice guidelines for CKD, 5 and (3) more than 20 years of age. Finally, 66 patients were excluded for incomplete data or not meeting the above criteria, and the study included data of 869 patients. The time from the initiation of SGLT2i therapy to the present study, that is, the median duration of treatment with SGLT2i was 13.0 months (range: 1–36). The estimated glomerular filtration rate (eGFR) was calculated using the following formula: [eGFR [mL/min/1.73(mL/min/1.73 m2) = 194 × age−0.287 × serum creatinine−1.094× [0.739(0.739 for)]. 6 The study was approved by the Human Ethics Committee of Kanagawa Medical Association (No. 1576, 23 Aug 2016).
Type of SGLT2i and combination treatments
Six types of SGLT2i were used by the patients; ipragliflozin (28.5%), dapagliflozin (17.8%), tofogliflozin (17.0%), luseogliflozin (7.5%), canagliflozin (12.0%) and empagliflozin (16.6%). Furthermore, the following glucose-lowering agents were used: SGLT2i alone (10.0% of the study patients), SGLT2i with dipeptidyl peptide-se-4 (DPP4) inhibitors (56.6%), sulphonylureas (32.2%), metformin (55.3%), insulin (24.6%), glucagon-like peptide-1(GLP-1) receptor agonists (12.5%) and pioglitazone (14.4%).
Statistical analysis
Data are reported as mean ± SD or median [lower quartile, upper quartile]. Differences between two groups were assessed by the t-paired test for parametric data and Wilcoxon signed-rank test for nonparametric data. A p value less than 0.05 was considered significant. Differences among more than two groups were tested by the Mann–Whitney test with Bonferroni correction. A p value <0.017 was considered significant on comparisons among three groups and <0.008 for comparison among four groups. Multiple linear regression analysis was performed to evaluate independent predictors of the change in ACR (ΔACR).
Results
Changes in clinical findings
Age varied widely with a mean of 60.1 ± 12.4 years, and this study included more males (n = 562) than females (n = 307). Body weight (kg), body mass index (BMI, kg/m2), BP at office (systolic/diastolic, mmHg), HbA1c level (mmol/mol) and eGFR (mL/min/1.73 m2) were 76.9 ± 16.7, 27.8 ± 5.0, 138 ± 19/79 ± 13, 64.1 ± 16.7 (8.0 ± 1.5%) and 77.7 ± 23.9 at initiation of SGLT2i, and 74.5 ± 16.2 (p < 0.01), 74.5 ± 16.2 (p < 0.01), 132 ± 17/77 ± 11 (P: NS), 56.5 ± 12.9 (7.3 ± 1.2%) (p < 0.01) and 75.0 ± 23.9, respectively, at the time of the study. The combination of weight loss and fall in HbA1c was identified in 71.5% of the patients. Treatment with SGLT2i was associated with a significant fall in ACR [from 47.1 mg/gCr (25% percentile: 18.1, 75% percentile: 153.4, n = 786) at initiation of SGLT2i to 41.1 (14.9, 127.0) at the time of the study (p < 0.01)] and creatinine clearance (p < 0.01).
Comparison of clinical findings based on age, eGFR and ACR
Age: Body weight, HbA1c, diastolic BP (DBP) and eGFR were significantly different among the three age groups (< 65, ⩾65 but < 75 and ⩾75 years) (Table 1). Furthermore, Δbody weight, ΔHbA1c, ΔSBP and ΔDBP, Δlog ACR and ΔeGFR following treatment were comparable (Table 1).
Effects of treatment based on age, eGFR and ACR.
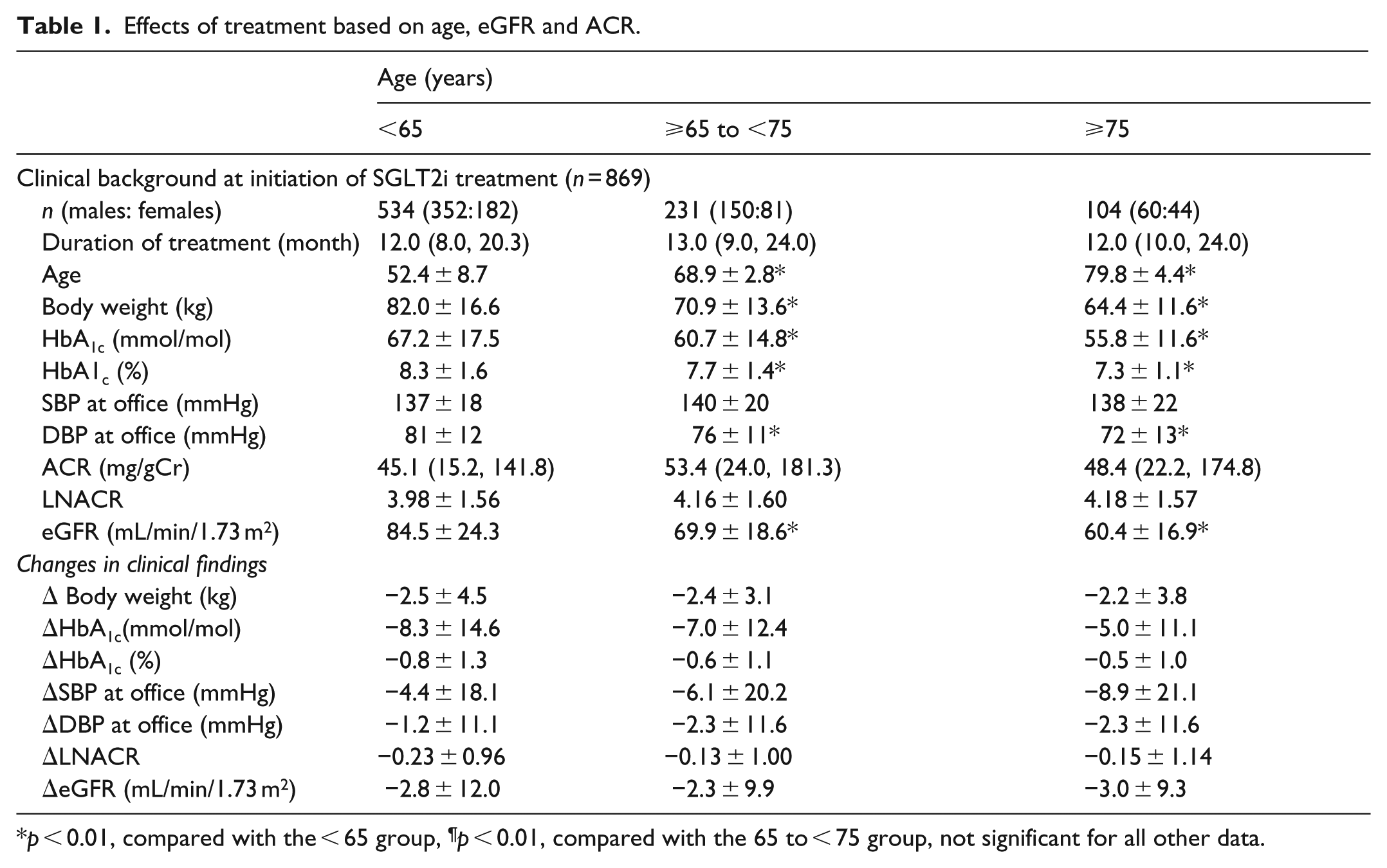
p < 0.01, compared with the < 65 group, ¶p < 0.01, compared with the 65 to < 75 group, not significant for all other data.
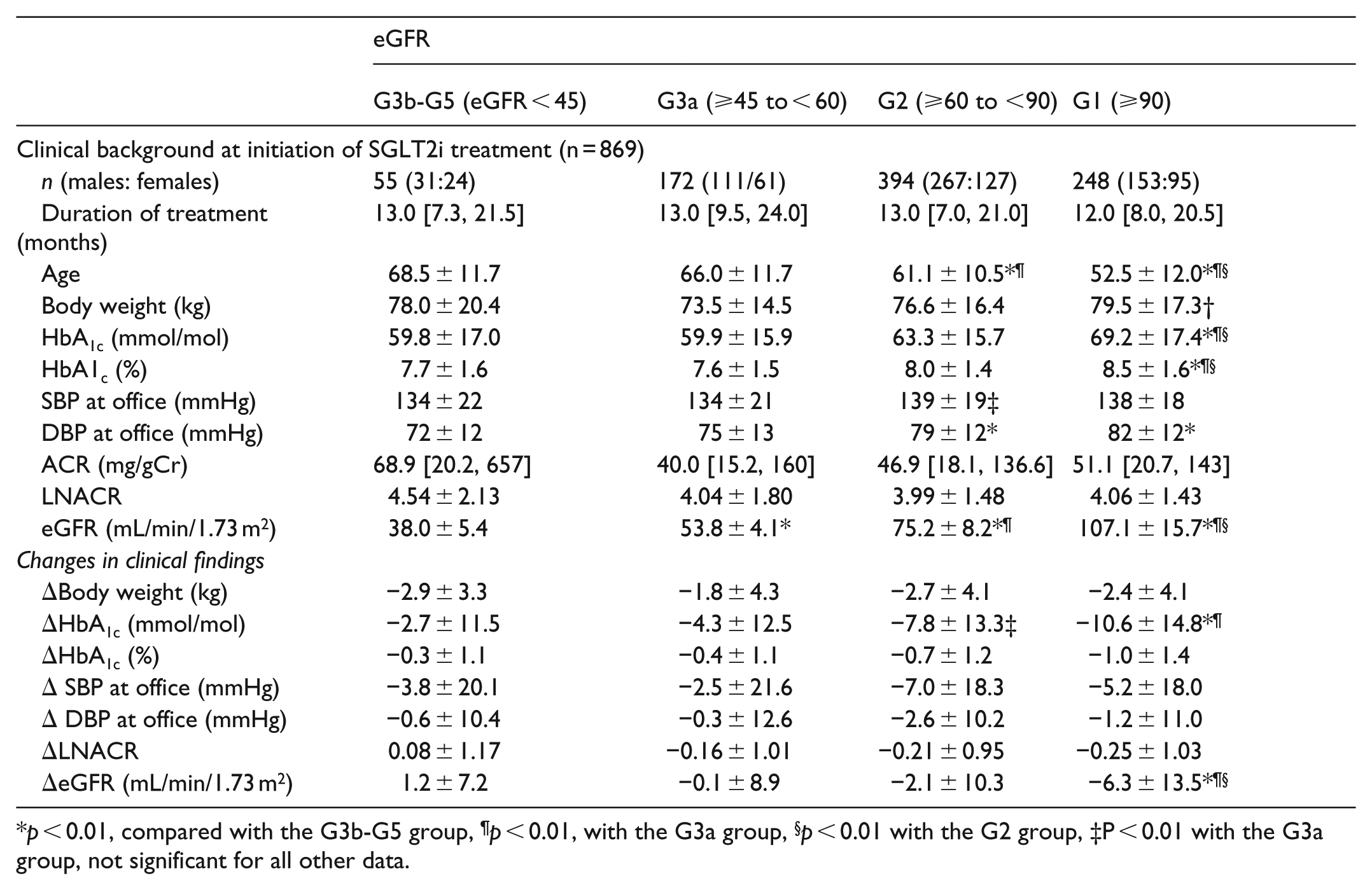
p < 0.01, compared with the G3b-G5 group, ¶p < 0.01, with the G3a group, §p < 0.01 with the G2 group, ‡P < 0.01 with the G3a group, not significant for all other data.
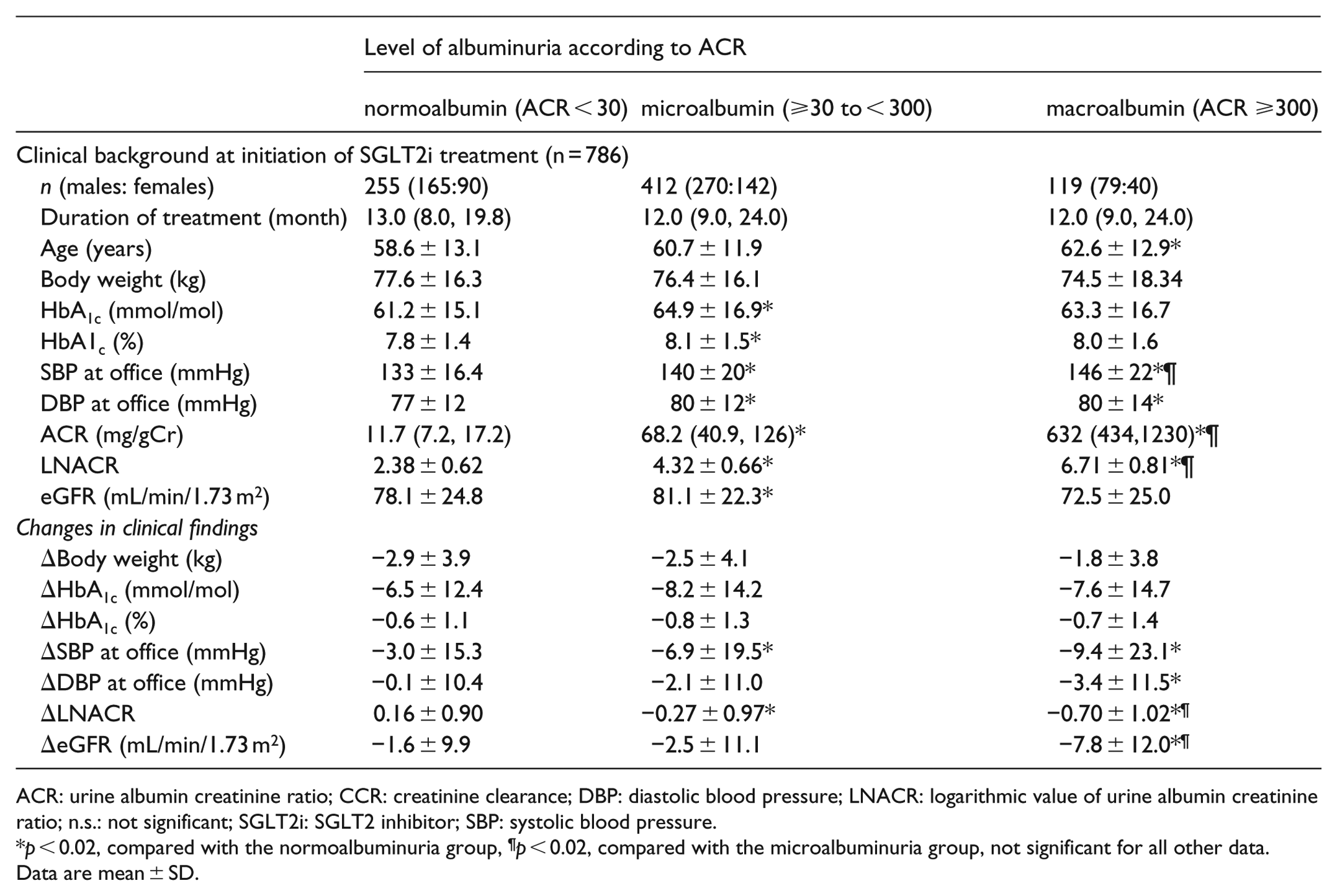
ACR: urine albumin creatinine ratio; CCR: creatinine clearance; DBP: diastolic blood pressure; LNACR: logarithmic value of urine albumin creatinine ratio; n.s.: not significant; SGLT2i: SGLT2 inhibitor; SBP: systolic blood pressure.
p < 0.02, compared with the normoalbuminuria group, ¶p < 0.02, compared with the microalbuminuria group, not significant for all other data.
Data are mean ± SD.
eGFR: eGFR tended to decrease with age. HbA1c was significantly higher in the G1 group (⩾90 mL/min/1.73 m2) compared with the other three groups. Both SBP and DBP tended to be higher in patients with high eGFR, with borderline significance. Progression of renal dysfunction was associated with little change in ACR. The ΔlogACR showed no significant changes after treatment in the four groups. However, the decrease in eGFR was significant in the G1 group compared with the other groups.
ACR: ACR correlated significantly with age and BP (Table 1). In all three ACR groups, SGLT2i treatment was neither associated with changes in weight nor HbA1c, although the ΔlogACR was significantly different in each group. There was a significant negative relationship between ΔACR and ACR at initiation of SGLT2i treatment. The decrease in both SBP and DBP was larger with higher level of albuminuria, although the change was sometimes statistically of borderline significance.
Independent predictors of ΔACR by multiple linear regression analysis
Multiple linear regression analysis identified serum Cr value at initiation of SGLT2i treatment, use of sulphonylureas, and ΔSBP were independent and significant determinants of ΔACR [regression coefficients: 0.38 (95% confidence interval (CI): 0.10; 0.67), 0.15 (95% CI: 0.01;0.30), and 0.01 (95% CI: 0.00; 0.01), respectively]. On the other hand, the use of β-blockers, ACR value at initiation of SGLT2i treatment, and use of empagliflozin correlated negatively with ΔACR [regression coefficients: −0.33 (95% CI: −0.62; −0.04), −0.22 (95% CI: −0.26; −0.17), and −0.20 (95% CI: −0.37; −0.02), respectively]. The use of renin-angiotensin system (RAS) inhibitors, DPP4 inhibitors and GLP-1 receptor agonists had no independent effect on ΔACR.
Discussion
The EMPA-REG renal OUTCOME trial 7 and CANVAS/CANVAS-R programme 2 concluded that treatment with empagliflozin and canagliflozin results in significant improvement in albuminuria and renal composite endpoints and that these SGLT2i have reno-protective effects. Our results showed that SGLT2i treatment was associated with a significant decrease in ACR. Whereas our results showed no relation between ΔACR and age or stage of eGFR, patients with progressive deterioration of renal function should be monitored carefully during the treatment since log ACR was worst in the G3b-G5 group (eGFR < 45), though this was of borderline statistical significance.
Our analysis showed that ACR decreased in patients with microalbuminuria and macroalbuminuria while it significantly increased in patients with normoalbuminuria. Other studies showed different results on improvement in ACR. For example, Heerspink et al. 8 investigated the renoprotective effects of canagliflozin and reported that ACR did not change significantly after a 2-year treatment, though it significantly decreased in patients with ACR ⩾30. Another study also reported that 4-week treatment with dapagliflozin at 10 mg/day reduced the median ACR from 75 to 47.5 mg/gCr in patients with ACR ⩾30. 9 On the other hand, Cherney et al. 10 reported recently that empagliflozin significantly reduced ACR in patients with diabetic nephropathy as well those with normoalbuminuria. Another study reported recently that the albumin-lowering effect of dapagliflozin varied among individual patients with ACR > 100 mg/gCr. 11 In the present study, although the increase in ACR of patients with normal albuminuria was significant, it was rather small, and its clinical value is not clear at this stage. Further examination is needed in patients with low ACR, especially those with normoalbuminuria.
The method used for measurement of ACR might provide explanation for the differences in the results with previous studies. Many GPs measure ACR using occasional spot urine at the hospital or clinic. In the INNOVATION study, which evaluated renal outcome following treatment with telmisartan, ACR was measured using first urine in the morning. 12 Others used 24-h urine 11 or first morning void urine 8 for measurement of ACR. For more accurate evaluation, it may be appropriate to use the first urine in the morning or 24-h urine.
This study is a retrospective observational research that included a heterogeneous group of patients who visited 30 clinical research facilities. The results need to be confirmed in a prospective study of carefully selected patients.
Conclusion
Although the use of concomitant drugs varies between Japan and other countries, the results of our retrospective study were similar to those of previous large-scale clinical trials. Based on the improvement in ACR, we recommend the use of SGLT2i in clinical practice for patients with diabetic nephropathy. The effect of SGLT2i on ACR was independent of age, and the use of SGLT2i seems an effective strategy for treatment of diabetes in advanced age, but attention should be given to patients with normoalbuminuria or progressively low renal function.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
