Abstract
Aim:
Type 2 diabetes mellitus is a polygenic metabolic disorder resulting from oxidative stress, the root cause of insulin resistance, β-cell dysfunction and impaired glucose tolerance. The aim of this study was to investigate the role of oxidative stress–related genes ALOX5, ALOX5AP, GPX1, GPX3 and MPO in type 2 diabetes mellitus susceptibility in the Chinese Han population.
Methods:
A total of 396 type 2 diabetes mellitus patients and 678 controls were recruited. The ALOX5 rs10900213, ALOX5AP rs4293222, GPX1 rs1050450, GPX3 rs3828599 and MPO rs2107545 gene polymorphisms were genotyped.
Results:
We found one single-nucleotide polymorphism in the MPO gene was associated with type 2 diabetes mellitus susceptibility [rs2107545: odds ratio = 1.563 (1.166–2.096); p = 0.003], after adjusting for covariates. Furthermore, we also considered the likely complexity of effects of genetic and conventional risk factors in type 2 diabetes mellitus–related vascular complications, such as carotid plaques. Our analysis revealed that the GPX1 rs1050450 and MPO rs2107545 were significantly associated with increased risk of carotid plaques in type 2 diabetes mellitus patients.
Conclusion:
Our study presents novel evidence for main effects of MPO gene on type 2 diabetes mellitus susceptibility. Furthermore, our study supported the association between variants of oxidative stress–related genes (GPX1 and MPO) and carotid plaques in type 2 diabetes mellitus patients, which indicated a modulation of type 2 diabetes mellitus–related vascular complication susceptibility by genetic predisposition.
Introduction
Type 2 diabetes mellitus (T2DM) encompasses individuals who have insulin resistance and usually have relative insulin deficiency. Intriguingly, accumulating evidence has shown that oxidative stress, which results from environmental stress and genetic predisposition, plays a pivotal role in the pathogenesis of T2DM. 1 During the past decade, genetic studies have put the spotlight on the association of the leukotriene generating 5-lipoxygenase (LOX) pathway with insulin resistance and T2DM. It is suggested that components of the 5-LOX pathway (such as ALOX5 and ALOX5AP genes) may provide a link between adipose tissue, inflammation and insulin resistance. 2 However, T2DM is regarded as a multi-factorial metabolic disorder resulting from oxidative stress due to impairment in antioxidant enzymes (AEs) and their scavenging activities against reactive metabolites. Evidence from related research indicates that genes encoding glutathione peroxidase (GPx) may play a pivotal role in the pathogenesis of T2DM. 3 In addition, the myeloperoxidase (MPO) gene is another intriguing candidate, and the variant of MPO is related to T2DM susceptibility in Caucasians in Turkey. 4 However, the association between MPO and T2DM has not been investigated in the Han Chinese population.
To explore the association between oxidative stress–related genes and T2DM in the Chinese Han population, we identified single-nucleotide polymorphisms (SNPs) in five candidate genes related to oxidative stress (such as ALOX5, ALOX5AP, GPX1, GPX3 and MPO genes). Our aim was to explore whether the variants of these oxidative stress genes contribute to the risk for T2DM in the Chinese Han population and further to evaluate the contribution of the genetic predispositions to T2DM-related vascular complications, such as carotid plaques, in order to elucidate the underlying genetic architecture.
Methods
Study population
A total of 396 patients (T2DM without carotid plaques: n = 230 and T2DM with carotid plaques: n = 166) and a control group including 678 age- and sex-matched healthy individuals without diabetes from the Third Xiangya hospital were recruited into our study. The following were documented: age, gender, weight, body mass index (BMI), blood pressure, fasting blood glucose (FBG) and blood lipids [including total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C)]. This study has been approved by the institutional Medical Ethics Committee at the Third Xiangya Hospital. Informed written consent was acquired from all participants.
SNP genotyping
Peripheral venous blood (5 mL) was collected from all participants, and genomic DNA was extracted from peripheral blood leukocytes using phenol-chloroform methods. The genotypes of SNPs (rs4293222 of ALOX5AP, rs10900213 of ALOX5, rs1050450 of GPX1, rs3828599 of GPX3 and rs2107545 of MPO) were determined using Sequenom iPLEX assays with allele detection by mass spectroscopy, using Sequenom MassARRAY technology (Sequenom, San Diego, CA, USA). The primers for polymerase chain reaction (PCR) amplification and extension reactions were designed using MassARRAY Assay Design Software (Sequenom; Supplement Table 1).
Statistical analysis
Statistical analyses were performed using SPSS version 18.0 for Windows (SPSS Inc., Chicago, IL, USA). An unpaired Student’s t-test was used to compare the quantitative data, the chi-square test was applied to identify deviations from Hardy–Weinberg equilibrium and the differences of genotype frequency distribution between T2DM patients and controls, and logistic regression analyses were used to verify the effect of the variant on disease risk after adjustment for the covariates (status of hypertension, hyperlipidaemia, obesity and smoking). p values <0.05 were considered statistically significant (two-tailed).
Results
Baseline characteristics
Supplement Table 2 summarises the baseline characteristics of the subjects. The distribution of age, sex, systolic blood pressure, diastolic blood pressure, serum level of HDL and LDL, and prevalence of smoking status between case and control were well matched. As expected, significance was noted for TC, TG, FBG and BMI (p < 0.05).
Association between specific SNP genotypes and risk of T2DM
Genotype frequency distribution and association with T2DM susceptibility for the SNPs are shown in Table 1. All of the five SNPs tested were in Hardy–Weinberg equilibrium in controls and were thus further analysed. Rs2107545 of the MPO gene was associated with T2DM susceptibility; with the TT genotype as reference, the presence of the CT genotype was associated with a significantly increased risk of T2DM after adjustment for hypertension, hyperlipidaemia, obesity and smoking status [adjusted odds ratio (OR) = 1.563 (1.166–2.096); p = 0.003, Table 1]. In contrast, the gene polymorphisms of ALOX5 rs10900213, ALOX5AP rs4293222, GPX1 rs1050450 and GPX3 rs3828599 did not relate to T2DM susceptibility (p > 0.05). Supplement Table 3 shows the risk estimates for carotid plaques among the diabetic patients. There was no risk found for ALOX5, ALOX5AP or GPX3 genotype. However, rs2107545 of MPO and rs1050450 of GPX1 were associated with carotid plaques risk in T2DM patients [p = 0.010, OR (95% confidence interval (CI)) = 1.380 (1.080–1.763) and p = 0.002, OR (95% CI) = 2.206 (1.333–3.652), respectively].
Genotype frequency distribution and association with T2DM susceptibility for the SNPs.
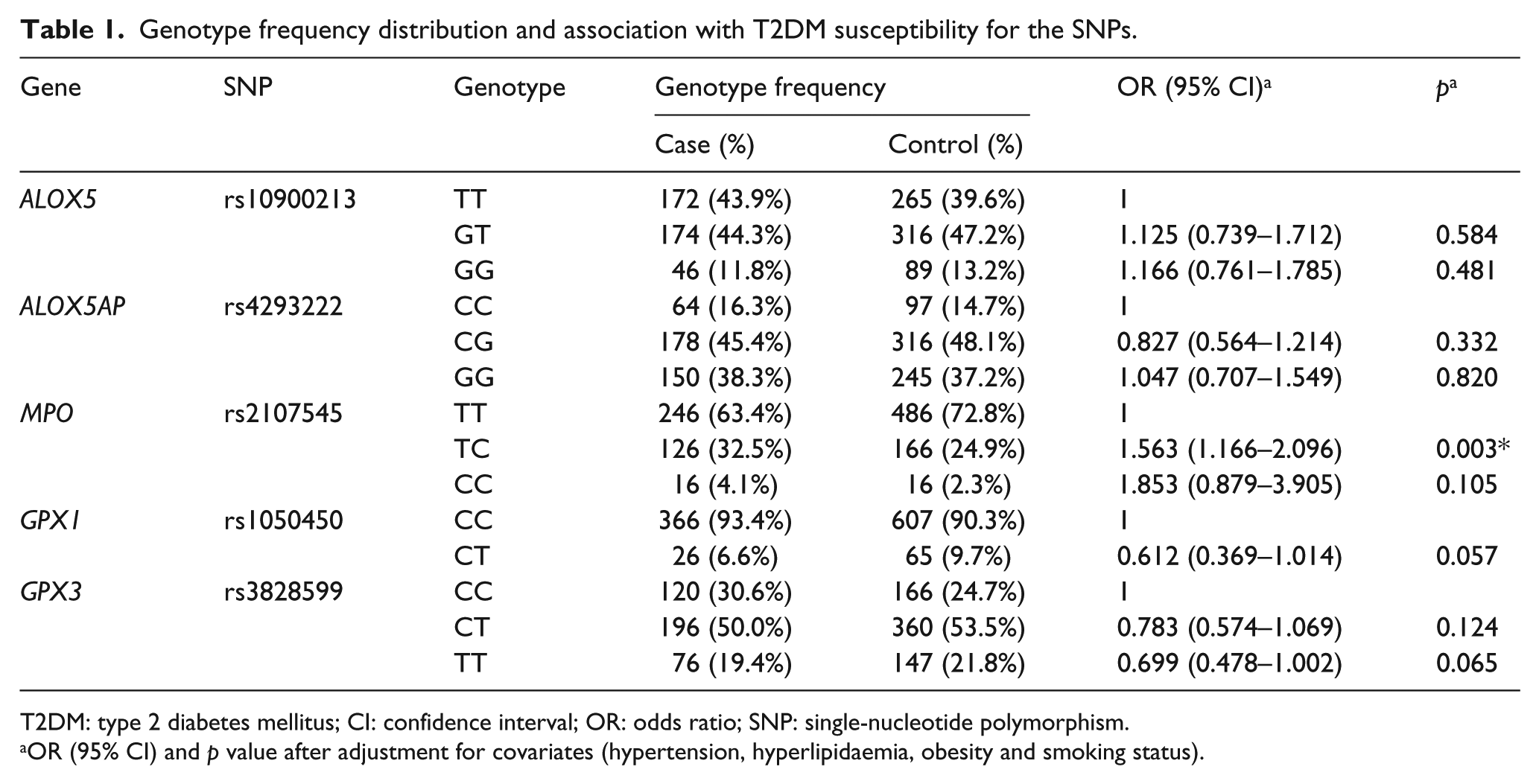
T2DM: type 2 diabetes mellitus; CI: confidence interval; OR: odds ratio; SNP: single-nucleotide polymorphism.
OR (95% CI) and p value after adjustment for covariates (hypertension, hyperlipidaemia, obesity and smoking status).
Discussion
T2DM is a complex multi-factorial disease that is thought to result from interactions between various environmental risk factors and an individual’s genetic predisposition. 1 In this study, we investigated the contribution of the hypothesised genetic and conventional factors involved in oxidative stress to susceptibility of T2DM. Our study disclosed that a gene variant of MPO played a significant role in the susceptibility to T2DM. Our results are in accordance with a previous study carried out in Turkey, which presented novel evidence for the involvement of MPO in T2DM susceptibility, suggesting a modulation of T2DM risk by an MPO polymorphism. 4 Validation of the association in independent populations appears to confirm the moderate effects of MPO gene polymorphism on T2DM risk. Although the underlying mechanism is poorly understood, the physiological activity of its encoded protein myeloperoxidase (MPO) in oxidative stress pathways might give a preliminary indication. Myeloperoxidase, which is generated by activated leukocytes (such as neutrophils and monocytes), is a pro-oxidant downstream effector in inflammation. It is well known that the pivotal physiological effect of myeloperoxidase is production of reactive oxygen species (ROS). Previous studies have demonstrated that ROS represent a crucial factor in insulin resistance, which especially occurs in the liver, muscle and adipose tissues. 5 It is believed that infiltration of neutrophils in adipose tissue is the major source of enhanced peroxides activity and that activation of MPO may be a key mechanism underlying oxidative stress and subsequent insulin resistance. 6 Furthermore, the study also revealed that MPO gene deletion prevented high-fat diet (HFD)-impaired insulin signalling via inhibition of insulin receptor (IR)-β protein nitration and reduction. 6 In our study, a prominent promoter polymorphism (rs2107545) of MPO was associated with susceptibility to T2DM. Our findings are compatible and strengthen previous genetic and biological observations, highlighting the need for further functional studies, particularly in view of the possible utility of MPO as a susceptibility biomarker and a potential drug target for treatment and prevention of T2DM in the future.
Furthermore, our results also revealed that variants of oxidative stress–related genes (GPX1 and MPO) contribute to increased susceptibility of carotid plaque in T2DM patients. The finding will provide new insight into the genetic architecture of T2DM-related vascular complications. It is shown in several studies that oxidant/antioxidant imbalance may contribute to the susceptibility of diabetes-related cardiovascular complications. 7 One important defence mechanism against endogenous ROS is the AE glutathione peroxidase-1 (GPX1). Experimental GPX1 deficiency amplifies the atherosclerotic process, such as endothelial dysfunction, vascular remodelling and invasion of leukocytes in cardiovascular tissue.8,9 Intriguingly, evidence from several studies revealed that the presence of Pro197Leu (rs1050450) substitution of the GPX1 may play a crucial role in determining genetic susceptibility to atherosclerosis in T2DM, 10 which is consistent with our result. On the contrary, the myeloperoxidase catalyses the formation of MPO-derived reactive species that may contribute to oxidative tissue damages, such as atherogenesis. Wiersma et al. 11 suggested that T2DM is associated with mildly increased plasma levels of MPO. Furthermore, it is revealed that plasma MPO level positively correlated with the degree of coronary artery stenosis in T2DM patients. 12 Our results present novel evidence for effects of the MPO gene on increased risk of carotid plaque in T2DM patients, which may support the pivotal role of the MPO gene in atherosclerosis susceptibility. In summary, our results indicated that the variants of oxidative stress–related genes (GPX1 and MPO) contribute to carotid plaque risk in T2DM patients. The MPO and GPX1 genes may contribute to the crosstalk between oxidant and antioxidant pathways and result in oxidant/antioxidant imbalance. Furthermore, the presence of chronic oxidative stress, which is the result of oxidant/antioxidant imbalance, may contribute significantly to the development and progression of diabetes-related vascular complications.
Conclusion
In conclusion, our study presented novel evidence for main effects of MPO gene on T2DM susceptibility. Furthermore, our study supported the association between variants of oxidative stress–related genes (GPX1 and MPO) and carotid plaque risk in T2DM patients, which indicated a modulation of T2DM-related vascular complication susceptibility by genetic predisposition. Our study has elucidated the insight into the genetic architecture of T2DM and related vascular complication, and implications regarding novel strategies for T2DM prevention and treatment in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by the New Xiangya Talent Project of the Third Xiangya Hospital of Central South University (JY201613) to D.L. and Natural Science Foundation of Hunan Province to Z.H. (2016JJ4107).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
