Abstract
Background:
The combined effect of vitamin D and parathyroid hormone on glucose homeostasis has not been adequately investigated. The aim of this study was to examine the role of parathyroid hormone/vitamin D axis on glucose homeostasis in elderly persons with prediabetes.
Methods:
Patients with prediabetes (n = 144) and healthy age-matched controls (n = 81) were included in this cross-sectional study. Study parameters included anthropometric characteristics, morning fasting glucose (fasting plasma glucose), insulin (fasting plasma insulin), parathyroid hormone, 25-hydroxyvitamin D, homeostasis model assessment of insulin resistance and homeostasis model assessment of β-cell function. Both groups were stratified into subgroups according to vitamin D status and tertiles of parathyroid hormone.
Results:
Both groups were comparable in terms of body mass index, 25-hydroxyvitamin D and parathyroid hormone status. In the prediabetes group, fasting plasma glucose differed significantly across parathyroid hormone tertiles, increasing from the first to the third tertile (p = 0.011). There were higher fasting plasma glucose values in participants with vitamin D deficiency/parathyroid hormone third tertile compared to all other groups (p = 0.031, 0.027 and 0.039, respectively).
Conclusion:
Parathyroid hormone status is associated with impaired glucose homeostasis; hypovitaminosis D combined with high parathyroid hormone concentrations are associated with glycaemic dysregulation in elderly patients with prediabetes.
Keywords
Introduction
During the last decades, accumulating evidence indicates a role for vitamin D (vit D) as a biological metabolic regulator. 1 Hypovitaminosis D seems to be implicated in the pathogenesis of type 1 and type 2 diabetes mellitus (Τ2DM) and an inverse association between serum 25-hydroxyvitamin D [25(OH)D] and fasting plasma glucose (FPG) concentrations, impaired glucose tolerance (IGT) and T2DM prevalence has been reported. 2 Available interventional studies have shown conflicting results, with regard to a potential beneficial effect of vit D supplementation in patients with diabetes mellitus (DM). 3 Furthermore, increased parathyroid hormone (PTH) concentrations are associated with lower insulin sensitivity and higher prevalence of metabolic syndrome (MetS), independently of vit D status. 4
Previous results indicated that the combination of hypovitaminosis D and increased PTH seems to independently predict β-cell dysfunction, insulin resistance and glycaemia. 5 However, the combined effect of vit D status and PTH in prediabetes (preDM) has not been assessed so far. The aim of this study was to evaluate vit D status and PTH concentrations in elderly patients with preDM, compared to healthy age-matched controls, trying to elucidate their potential combined role in glucose dysregulation and insulin resistance.
Materials and methods
A group of elderly (⩾65 years old) with preDM and a healthy, age-matched control group [normal fasting glucose (NFG) group] were included. PreDM was diagnosed based on the American Diabetes Association criteria, either as impaired fasting plasma glucose (IFG) (FPG: 101–125 mg/dL) or IGT [2-h plasma glucose in the 75-g oral glucose tolerance test (OGTT): 140–199 mg/dL]. 6 Exclusion criteria were (a) age < 65 years old, (b) patients with pre-existing DM, (c) patients on any medication that could affect glucose metabolism or history of thyroidectomy or parathyroidectomy, (d) patients on vit D supplements, (e) conditions that affect vit D metabolism, such as malabsorption syndromes and chronic renal failure, (f) patients with body mass index (BMI) ⩾35 kg/m2.
This study was conducted in accordance with the Helsinki Declaration principles. Written informed consent was obtained from all participants. The research protocol was approved by the Ethics Committee of AHEPA University Hospital.
FPG, fasting plasma insulin (FPI), HbA1c, 25(OH)D and PTH were measured. All participants underwent an OGTT. Reference ranges were 70–110 mg/dL for glucose and <25 mIU/L for insulin. Insulin resistance was estimated with the homeostasis model assessment of insulin resistance (HOMA-IR): fasting insulin concentration (mU/mL) × FPG (mmol/L)/22.5. 7 The reference range for PTH was 15–65 pg/mL. Vit D status was assessed with measurement of serum 25(OH)D [including both 25(OH)D2 and 25(OH)D3].
Statistical analysis
Characteristics of preDM and NFG groups as well as 25(OH)D deficient and 25(OH)D sufficient groups were compared by independent samples t-test or x2 test. Vit D status was classified as ‘sufficient’ or ‘insufficient’ according to Institute of Medicine criteria [sufficiency: serum 25(OH)D 20–40 ng/mL (50–100 nmol/L); insufficiency < 20 ng/mL (50 nmol/L)]. 8 The tertiles of PTH were first tertile ⩾ 20 pg/mL, second tertile ⩾ 20 pg/mL and ⩾40 pg/mL, and third tertile > 40 pg/mL. The two-factor analysis of covariance (ANCOVA) models were used to evaluate the main effects of preDM versus NFG groups, vit D status (deficient vs sufficient), and PTH status (tertile) and their interaction. The one-factor ANCOVA models were used to compare FPG, FPI, HOMA-IR and homeostasis model assessment of β-cell function (HOMA-β) in the subgroups. Participants were stratified into four subgroups: (a) vit D deficient/PTH first to second tertile, (2) vit D deficient/PTH third tertile; (3) vit D sufficient/PTH first to second tertile; (4) vit D sufficient/PTH third tertile. Means were compared using one-factor ANCOVA adjusting for age, sex, BMI and season of sample collection (summer to fall: 1 June–30 November vs winter to spring: 1 December–31 May). Statistical analyses were performed with IBM SPSS Statistics software, version 22.0.
Results
Demographic and metabolic characteristics of study participants, according to glycaemic status are presented in Table 1. Βoth groups were comparable in terms of age, gender distribution and BMI. PreDM group manifested significantly higher FPG concentrations and HOMA-IR compared with NFG group [101.0 (99.0–108.0) vs 93.5 (91.5–95.5) mg/dL, p < 0.0005 and 2.85 ± 1.35 vs 2.28 ± 1.43, p = 0.008, respectively], whereas no differences were observed in FPI concentrations and HOMA-β [11.1 ± 5.4 vs 9.6 ± 5.9 μU/mL, p = 0.054 and 96.34% (94.24%–98.44%) vs 96.32% (94.18%–98.46%), p = 0.999]. No differences were observed in PTH (53.2 ± 22.1 vs 49.6 ± 21.8 pg/mL, p = 0.262) and 25(OH)D (18.6 ± 5.7 vs 17.9 ± 5.1 ng/mL, p = 0.365).
Demographic and metabolic characteristics of study participants, according to glycaemic status (preDM vs NFG).
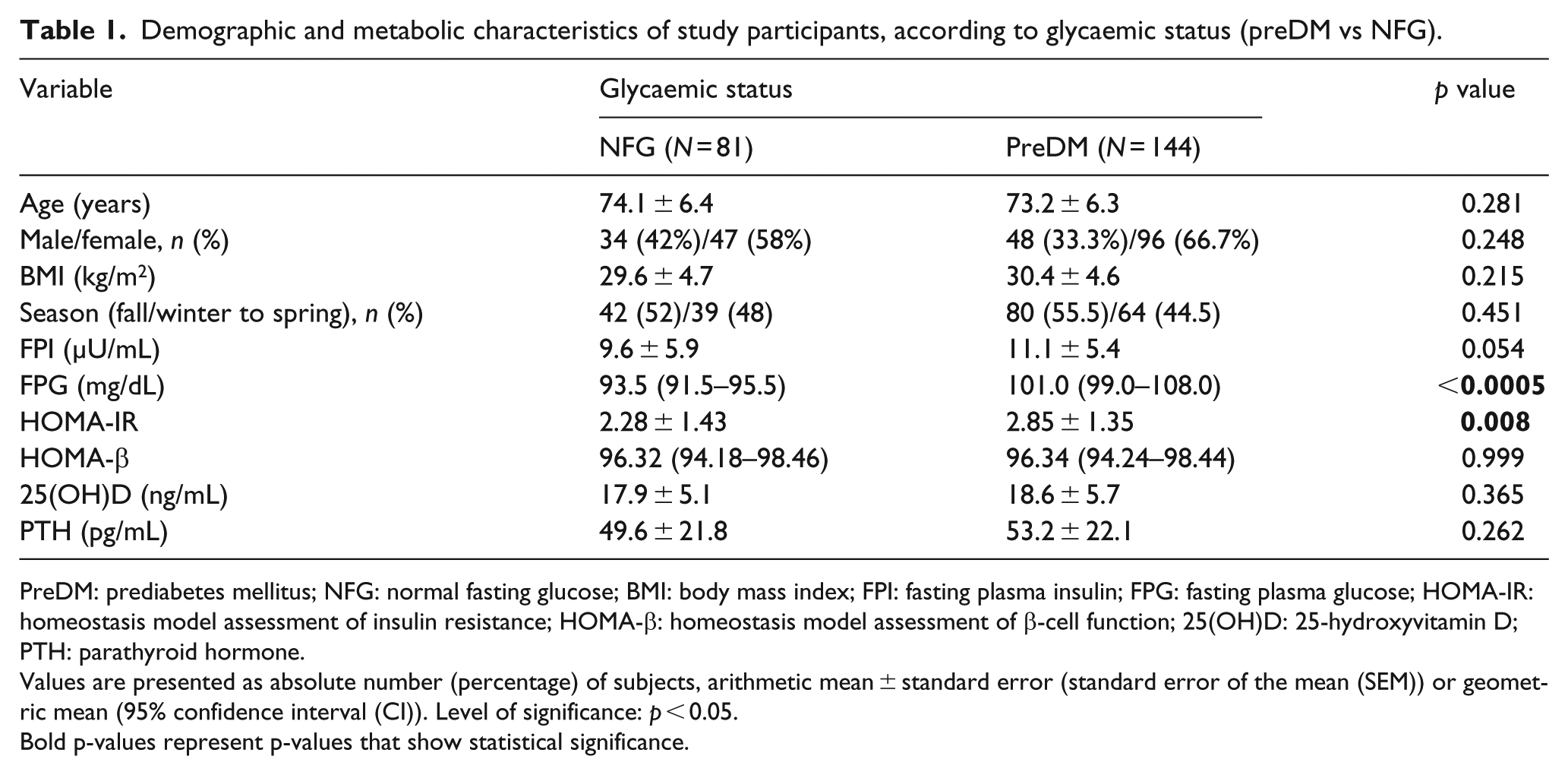
PreDM: prediabetes mellitus; NFG: normal fasting glucose; BMI: body mass index; FPI: fasting plasma insulin; FPG: fasting plasma glucose; HOMA-IR: homeostasis model assessment of insulin resistance; HOMA-β: homeostasis model assessment of β-cell function; 25(OH)D: 25-hydroxyvitamin D; PTH: parathyroid hormone.
Values are presented as absolute number (percentage) of subjects, arithmetic mean ± standard error (standard error of the mean (SEM)) or geometric mean (95% confidence interval (CI)). Level of significance: p < 0.05.
Bold p-values represent p-values that show statistical significance.
Vit D deficiency was prevalent in both groups. A total of 82 participants (57%) in the preDM group and 56 in NFG group (69%) were 25(OH)D deficient (p = 0.451). Participants, with vit D deficiency, did not differ according to FPG and FPI as well as HOMA-IR and HOMA-β, compared with those who were vit D sufficient, for both preDM and NFG groups.
PTH subgroups did not differ in relation to HOMA-IR, HOMA-β and insulin, concentrations, after accounting for the demographic covariates for NFG group. However, in the preDM group, FPG differed significantly across PTH tertiles, increasing from the first to second to third tertile (p = 0.011, across all groups), after adjusting for age, gender, BMI and season of sampling. No differences in parameters of glycaemic homeostasis were observed among other subgroups of PTH tertiles.
There was an increasing trend for both FPI and FPG according to PTH tertiles, which resulted in significantly higher concentrations in participants classified as vit D deficiency/PTH third tertile compared to all other groups (p = 0.031 and 0.027, respectively). Participants with vit D sufficiency/PTH third tertile demonstrated increased FPI concentrations (p = 0.015), compared with those with vit D sufficiency/PTH first to second tertile, after adjustment for age, gender, BMI and season of sampling. Participants with vit D sufficiency/PTH third tertile had increased FPG compared with those with vit D sufficiency/PTH first to second tertile (p = 0.024) and those with vit D deficiency/PTH first to second tertile (p = 0.018). HOMA-IR was significantly higher in preDM group in the vit D deficiency/PTH third tertile (p = 0.039) compared to all other groups. These results were also evident (p = 0.038) for participants with vit D sufficiency/PTH third tertile, compared with participants with vit D sufficiency/PTH first to second tertile. No statistical differences for HOMA-β were observed among groups.
Discussion
The results of this study indicate that PTH status plays an important role in glucose homeostasis in elderly patients with preDM and vit D deficiency. PTH concentrations in the third tertile were associated with insulin resistance, especially when vit D deficiency co-existed. In addition, PTH in combination with vit D may affect the glucose regulation in elderly people with preDM, as vit D deficiency/PTH third tertile subgroup had higher FPG compared with the all other groups. Vit D sufficiency/PTH third tertile subgroup demonstrated increased FPG, FPI and HOMA-IR, compared with those with vit D sufficiency/PTH first to second tertile, considered as the reference population group. This finding indicates that PTH plays a role in the development of insulin resistance and glucose dyshomeostasis in preDM, independently of vit D status.
The pathogenic mechanism between vit D/PTH and glucose intolerance is not yet clarified. It has been suggested that PTH acts directly on β-cell or indirectly through augmentation of extracellular calcium intake and increase of peripheral insulin resistance. 9 PTH increases cytosolic calcium in pancreatic cells and induces adipocyte tissue lipolysis in a dose-dependent manner through protein kinase A phosphorylation of the hormone-sensitive lipase (HLA), results in insulin resistance. 10 Vit D status has been also hypothesized to be involved in the regulation of β-cell function. Still, a recent meta-analysis of 10 randomized control trials with vit D supplementation in patients with preDM did not show any effect on insulin resistance (HOMA-IR). 11
Although previous studies have confirmed that vit D and PTH interact through a tightly regulated negative feedback network, this is the first study to examine the combined effect of vit D/PTH on glucose homeostasis in preDM. Its main limitation is the small study sample and its cross-sectional design, which cannot establish a causative relationship.
Conclusively, this study indicates that PTH status could independently affect glucose homeostasis in elderly patients with preDM. Assessment of both 25(OH)D and PTH concentrations is a more reliable index of glucose homeostasis than either marker alone. Vit D deficiency with PTH concentrations ⩾40 pg/mL exerts the worst dysglycaemic effects in patients with preDM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
