Abstract
Purpose:
This single centre study investigates the influence of diabetes mellitus on outcomes following carotid artery endarterectomy or stenting.
Methods:
In total, 752 carotid revascularizations (58.2% carotid artery stenting and 41.8% carotid endarterectomy) were performed in 221 (29.4%) patients with diabetes and 532 (70.6%) patients without diabetes. The study outcomes were death, disabling and non-disabling stroke, transient ischaemic attack and restenosis within 36 months after the procedure.
Results:
Patients with diabetes had higher periprocedural risk of any stroke or death (3.6% diabetes vs 0.6% no diabetes; p < 0.05), transient ischaemic attack (1.8% diabetes vs 0.2% no diabetes; p > 0.05) and restenosis (2.7% diabetes vs 0.6% no diabetes; p < 0.05). During long-term follow-up, there were no significant differences in Kaplan–Meier estimates of freedom from death, any stroke and transient ischaemic attack, between people with and without diabetes for each carotid artery stenting and carotid endarterectomy subgroup. Patients with diabetes showed higher rates of restenosis during follow-up than patients without diabetes (36-months estimate risk of restenosis: 21.2% diabetes vs 12.5% no diabetes; p < 0.05).
Conclusion:
The presence of diabetes was associated with increased periprocedural risk, but no further additional risk emerged during longer term follow-up. Restenosis rates were higher among patients with diabetes.
Introduction
Diabetes mellitus (DM) is as a major risk factor for both stroke and cardiovascular disease, and its prevalence is high among patients undergoing carotid revascularization.1,2 In addition, stroke in patients with diabetes is associated with a worse functional outcome and higher mortality compared to their counterparts. 3
There is conflicting evidence on the effect of DM on the outcomes of patients undergoing carotid endarterectomy (CEA). A number of studies reported increased perioperative risk of stroke and death in patients with diabetes undergoing surgery,4,5 and in some series, it was a marker of coronary insufficiency and a predictor of cardiac adverse events. 6 On the other hand, other studies have documented similar perioperative outcomes during CEA in both patients with and without diabetes. 7 Moreover, there are few data in the literature concerning the analysis of risk factors influencing early and late results in patient with diabetes undergoing carotid artery stenting (CAS), despite the growing emergence of CAS into clinical practice as an appealing therapeutic alternative.8–11
Given the increasing prevalence of DM disease in Western countries, 12 the aim of this study was to investigate whether the 30-day and 3-year outcomes of patients undergoing CAS and CEA differed depending on the presence of DM in a single centre experience. In addition, the presence of DM was evaluated as a predictor factor of stenosis recurrence after carotid revascularization.
Methods
Study design
A database of 752 consecutive patients with symptomatic or asymptomatic carotid artery stenosis undergoing CEA or CAS for carotid revascularization in the Vascular Surgery Department of a single institution from 2010 to 2016 was analysed. Carotid stenosis was ⩾80% for asymptomatic or ⩾50% for symptomatic patients, as detected by duplex ultrasound scan (DUS) imaging and confirmed by Computer Tomography Angiography (CTA) or Magnetic Resonance Angiography (MRA) using North American Symptomatic Carotid Endarterectomy Trial (NASCET) criteria. 13
The choice of revascularization technique (CAS/CEA) was based on general guideline recommendations, for example, ESVS (European Society for Vascular Surgery), ACC/AHA (American College of Cardiology/American Heart Association) and the team centre experience according to morphologic and clinical data indicating best suitability with the aim of performing CAS and CEA with low procedural risks. Usually, patients with unfavourable aortic arch anatomy, severe peripheral vascular disease precluding femoral access or extremely tortuous carotid anatomy were excluded from CAS. Similarly, old age; unstable plaque; known allergies to aspirin, clopidogrel or contrast media; and renal insufficiency (creatinine ⩾ 1.5 mg/dL) were considered as exclusion criteria for CAS. 14
CAS protocol
For CAS, the patient was given dual antiplatelet therapy beginning 1 day before the procedure. All patients received a 300 mg loading dose of clopidogrel 1 h before the procedure.
Clinical investigation included a baseline assessment of physical examination, carotid DUS of the supra-aortic vessels, procedural angiography and neurological assessment measured using the National Institutes of Health Stroke Scale (NIHSS). 15 All aortic arch types were included.
All procedures were performed following a standardized protocol in the operating room, equipped with a portable imaging fluoroscopic C-arm (OEC 9900 elite; GE Medical Siemens, Waukesha WI, USA), by a single vascular team. Two skilled operators with high-volume experience (>50 CEA/CAS procedures per year as first operator) performed all procedures.8,16,17 All patients received an intravenous heparin bolus (100 units/kg heparin) to achieve intraoperative anticoagulation [activated clotting time (ACT) ⩾250 s throughout the procedure]. In our centre, with increasing experience, the number of CAS increased over time allowing CEA to be used for fewer and more complex cases in recent years. The carotid stenting procedure has been described previously. 8 Variable models of carotid stents (open-cell, closed-cell, hybrid and micromesh stents) were employed, as a function of lesion characteristics and vessel anatomy. Temporary distal (Emboshield NAV6; Abbott Vascular, Santa Clara, CA, USA) or proximal (Mo.Ma.; Medtronic, Minneapolis, MI, USA) cerebral protection devices were used as per internal protocol. Post-dilation was performed with a 4.5- or 5.5-mm-diameter Rx Viatrac 14 Plus balloons (Abbott Vascular, Santa Clara, CA, USA). CAS postoperative medical therapy included clopidogrel (75 mg once daily) for 1 month and aspirin (100 mg once daily) for a lifelong period.
CEA protocol
Patients were usually maintained on aspirin therapy. CEA was performed under local anaesthesia under electroencephalography (EEG) monitoring or transcutaneous oxygen saturation monitoring. Both Dacron or bovine pericardium graft angioplasty, or eversion endarterectomy were performed.
Procedural follow-up
Patients after both CAS and CEA were followed at the perioperative (30 days) and at the postoperative periods; at 3 months, 6 months and yearly thereafter within 3 years; and symptom status and exact information about clinical adverse events were obtained.
Endpoints and definitions
Primary endpoints were the rate of death, disabling and non-disabling stroke, and transient ischaemic attack (TIA) within 30 days.
Secondary endpoints were the rate of death, disabling and non-disabling stroke, and TIA, and restenosis during the follow-up period, within 36 months after the procedure.
A disabling stroke was considered a stroke that was associated with disability 6 months after the procedure with a modified ranking score of at least 3 (i.e. at least moderate disability from the index score, with the need for some help in daily activities) as assessed by the neurologist. Otherwise, stroke was defined as non-disabling, if after 6 months it was associated with a modified ranking score of less than 3 (i.e. at most only slight disability from the index stroke, without functional impairment in daily affairs). 9 The exposure variable for this study was the presence of DM at the time of carotid procedure.
Patients with diabetes were defined as those with an established diagnosis and/or receiving oral hypoglycaemic or insulin therapy at the time of carotid procedures according to International Classification of Diseases, 9th revision, Clinical Modification (ICD-9-CM 250).
The diagnosis and quantification of restenosis was performed using carotid duplex ultrasound scan. Carotid restenosis was set at >40%.4,18
Statistical analysis
Continuous data are presented as percentages or mean ± standard deviation (SD). Rates for comorbidities, complications and 30 days outcomes were compared between patients with and without diabetes and between those undergoing CAS and CEA by chi-square test. Survival, stroke, TIA and restenosis rates were calculated using Kaplan–Meier analysis to compensate for patient dropouts and are reported using current Society for Vascular Surgery (SVS) criteria. 19 Standard errors (SEs) are reported in Kaplan–Meier analyses. The log-rank test was used to determine differences among patients submitted to CEA and CAS with and without DM. The Cox proportional hazard models were used to examine the associations between DM and covariates with long-term outcome measures, that is, death and stroke (both disabling and non-disabling), TIA and restenosis, by including time-independent interaction of each covariate with survival time.
A value of p < 0.05 was considered statistically significant for all measurements. All analyses were performed using STATA™ (STATA Corp., version 14.0, College Station, TX, USA).
Ethics
The study was approved by the local ethic committee of this institution (statement CE 30.05.2006). Written consent was obtained from all patients before both CAS and CEA revascularization.
Results
Patient population
From 2010 through 2016, a total of 752 patients underwent interventions for carotid stenosis. There were 488 (64.9%) males and 264 (35.1%) females. Mean age was 70.1 (range: 55–87); 215 subjects (28.6%) were symptomatic.
About 221 (29.4%) procedures were performed in patients with diabetes and 531 (70.6%) in patients without diabetes. Of the 752 carotid revascularization, 438 (58.2%) were by CAS and 314 (41.8%) were by CEA.
Demographic and baseline characteristics of study participants with and without diabetes are displayed by procedure in Table 1.
Baseline characteristics of study participants (n = 752).
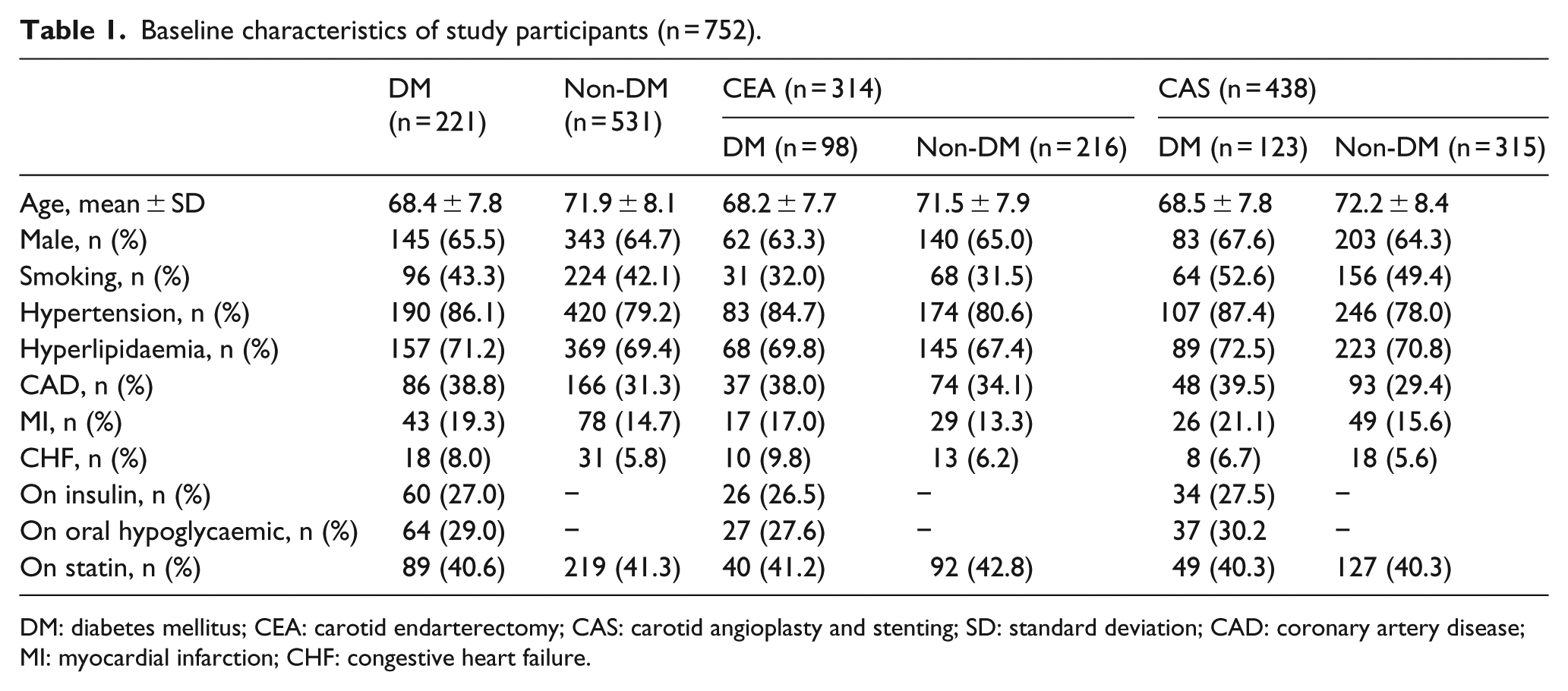
DM: diabetes mellitus; CEA: carotid endarterectomy; CAS: carotid angioplasty and stenting; SD: standard deviation; CAD: coronary artery disease; MI: myocardial infarction; CHF: congestive heart failure.
DM was more common in CEA than in CAS patients (31.2% vs 28.0%). Patients with diabetes were younger (68.4 ± 7.8 vs 71.9 ± 8.1) and more likely to have a history of coronary disease (38.8% vs 31.3%), hyperlipidaemia (71.2% vs 69.4%) and hypertension (86.1% vs 79.2%), with respect to population without diabetes. There were no substantial imbalances in the distribution of comorbidities between the two procedural groups, except with respect to smokers (31.6% in the endarterectomy group vs 50.2% in the stenting group, p < 0.05).
Procedural follow-up
Patients were required to undergo DUS and neurological examination at 1 month and subsequently at 3, 6, 12, 24 and 36 months after the procedure.
Periprocedural outcome measures in patients with diabetes compared with those without diabetes are reported in Table 2. Patients with diabetes had higher periprocedural risk of any stroke or death (3.6% DM vs 0.6% NON-DM; p < 0.05). Perioperative restenosis occurred in 2.7% of patients with diabetes and 0.6% of patients without diabetes (p < 0.05; Table 2). Periprocedural outcomes were similar for CEA and CAS for 30-day risk of disabling or non-disabling stroke and death. The periprocedural risk of any stroke or death in overall population was 1.5% (11/752), with no significant differences in rates between the two procedures: 1.0% (3/314) in CEA and 1.8% (8/438) in CAS. Patients undergoing CEA has a slightly lower 30-day TIA (1.0% vs 1.4%) and restenosis (0.3% vs 0.9%) rate with respect to those undergoing CAS (Table 2).
Periprocedural (30 days) outcomes after carotid revascularization in patients with and without diabetes receiving CEA or CAS.
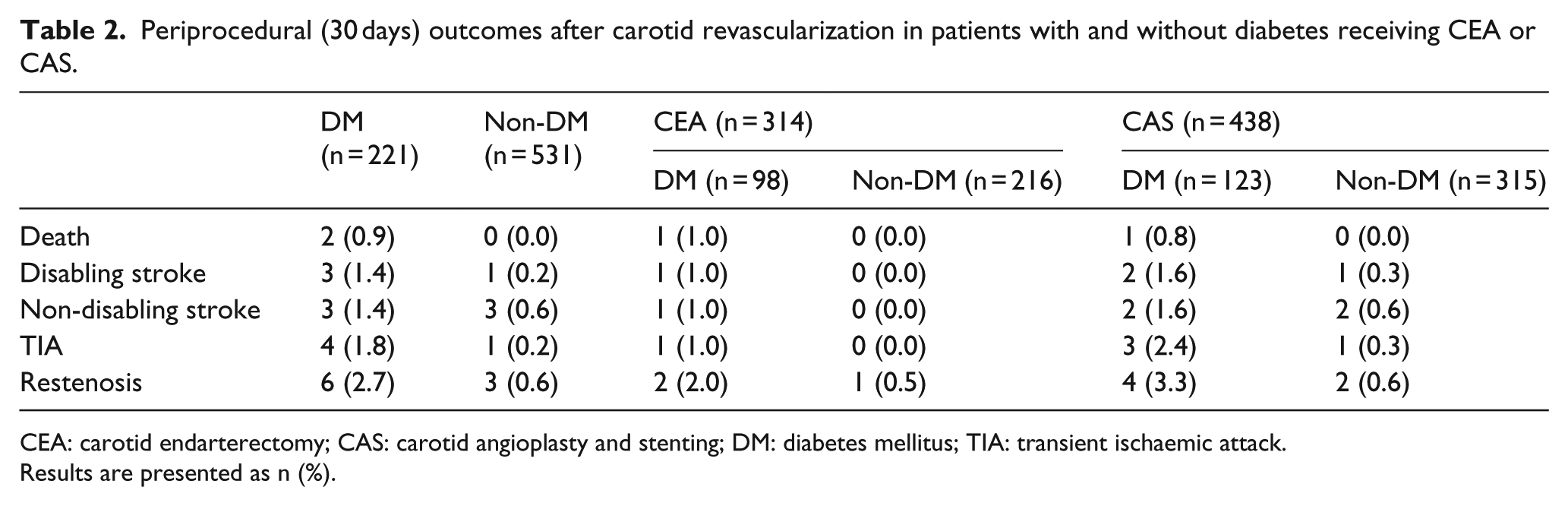
CEA: carotid endarterectomy; CAS: carotid angioplasty and stenting; DM: diabetes mellitus; TIA: transient ischaemic attack.
Results are presented as n (%).
Long-term follow-up
For long-term follow-up, 98.5%, 95%, 79%, 76% and 73% patients attended their 3-, 6-, 12-, 24- and 36-month follow-up appointments. Patients were not able to attend follow-up mainly because they moved to remote locations or because they declined further visits for personal reasons. Most recent patients could not attend their 24- and 36-month appointment yet. Patients with diabetes undergoing CAS (DM CAS) tended to have a lower survival rate. The 36-month risk of any cause mortality was 19.6 (4.1) % for patients with diabetes (DM), and 16.6 (2.4) % for patients without diabetes (non-DM), without significant difference in Kaplan–Meier estimates at 36-months between the two groups according to log-rank test (Figure 1).
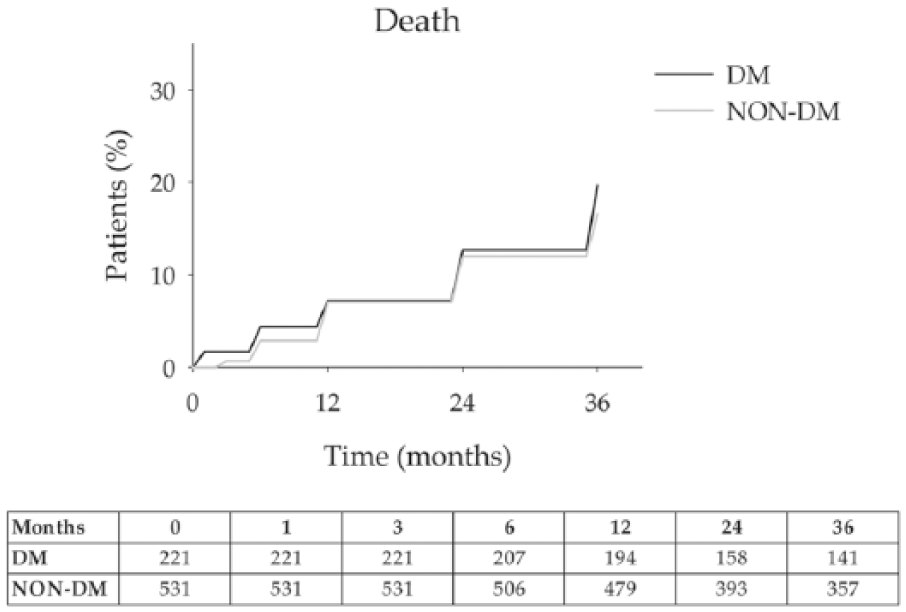
Death: 202 (91.4%) and 491 (92.5%) patients with and without diabetes survived during follow-up (p > 0.05). The 36-month survival rates were 80.3% for DM and 83.4% for non-DM. Error bars are omitted for clarity. Standard errors did not exceed 10% at all-time intervals that were analysed. The number of patients at risk at each time interval is shown below the figure.
The risk of disabling stroke at 36-months was 19.0 (4.0) % (mean (SD)) for DM and 14.0 (2.3)% for non-DM (Figure 2).
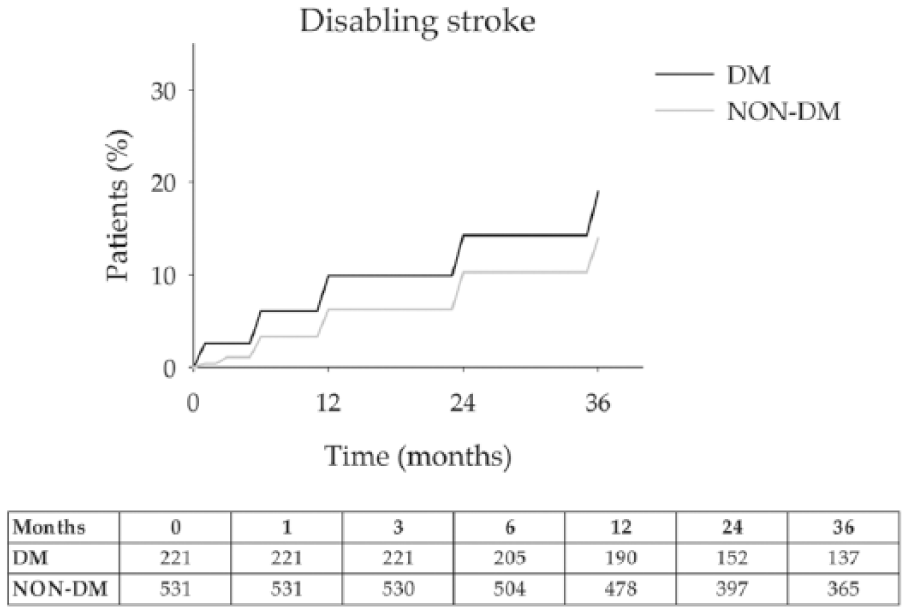
Disabling stroke: 19 (8.8%) and 35 (6.6%) patients with and without diabetes experienced a disabling stroke during follow-up (p > 0.05). The 36-month estimate risk of disabling stroke was 19.0% for DM and 14.0% for non-DM. Error bars are omitted for clarity. Standard errors did not exceed 10% at all-time intervals that were analysed. The number of patients at risk at each time interval is shown below the figure.
Figure 3 showed estimates of non-disabling stroke rates at 36-months of 19.7 (4.1) % for DM and 12.3 (2.2) % for non-DM.
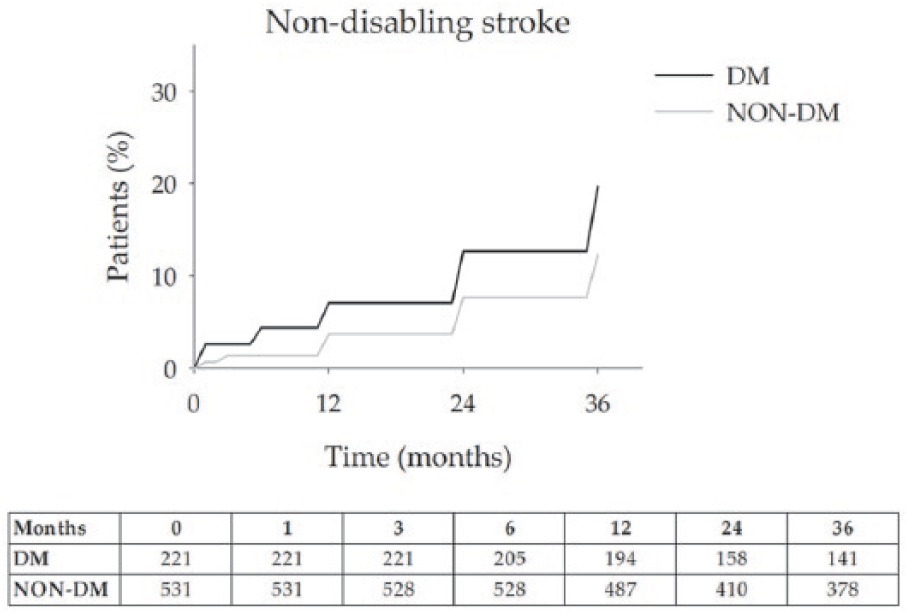
Non-disabling stroke: 18 (8.0%) and 28 (5.2%) patients with and without diabetes experienced a non-disabling stroke during follow-up (p > 0.05). The 36-month estimate risk of non-disabling stroke rates was 19.7% for DM and 12.3% for non-DM. Error bars are omitted for clarity. Standard errors did not exceed 10% at all-time intervals that were analysed. The number of patients at risk at each time interval is shown below the figure.
Patients with diabetes had a greater risk of TIA than those without diabetes. The 12-, 24- and 36-month estimates of TIA rates were 8.9 (2.7) %, 13.3 (3.4) % and 18.8 (3.9) % for DM and 4.5 (1.3) %, 7.6 (1.7) % and 11.4 (2.1) % for non-DM (p > 0.05; Figure 4).
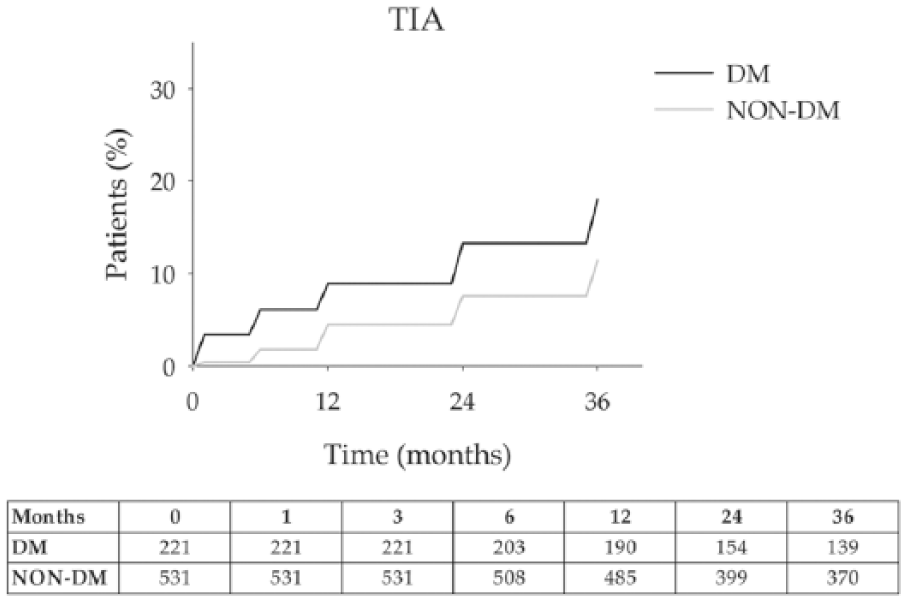
TIA: 17 (7.7%) and 27 (5.0%) patients with and without diabetes experienced a TIA during follow-up (p > 0.05). The 36-month estimate risk of TIA rates was 18.0% for DM and 11.4% for non-DM. Error bars are omitted for clarity. Standard errors did not exceed 10% at all-time intervals that were analysed. The number of patients at risk at each time interval is shown below the figure.
There were no significant differences between populations with and without diabetes for each CAS or CEA subgroup in Kaplan–Meier estimates of event rates for death, disabling stroke, non-disabling stroke and TIA (p > 0.05; Figures S1–S4, Supporting Information).
During follow-up, recurrent stenosis of 40% or more was detected in 44 (5.9) % patients [17 (7.7) % with diabetes and 27 (5.0) % without diabetes] (p > 0.05). Patients with diabetes exhibited higher restenosis risk with respect to those without diabetes [9.7 (2.2) %, 15.3 (3.6) % and 21.2 (4.2) % vs 3.3 (1.1) %, 7.4 (1.7) % and 12.5 (2.2) % for 12-, 24- and 36-month rates, for patients with and without diabetes, respectively (p < 0.05)] (Figure 5). Restenosis occurred in 19 (6.1%) patients treated with endarterectomy and 34 (7.7%) patients treated with stenting. No significant differences between the two treatment groups were observed in the percentage of patients who had restenosis (Figure S5, Supporting Information).
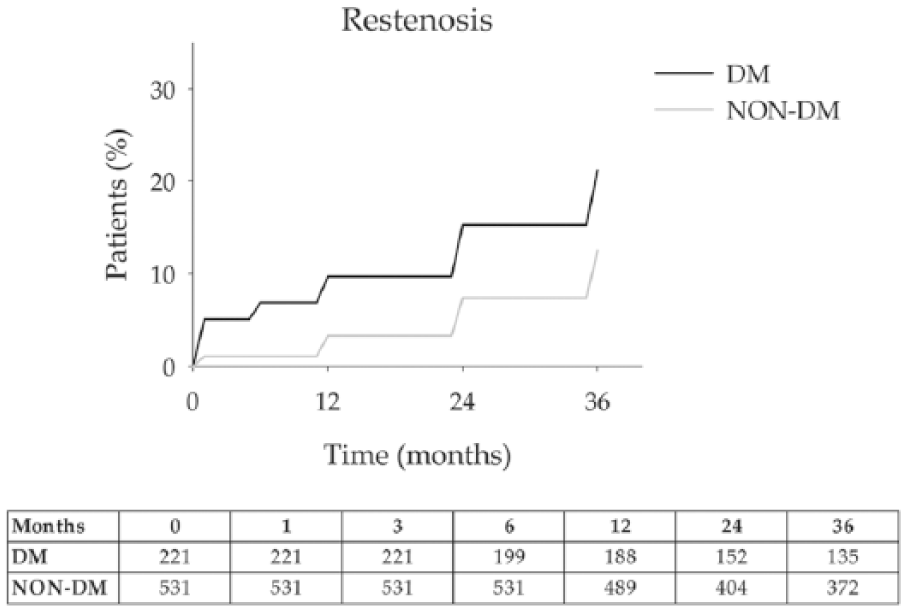
Restenosis: 22 (10.2%) and 30 (5.7%) patients with and without diabetes experienced a TIA during follow-up (p < 0.05). The 36-month freedom from restenosis rates was 21.2% for DM and 12.5% for non-DM. Error bars are omitted for clarity. Standard errors did not exceed 10% at all-time intervals that were analysed. The number of patients at risk at each time interval is shown below the figure.
Results of Cox proportional hazards models are listed in Table 3. Among patients with diabetes, age, hyperlipidaemia, history of coronary artery disease (CAD) and complex plaque were significantly associated with risk of developing death and stroke (both disabling and non-disabling), TIA and restenosis. For patients without diabetes, no factors were found to be significantly associated with risk of death and stroke (both disabling and non-disabling), TIA and restenosis.
HRs of death and stroke (both disabling and non-disabling), TIA and restenosis.
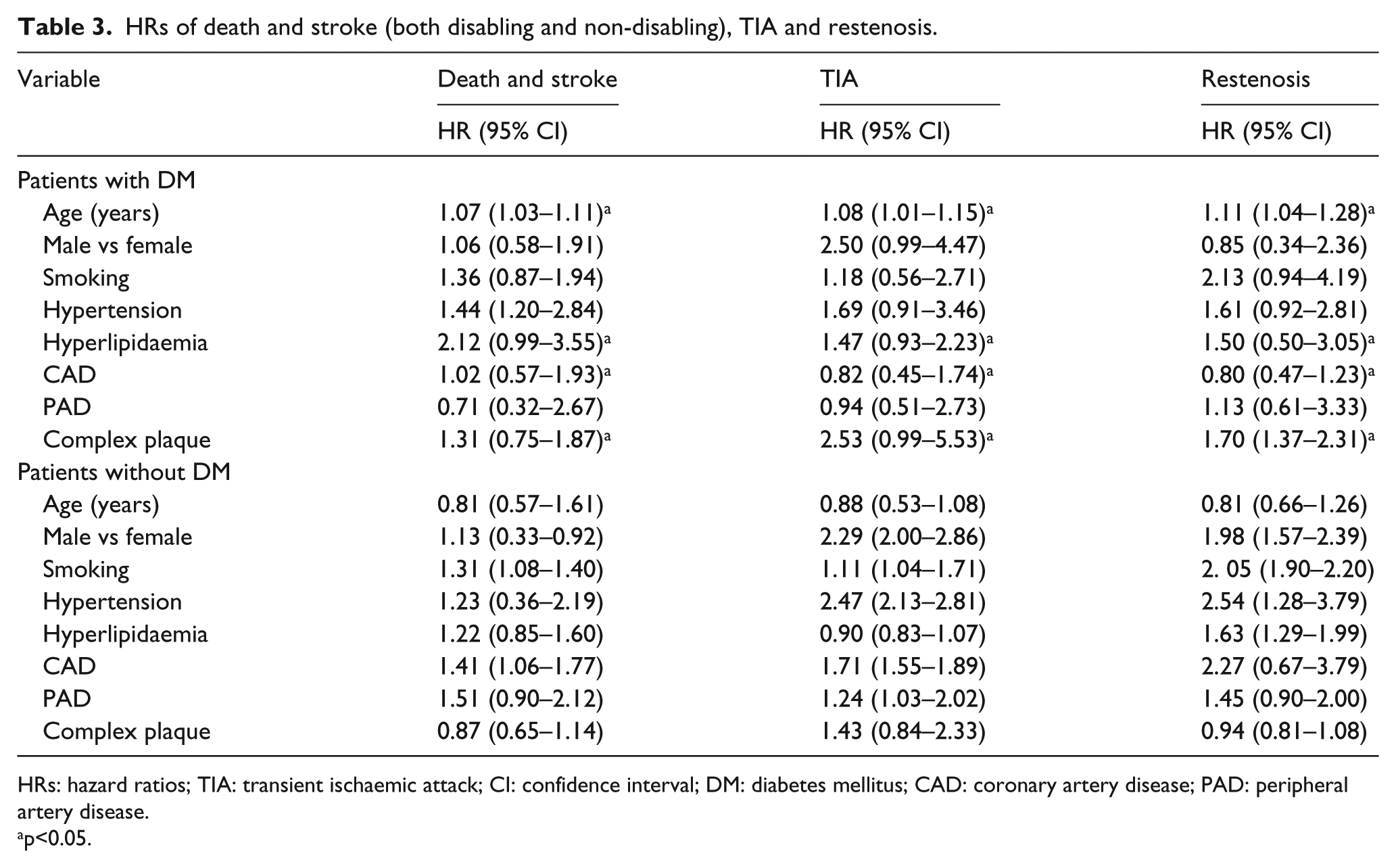
HRs: hazard ratios; TIA: transient ischaemic attack; CI: confidence interval; DM: diabetes mellitus; CAD: coronary artery disease; PAD: peripheral artery disease.
p<0.05.
Discussion
In the present single centre study, patients with diabetes and severe carotid stenosis had higher 30-day death and stroke (disabling and non-disabling) rates than patients without diabetes, both when surgical approach is applied as treatment and with carotid stenting. The perioperative restenosis was twofold higher in patients with diabetes. During long-term follow-up, the rates of death and disabling and non-disabling stroke at 36-months were similar between patients with and without diabetes with both procedures confirming that the efficacy of carotid revascularization in prevention of major cardiovascular adverse events may persist in the long term also in diabetic settings. Patients with diabetes were more likely to experience TIA than those without diabetes, irrespectively of the procedure. Of note, patients with diabetes showed higher rates of recurrent stenosis during follow-up.
DM is the leading cause of disability in Western countries affecting about one of every five adults aged ⩾60 years with a strong cardiovascular burden (Italian total prevalence rate = 6.2%; in adults aged ⩾75 years = 20.3%; in 2015 in a large, representative multiregional population-based cohort of people, the ARNO Observatory).12,20 Previous epidemiologic studies have shown that diabetes independently increases risks of ischaemic stroke (with relative risk ranging from 1.8-fold to 6-fold) and that stroke functional limitations and stroke-related mortality are higher in patients with diabetes.21,22 Therefore, it is expected that patients with diabetes with severe carotid stenosis may derive considerable benefits from stroke prevention measures, such as carotid revascularization.
In order to minimize perioperative outcomes in patients with diabetes, it is important to identify whether diabetes is a predictor of adverse outcomes and whether such events are affected by revascularization techniques, as this information would be critical in assessing the health benefits of CAS as a valid alternative to open vascular procedures in this subgroup of patients.
There is no uniform consensus in the literature on the potentially increased risks that DM poses on carotid revascularization procedures. Previous studies have shown that patients with diabetes undergoing CEA have a higher incidence of death and neurologic or cardiovascular events.5,23 In a recent study of 1116 CEA procedures, the perioperative risk of stroke and death was threefold higher in patients with diabetes with respect to those without diabetes. 4 On the contrary, in another study, patients with diabetes had higher cardiac-related mortality, but there was no significant difference between the two groups of patients in neurologic postoperative morbidity. 3
Few studies have analysed the influence of DM on outcomes among patients who underwent CAS. A pooled analysis of outcomes from 2196 procedures found no higher perioperative risk of major adverse cardiac events in patients with diabetes who underwent CAS procedure. 4 Siewiorek et al. 24 found that diabetes was significantly associated with increased combined rate of 30-day stroke, TIA and death in 203 CAS procedures, while rates of stroke alone and mortality alone were not significantly higher in patients with diabetes. Data from 3433 patients with symptomatic carotid stenosis who were randomly assigned in the Endarterectomy versus Angioplasty in Patients with Symptomatic Severe Carotid Stenosis (EVA-3S) trial, the Stent-Protected Angioplasty versus Carotid Endarterectomy (SPACE) trial and the International Carotid Stenting Study (ICSS) have shown that CAS increased perioperative cumulative stroke and death risk in patients with diabetes but not within 120 days. 25
A number of differences in techniques and approaches between procedures could explain the conflicting results arising from these studies. Moreover, significant heterogeneity in operator experience, study population characteristics, measure of diabetes used in the study, the presence of other comorbid conditions and physician and hospital characteristics could also justify the different perioperative exposure risk of patients with diabetes during CAS and CEA. 7 Adegbala et al. demonstrated that DM with or without chronic complications does not confer significantly increased perioperative complications among patients undergoing CAS, despite the increased incidence of comorbid conditions in the diabetes population. However, in the CEA subgroup, they found that diabetes with chronic complications was an important risk factor. 26 Protack et al. 27 specifically investigated metabolic syndrome as a predictor of adverse outcomes after carotid revascularization and found no differences in 30-day ipsilateral stroke and cardiac complications between subjects with and without diabetes, but during long-term follow-up, patients with diabetes had statistically lower survival and higher rates of occurrence of any of stroke, myocardial infarction or death.
In the present study, despite the slightly lower life expectancy due to increased all-cause mortality (36-month survival: 91.4% vs 92.5% for patients with and without diabetes, respectively), in the long term, patients with diabetes did not perform worse in terms of stroke (both disabling and non-disabling) risk at 36 months after carotid revascularization. These findings confirm the efficacy of stroke prevention of carotid revascularization and the durability of the carotid repair (whichever the treatment applied). These long-term data are of relevance especially for the CAS group since few data are available from the current literature. According to this analysis, low disabling (8.2%) and non-disabling stroke rates (9.1%) can be achieved with CAS within 3 years also in a subgroup of patients with higher cardiovascular mortality and morbidity. The presence of diabetes might both increase neointimal hyperplasia and accelerate the growth of new carotid plaques at the site of arterial injury, thereby supporting the increased restenosis risk whichever treatment was applied to carotid stenosis.5,28
Limitations
This study is retrospective and not a randomized trial. A biochemical assessment to evaluate glycaemic or metabolic control in patients with and without diabetes has not been performed, and patients’ adherence to prescribed therapy had not been supervised. Moreover, it is known that the presence of diabetes is usually associated with multiple risk factors for cardiovascular diseases, such as hypertension, hyperlipidaemia, reduced high-density lipoprotein cholesterol and high body mass index, which all together could lead to a clinical entity known as metabolic syndrome. It is a general consensus of opinion that the various risk factors together could lead to synergy and potentially greater risk than the individual risk factor. For this reason, it could be interesting in the future to investigate metabolic syndrome as a risk factor for short- and long-term operative events during carotid revascularization.
In conclusion, although these findings suggest that diabetes overall does not appear to be a predictor of adverse outcomes on patients undergoing CEA and CAS, the presence of diabetes might convey higher risk of recurrent stenosis in the long-term follow-up, and thus it might be an indicator for refining cardiovascular risk estimation and making therapeutic decision.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
