Abstract
Background:
The role of long non-coding RNA in diabetic retinopathy, a serious complication of diabetes mellitus, has attracted increasing attention in recent years. The purpose of this study was to explore whether long non-coding RNA nuclear paraspeckle assembly transcript 1 was involved in the context of diabetic retinopathy and its underlying mechanisms.
Results:
Our results revealed that nuclear paraspeckle assembly transcript 1 was significantly downregulated in the retina of diabetes mellitus rats. Meanwhile, miR-497 was significantly increased in diabetes mellitus rats’ retina and high glucose–treated Müller cells, but brain-derived neurotrophic factor was increased. We also found that high glucose–induced apoptosis of Müller cells was accompanied by the significant downregulation of nuclear paraspeckle assembly transcript 1 in vitro. Further study demonstrated that high glucose–promoted Müller cells apoptosis through downregulating nuclear paraspeckle assembly transcript 1 and downregulated nuclear paraspeckle assembly transcript 1 mediated this effect via negative regulating miR-497. Moreover, brain-derived neurotrophic factor was negatively regulated by miR-497 and associated with the apoptosis of Müller cells under high glucose.
Conclusion:
Our results suggested that under diabetic conditions, downregulated nuclear paraspeckle assembly transcript 1 decreased the expression of brain-derived neurotrophic factor through elevating miR-497, thereby promoting Müller cells apoptosis and aggravating diabetic retinopathy.
Keywords
Introduction
Diabetic retinopathy (DR) is the most common and serious complication of diabetes mellitus (DM) in the eye, and its incidence increases with the prolongation of the DM course. 1 DR, characterized by retinal microangiopathy, has become the leading cause of blindness in adults, which seriously affects the quality of life of DM patients. The pathogenesis of DR is complicated, in which retinal nerve degeneration occurs in the early stage of DR and plays an important role in the progression of DR. 2 Müller cells, as the main glial cell in the retina, provide the structural support and energy required by energy metabolism for retinal neurons and accordingly have a vital role in the growth, injury, repair and regeneration of retinal neurons. 3 The high glucose (HG)-induced oxidative stress, mitochondrial dysfunction and inflammatory factors can cause Müller cells dysfunction or even apoptosis. 4 Therefore, investigation of the molecular mechanism of Müller cells injury in the development of DR is conducive to explore the effective target of preventing and treating the DR-induced retinal nerve injury.
Glutamate is an important neurotransmitter for retina cells, including cone cells and rod cells. In the physiological condition, Müller cells provided the precursor of glutamate synthesis for retinal cells and removed excess glutamate to protect them from neurotoxic effects. 5 Under the pathological conditions of DM, the capacity of transporting and metabolizing glutamate of Müller cells was decreased, resulting in extracellular glutamate accumulation, which led to the lethal damage to nerve cells. 6 HG aggravated mitochondrial damage, then increased cytochrome C level and induced the apoptosis of Müller cells in vitro. 7 Detailed understanding on the pathological changes and injury prevention of Müller cells in early DR has become the research focus currently.
MicroRNAs (miRNAs), a class of non-coding small RNAs with a length of about 21–23 nucleotides, are widely involved in the regulation of the physiological processes of the cells such as cell growth, differentiation and apoptosis. 8 Recent studies identified that miRNAs were closely related to the development of DR that could be used as the potential targets for DR diagnosis and treatment. 9 However, some miRNAs have been suggested to be abnormally expressed in the course of DM, but the role and specific mechanism have not been established, such as miR-497. MiR-497 was upregulated in pancreas islets of spontaneously diabetic rats, indicating a certain role of miR-497 in pathogenic processes. 10 In addition, miR-497 was demonstrated to decrease in glaucomatous retinae and was related to the inflammatory and apoptotic signal. 11 Thus, miR-497 may be an important regulator involved in the context associated with the development of DR. More importantly, the bioinformatics analysis (microrna.org) showed that the binding sites of miR-497 exist in the 3′-untranslated region (UTR) of brain-derived neurotrophic factor (BDNF) who has a significant role in maintaining retinal neurons function and regeneration. 12 BDNF derived from Müller cells could protect photoreceptors and ganglion neurons and was essential for avoiding retinal neuronal degeneration under diabetic condition. 13 The above findings prompted the inference that miR-497 may participate in pathogenesis of DR through regulating BDNF in Müller cells.
Recent articles warranted that long non-coding RNAs (lncRNAs) were also implicated in DR. LncRNA MALAT1, myocardial infarction–associated transcript (MIAT), Sox2OT, RNCR3 were uncovered to exert a broad effect on DR progression, implying that lncRNA is a potential target for DR treatment.14–16 To date, little is known about lncRNA functions and regulation network under the pathological conditions of DM. As miR-497 is a potential target of lncRNA nuclear paraspeckle assembly transcript 1 (NEAT1) (DIANA tools), we explored the interaction between miR-497 and NEAT1 in DR derived Müller cells and further clarified the role of miR-497 and NEAT1 in DR.
Materials and methods
Animal study
All animal experiments were approved by the ethics committee of The First Affiliated Hospital of Zhengzhou University. Male Sprague Dawley rats (200–250 g) were purchased from the animal center of Zhengzhou University. After routine feeding for 3 days, the rats were randomly divided into two groups (n = 6/group): control and DM group. The rats in DM group received streptozotocin (STZ, 65 mg/kg in citrate buffer) (Sigma, St Louis, MO, USA) by intravenous injection once, and the control rats were administrated with an equal volume of citrate buffer (10 mmol/L). The rat with blood glucose levels >16.7 mmol/L after 5 days of injection was considered successful. After 4 weeks of induction, all rats were sacrificed to isolate the retinal tissues, and the collected tissues were stored in –80°C until use.
For pcDNA-NEAT1 administration, pcDNA-NEAT1 (5′-gccactatcggatttataaat-3′) (5 µg/eye/week for 3 weeks) was injected into the left eye of DM rats by intravitreal injection as previously described, 17 and an equal dose of the negative control was injected into the right eye. After 4 weeks of DM, all rats were sacrificed to collect the retinal tissues as above described.
Quantitative reverse transcription–polymerase chain reaction
The expression of NEAT1, miR-497 and BDNF messenger RNA (mRNA) in retinal tissues or Müller cells were determined by quantitative reverse transcription–polymerase chain reaction (qRT-PCR). Total RNA was extracted from retinal tissues or Müller cells using TRIzol reagent (Invitrogen, Waltham, MA, USA) according to reagent instructions and previously reported. For detection of miR-497, the first-strand cDNA was synthetized from total RNA by miRNA cDNA Synthesis Kit (TaKaRa, Dalian, China). For NEAT1 and BDNF mRNA analysis, the cDNA synthesis was performed with SuperScript first-strand synthesis kit (Invitrogen, Waltham, MA, USA) according to kit instructions. The first-strand cDNA was amplified using Script SYBR Green PCR Kit (Qiagen, Hilden, Germany) and specific primers. The expression of NEAT1 and BDNF mRNA were normalized to the endogenous control glyceraldehyde 3-phosphate dehydrogenase (GAPDH), and U6 was used as the control for miR-497. The specific primers used in this study were as follows:
NEAT1 – F: GTTCCGTGCTTCCTCTTCTG; R: GTGTCCTCCGACTTTACCAG;
BDNF – F: AGGACAGCAAAGCCACAATGTTCC; R: TGGACGTTTGCTTCTTTCATGGGC;
miR-497 – F: GTCGTATCCAGTGCAGGGTCCGAGGT; R: ATTCGCACTGGATACGACTACAAACC;
GAPDH – F: GTGAACCATGAGAAGTATGACAAC; R: CATGAGTCCTTCCACGATACC;
U6 – F: GCTTCGGCAGCACATATACTAAAAT; R: CGCTTCACGAATTTGCGTGTCAT.
Western blot analysis
Total protein was extracted from retinal tissues or Müller cells using lysis buffer with protease inhibitors (Sigma, St Louis, MO, USA) according to the manufacturer’s instructions. After determining the protein concentration, equal amount (25 μg) of protein of each sample was analysed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then the seperated proteins were electrotransferred to PVDF membranes (Millipore, Billerica, MA, USA). After blocking in skim milk (5%, w/v), the PVDF membranes were incubated with primary antibody against BDNF (1:1000, Santa Cruz Biotechnology, Dallas, TX, USA) and β-actin (1:500, Santa Cruz Biotechnology, Dallas, TX, USA), followed by incubation with horseradish peroxidase (HRP) secondary antibodies. The target protein bands were visualized using chemiluminescence substrate (Thermo Scientific, Waltham, MA, USA). The β-actin band was served as a loading control.
Cell culture and treatment
Primary rat Müller cells were isolated and cultured as in previous studies. 18 Rats were killed by cervical dislocation, and the eyes were isolated and washed with phosphate-buffered saline (PBS). After removing, 0.5 mm neuroretina was dissected and cut in pieces, followed by trypsin digestion for 5 min. The mixture of trypsin and cells were moved into a sterile centrifuge tube and mixed fully with Dulbecco’s Modified Eagle’s Medium (DMEM)/F12 medium (HyClone, Logan, UT, USA) containing 15% fetal bovine serum (FBS, Gibco, Grand Island, NY, USA). After filtration with the stainless steel sieve, the cell pellets were collected by centrifugation and then resuspended in complete DMEM/F12 medium and cultured in the cell culture flask (Corning New York, USA) at 37°C with 5% CO2. After 72 h, the medium was replaced with fresh medium. The 2–3 passages of Müller cells were used in this study.
Cell transfection
In this study, pcDNA-NEAT1 and its control were adopted and synthesized by Genechem (Shanghai, China), and miR-497 inhibitor/mimic was purchased from RiboBio Co. (Guangzhou, China). The sequences of miR-497 mimic and inhibitor were as follows: mimic, 5′-CAGCAGCACACUGUGGUUUGUA-3′; inhibitor, 5-ACAAACCACAGUGGUCGUCUG-3. For transfection, Müller cells were cultured in 24-well plate (1×105 cells/well) and transfected with pcDNA-NEAT1 or miR-497 mimic or inhibitor (50 pmol/L/well) by Lipofectamine 2000 (Invitrogen, Waltham, MA, USA) according to manufacturer’s instructions. After 48 h of transfection, the cells were collected for detection.
RNA immunoprecipitation assays
Müller cells were collected and lysed in RNA immunoprecipitation assays (RIPA) buffer containing protease inhibitor cocktail. After centrifugation at 12,000 r/min for 10 min, the supernatants were collected and incubated with the antibody for Ago2 or IgG (Sigma, St Louis, MO, USA) coated with magnetic beads (Life Technologies, Gaithersburg, MD, USA) in RNA immunoprecipitation (RIP) immunoprecipitation buffer at 4°C for 1 h. The immunoprecipitated complex was eluted in elution buffer. The RNAs were isolated from the immunoprecipitated complex and were analysed using qRT-PCR with the appropriate primers showed above.
RNA pull-down assays
To test whether NEAT1 interacts with miR-497, the RNA pull-down assay was performed. The DNA sequence of NEAT1 was amplified and transcribed into RNA using T7 RNA polymerase (Roche, Pleasanton, CA, USA) and biotin-labelled UTP (Perkin Elmer). The cellular protein extract was prepared using protein lysis buffer. The labelled RNA, protein extract and streptavidin agarose beads (Invitrogen, Waltham, MA, USA) were added in binding reaction and incubated at 4°C for 1 h. The retrieved pellets from the beads were used for analysis of the AGO2 protein and miR-497 level. LncRNA loc285194 was used as the negative control.
Luciferase report gene assays
According to the predicted sequences of the binding sites of BDNF 3-UTR and miR-497, the mutant/wild-type sequences of BDNF 3-UTR were amplified and inserted into pmiR-RB-REPORT luciferase vector (RiboBio). Müller cells were cultured in 24-well plates (2×105 cells/well) for 24 h. The recombinant luciferase vectors (500 ng) and miR-497 inhibitor/mimic were co-transfected into Müller cells using Lipofectamine 2000 (Invitrogen, Waltham, MA, USA). After culture for 48 h, Müller cells were lysed to detect the Firefly and Renilla luciferase activity in cell lysates by the luciferase reporter assay system (Promega, Madison, WI, USA) according to the manufacturer’s instructions.
Cell apoptosis analysis
The apoptosis of Müller cells was assessed using flow cytometry with Annexin V-fluorescein isothiocyante (FITC)/propidium iodide (PI) apoptosis kit (Sigma Aldrich, St Louis, MO, USA). After transfection or HG treatment, Müller cells were harvested and stained with Annexin V-FITC and PI according to the manufacturer’s instructions. The apoptosis was quantified on a flow cytometry (FACScan, BD Biosciences, Franklin Lakes, NJ, USA).
Detection of retinal thickness
The retinal thickness was measured as previously described. 19 The eye was enucleated and fixated with formalin. The retina was separated and embedded in paraffin. The retina pieces were prepared and stained toluidine blue. The retinal thickness was calculated as the length (μm) from the retinal pigment epithelium (RPE) layer to the inner limiting membrane with the microscope.
Statistical analysis
All data from at least three independent experiments were expressed as the mean ± standard deviation (SD) and analysed by SPSS 18.0 software and GraphPad Prism. Student’s t-test was used to analyse the statistical difference (p < 0.05).
Results
The expression changes of NEAT1, BDNF and miR-497 in the retina tissues of DM rats
We first compared the differential expression of NEAT1, BDNF and miR-497 in DM retina and normal retina of rats. The results showed that the relative level of NEAT1 in the retina of DM rats was significantly lower than normal rat retina (Figure 1(a)). Moreover, we found a significant increase in miR-497 expression in the retina of DM rats (Figure 1(b)). Similar to NEAT1, both BDNF mRNA and protein levels were downregulated in the DM rat retina (Figure 1(c)).
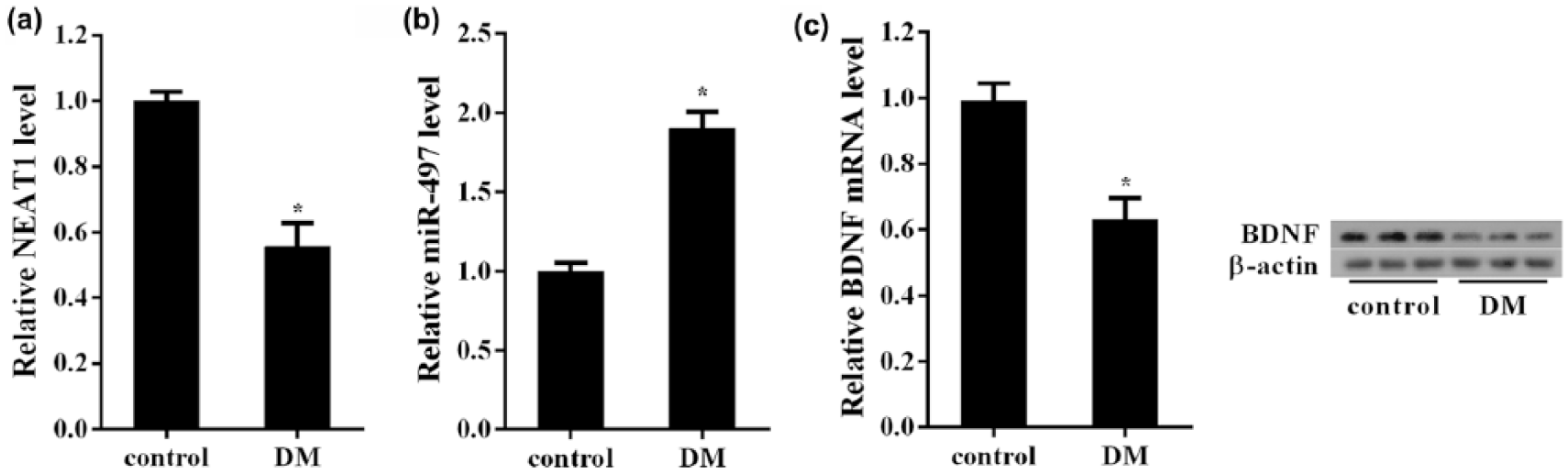
The expression changes of NEAT1, BDNF and miR-497 in the retina tissues of DM rats: (a) the relative expression of NEAT1 in the retina tissues was determined using qRT-PCR. NEAT1 was significantly downregulated in the retina tissues of DM rats (n = 6). (b) MiR-497 determined by qRT-PCR had significantly higher levels in DM retina tissues (n = 6) and (c) BDNF was significantly decreased in DM retina tissues (n = 6) at both mRNA and protein levels. *p < 0.05 versus control.
The effects of HG treatment on the expression of NEAT1, BDNF and miR-497 in Müller cells
We further examined the expression changes of NEAT1, BDNF and miR-497 in HG-treated Müller cells, the model in vitro for DR. As shown in Figure 2(a), HG (25 mM) treatment for 72 h significantly decreased the expression of NEAT1 in Müller cells when compared to Müller cells with 5 mM glucose [normal glucose (NG)]. Meantime, there was a significant increase in miR-497 expression in HG-treated Müller cells (Figure 2(b)). The reduction of BDNF mRNA and protein levels was detected in the Müller cells with HG treatment (Figure 2(c)).
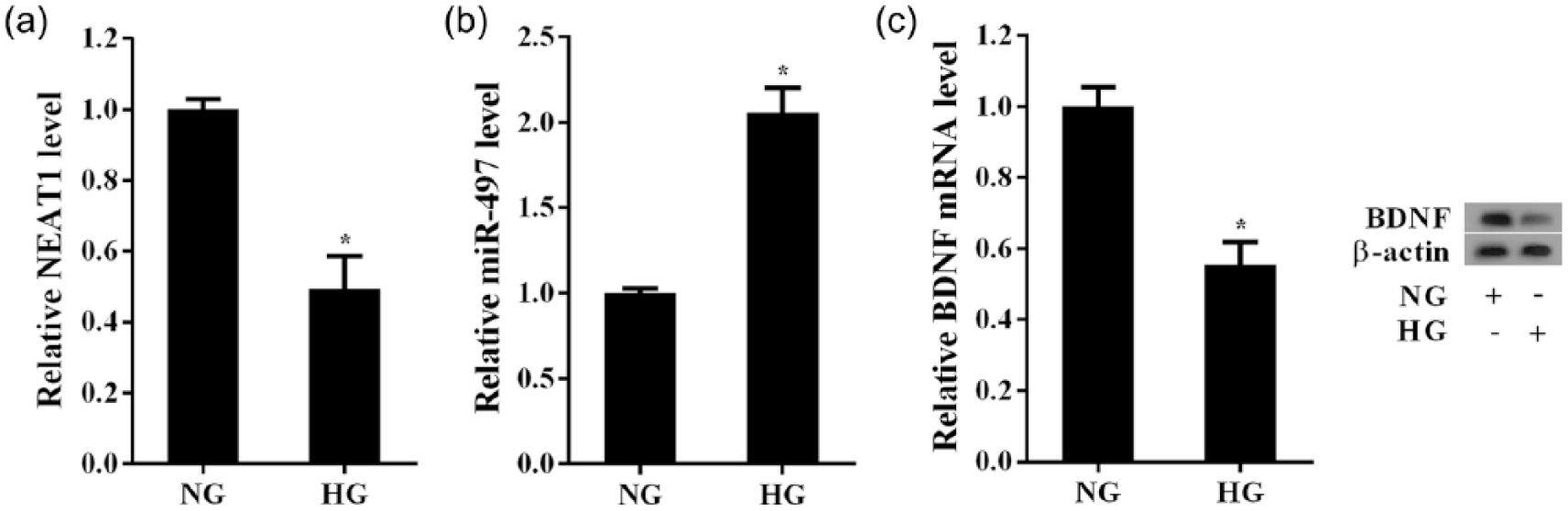
The effects of HG treatment on the expression of NEAT1, BDNF and miR-497 in Müller cells. Müller cells exposured to HG (25 mM) for 72 h was used to simulate the DR conditions. Müller cells cultured in the medium containing 5 mM glucose (NG) were used as the control. Analysis of the relative expression of: (a) NEAT1 and (b) miR-497 in Müller cells with or without HG. (c) Analysis of the level of BDNF mRNA and protein in Müller cells with or without HG. *p < 0.05 versus control.
The effects of NEAT1 overexpression on Müller cells apoptosis with HG treatment
As NEAT1 was downregulated in both DM rat retina and HG-treated Müller cells, we explored the effects of NEAT1 overexpression on the apoptosis of Müller cells with HG treatment. The results revealed that the transfection of pcDNA-NEAT1 effectively promoted the expression of NEAT1 in Müller cells and almost completely reversed the inhibition of HG on NEAT1 expression (Figure 3(a)). Pre-treatment with pcDNA-NEAT1 also repressed the increase of miR-497 expression in Müller cells which induced by HG treatment (Figure 3(b)). Flow cytometry analysis showed that NEAT1 overexpression attenuated the promoting apoptosis of HG treatment on Müller cells (Figure 3(c)). These results suggested that downregulated NEAT1 could mediate the promoting apoptosis of HG on Müller cells, which might be related to regulating miR-497 expression in Müller cells.
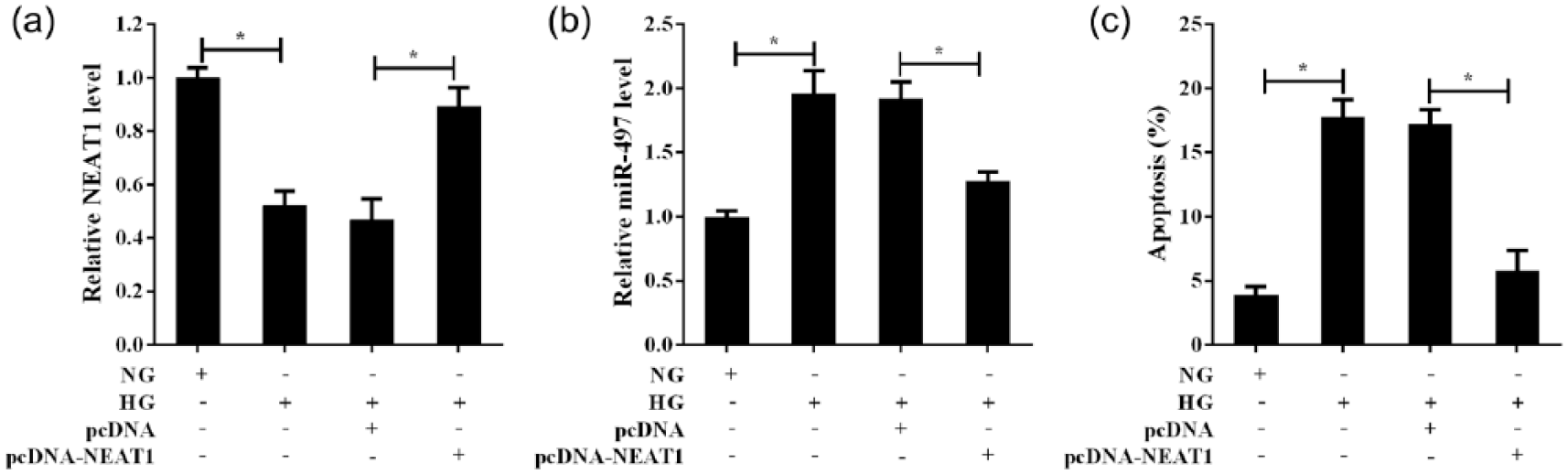
The effects of NEAT1 overexpression on Müller cells apoptosis with HG treatment. Müller cells were treated with NG or HG or HG+pcDNA or HG+pcDNA-NEAT1 for 72 h: (a) the relative expression of NEAT1 in the Müller cells with treatment as indicated was determined using qRT-PCR, (b) NEAT1 regulated miR-497 expression in Müller cells with HG treatment and (c) the apoptosis of Müller cells was analysed by flow cytometry methods. *p < 0.05 by Student’s t-test.
Interaction between NEAT1 and miR-497 in Müller cells
Bioinformatics analysis (DIANA tools) indicated that the potential NEAT1 binding sites were presented in miR-497 sequences (Figure 4(a)). RIP assays showed that NEAT1 and miR-497 were presented in AGO2 precipitates, confirming that NEAT1 might interact with miR-497 (Figure 4(b)). RNA pull-down assays and qRT-PCR analysis verified that NEAT1 interacted with miR-497 (Figure 4(c)). These findings further indicated that HG inhibited NEAT1 expression, which in turn increased the level of miR-497 in Müller cells.
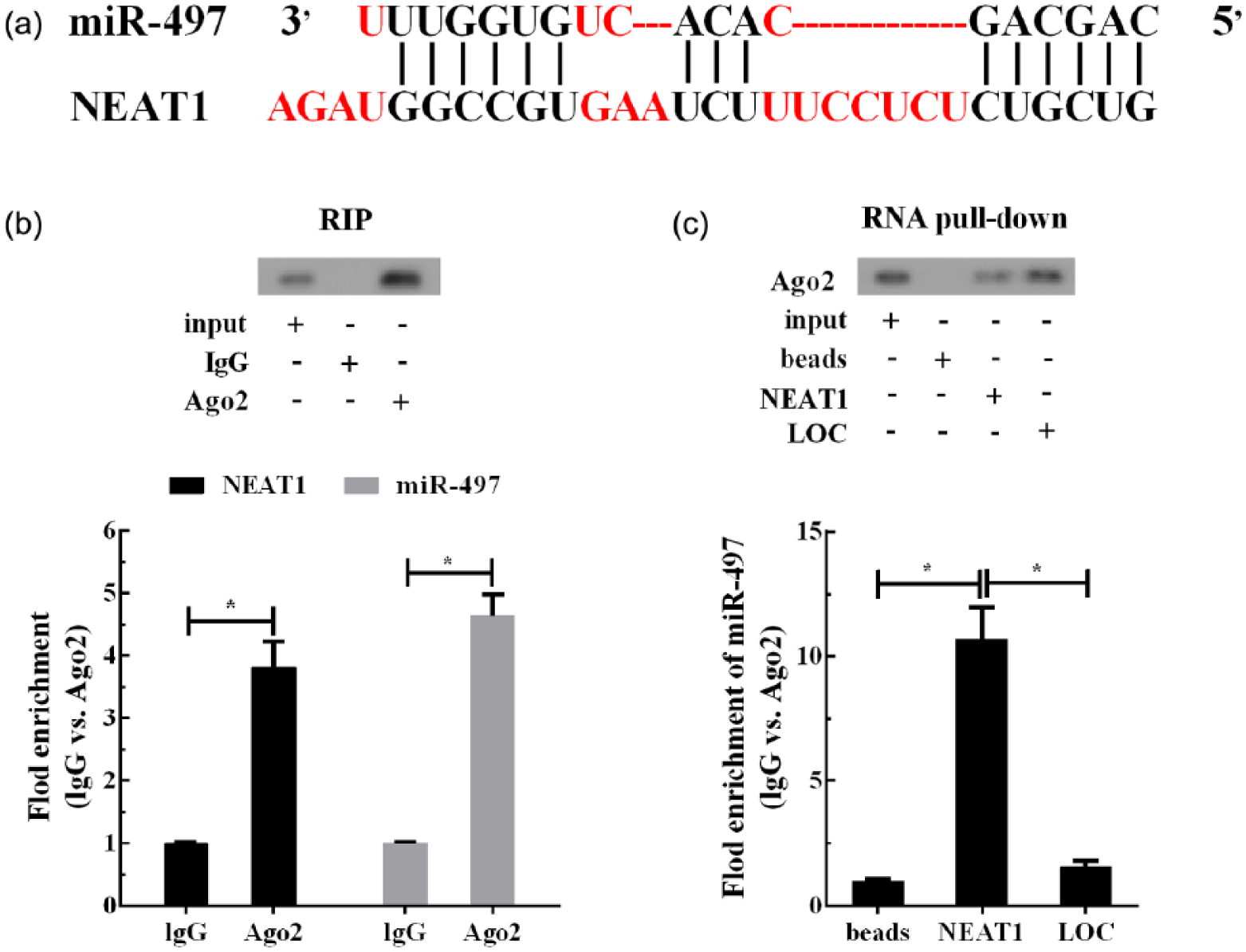
Interaction between NEAT1 and miR-497 in Müller cells: (a) the potential NEAT1 binding sites were presented in miR-497 sequences, (b) RNA immunoprecipitation (RIP) assays were performed to explore the possible correlation between NEAT1 and miR-497 and (c) NEAT1 directly combined with miR-497 was supported by RNA pull-down assays. *p < 0.05 by Student’s t-test.
MiR-497 negatively regulated BDNF expression in Müller cells
To identify the targets of miR-497 in Müller cells apoptosis, luciferase report gene assays were designed to investigate the regulation of miR-497 on BDNF expression based on bioinformatic analysis (microrna.org). The putative binding sites of miR-497 and BDNF 3-UTR and the mutant sequences of the binding sites were shown in Figure 5(a). MiR-497 overexpression induced by miR-497 mimic dramatically reduced the relative luciferase activity in Müller cells transfected with the recombinant luciferase vectors carrying the wild-type sequences of putative binding sites, whereas no effect of miR-497 overexpression was observed in Müller cells transfected with the recombinant luciferase vectors carrying the mutant sequences of putative binding sites (Figure 5(b)). MiR-497 silencing consistent with miR-497 overexpression showed that miR-497 negatively regulated the relative luciferase activity in Müller cells transfected with the wild-type sequences of BDNF 3-UTR, but it had no effect on the relative luciferase activity of Müller cells transfected with the recombinant luciferase vectors carrying the mutant sequences of putative binding sites (Figure 5(b)). The findings were further confirmed by the results that miR-497 negatively regulated BDNF expression both at mRNA and protein levels in Müller cells (Figure 5(c)).
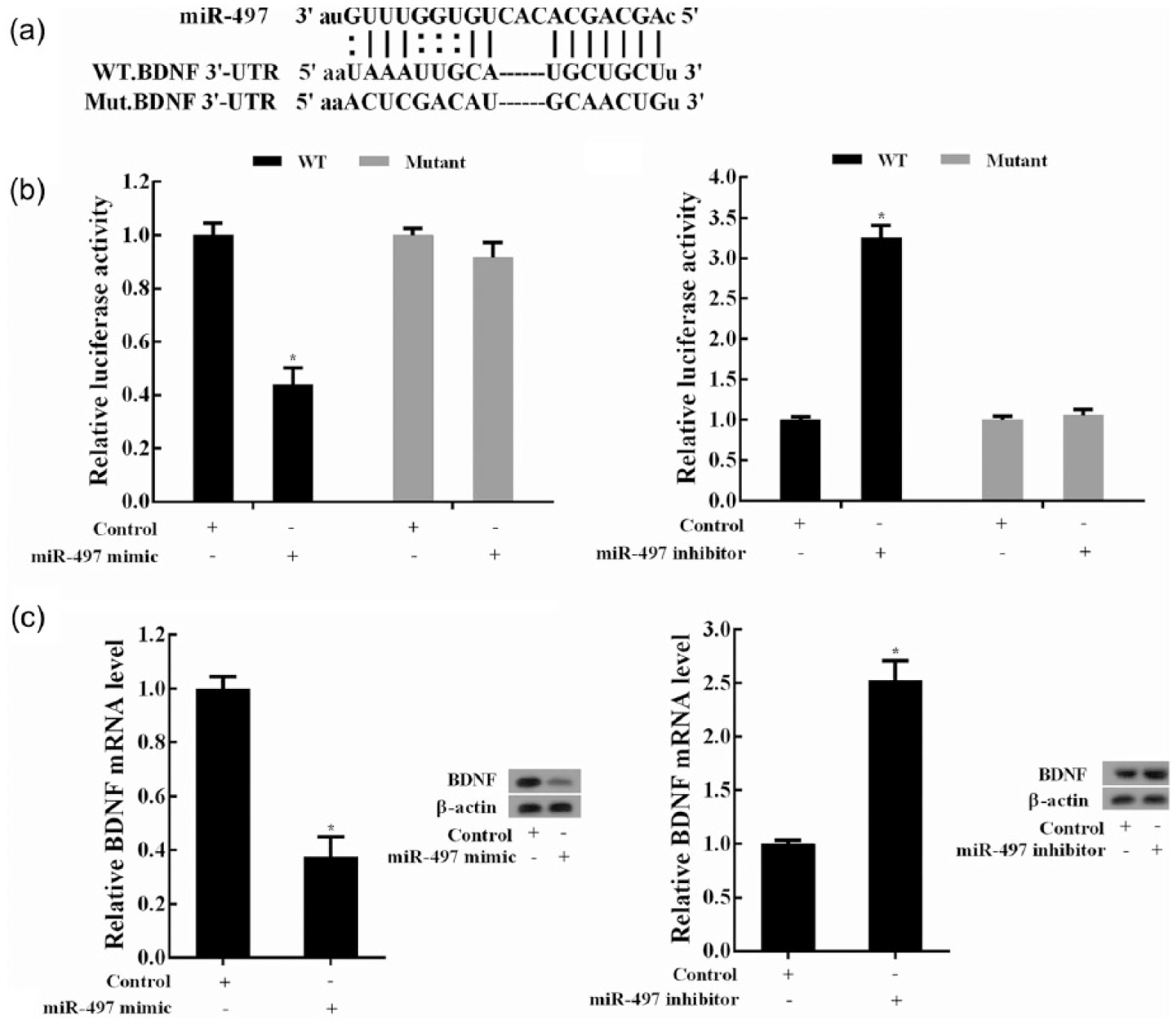
MiR-497 negatively regulated BDNF expression: (a) putative binding sites of miR-497 and BDNF. According to the predicted sequences of the binding sites, the mutant sequences were shown. (b) Luciferase report gene assays were used to explore the interaction between miR-497 and BDNF 3-UTR in Müller cells. *p < 0.05 versus control. (c) Effects of miR-497 expression on BDNF mRNA and protein levels. Müller cells were transfected with miR-497 inhibitors or mimics, and the expression changes of BDNF mRNA and protein were analysed and compared. *p < 0.05 versus control.
Downregulated NEAT1 mediated the HG-induced apoptosis of Müller cells through regulating miR-497/BDNF
To test the role of miR-497 in the HG-induced apoptosis of Müller cells, NEAT1 and miR-497 were co-expressed in Müller cells. It was observed that miR-497 mimic rescued the NEAT1-induced reduction of miR-497 in Müller cells, and miR-497 level was restored completely (Figure 6(a)). Meanwhile, miR-497 overexpression eliminated the NEAT1-induced inhibition on BDNF, suggesting that NEAT1 could positively regulate BDNF expression through miR-497 (Figure 6(b)). More importantly, miR-497 overexpression also obviously brought down the effects of protecting Müller cells apoptosis induced by NEAT1 and promoted cell apoptosis (Figure 6(c)).
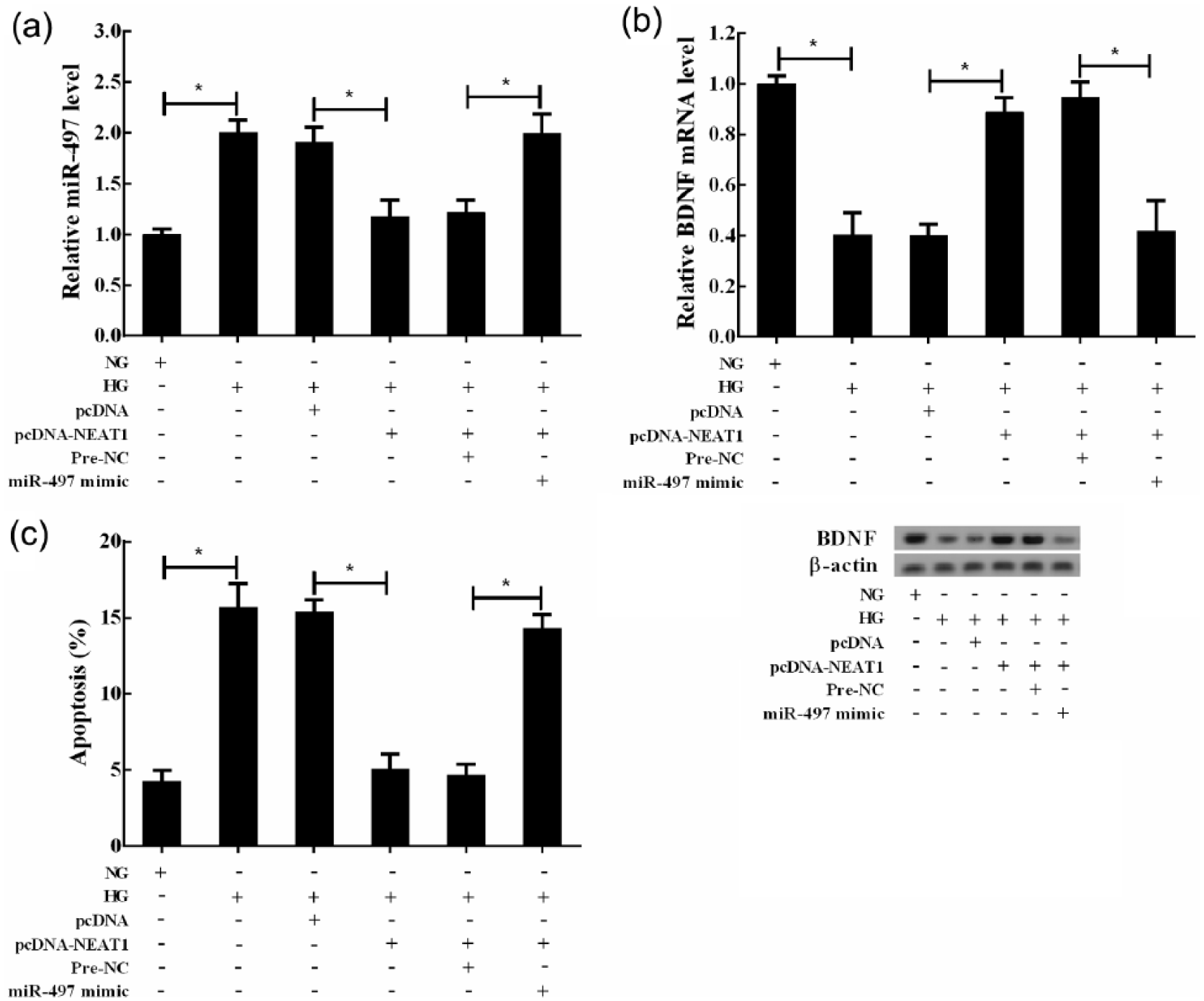
Downregulated NEAT1 mediated the HG-induced apoptosis of Müller cells through regulating miR-497/BDNF. Müller cells were divided into six groups: NG, HG, HG+pcDNA, HG+pcDNA-NEAT1, HG+pcDNA-NEAT1+Pre-NC and HG+pcDNA-NEAT1+miR-497 mimic. (a) Analysis of the relative expression of miR-497 in Müller cells, (b) analysis of the level of BDNF mRNA and protein in Müller cells and (c) the apoptosis of Müller cells was analysed by flow cytometry methods. *p < 0.05 by Student’s t-test.
The protective effects of NEAT1 on retinal function in DM rats
Based on the results of the experiment in vitro, the pcDNA-NEAT1 was injected into the eye of DM rats by intravitreal injection to observe the protective effects of NEAT1 on DR retina function. The intravitreal injection of pcDNA-NEAT1 significantly upregulated NEAT1 level in the retina of DM rats (Figure 7(a)) but significantly inhibited miR-497 expression (Figure 7(b)). Furthermore, BDNF mRNA and protein expressions were increased significantly with NEAT1 expression in DM retina (Figure 7(c)). Upregulated NEAT1 in DM retina effectively alleviated the retina thickening of DM rats (Figure 7(d)). Moreover, NEAT1 overexpression increased the expression of glutamine synthetase (GS) in DM retina, suggesting the beneficial effect of NEAT1 on retinal function of DM rats (Figure 7(e)). And cleaved caspase-3 was decreased with NEAT1 expression in DM retina, suggesting that the DM-induced apoptosis was alleviated by NEAT1 overexpression (Figure 7(e)).
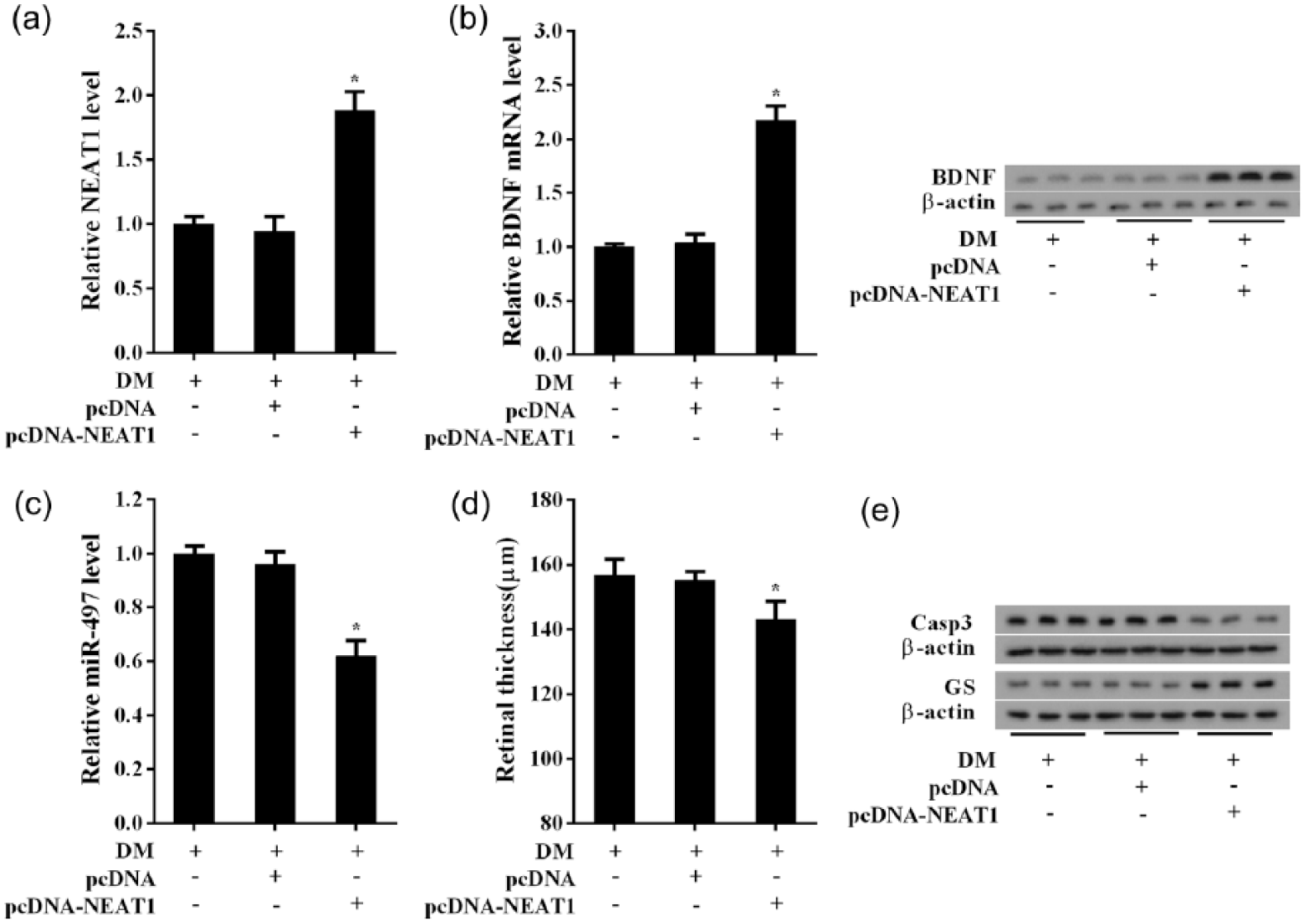
The protective effects of NEAT1 on retinal function in DM rats. The DM rats (n = 18) induced by STZ had pcDNA-NEAT1 administration at the left eyes (n = 12) by intravitreal injection, the right eyes (n = 12) as the control. The retina tissues were collected for detection. (a) The pcDNA-NEAT1 upregulated NEAT1 expression in the retina of DM rats, (b) but significantly inhibited miR-497 expression, (c) BDNF mRNA and protein were also induced by pcDNA-NEAT1 in DM retina, (d) the pcDNA-NEAT1 relieved the retina thickening in DM rats and (e) NEAT1 overexpression increased the expression of glutamine synthetase (GS) in DM retina, but cleaved caspase-3 was decreased with NEAT1 expression in DM retina. *p < 0.05 versus DM+pcDNA.
Discussion
The role of non-coding RNAs (ncRNAs) in human diseases, genetic diseases, tumours, metabolic diseases and so on has attracted increasing attention in recent years. Extensive evidence suggested that ncRNAs were implicated in the pathological micro-environment, cell proliferation and apoptosis, systemic or local inflammatory response that directly involved in the genesis and progression of human diseases. NcRNAs, including long ncRNAs (lncRNAs) and microRNAs (miRNAs), have been shown to participate in the pathogenesis of DM and the related complications. 20 For instance, lncRNA maternally expressed gene 3 (Meg3), an imprinted gene, was downregulated in type 1 and type 2 diabetes mellitus (T1DM and T2DM) mice, as well as in the islets of T2DM patients.21,22 Low-expressed Meg3 inhibited insulin synthesis and secretion and promoted beta cell apoptosis. 21 MEG3 knockdown further aggravated the microvascular dysfunction of DR. 23 In this study, we observed a decrease in lncRNA NEAT1 expression in the retina of DM rats. The abnormal NEAT1 expression in DM retina might be related to the changes of DR retina structure and function. LncRNA NEAT1 has been shown to be involved in many human diseases, including tumours and nervous system diseases. 24 Herein, this work, for the first time, explored the role of NEAT1 in DR and its underlying mechanism. NEAT1 has also been indicated as a regulatory molecule closely related to inflammation. 25 As the main glial cells of the retina, Müller cells–mediated inflammation plays vital role in the development process of DR. 26 So, we aimed to clarify whether NEAT1 was involved in DR through regulating physiological activity of Müller cells. In this work, HG-induced apoptosis of Müller cells was accompanied by the significant downregulation of NEAT1 in vitro. Further study demonstrated that HG promoted Müller cells apoptosis through downregulating NEAT1. Müller cells were essential for the maintenance of normal retina function, which supported retinal neurons and blood–retinal barrier. Studies have shown that Müller cell loss induced by apoptosis was one of the basic pathogenesis of DR, which penetrated the course of DR. 27 Both in vivo and in vitro studies confirmed that the diabetic conditions induced the apoptosis of Müller cells. 7
It has been well known that NEAT1 could regulate the downstream molecules and signalling pathways by miRNAs and thereby influenced cell function or survival. As documented in previous studies, miR-101, miRNA-214, miR-107 and miR-449b-5p were identified as the target of NEAT1.28–31 Based on bioinformatic analysis, we confirmed that miR-497 was a new target of NEAT1 in Müller cells. Previous studies shown that miR-497 was upregulated in the pancreatic islets of T2DM rats. 10 Our results revealed that the expression of miR-497 was elevated in the retina of DM rats and the HG-treated Müller cells, suggesting a fundamental role of miR-497 in DR. In a further experiment, it was confirmed that downregulated NEAT1 mediated the promoting apoptosis of HG in Müller cells via negative regulating miR-497. Current evidence indicated that the interaction between miRNA and lncRNA was achieved by a variety of mechanisms: (1) lncRNA, as a ‘sponge’, adsorbs miRNA; (2) miRNA decays lncRNA; (3) lncRNA competes with miRNA for interacting with target molecules; and (4) lncRNA affects the stability of miRNA.32–35 In this study, miR-497 expression was negatively regulated by NEAT1. And RIP assays together with RNA pull-down assays identified the interaction between NEAT1 and miR-497, but the precise interactions between NEAT1 and miR-497 need more work.
BDNF, as a retinal neuronal protection factor, was important for the survival of retinal ganglion cells after DR. 36 In addition, BDNF was conducive to maintain Müller cell function, and Müller cells–derived BDNF could protect photoreceptors and ganglion neurons, and avoid retinae neuronal degeneration under diabetic condition.13,37 In this study, BDNF was detected to decrease in the retina of DM rats and the HG-treated Müller cells, which were consistent with the published studies. Moreover, we analysed and confirmed that BDNF was negatively regulated by miR-497 and associated with the apoptosis of Müller cells under HG.
Conclusion
In conclusion, our results suggested that under diabetic conditions, downregulated NEAT1 decreased the expression of BDNF through elevating miR-497, thereby promoting Müller cells apoptosis and aggravating DR (Figure 8). Constantly exploring and revealing the molecular basis of DR will be helpful in the development of therapeutic method for DR. Our study needs to be improved from the following: (1) further experiments are needed to explore the concrete mechanism of BDNF regulating Müller cells apoptosis; (2) more studies are needed to clarify whether NEAT1 can involve in DR through directly regulating other retinal cells, including retinal ganglion cells, RPE and retinal endothelial cells.
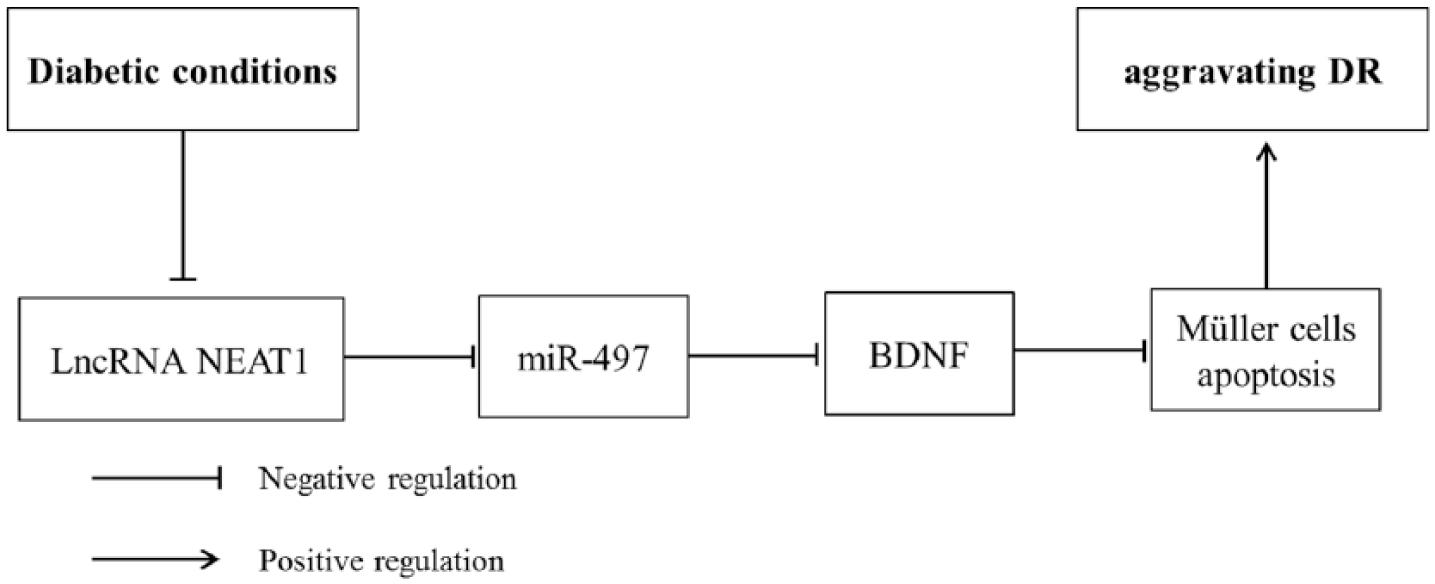
The pictorial representation of NEAT1 correlation with miR-497 and BDNF in DR.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
