Abstract
The present study was conducted to ascertain how cigarette smoke affects the exocrine–endocrine interactions of the human pancreas with diabetes mellitus secondary to pancreatic diseases (type 3c). Blood has been collected from smoking and non-smoking healthy individuals as well as from patients with diagnosed chronic pancreatitis and diabetes type 3c. The concentrations of interleukin-6, endothelin-1 and insulin in the plasma were determined by enzyme-linked immunosorbent assay (ELISA) tests. The activities of amylase and lipase in the serum, as well as the lipid profile, creatinine, uric acid and urea concentrations, were measured using colorimetric methods. Samples of normal pancreatic tissue and chronic pancreatitis were verified histopathologically and then interleukin-6, endothelin-1, insulin and glucagon were localized by immunohistochemical staining using a monoclonal anti-human antibody. The highest levels of interleukin-6 and endothelin-1 and the lowest levels of insulin and glucagon intensity from the immunostaining were observed in smoking patients with diabetes. In all smoking patients with pancreatitis and diabetes, there was a significant elevation in interleukin-6 and endothelin-1 concentration and amylase and lipase activities, hyperlipidaemia and a lower value of estimated glomerular filtration rate and blood urea nitrogen when compared to non-smokers. Our study confirmed that smoking exerts a pro-inflammatory effect and disturbs the exocrine–endocrine interactions of the pancreas.
Introduction
Diabetes mellitus is defined as a regulatory dysfunction of metabolism mainly characterized by chronic hyperglycaemia. The underlying causes may be impaired insulin secretion or insulin resistance, or both. Diabetes can also develop as a direct consequence of other diseases, including diseases of the exocrine pancreas. Because of this heterogeneity in underlying causes, diabetes mellitus is currently classified into different types, comprising types 1–4.1–3 The awareness of type 1 and type 2 diabetes mellitus among physicians and in the general population is rather broad. Diabetes mellitus secondary to pancreatic diseases (type 3c), however, is a condition rarely considered in everyday practice. Yet, recent data on type 3c diabetes mellitus (T3cDM) show that it might be more common than previously thought. Studies propose that this clinically important condition might be consistently underdiagnosed and misdiagnosed.1,4 T3cDM is a frequent comorbidity of chronic pancreatitis (CP), with prevalence estimates ranging from 25% to 80%.3–7 Pancreatic cancer (8% of all T3cDM) seems to be the second most common cause according to a recent study. 4 The risk of diabetes in patients with CP increases the longer the duration of the disease, with worsening pancreatic damage, especially early-onset pancreatic calcification. It also rises with prior distal pancreatectomy. 5 Multiple clinical studies have shown that using tobacco, particularly via smoking cigarettes, elevates the risk of developing pancreatic diseases such as pancreatitis and cancer.8–10 Furthermore, the risk increases as a function of the amount of tobacco consumed. Smoking tobacco has often been linked as a co-factor with alcohol abuse in predisposition to pancreatic disorders. However, the inclusion of smokers who do not drink alcohol in some of these studies has highlighted the fact that cigarette smoking can also be considered an independent risk factor. 11 Despite significant clinical advancements in this field, scientific data exploring how tobacco toxins affect the pancreas at the cellular level are scarce. 9 Smoking has an influence on changes in hormonal secretion in the pancreas.12–15 Endocrine dysfunction of pancreatic islet beta (β) cells is a common complication of pancreatic inflammation resulting from long-term smoking, and as a consequence, diabetes occurs. Substances present in tobacco smoke undoubtedly trigger free radical processes, interfere with vascular homeostasis and the proper functioning of the vascular endothelium,16,17 and also increase inflammation and oxidative stress. 18 Because higher levels of inflammatory markers [C-reactive protein (CRP) and interleukin-6 (IL-6)] herald the development of diabetes, 19 smoking may also contribute to the development of diabetes by increasing levels of inflammatory markers. 18
The main characteristic of T3cDM is the gradual loss of β-cell function leading to impairment in or lack of insulin secretion.3–7 However, adipose tissue, gastro-intestinal tract, pancreatic β-cell activity and kidney function may also be involved in disease development. 20
In parallel to impairment in endocrine pancreatic function, a reduction in exocrine function has also been observed in all types of diabetes. 21 A decrease in amylase and lipase activity has been mentioned by Yadav et al., 22 although cases with elevated amylase have also been reported. And studies evaluating the pancreatic exocrine function in smoking patients with CP showed a significant impairment in the activity of pancreatic enzymes.10,23–27 Another study found that cigarette smoking may be an important factor in potential changes in the lipid profile in healthy young people. 26
Most recently, a trend of positive correlations between high-density lipoprotein (HDL) cholesterol and amylase in diabetes type 2 patients has been discovered. 22 To our knowledge, no study correlating cholesterol levels and amylase in T3cDM patients has been conducted. Increased hyperlipidaemia combined with oxidative stress (resulting from hyperglycaemia and cigarette smoking) are among the main factors inducing atheromatosis, diabetic angiopathy, thrombotic incidents and cardiovascular problems, all of which are commonly observed diabetic complications. 27
In addition, CP is a disease with poor clinical prognosis. Almost half of the patients with alcoholic pancreatitis die after 20–25 years of disease duration. 28 The development of secondary diabetes in the course of CP is an independent risk factor for death in this group of patients. 29 As it is chronic, it may also affect other organs in the body. So far, little is known about the long-term consequences of ongoing CP and diabetes in smoking patients. In smoking patients, episodes of acute pancreatitis (AP) occur often enough, 18 resulting in organ ischaemia that may cause complications typical of the disease (shock, acute respiratory failure, acute renal failure and others). 30 After stabilization of the acute state in the course of CP, exocrine and endocrine functions of the pancreas are primarily monitored; distant organs are rarely diagnosed.
In our earlier studies, we have shown that tobacco smoke disorders pro-oxidant and antioxidant parameters in pancreatic fluids. 18 However, there are no reports on the localization of IL-6, endothelin-1 (ET-1) or the major pancreatic hormones insulin and glucagon in the pancreatic tissue of non-smoking and smoking patients with CP and diabetes. The concentrations of IL-6 and ET-1, amylase and lipase activity, changes in the lipid profile and changes in the parameters responsible for renal function in the blood of non-smoking and smoking patients with CP and diabetes are also unknown. Therefore, the objective of this study was to summarize clinical and scientific knowledge of the effects tobacco has upon the pancreas, and how tobacco may contribute to CP, the development of diabetes and the complications associated with it.
Materials and methods
Materials
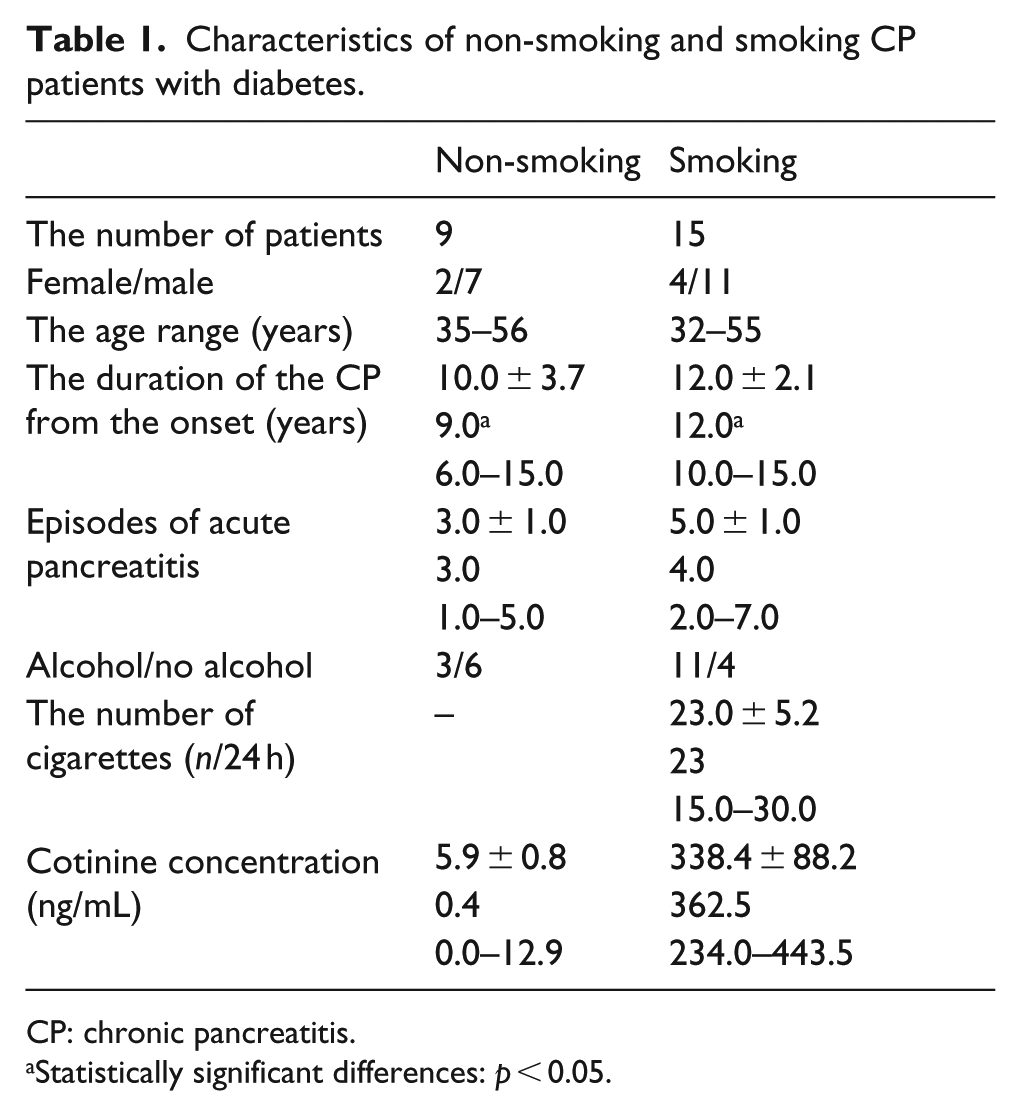
The study was conducted on 24 patients with CP and diabetes (18 men, 6 women; aged 32–56 years; mean age 44 years) admitted to the Department of Gastrointestinal and General Surgery, Wrocław Medical University, Poland, for evaluation of their disease (Table 1). This work was carried out in accordance with the Declaration of Helsinki (2000) of the World Medical Association. The study was approved by the Bioethics Commission at Wrocław Medical University (No. KB: 4353/2011). All healthy individuals and patients provided their informed consent in writing.
Characteristics of non-smoking and smoking CP patients with diabetes.
CP: chronic pancreatitis.
Statistically significant differences:
The patient classification and assessment of pancreatitis were based on the following: routine clinical examination and laboratory tests (medical history: pain appearance background, disease duration; physical examinations); ultrasonography (US) and computed tomography (CT) of the abdomen and, in special cases, endoscopic retrograde cholangiopancreatography (ERCP) and magnetic resonance cholangiopancreatography (MRCP) of the pancreas and bile ducts. The physical examination, supplemented by the aforementioned diagnostic procedures, permitted the identification of the pancreatitis aetiology in most cases.
The diagnosis of CP was combined with a history of alcohol abuse, where the patient consumed more than 40 g of alcohol per day for more than 5 years. The obstructive pancreatitis aetiology was defined on the basis of ERCP and US tests. The family type of pancreatitis was not diagnosed. Cases which could not be classified according to the above criteria were defined as cases of idiopathic pancreatitis.
Data on smoking and alcohol consumption were obtained during direct personal interviews conducted with each patient. Based on the interview about smoking habits and on the elevated levels of cotinine – a nicotine metabolite in the plasma – CP patients were divided into two groups: non-smokers and smokers. Among the study group of patients, 9 CP non-smokers and 15 CP smokers were diagnosed with diabetes. The diagnosis was made based on the diagnostic criteria for T3cDM proposed by Ewald and Bretzel 1 (Table 1).
A control group was selected based on interviews by employees of the Medical University and carried out during regular medical examinations for the Polish social security system. The control group comprised 53 healthy individuals, aged 21–62 years (mean age 41 years). Among the control subjects, no cases of diabetes mellitus nor individuals with a history or symptoms of pancreatitis were observed. All hospitalized patients and the control group of volunteers gave informed consent to participate in the study.
Blood and tissues served as the study material. Venous blood was collected from fasting patients during the first 24 h of hospitalization. Serum was obtained according to the standard procedure by collecting venous blood in disposable trace element-free tubes (No. Cat. 03.1524.001, Sarstedt, Germany) with serum clotting activator, left at 25°C to complete thrombosis and centrifuged (1.200g/20 min). In order to obtain plasma, blood was collected into trace element-free tubes containing EDTA-K 2 (No. Cat. 04.1931.001, Sarstedt, Germany) and lithium heparin (No. Cat. 05.1106.001, Sarstedt, Germany), immediately gently mixed and centrifuged (2.500g/15 min). The obtained serum and plasma were portioned and stored in sealed tubes (No. Cat. 0030102.002, Eppendorf, Germany) at −80°C until used.
Fourteen CP patients with diabetes (non-smoking,
All tissue sections were routinely fixed in a phosphate-buffered 10% formaldehyde solution and embedded in paraffin. Serial 4-µm-thick sections were placed on histological slides (Super Frost Plus, Cat No.: 041300, Menzel-Gloser, Germany). Preparations stained with haematoxylin and eosin were verified histologically in the Department of Pathomorphology, Wrocław Medical University.
Methods
Method for the determination of concentration and activity parameters
The cotinine determination was carried out by a method described earlier. 25 Immunoenzymatic methods – an IL-6 DuoSet ELISA Development System (Cat. No. DY, R&D Systems; 206) and Human ET-1 Immunoassay (Code: BBE5; R&D Systems, Minneapolis, MN, USA) – were used to determine plasma IL-6 and ET-1 levels with specific antibodies against human IL-6 and ET-1. The IL-6 and ET-1 values were expressed in picograms per millilitre.
Fasting glucose levels were measured in plasma using an oxidation method with a reagent for the quantitative determination of glucose (Cat. No. 80009, BIOLABO, France). Glucose is oxidized by glucose oxidase to gluconic acid and hydrogen peroxide. Hydrogen peroxide involving peroxidase reacts with 4-chlorophenol and 4-aminoantipyrine to form a red quinone-imine. The absorbance of the coloured complex proportional to the concentration glucose in the sample is measured at 500 nm.
Another immunoenzymatic method – an Insulin ELISA Kit (Cat. No. 6219, DAKO, UK) – was used to determine plasma insulin levels with specific antibodies against human insulin. The insulin values were expressed in units per millilitre. The enzyme activity was determined using a colorimetric method with substrates – for amylase: ethylidene-G7-PNP (4,6-ethylidene-p-nitrophenyl-alpha,D-malthoheptozyde) and for lipase: 1,2-o-dilauryl-rac-glycero-3-glutaric acid-(6-methylresorufin) ester.
Total cholesterol was measured in the serum using a reagent (Cat. No: 5017.1; BioMaxima, Poland). Cholesterol was determined using an enzymatic method. One of the reaction by-products, H2O2, was measured quantitatively in a peroxidase-catalysed reaction that produces a colour, with intensity proportional to cholesterol concentration. Absorbance was measured at
Triglycerides (TGs) were determined in the serum using a reagent (Cat. No: 5031.1; BioMaxima, Poland). TGs were hydrolysed to produce glycerol, then glycerol was oxidized using glycerol oxidase, and H2O2, one of the reaction products, was determined as described above for cholesterol. Absorbance was measured at
HDL concentration was determined in the serum using a direct method (Cat. No: 10300060, BioMaxima, Poland). In this test, all cholesterol fractions except HDL were blocked. HDL was subjected to an enzymatic reaction that involved the formation of colour compounds. The amount of colour product was proportional to the HDL concentration in the sample. The absorbance was measured at
The value for the Castelli risk index I [total cholesterol (TC)/HDL ratio] was calculated according to Asare et al. 32 The marker of lipid peroxidation, thiobarbituric acid reactive substances (TBARS), was measured in the plasma using thiobarbituric acid, according to the method described earlier. 33
Serum creatinine concentration was determined by the Jaffe colorimetric method (Cat. No.: 1 1711 99 90 10 026, BioMaxima, Poland). Uric acid levels were determined by a colorimetric assay of urokinase (Cat. No.: 1 3001 99 10 021, BioMaxima, Poland). Urea concentration was determined by the Urease–Glutamate dehydrogenase (GLDH) method. For diagnostic purposes and analysis of results, urea concentration was converted to urea nitrogen concentration (blood urea nitrogen, BUN). The estimated glomerular filtration rate (eGFR) was calculated using the CKD-EPI Creatinine Equation (as recommended by the National Kidney Foundation).
Immunohistochemical analysis
The tissue sections were deparaffinized and rehydrated in an alcohol series. All sections were then incubated with 3% hydrogen peroxide to block the intracellular activity of peroxidase. The non‑specific binding was blocked with a 0.05 M Tris–HCl buffer, pH 7.4 with 1% bovine serum albumin (Cat. No.: S0809, Antibody Diluent, DakoCytomation, Denmark). The tissue sections were subsequently incubated for 30 min at room temperature with specific monoclonal mouse antibodies against IL-6 (Cat. No.: MAB206, R&D Systems, USA), ET-1 (Cat. No.: ABIN120978, Abcam, Cambridge, UK), insulin (Cat. No.: N1542; DakoCytomation, Denmark) and glucagon (Cat. No.: N1541; DakoCytomation, Denmark). After washing in 0.05 M Tris–HCL saline (Cat. No.: 170-6435, Bio-Rad Laboratories; Hercules, USA) with 0.1% Tween 20 (Cat. No.: P1379, Sigma Chemical, St. Louis, MO, USA), the IL-6, ET-1, insulin and glucagon complexes were visualized using the LSAB2-HRP test (Cat. No.: K0673, DakoCytomation, Carpinteria, CA, USA). Peroxidase activity was localized against 3,3′-diaminobenzidine in an imidazole–HCl buffer, pH 7.5 (Cat. No.: K0637, DAB, DakoCytomation, Denmark). Consequently, the sections were washed in distilled water, contrasted with haematoxylin (Cat. No.: S2020, Chem Mate™, DakoCytomation, Denmark) and finally closed in glycerine gel and left until dry. A negative control was performed for each tissue section, replacing the primary antibody with the anti-rabbit immunoglobulin control IgG antibody (Cat. No.: X0903, Negative Control DakoCytomation, Carpinteria, CA, USA).
The incubation time of individual stages of the test were matched experimentally in laboratory conditions. The stained tissue sections were viewed under high power using an Olympus BX41 light microscope (Olympus Optical Co. Ltd, Japan) that interfaced with an Olympus DP70 digital camera (Olympus Optical Co. Ltd, Japan) in order to digitize the light microscopic image.
Evaluation of the immunohistochemical staining results
The intensity of the parameters IL-6, ET-1, insulin and glucagon immunoreactions within the cells was graded semi-quantitatively in the following manner: negative (no immunohistochemical reaction: 0), weakly positive (lightly stained but clearly differentiated from negative background: +/–), moderately positive (between weak and strong: +), strongly positive (dark brown with high contrast: + +) and very strongly positive (+ + +).
Digital imaging methodology
In order to determine the parameters’ content in the immunohistochemically stained sections, a digital imaging method for DAB quantification on slides with haematoxylin counterstain was used. The image analysis allows for quantitative numerical comparisons of staining intensity based on the value of signals read from the DAB staining, which is possible due to the existence of a linear relationship between staining intensity and antigen concentration. Stained tissue sections were viewed under a light microscope that interfaced with a camera for scientific applications. Both units were controlled by a personal computer. The Fiji measurement system (operates in the Windows environment) was used to perform the quantitative analysis of the digitized images. The system quantified the average darkness of the image due to the DAB signal from antigen staining. Then, it converted the signal staining into a mean gray value, and the results were quantified in DAB signal intensity units. The reaction intensity was evaluated in arbitrary unit (AU) of optical density (OD). A total of 10 areas were acquired from each tissue section of the control patients as well as the smoking and non-smoking CP patients with diabetes.
Statistical analysis
Participants were categorized into four groups: non-smoking and smoking control groups, and non-smoking and smoking CP patients with diabetes. The normality of the variables was tested by the Shapiro–Wilk test. The differences between analysed groups were tested using the Mann–Whitney U test. Spearman rank correlation analysis was used to evaluate the correlations. In all instances,
Results
Concentration of IL-6, ET-1 and insulin
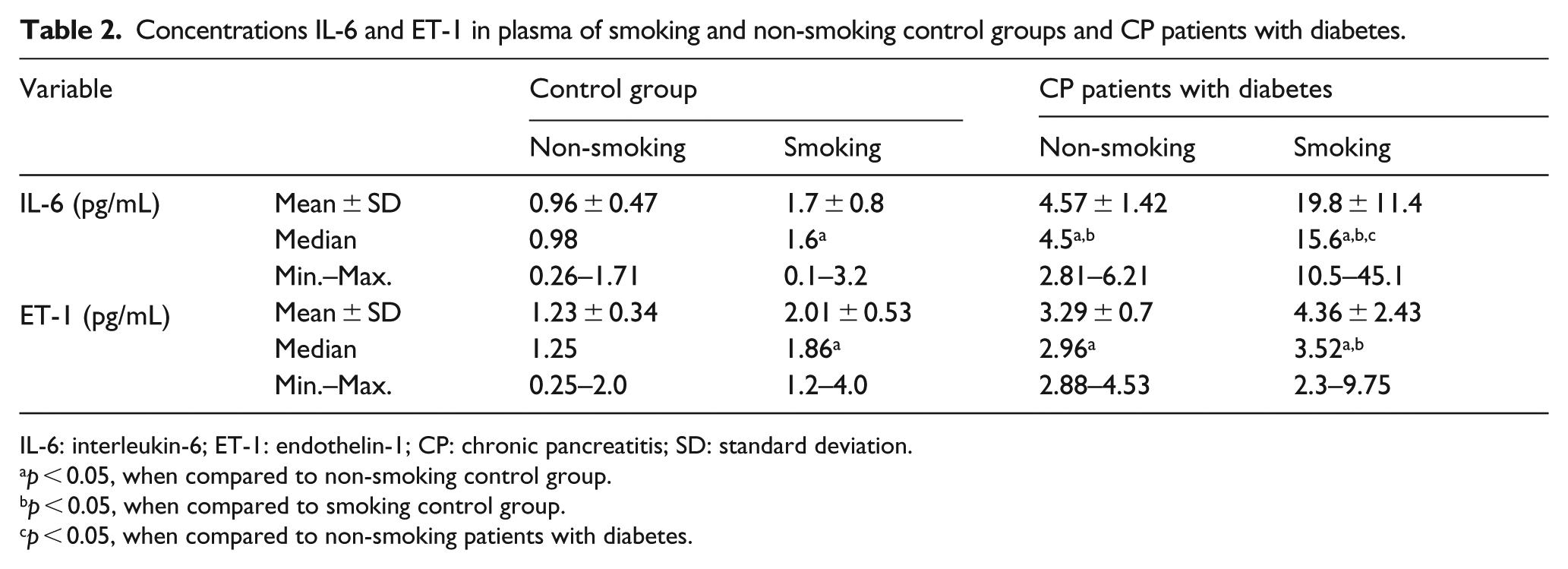
The concentration of IL-6, an established mediator of inflammation, was found to be higher in the plasma of both the control group and CP patients with diabetes who had been exposed to cigarette smoke. The study confirmed significant differences in the concentration of plasma IL-6 between the non-smoking and smoking control groups, and non-smoking and smoking patients with diabetes (Table 2).
Concentrations IL-6 and ET-1 in plasma of smoking and non-smoking control groups and CP patients with diabetes.
IL-6: interleukin-6; ET-1: endothelin-1; CP: chronic pancreatitis; SD: standard deviation.
The concentration of serum IL-6 was found to be four times higher in smoking than in non-smoking CP patients with diabetes. Interestingly, the IL-6 levels were found to be only twice higher in the plasma of the smoking control than in that of non-smoking control. The highest level of IL-6 was found in smokers with diabetes.
The mean values of plasma ET-1 concentration along with standard deviations in the non-smoking and smoking control groups and patients with CP are summarized in Table 2. The highest concentration of ET-1 was observed in the plasma of smoking patients with diabetes followed by non-smoking patients with diabetes. The differences in ET-1 concentration between the non-smoking control group, the smoking control group and diabetic patients were statistically significant (
The mean values of plasma insulin concentration along with standard deviations in non-smoking and smoking CP patients with diabetes and in the control groups are summarized in Table 2. The highest level of insulin was found in the non-smoking control, the lowest in smoking CP patients with diabetes. The differences in insulin concentration between non-smoking and smoking controls and non-smoking and smoking patients with diabetes were statistically significant (Table 3). A lower, similar value of insulin concentration was observed in non-smoking and smoking CP patients with diabetes compared with the non-smoking control.
Concentrations and activities of analysed parameters in the blood of smoking and non-smoking control groups and CP patients with diabetes.
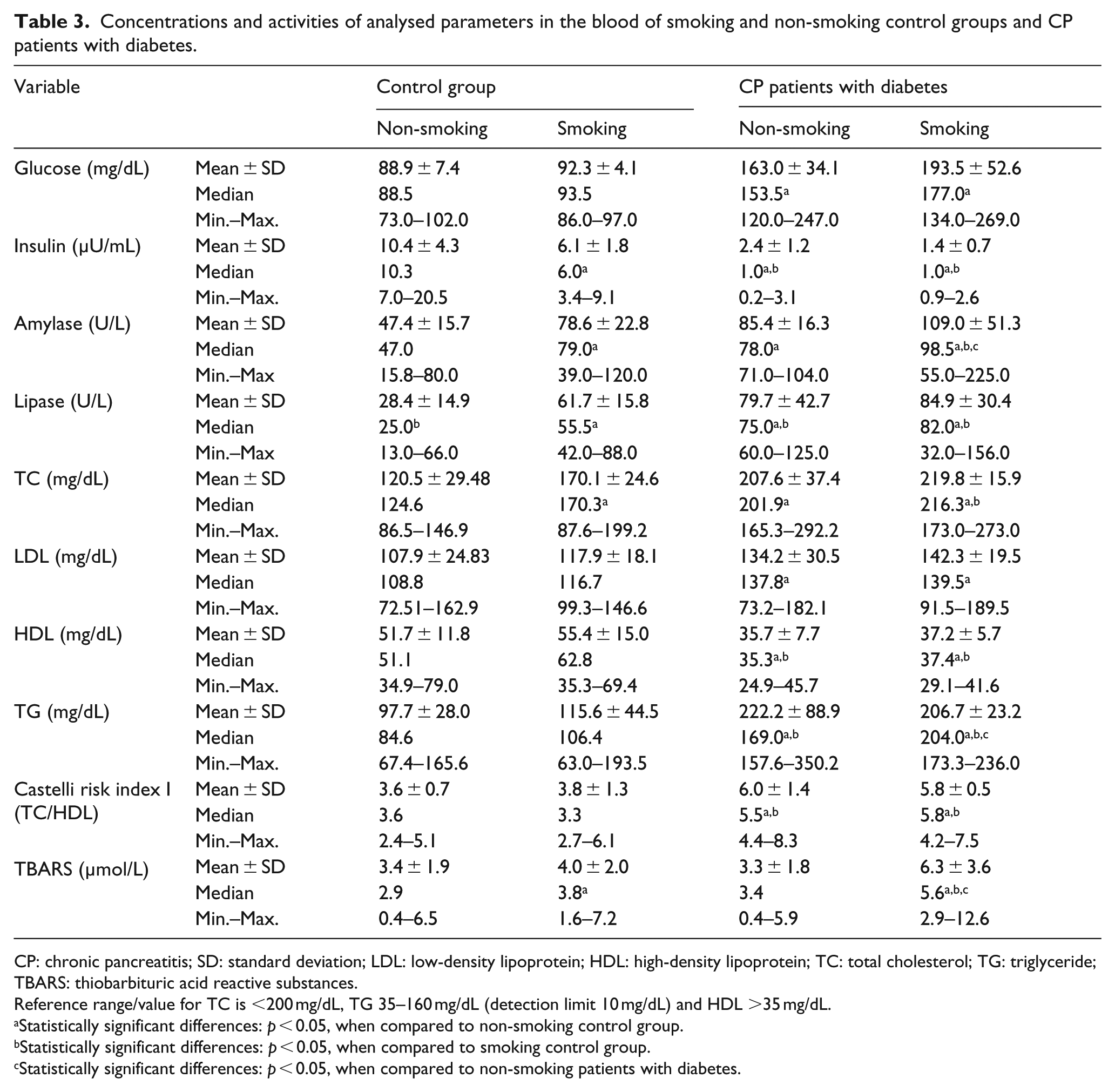
CP: chronic pancreatitis; SD: standard deviation; LDL: low-density lipoprotein; HDL: high-density lipoprotein; TC: total cholesterol; TG: triglyceride; TBARS: thiobarbituric acid reactive substances.
Reference range/value for TC is <200 mg/dL, TG 35–160 mg/dL (detection limit 10 mg/dL) and HDL >35 mg/dL.
Statistically significant differences:
Statistically significant differences:
Statistically significant differences:
Amylase and lipase activity
The highest amylase and lipase activity was found in smoking CP patients with diabetes. It was noted that the serum enzyme activity was significantly higher in the smoking control than in the non-smoking control. A statistically significant increase in amylase and lipase activity in non-smoking and smoking CP patients with diabetes was observed compared to the non-smoking control. Higher enzyme activity was found in the serum of smoking patients than in non-smoking patients (Table 3).
The lipid profile, TBARS, creatinine and eGFR
Changes in lipid peroxidation and the lipid profile were observed in the serum of non-smoking and smoking CP patients with diabetes. The concentration of HDL was statistically lower in diabetic CP patients, non-smoking or smoking, compared to the control group, while the concentrations of TC, TG and LDL were statistically highest in non-smoking and smoking diabetics, compared to control groups (Table 3). When we analysed the value of the Castelli risk index I (TC/HDL), we found a higher value in CP patients with diabetes when compared to the control group (non-smoking and smoking) (Table 3).
It was also observed that the concentration of TBARS was statistically significantly higher in the smoking control and smoking CP patients with diabetes (4.0 ± 2.0 and 6.3 ± 3.6 μmol/L), compared to the non-smoking control group (3.4 ± 1.9 μmol/L). The highest TBARS concentration was found in smoking CP patients with diabetes (Table 3).
The highest serum creatinine level was found in the smoking group of diabetic patients: 1.3 ± 0.2 mg/dL. A statistically significant increase in creatinine and uric acid concentrations in smoking CP patients with diabetes was present when compared to the non-smoking control. A significant decrease in the values of BUN and the BUN/creatinine ratio was observed in non-smoking and smoking patients with diabetes as compared to non-smoking and smoking controls. The lowest value of eGFR was observed in smoking patients with diabetes. A statistical decrease in the value of eGFR in smoking diabetics compared to the non-smoking control was also observed (Table 4).
Creatinine and uric acid concentrations, and values of eGFR in serum non-smoking and smoking healthy persons and patients with chronic pancreatitis.
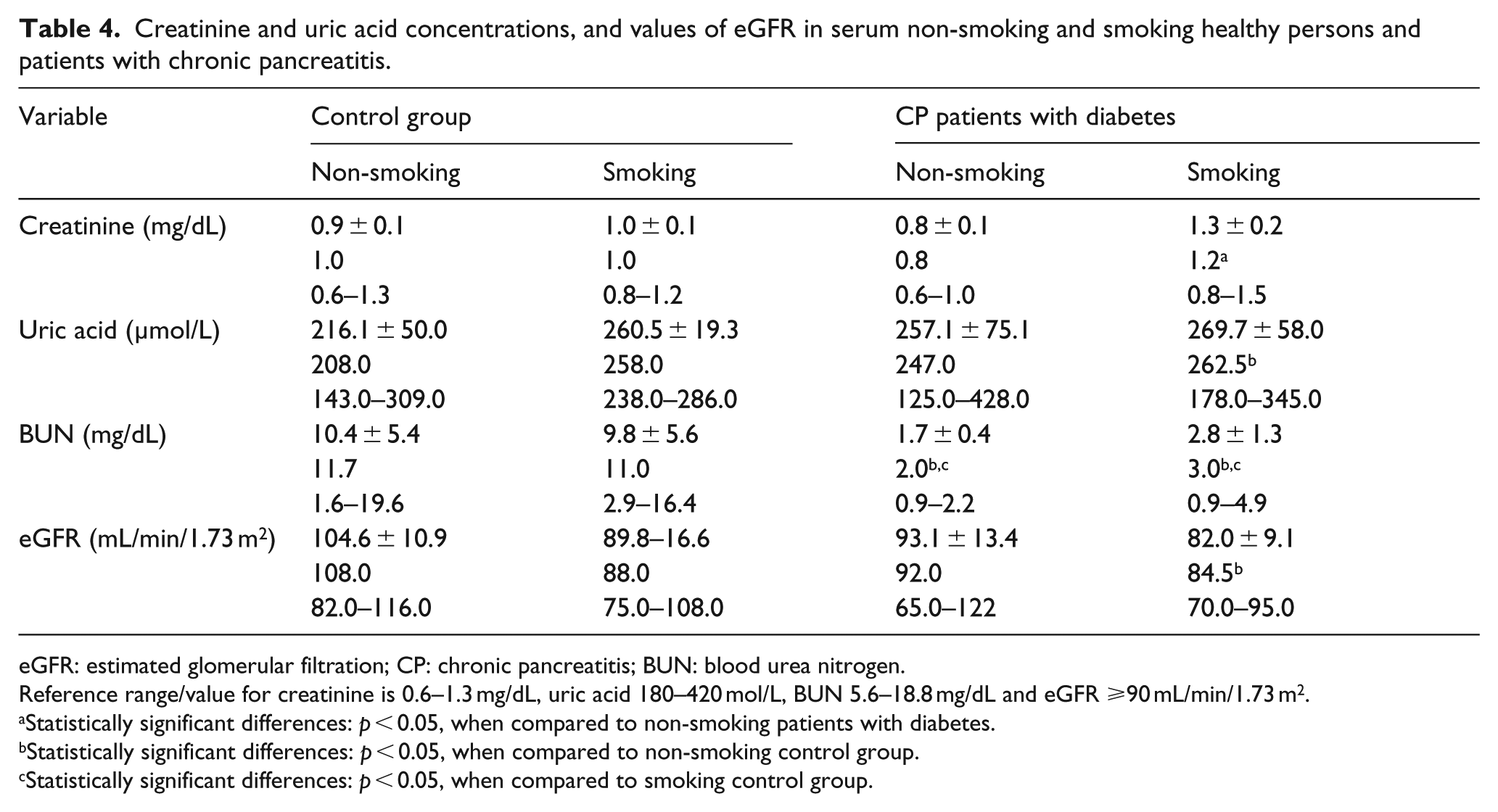
eGFR: estimated glomerular filtration; CP: chronic pancreatitis; BUN: blood urea nitrogen.
Reference range/value for creatinine is 0.6–1.3 mg/dL, uric acid 180–420 mol/L, BUN 5.6–18.8 mg/dL and eGFR ⩾90 mL/min/1.73 m2.
Statistically significant differences:
Statistically significant differences:
Statistically significant differences:
Correlations between parameters
Significant correlations in the non-smoking group of patients with diabetes
The correlation coefficients of specific biochemical parameters as determined in non-smoking CP patients with diabetes are summarized in Tables 5 and 6.
Correlations of the biochemical parameters in the non-smoking group of patients with diabetes.
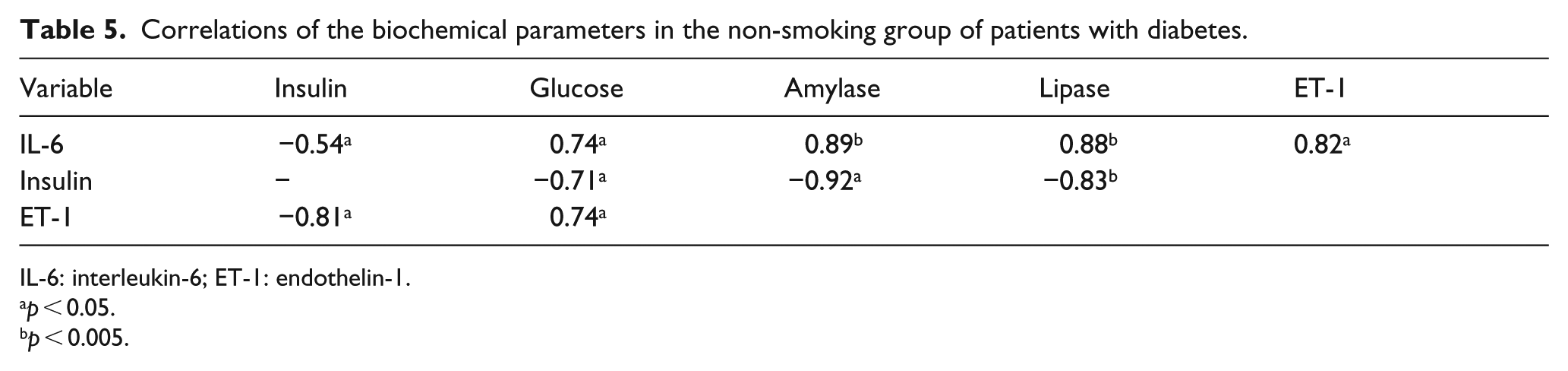
IL-6: interleukin-6; ET-1: endothelin-1.
Correlations of the lipid profile concentrations with the blood glucose and insulin concentrations and the enzymes activities of the non-smoking patients with diabetes.
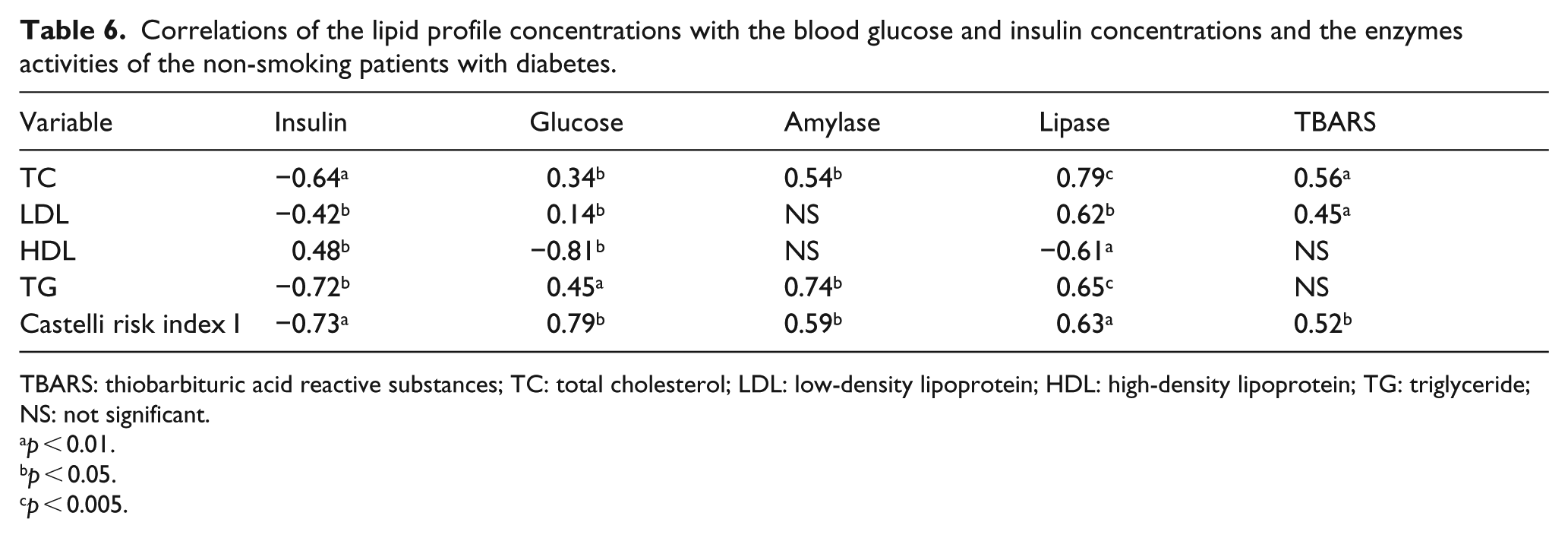
TBARS: thiobarbituric acid reactive substances; TC: total cholesterol; LDL: low-density lipoprotein; HDL: high-density lipoprotein; TG: triglyceride; NS: not significant.
There were strong positive correlations between the enzyme activity of lipase and amylase, between these enzymes and IL-6 and between ET-1 and IL-6 and glucose. The results of the study confirmed the existence of a correlation between decreased insulin levels and increased pancreatic enzyme activity and between elevated ET-1 and glucose levels in patients with diabetes (negative correlation). In addition, there was a significant positive correlation between lipase activity and amylase activity, which confirms the relationship between the parameters (
A negative correlation between insulin and the TC, LDL and TG levels as well as the value of the Castelli risk index I was found. A lower insulin level correlated with a lower HDL level. A negative correlation between HDL level and glucose concentration and lipase activity was detected. A higher glucose concentration was accompanied by higher TC, LDL and TG levels and higher values of the Castelli risk index I. Higher amylase activity correlated with higher TC and TG levels and higher values of the Castelli risk index I.
Positive correlation coefficients between lipase activity and the analysed parameters were noted in Table 6. Positive correlation coefficients were observed between TBARS concentration and TC and LDL levels and the value of the Castelli risk index I.
Significant correlations in the smoking group of patients with diabetes
The correlation coefficients of specific biochemical parameters determined in smoking CP patients with diabetes are summarized in Tables 7 and 8.
Correlations of the biochemical parameters in the smoking group of patients with diabetes.
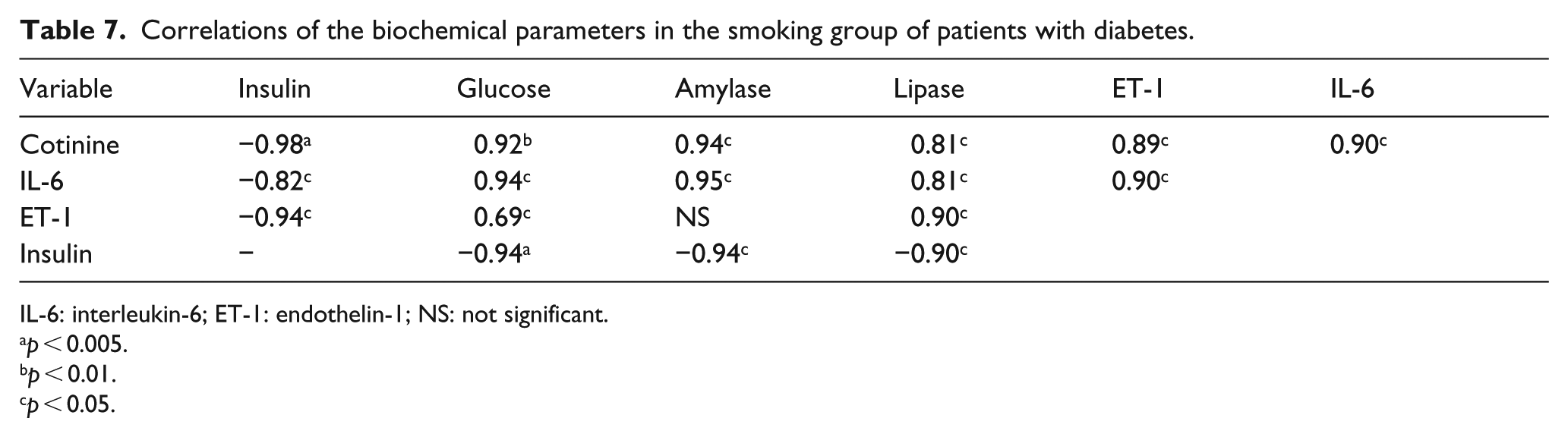
IL-6: interleukin-6; ET-1: endothelin-1; NS: not significant.
Correlations of the lipid profile concentrations with the blood glucose and insulin concentrations and the enzymes activities of the smoking patients with diabetes.
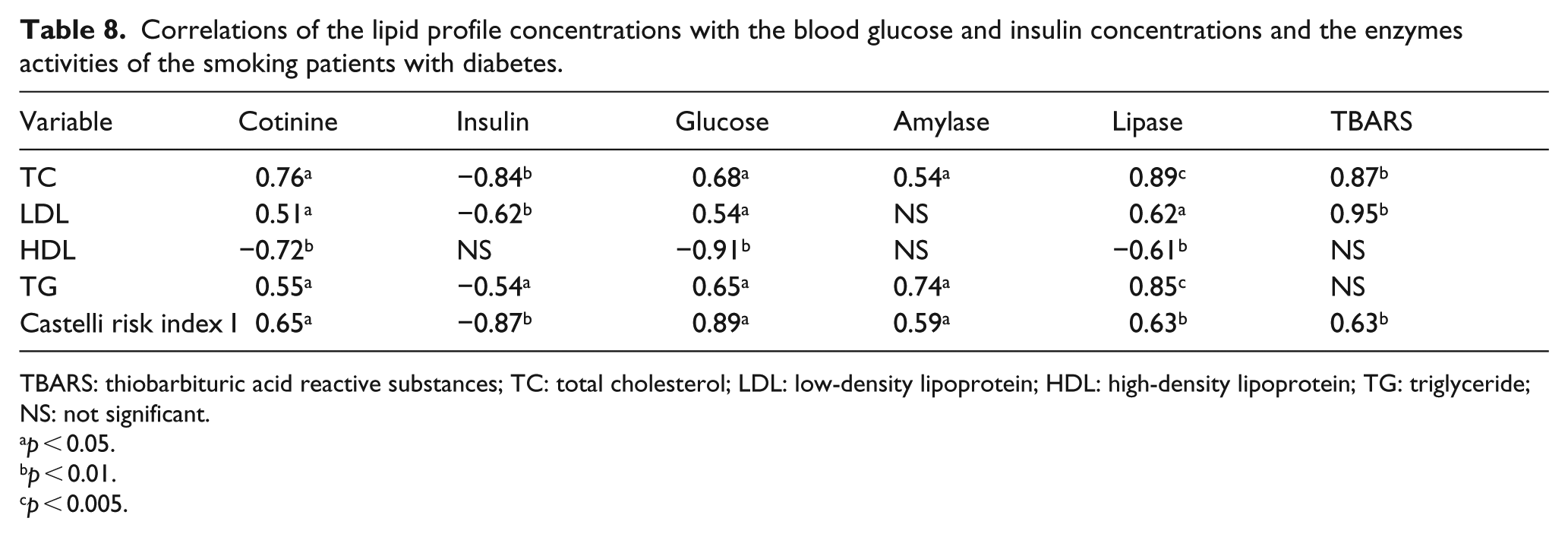
TBARS: thiobarbituric acid reactive substances; TC: total cholesterol; LDL: low-density lipoprotein; HDL: high-density lipoprotein; TG: triglyceride; NS: not significant.
Cigarette smoking in diabetic patients resulted in increased secretion of the cytokine of inflammatory IL-6, with simultaneous pancreatic blood flow disorder, increased lipase and amylase enzyme activity and decreased insulin secretion. Evidence of this is found in the positive correlations between cotinine and IL-6, pancreatic enzymes and ET-1, and the negative correlation between cotinine and insulin. A negative correlation was also found between the cytokine level and insulin (the increase in IL-6 correlated with a decrease in insulin concentration). In addition, the reduction in insulin levels correlated with the intensity of lipase and amylase activity. In contrast, pancreatic circulation disorders in smoking diabetic patients were confirmed by an increase in endothelin concentration, which positively correlated with an increase in lipase activity and glucose concentration, and negatively correlated with insulin secretion from pancreatic B cells. There was a positive significant correlation between lipase activity and amylase activity, which confirms the relationship between the parameters (
A positive correlation between cotinine and the TC, LDL and TG levels as well as the value of the Castelli risk index I was found. A higher cotinine level correlated with a lower HDL level. A negative correlation between insulin concentration and TC, LDL and TG levels and the value of the Castelli risk index I was found. Higher TC and TG levels were correlated with higher glucose levels, higher TBARS levels and higher amylase and lipase activity. A negative correlation between HDL level and glucose level and lipase activity was detected. A higher value of the Castelli risk index I was accompanied by higher levels of the analysed parameters (except insulin). Positive correlation coefficients between LDL levels and glucose levels, TBARS levels and lipase activity were noted (Table 8). A strongly positive correlation between cotinine and creatinine and BUN concentrations (
Histological diagnosis of pancreas
Normal tissue
The histopathologic examination confirmed the regularity of the pancreatic exocrine and endocrine cells. Pancreatic islets were clearly separated. There were no changes indicating the existence of chronic inflammation, that is, no calcification or fibrosis. In the four samples collected from healthy subjects, a lack of or a weak immunohistochemical reaction to IL-6 and ET-1 was observed in the pancreatic islet and exocrine cells (Table 9). The analysis of IL-6- and ET-1-immunostained sections of the normal, semi-quantitative digital image was positive for the pancreatic islet and acinar cells (81.5 ± 13.5 and 121.5 ± 23.5 AU, respectively) (Figure 1(a) and (d)). Isolated islet cells clearly showed a strong and very strong expression of insulin in the tissue of the pancreas on their whole surface (Figure 2(a)). The analysis of the insulin-immunostained sections of the control tissues’ semi-quantitative digital image resulted in a very intense positive identification of beta cells (282.5 ± 53.3 AU). In the control pancreas, a specific response to glucagon was observed in cells localized mainly at the periphery of the pancreatic islets with single cells in the central part of the islet (Figure 2(d)). In the control group, the intensity of the immunohistochemical reaction ranged from 87.4 to 205.8 AU. The glucagon expression was about two times lower than that of insulin (Table 9).
Parameters immunostaining of the pancreatic tissues control groups and non-smoking and smoking CP patients with diabetes.
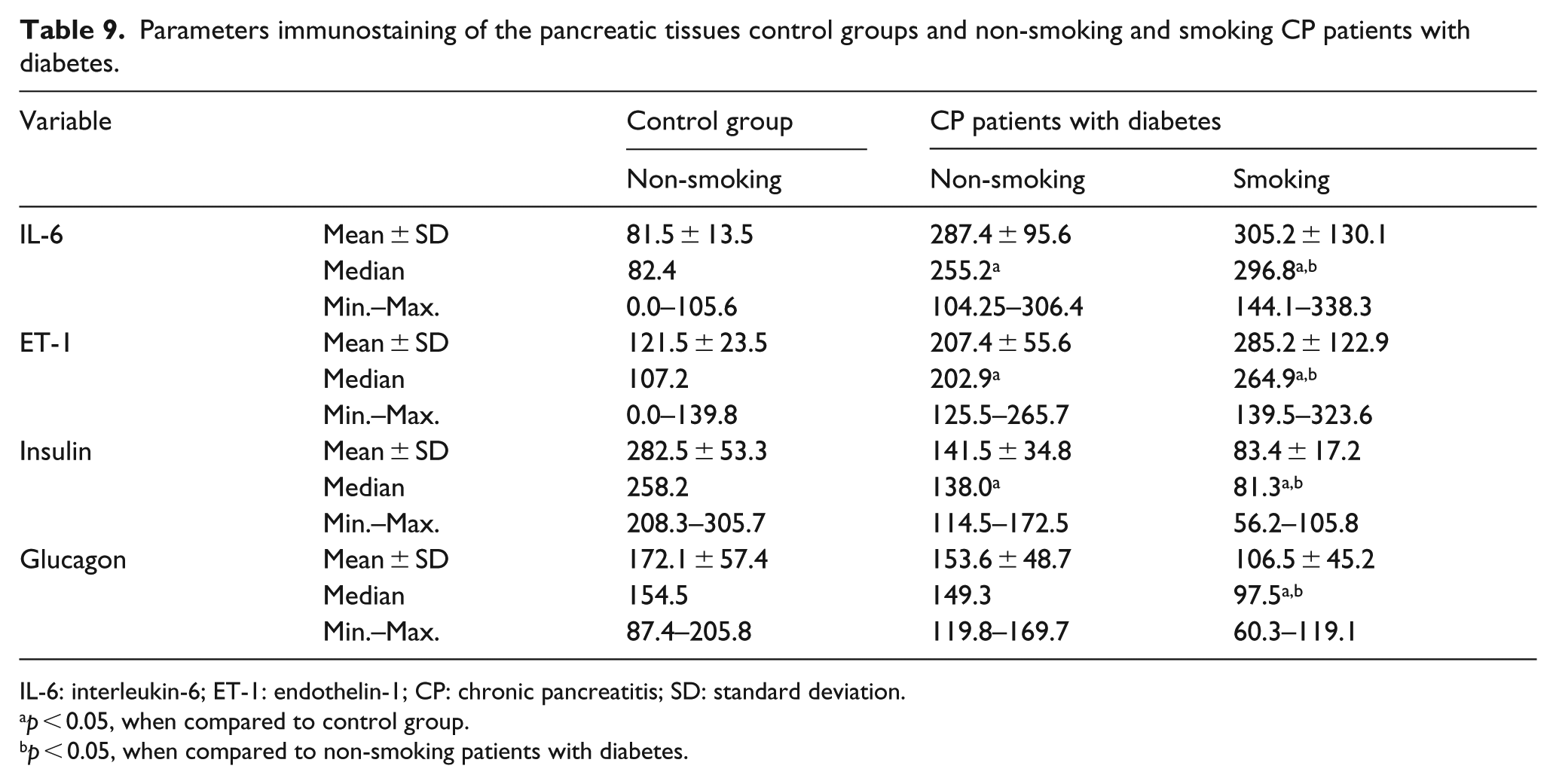
IL-6: interleukin-6; ET-1: endothelin-1; CP: chronic pancreatitis; SD: standard deviation.
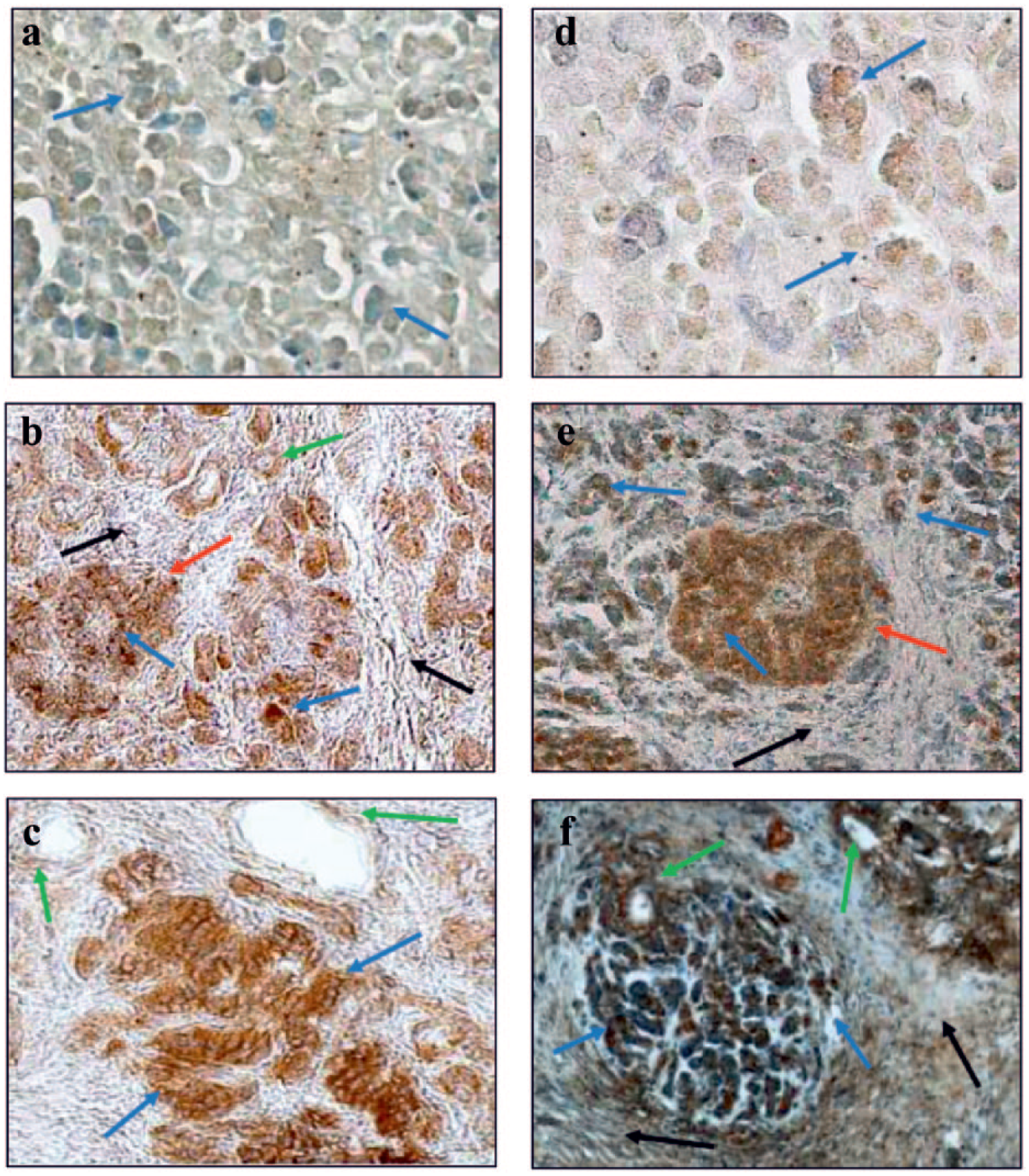
Immunohistochemical localization of IL-6 (a, b, c) and ET-1 (d, e, f) in normal pancreas and pancreas from non-smoking and smoking CP patients with diabetes. IL-6: (a) (210×): No immunohistochemical reaction in the acinar cells. (b) (210×): A strong expression in acinar, pancreatic duct and islet cells. (c) (210×): A very strong reaction in acinar and a moderate reaction in pancreatic duct cells. ET-1: (d) (210×): No and weak immunohistochemical reaction in the acinar cells. (e) (175×): A strong immunohistochemical reaction in islet and acinar cells. (f) (137×): A strong and very strong reaction in acinar and pancreatic duct cells (arrows – red: pancreatic islets; blue: acinar cells; green: pancreatic duct cells; black: fibrous lesions in the sublayer).
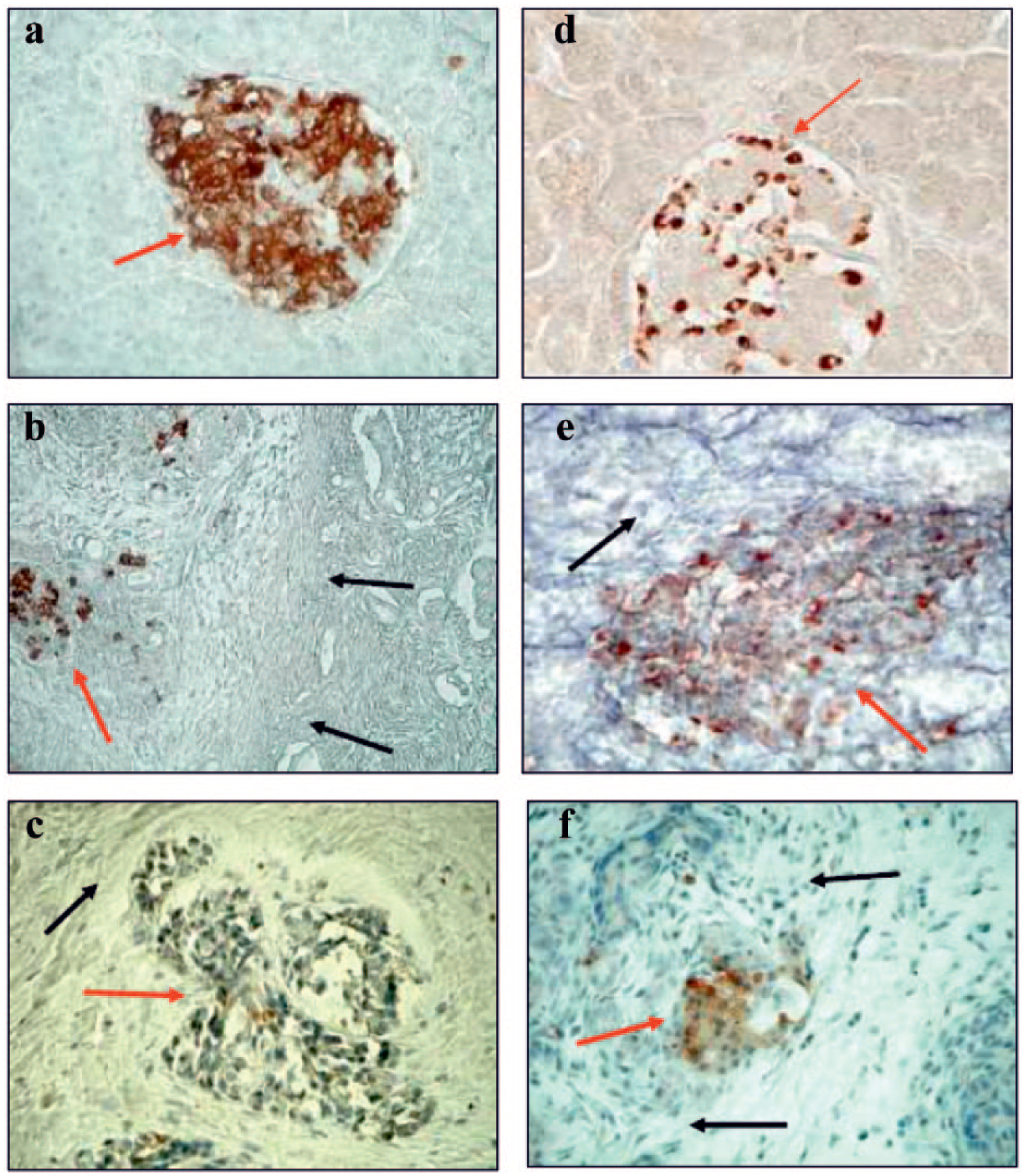
Immunohistochemical localization of insulin (a, b, c) and glucagon (d, e, f) in normal pancreas and pancreas from non-smoking and smoking CP patients with diabetes. Insulin: (a) (210×) A strong and very strong reaction in islet B cells. (b) (137×) A strong reaction in B cells of damage islet. (c) (210×) A weak immunohistochemical reaction in the B cells in damage islet. Glucagon: (d) (210×) A strong reaction in islet A cells. (e) (265×) A diffuse strong and moderate reaction in A cells of damage islet. (f) (210×) A diffuse and moderate immunohistochemical reaction of glucagon in of damage islet (arrows – red: pancreatic islets; blue: acinar cells; green: pancreatic duct cells; black: fibrous lesions in the sublayer).
Non-smoking and smoking CP patients with diabetes
The histopathologic diagnosis confirmed the occurrence of characteristics of the inflammatory process in all the tissues of CP patients. In the exocrine and endocrine parts of the pancreas, fibrous connective tissues were observed.
In all sections taken from non-smoking and smoking patients with CP and diabetes, a much stronger immunohistochemical reaction to IL-6 and ET-1 in the acinar, islet and ductal cells was found when compared to the control sections. Significant differences were detected between non-smoking and smoking patients with diabetes regarding the IL-6 and ET-1 staining intensity in the pancreatic tissue (Table 9).
In smoking CP patients with diabetes, fibrosis and progressive atrophy of cells and a functional defect of these cells was observed, characterized by the much stronger immunohistochemical reaction of IL-6 (diffused and focal) relative to control and non-smoking patients with CP. Interestingly, the highest IL-6 concentrations were detected in the plasma of smoking patients with diabetes.
A less diffuse and less intense staining pattern was seen in the beta cells of specimens retrieved from non-smoking and smoking CP patients with diabetes compared to the control. Finally, in specimens of smoking CP patients, staining in the beta cells was markedly decreased when compared with both the normal pancreas from the control and the pancreas from non-smoking CP patients with diabetes. There was a statistical decrease in the immunohistochemical reaction for insulin in specimens from non-smoking and smoking patients with diabetes compared with the control.
There was a marked decrease in the number of beta cells and the intensity of insulin in tissues from smoking CP specimens with diabetes compared to non-smoking CP specimens with diabetes and the controls (Table 9). Compared to healthy subjects, the weakest reaction for glucagon occurred in the pancreas of the group of smoking CP patients with diabetes (Figure 2(f)). A comparative analysis between the intensity of the immunohistochemical response to glucagon in non-smoking and smoking CP patients with diabetes not showed significant differences. On the other hand, a significant decrease in the intensity of the immunohistochemical reaction for glucagon in smoking diabetic patients was observed compared to non-smoking diabetic patients (Table 9).
Discussion
Diabetes is one of the most common metabolic disorders and the reasons underlying its development are genetic, environmental and lifestyle-related. Many epidemiological studies indicate that cigarette smoking is an independent risk factor for type 2 diabetes.30,34–36
It was found that cigarette smoking can also be an independent risk factor for CP, a major cause of T3cDM. 37 Each cigarette smoked leads to an inflammatory response in the body and if continued, CP develops. Because higher levels of inflammatory markers (CRP and IL-6) herald the development of diabetes 19 and CP, 38 smoking may also contribute to the development of diabetes by increasing levels of inflammatory markers. 18 The presented studies showed that IL-6 concentration in plasma was significantly higher in non-smoking and smoking CP patients with diabetes than in the non-smoking control (Table 2). There was also a statistically higher IL-6 concentration in smoking patients with diabetes compared to non-smoking patients with diabetes. But the highest level of IL-6 was found in smokers with diabetes. In smoking CP patients with diabetes, fibrosis, progressive atrophy of cells and a functional defect of these cells characterized by a much stronger immunohistochemical reaction of IL-6 (diffused and focal) were observed relative to the controls and to non-smoking patients with diabetes. The highest intensity of immunostaining for IL-6 in smoking CP with diabetes coincided with the highest marked cytokine concentrations in the blood of these patients. These findings are consistent with earlier studies. 18 A drastic increase in the level of the investigated cytokine in smoking patients points to a pro-inflammatory effect of cigarette smoke. Experimental rats showed significantly increased levels of serum IL-6 as a consequence of oxidative stress following a 12-week exposure to cigarette smoke. 37
Cigarette smoking is a factor that triggers inflammation in the pancreas and induces fibrosis of the organ, as evidenced by the increase in plasma IL-6 concentration, the increase in IL-6 expression in the damaged tissue of smoking patients (Figure 1(c)) and the strongly positive correlation between the concentration of cotinine (a nicotine metabolite) and the IL-6 concentration (Table 7). According to a recent hypothesis, an initiating event is required for the development of CP; it is first AP until activation of pancreatic stellate cells (PSCs).
The cigarette smoke components activate the anti-inflammatory response (with the release of pro-inflammatory cytokines – IL-6) and stimulate PSCs to produce an extracellular matrix, mainly collagen, fibronectin and proteoglycans. Organ fibrosis comes as a result of continuous inflammation and continuous activation of PSCs.
PSCs’ transformation into myofibroblasts, that is, active forms capable of producing an extracellular matrix and various adhesion molecules, also occurs in response to various inflammatory cytokines and chemokines such as tumor necrosis factor alpha (TNFα), IL-1β, IL-6, IL-8; the growth factors transforming growth factor beta 1 (TGF-β1) and platelet-derived growth factor (PDGF) released by both pancreatic cells (damaged parenchyma cells) and by inflammatory cells.39–41 Potential paracrine activators of PSCs acting in vivo also include reactive oxygen species and ET-1. Active PSCs can produce autocrine agents such as growth factors (PDGF, TGF-β1), cytokines (IL-1, IL-6, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)), activin-A (which increases collagen production and increases TGF-β1 expression and secretion) and ET-1 (which can stimulate the migration and contraction of PSCs). 40 ET-1 is a hormone with strong vasoconstrictor properties, secreted in excessive amounts by damaged endothelial cells.16,42 Borissova et al. 42 demonstrated that smoking increases the ET-1 level in plasma. ET-1 reduces peripheral glucose utilization42,43 probably by inducing vasoconstriction and subsequent tissue hypoxia. Tobacco smoke has long been recognized as a factor that is an impediment to normal endothelial functioning of vessels, including small vessels in the pancreas. The last study has demonstrated that the significantly higher concentration of ET-1 in the plasma of smoking healthy individuals and in patients with pancreatitis, when compared with non-smokers, is consistent with the strong degree of ET-1 expression in pancreatic tissue.16,42 The current study confirmed the previous results. Significant differences were found between ET-1 levels in the plasma of the non-smoking and smoking control groups. An increase in the level of this peptide in the smoking control group and in CP patients is evidence of the impact of cigarette smoking on the functioning of the blood vessel endothelium. This thesis is confirmed by the strongly positive correlation between the cotinine concentration and the ET-1 concentration (Table 7). The highest plasma concentrations of ET-1 were observed in non-smoking and smoking patients with diabetes mellitus (Table 2). Harris et al. 44 also demonstrated a significant correlation between plasma ET-1 levels and diabetic complications. The high level of ET-1 in patients with diabetes can be the result of an imbalance between the factors secreted by the endothelium responsible for the maintenance of normal vascular wall tension (reduction of NO synthesis in favour of ET-1). This phenomenon is particularly evident in the group of smokers, where in addition to the developing state of insulin resistance, the substances contained in the cigarette smoke cause the decomposition of nitric oxide, leading to excessive production of ET-1. 45 The immunohistochemical ET-1 reaction was negative or weakly positive in histologic specimens from the control group, whereas in smoking patients with diabetes, it was strong or very strong in acinar, ductal and islet cells. The difference in the intensity of immunostaining was confirmed by densitometric analysis. Statistically higher ET-1 immunoreactivity was found in non-smoking and smoking patients with diabetes compared to the control group (Table 9). In addition, the degree of immunoreactivity of ET-1 is also dependent on the severity of fibrogenesis of the study tissues (Figure 1(e) and (f)). ET-1 is an activator of PSCs for collagen synthesis, suggesting how important this hormone is in the development of CP. ET-1 also strongly stimulates expression of IL-6, which, in addition to stimulating PSCs, is primarily a pro-inflammatory cytokine. In addition, a strong correlation was observed between ET-1 and IL-6 in smokers, which confirms the relationship between these two factors. It follows from this that ET-1 enhances the local inflammatory response in the pancreas. Due to the strong vasoconstrictor ET-1, a high level of the peptide contributes to local tissue ischaemia, leading to damage in pancreatic tissue.
Ongoing inflammation, which translates into some degree of tissue destruction, leads to permanent histological changes that prevent proper functioning of the organ, among other things, and affect the development of diabetes. It was also observed that as the development of pancreatic fibrosis occurs in patients with CP, the ratio of beta cells to alpha cells decreases. 12 In the present study, a lower number of beta and alpha cells in the islets of Langerhans were found in smoking patients with diabetes compared to the controls (Figure 2(c) and (f)). The lowest intensity of immunostaining for insulin and glucagon was observed in smoking CP patients with diabetes (Table 9). Tissues of these patients were characterized by a significant reduction in the stroma. In addition, severe inflammation and multifocal intra- and inter-lobular fibrosis and severely damaged islets were observed in non-smoking and smoking patients with diabetes (Figure 2(b), (c), (e) and (f)).
The pathogenesis of T3cDM is ultimately due to decreased insulin secretion caused by both a reduction in the number of islets and their functional capacity as a consequence of extensive fibrosis and sclerosis. This theory of T3cDM was also described by other researchers.46,47 The mechanism by which the scarring of pancreatic tissue induces insulin deficiency can be explained by the anatomical and functional interplay between the pancreatic islets and acinar tissue. In earlier studies, it was found that prolonged smoking destroys the exocrine part of the pancreas (i.e. the pancreas decreases morphologically), while the pancreatic islets become restricted and then destroyed.12,15–18 The studies conducted by Sarles et al. 48 showed that addicted smokers are more predisposed to pancreatic calcification than non-smokers.
In the present study, the significantly lower levels of insulin and the higher blood glucose levels in smoking patients with CP in comparison to the controls coincided with the progression of changes in the structure and dysfunction of the endocrine organ. The suppressive effect of cigarette smoking on insulin secretion has been revealed in heavy male smokers. 49
In recent years, it has also been shown that smoking cigarettes affects the production and secretion of other pancreatic hormones such as glucagon, somatostatin and pancreatic polypeptide. Although the precise role of cigarette smoking in the pathogenesis of the decreased excretion of hormones in CP still remains unclear, the presented data provide evidence that tobacco smoking may be regarded as a major causative factor of endocrine disorders in CP.
There is also morphological evidence that indicates that pancreatic exocrine functioning may be influenced by the pancreatic endocrine hormones. Insulin has a trophic effect on the exocrine pancreas, especially on the peri-insular acini. Progressive damage to the pancreatic acinar cells is seen in insulin deficiency. The pancreatic exocrine tissue in a diabetic patient becomes fibrosed and shows a reduced response to the hormonal stimulation. 50 This mechanism has been confirmed in the present study. In the histopathological examination of pancreatic tissues from smoking diabetic patients, the exocrine and endocrine parts of the pancreas were observed to have been severely destroyed (Figure 2(c) and (f)).
Low serum amylase and lipase activity observed in diabetes type 2 may reflect the impaired exocrine–endocrine interactions of the pancreas.22,50 However, only a few clinical studies have addressed this complex relationship.
In our study, we found significantly higher amylase and lipase activity in diabetic patients (especially in smokers) compared to those in the non-smoking control. Another study has revealed that the differences in amylase activity in both the serum and urine of smoking patients compared to non-smoking patients with pancreatitis may prove the significant influence of tobacco smoking on the exocrine functioning of the pancreas. 51
Also, several animal and cellular studies have tried to find out the pathogenic features and the underlying mechanisms which link the islet cells and the acinar cells. 50 But the estimation of the serum amylase activity, along with the blood glucose to assess this relationship, consolidates its clinical relevance in humans. There is evidence that the pancreatic hormones insulin and glucagon influence enzyme synthesis and release in the exocrine pancreas. Insulin has a trophic effect on the acinar cells, whereas glucagon has been found to have an inhibitory influence on the exocrine secretions; moreover, there is a decrease in the sensitivity of the diabetic pancreatic acini to secretagogues. The insulin deficiency and the glucagon excess in diabetes affect the normal atmosphere of the pancreas, thereby decreasing the total volume, the amylase secretions and the bicarbonate content of its exocrine secretions. 50
In our work, we showed a simultaneous decrease in insulin and glucagon secretion from the pancreatic cells of the islets due to their destruction. Our results suggest that the low level of glucagon did not inhibit the secretion of pancreatic enzymes in smoking patients with diabetes. On the other hand, the inflammatory process stimulated by the components of cigarette smoke destroys the acinar cells, which results in the increased secretion of enzymes. Confirmation of this can be found in the strongly positive correlation between cotinine concentration and amylase and lipase activity in smoking patients with diabetes. Several studies evaluating the pancreatic exocrine function in smoking patients with CP showed a significant increase in the activity of the pancreatic enzymes.10,23–26,51
In patients with type 1 diabetes, an increase in serum amylase levels was found. In type 1 diabetes, the clinical and the pathological pictures are different, with the presence of insulitis and the diffuse lymphocytic infiltration of the exocrine pancreas. Also, the difference in serum amylase activity compared to that in the controls was more pronounced in patients with type 1 diabetes who presented with acute fulminant ketoacidosis.52–54 In patients with T3cDM, the change in glucose metabolism ranges from only mild impairment to a severe form which is characterized by frequent episodes of hypoglycaemia, commonly referred to as brittle diabetes. 1
It is generally reported that T3cDM is difficult to control, although there are only a few older studies on this topic.55,56 The hypotheses put forward for the difficulty in achieving good glycaemic control in patients with T3cDM include the loss of pancreatic counter-regulatory hormones (e.g. somatostatin and glucagon), impaired and irregular nutrient absorption due to exocrine pancreatic insufficiency and poor compliance with lifestyle modifications and medication use in patients with cigarette smoking- and/or alcohol-induced pancreatitis.13,14
Since diabetes is a multifactorial disorder, apart from the major pancreatic hormones, the interplay between a number of other factors has an important role in carbohydrate, protein and lipid metabolisms. Hypercholesterolaemia and hypertriglyceridaemia are mostly observed and they are related largely to the degree of diabetic control. 57 Hyperglycaemia may alter the lipoproteins to a form that promotes atherogenesis. Dyslipidaemia, which is characterized by raised levels of TGs, low HDL and high levels of small, dense LDL particles, is very frequently seen in diabetes. This contributes to the risk of coronary artery disease in these patients; the determination of the serum lipid profile in diabetes patients is now considered a standard of diabetic care.22,58 Also, cigarette smoking, which is considered a major risk factor for atherosclerosis and coronary heart disease, may lead to changes in the normal plasma lipid profile even in healthy young people. 59
In general, although many publications on lipid concentration in diabetes type 1 i 2 exist, fewer studies refer to the lipid profile of T3cDM. A study of the lipid profile in patients with T3cDM which also takes into account risk factors such as cigarette smoking has not come to our knowledge.
In our study, we found a dyslipidaemic picture of the lipid profile in non-smoking and smoking diabetic patients as compared to the healthy controls. We observed a significant increase in TC, LDL and TG concentrations in the serum of non-smoking and smoking diabetic patients compared to the non-smoking control group. The relationship of elevated TC and TGs to the risk of ischaemic heart disease has been described in several studies.60,61 The results suggest that an increase in TGs is the most important factor in cardiovascular diseases. 62 A strong positive correlation was also found between cotinine concentration and TC, LDL and TG in the serum of smoking patients with diabetes. The obtained results suggest a significant effect of cigarette smoking on the risk of cardiovascular diseases in these patients.
The cardio-metabolic protective factors like HDL and the cholesterol/HDL ratio in our study were found to be lower in diabetic patients compared to healthy controls. This further confirmed the well-known risk of cardio-metabolic abnormalities and dyslipidaemia in diabetic patients.
Interestingly, cholesterol and TG levels exhibited a positive correlation with serum amylase and lipase activity. It seems that high enzyme activity in diabetic patients (type 3c), even when within normal rage, may be associated with an increased risk for atheromatosis and probably indicates a more extended effect of impaired secretion of insulin and glucagon and impaired pancreatic functioning.
Cigarette smoking results in the induction of lipid peroxidation processes and also increases the levels of substrates of this reaction. The main products of lipid peroxidation are aldehydes, which are a recognized marker of the severity of this process. Nakhjavani et al. 63 suggested that the chronicity of diabetes promotes lipid peroxidation and malondialdehyde production, independent of glycaemic control and antioxidant activity. In our study, we found differences in the TBARS only between non-smoking healthy individuals (3.4 ± 1.9 μmol/L) and smoking patients with diabetes (6.3 ± 3.6 μmol/L). The highest TBARS concentration was observed in smoking patients with diabetes, which confirms that cigarette smoking results in higher oxidative stress markers in the blood of patients with diabetes. Data on acute metabolic complications, such as hypoglycaemia and diabetic ketoacidosis, in pancreatic disorders are scarce. In the only prospective study to date, 54 patients with T3cDM due to CP or a total pancreatectomy were studied. 64 The risk of diabetic retinopathy was 31% and correlated with the duration of the diabetes. In separate studies, diabetic retinopathy (37%), diabetic nephropathy (29%) and peripheral arterial disease (26%) developed in patients from the respective cohorts.3,65 Additionally, complications due to CP are more common in smokers than in non-smokers. 19
In our work, we showed statistically higher creatinine and uric acid levels in smoking diabetic patients compared to non-smoking controls (Table 4). Uric acid – the indicator of kidney function – is also described as a low-molecular antioxidant in pancreatitis. 66 Increased serum uric acid levels of smoking diabetic patients compared to non-smokers may be the result of oxidative stress induced by smoking cigarettes.
It has been proven that the consequence of CP is malnutrition and fatty diarrhoea resulting from impaired digestion. In patients with T3cDM, exocrine pancreatic insufficiency is ubiquitous. Clinically, overt protein and fat malabsorption does usually not occur until over 90% of exocrine pancreatic function has vanished; yet clinical studies clearly indicate that a majority of patients with chronic pancreatic disease have some degree of malabsorption, which might remain undetected by patients and their physicians. 67 This maldigestion, however, can lead to qualitative malnutrition with regard to the absorption of fat-soluble vitamins (A, D, E and K), especially vitamin D. In addition, secondary malnutrition to pancreatic exocrine insufficiency plays a prognostic role in CP patients. The main nitrogenous product of protein catabolism in the human body is urea. Serum urea concentration depends on kidney perfusion and diuresis size, glomerular filtration rate (GFR) and urea synthesis rate, which is directly related to daily protein intake in the diet and protein catabolism. In our work, we showed a significant difference between the average value of urea nitrogen (BUN) in non-smoking and smoking patients with diabetes, and non-smoking and smoking healthy individuals (Table 4). The low concentration of urea found in non-smoking and smoking CP patients with diabetes is associated with reduced urea synthesis caused by the small amount of amino acids available for deamination. However, the small supply of amino acids is due to malabsorption or starvation. The creatinine concentration, which is observed to be within the reference range, and the low urea concentration in the serum of patients with CP and diabetes indicate reduced catabolism of proteins. The long-lasting disease process, or CP, leads to disturbance of the nutritional status.
Interestingly, the lowest value of eGFR and a negative correlation between cotinine and eGFR (
As shown before, 75% of all T3cDM patients suffer from CP as the underlying disease. Furthermore, CP and diabetes are both well-accepted risk factors for the development of pancreatic cancer. T3cDM may be seen as a premalignant condition,70,71 and cigarette smoking is an established risk factor for these diseases,10,72 which is why attempts to reduce the toxic and modifiable contributors to CP, such as abstaining from alcohol and smoking cessation, are highly recommended. Both exacerbate the progression of underlying pancreatic inflammation and fibrosis and contribute to pain.
Our study of T3cDM fills in several important knowledge gaps. At a foundational level, a better understanding of the pathogenesis is needed to more accurately define and distinguish T3cDM from other diabetes subtypes because its prevalence and clinical importance has been underestimated and underappreciated so far. In contrast to the management of type 1 or type 2 diabetes, the endocrinopathy in T3cDM is very complex and complicated by additional present comorbidities such as maldigestion and concomitant qualitative malnutrition. Also important are risk factors for CP and consequent diabetes, such as alcohol and cigarette smoking. Cigarette smoking in patients with CP and diabetes increases inflammation and its associated consequences, impairs the function of the endothelium of the blood vessels leading to ischaemia of the organ and stimulates PSCs for the production and secretion of collagen. These mechanisms lead to the destruction of the exocrine and endocrine parts of the pancreas. In the end, based on the obtained results, it can be concluded that smoking, hyperlipidaemia and a lowered eGFR are expected to influence target organ complications and should be studied concurrently with T3cDM. Furthermore, in smoking patients with T3cDM due to CP, special considerations are needed for management of the disease due to the elevated risk of pancreatic cancer.
Cigarette smoking as a risk factor for pancreatic diabetes mellitus secondary.
Smoking and exocrine–endocrine interactions of the pancreas.
Smoking, chronic pancreatitis, diabetes type 3c and complications.
Footnotes
Acknowledgements
The authors thank Professor Jerzy Rabczynski from the Department of Pathological Anatomy, Wrocław Medical University, for his help in the immunohistochemistry assessment of tissues.
Declaration of conflicting interests
The author(s) received no financial support for the research, authorship and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
