Abstract
Objective:
To investigate the relationship between 1-h post-load plasma glucose, measured during an oral glucose tolerance test, and arterial stiffness, determined by brachial–ankle pulse-wave velocity, in normotensive subjects with normal glucose tolerance.
Methods:
Study subjects were non-industrial workers aged 25–55 years (n = 8381) who underwent a regular health check-up every 5 years. We included only normotensive subjects with normal glucose tolerance based on the American Diabetes Association criteria. Subjects taking medication and having an abnormal ankle–brachial index (⩽1.0 or ⩾1.3) were excluded. The final sample comprised 4970 participants (mean age: 38.8 ± 9.4 years; women: n = 2048).
Results:
1-h post-load plasma glucose correlated with brachial–ankle pulse-wave velocity in men (β = 0.04, p = 0.01), but not women (β = –0.03, p = 0.13) in multivariate linear regression analysis. We found a significant interaction between 1-h post-load plasma glucose and age in men (p = 0.04); therefore, a subgroup analysis was performed in each 5-year age group. The correlation between 1-h post-load plasma glucose and brachial–ankle pulse-wave velocity was significant in the 55-year-old age group (β = 0.12, p = 0.01) and neared significant in 45-year-old (β = 0.08, p = 0.07) and 50-year-old (β = 0.09, p = 0.07) age groups.
Conclusion:
Elevated 1-h post-load plasma glucose levels were associated with arterial stiffness in normotensive, middle-aged men with normal glucose tolerance.
Keywords
Introduction
Diabetes mellitus is an established risk factor for cardiovascular events and mortality. 1 Accumulating evidence suggests that arterial stiffness is one of the key mechanisms in the pathologies. 2 Importantly, the progress of arterial stiffness occurs even before the development of overt diabetes; it is seen in subjects with impaired glucose tolerance (IGT)3–5 who are reportedly at high risk of cardiovascular disease and mortality. 6
Brachial–ankle pulse-wave velocity (baPWV), a recently established marker of arterial stiffness, 7 has been reported as a surrogate end-point for cardiovascular events and all-cause mortality.8,9 A major advantage of baPWV is that the result obtained is operator independent. As well as other markers of arterial stiffness,3–5 a higher baPWV was observed in IGT subjects than in normal glucose tolerance (NGT) subjects.10,11
NGT is considered a low cardiovascular risk state; however, a cut-off point of 155 mg/dL for the 1-h post-load plasma glucose (1h-PG) during an oral glucose tolerance test (OGTT) in NGT subjects predicts type 2 diabetes 12 and mortality. 13 Although the cause of mortality is unclear, a 1h-PG ⩾ 155 mg/dL is associated with subclinical cardiovascular organ damage such as carotid atherosclerosis and left ventricular hypertrophy.14,15 With respect to arterial stiffness, Sciacqua et al. 16 reported that NGT subjects with 1h-PG level ⩾155 mg/dL had higher carotid–femoral PWV values than did those with a 1h-PG < 155 mg/dL. However, their study included only hypertensive subjects, although blood pressure (BP) may fluctuate PWV values because PWV is strongly influenced by BP levels.17,18 Niijima et al. 19 reported that 1h-PG positively correlated with baPWV in normotensive subjects with NGT. However, potential confounders, such as age and sex, 20 were not considered.
Therefore, the aim of this study was to investigate the association between 1h-PG value and arterial stiffness, evaluated by baPWV, in non-hypertensive subjects with NGT.
Patients and methods
Study population
The study subjects were 25- to 55-year-old non-industrial workers in main and branch offices in Japan. A total of 8381 employees underwent a 5-year health check-up at the Chiba Foundation for Health Promotion & Disease Prevention between April 2011 and March 2016. We included subjects with NGT per the American Diabetes Association (ADA) criteria, 21 namely, a fasting plasma glucose (FPG) concentration <100 mg/dL, a 2-h post-load plasma glucose (2h-PG) concentrations of <140 mg/dL and an HbA1c level <5.7%. We excluded subjects with hypertension, those taking medications and those with an abnormal ankle–brachial index (ABI) (⩽1.0 or ⩾1.3). Hypertension was defined as a systolic blood pressure (SBP) ⩾140 mmHg or diastolic BP (DBP) ⩾90 mmHg. The final sample comprised 4970 participants (2048 women and 2922 men). The institutional ethics committee approved the study protocol. Written informed consent was obtained from each subject.
Measurements
The brachial–ankle PWV in subjects at rest for ⩾5 min in a supine position was measured using a commercially available device (Form PWV/ABI; Omron Colin, Tokyo, Japan) that provides simultaneous measurements of heart rate, bilateral brachial and ankle BPs and ABI: the methodology has been described previously. 7 The baPWV was calculated as the mean of the bilateral baPWV values. The ABI was calculated as the ratio of the ankle SBP to the brachial SBP.
Venous blood samples were collected from all participants after an overnight fast (0 min) and 60 and 120 min after a 75 g glucose load. HbA1c was measured using high-performance liquid chromatography (HPLC; ADAMS A1C HA-8181; Arkray, Kyoto, Japan) per the standardized method. Plasma glucose was measured using the glucose oxidase/hydrogen peroxide electrode method (ADAMS glucose GA-1171 system; Arkray, Kyoto, Japan). The levels of total cholesterol, triglyceride, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, creatinine, and uric acid were determined enzymatically using an autoanalyzer (Type 7700; Hitachi Ltd., Tokyo, Japan). The estimated glomerular filtration rate was calculated according to the Modification of Diet in Renal Disease Study equation. Body mass index (BMI) was calculated as weight (kg) divided by the square of the height (m).
Statistical analysis
All data were analysed using STATA 14.2 software (StataCorp LP, College Station, TX, USA). All continuous values are expressed as mean ± standard deviation (SD), and categorical variables are reported as percentages. Normally distributed continuous variables were assessed by t-test, non-normally distributed continuous variables by Mann–Whitney U test. Pearson’s chi-square test was used to analyse categorical variables. Univariate and multivariate linear regression analyses were used to assess correlations between baPWV and other variables (age, BMI, SBP, heart rate, Haemoglobin A1C, FPG, 1h-PG, 2h-PG, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglyceride, estimated glomerular filtration rate, uric acid and current smoking status). A variance inflation factor was assessed to identify multicollinearity among explanatory variables. Product interaction terms were built to test interactions between 5-year age groups and 1h-PG levels in relation to baPWV. To assess baPWV values according to a cut-off point of 155 mg/dL for 1h-PG, adjustment for the same confounders as in the linear regression analysis was made in analysis of covariance (ANCOVA). All p values were two-tailed; p values less than 0.05 was considered statistically significant.
Results
The baseline clinical characteristics of the study population are shown in Table 1. The mean ages of the male and female subjects were 39.3 ± 9.6 years and 38.0 ± 9.1 years, respectively. Metabolic profiles, such as lipid, uric acid and glucose profiles, including 1h-PG (1h-PG: 115.3 ± 33.5 mg/dL in men vs 107.5 ± 31.3 mg/dL in women, p < 0.001), were slightly impaired in men compared with women. Of 2922 male subjects, 384 had 1h-PG ⩾ 155 mg/dL, and of 2048 female subjects, 155 had 1h-PG ⩾ 155 mg/dL (13.1% vs 7.6%, p < 0.001). The baPWV value of male subjects was significantly higher than that of female subjects (1244 ± 152 cm/s vs 1129 ± 156 cm/s, p < 0.001).
Baseline clinical characteristics of the study population.
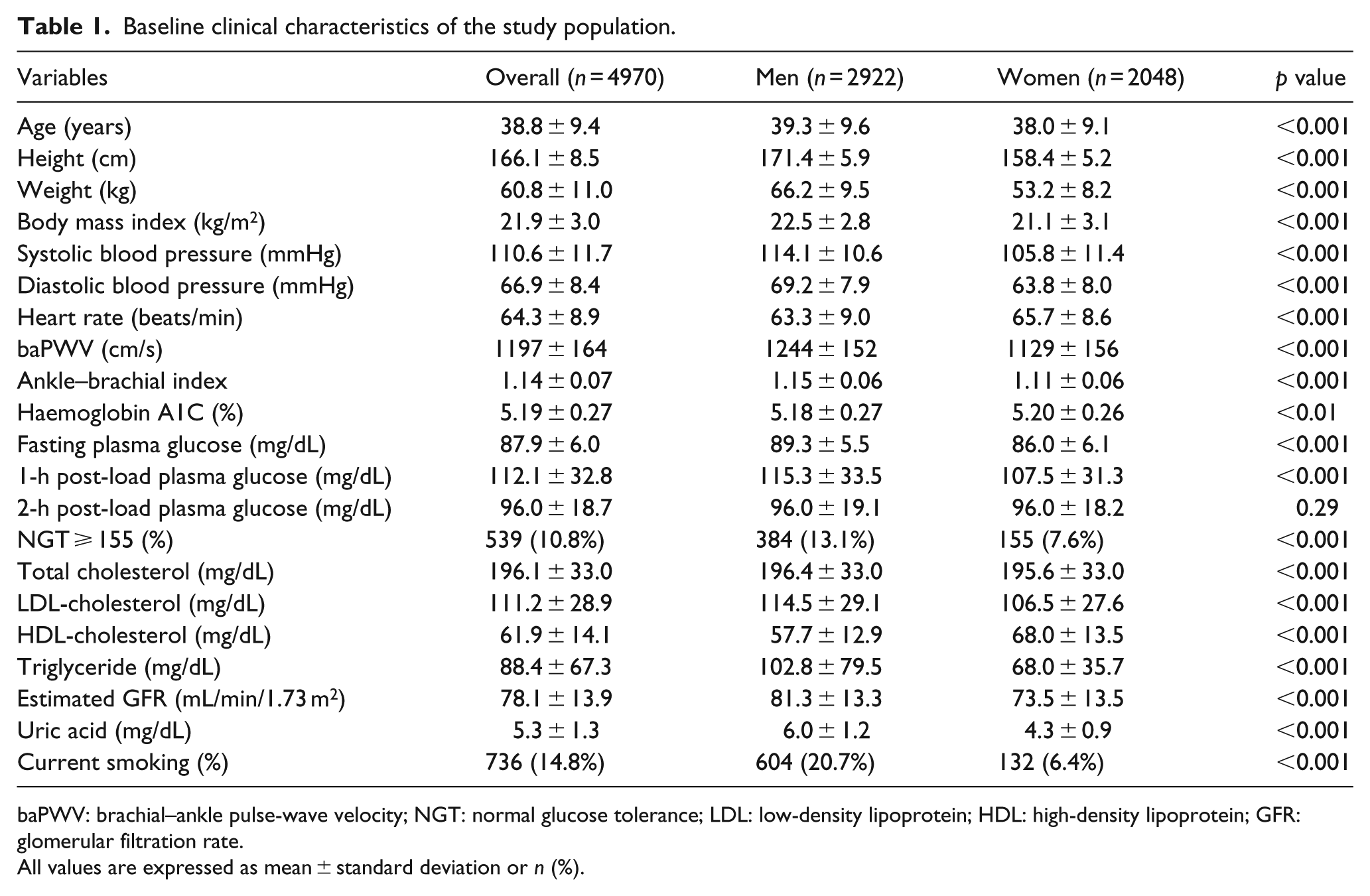
baPWV: brachial–ankle pulse-wave velocity; NGT: normal glucose tolerance; LDL: low-density lipoprotein; HDL: high-density lipoprotein; GFR: glomerular filtration rate.
All values are expressed as mean ± standard deviation or n (%).
Correlation between baPWV and 1h-PG
The 1h-PG value significantly correlated with baPWV in men, but not in women (men: r = 0.18, p < 0.001, women: r = 0.02, p = 0.30, as shown in Figure 1). In the univariate analysis, the FPG and 2h-PG values also significantly correlated with baPWV in men (FPG: r = 0.16, p < 0.001; 2h-PG: r = 0.13, p < 0.001), but multivariate analysis revealed that the relationship remained significant only for 1h-PG (Table 2). The FPG and 2h-PG significantly correlated with baPWV in the univariate analysis in women (FPG: r = 0.20, p < 0.001; 2h-PG: r = 0.07, p < 0.01). Multivariate analysis revealed FPG, but not 1h-PG or 2h-PG, significantly correlated with baPWV (Table 2).
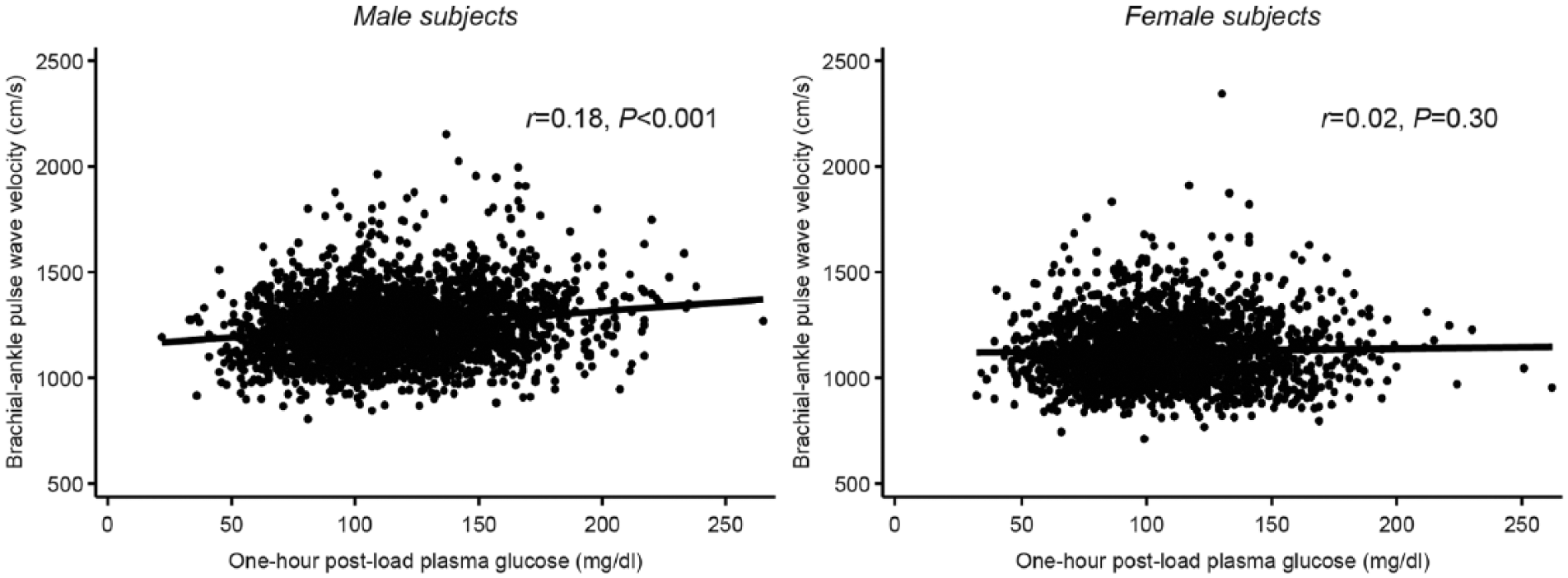
Correlation between brachial–ankle pulse-wave velocity and 1-h post-load plasma glucose during an oral glucose tolerance test in male and female subjects.
Multiple linear regression analysis on baPWV as dependent variable in men and women.
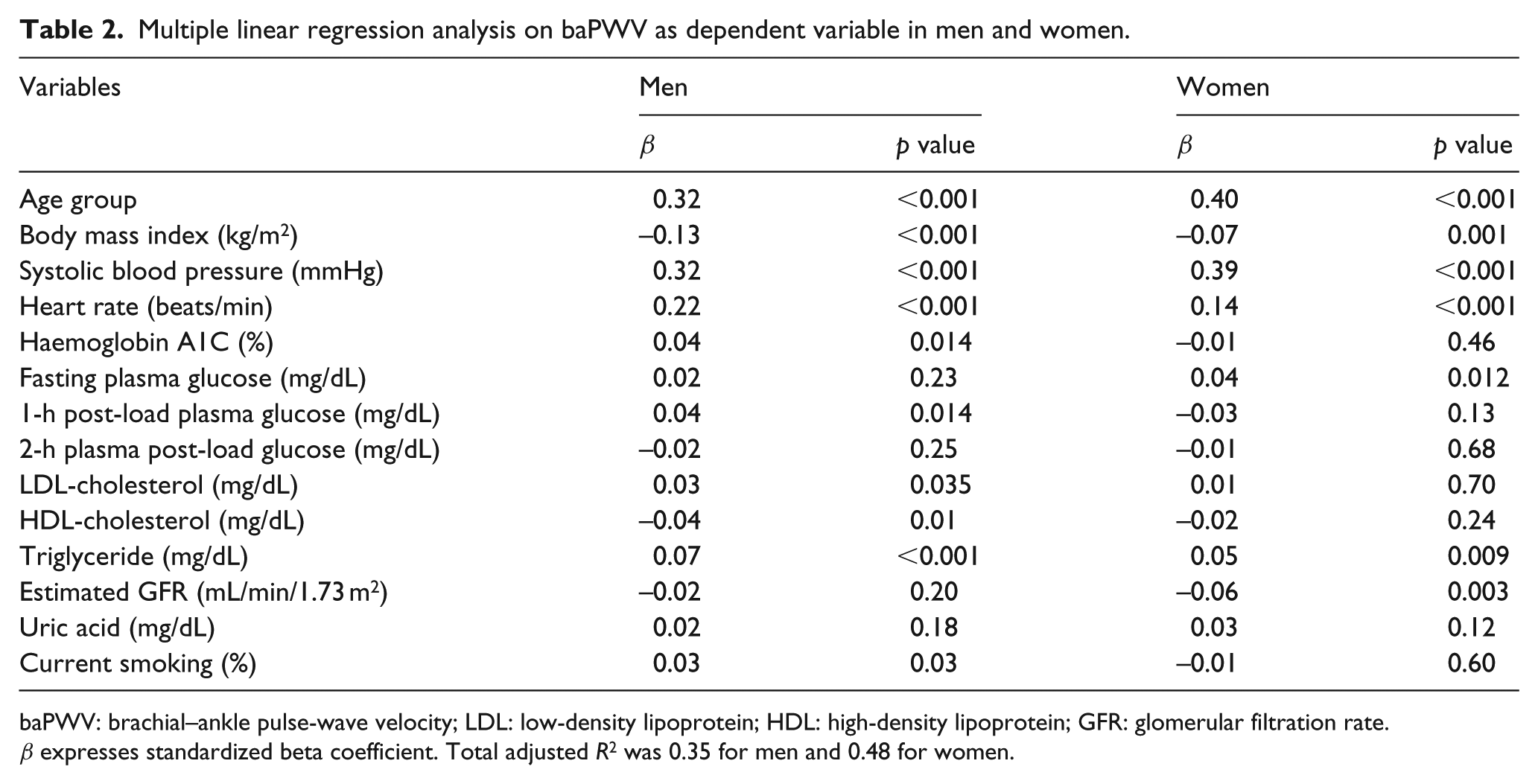
baPWV: brachial–ankle pulse-wave velocity; LDL: low-density lipoprotein; HDL: high-density lipoprotein; GFR: glomerular filtration rate.
β expresses standardized beta coefficient. Total adjusted R2 was 0.35 for men and 0.48 for women.
Because we found a significant interaction between age group and 1h-PG levels in relation to baPWV (p = 0.04) in male subjects, the results were stratified by age groups. The simple correlation between baPWV and 1h-PG level was significant in 45-, 50-, and 55-year-old age groups (Table 3). Multivariate analysis revealed that the correlation was significant in the 55-year-old age group (β = 0.12, p = 0.014), but not in the 45- or 50-year-old age groups (β = 0.08, p = 0.07; β = 0.09, p = 0.07, respectively; Table 3). We performed an additional analysis using wider age groups: 25- to 40-year-old age group (n = 1796) and 45- to 55-year-old age group (n = 1126). The association between baPWV and 1h-PG level was significant in the latter, middle-aged group (β = 0.10, p < 0.001) but not in the younger group (β = 0.03, p = 0.22). No significant interaction was found between age groups and 1h-PG level in relation to baPWV. A significant correlation was found between baPWV and 1h-PG in the 55-year-old age group in women in the univariate analysis (r = 0.18, p = 0.03), but no significant associations were found in any female age group in the multivariate analysis (55-year-old age group: β = –0.03, p = 0.61).
Correlation of 1-h post-load plasma glucose with baPWV as dependent variable in male age subgroups.
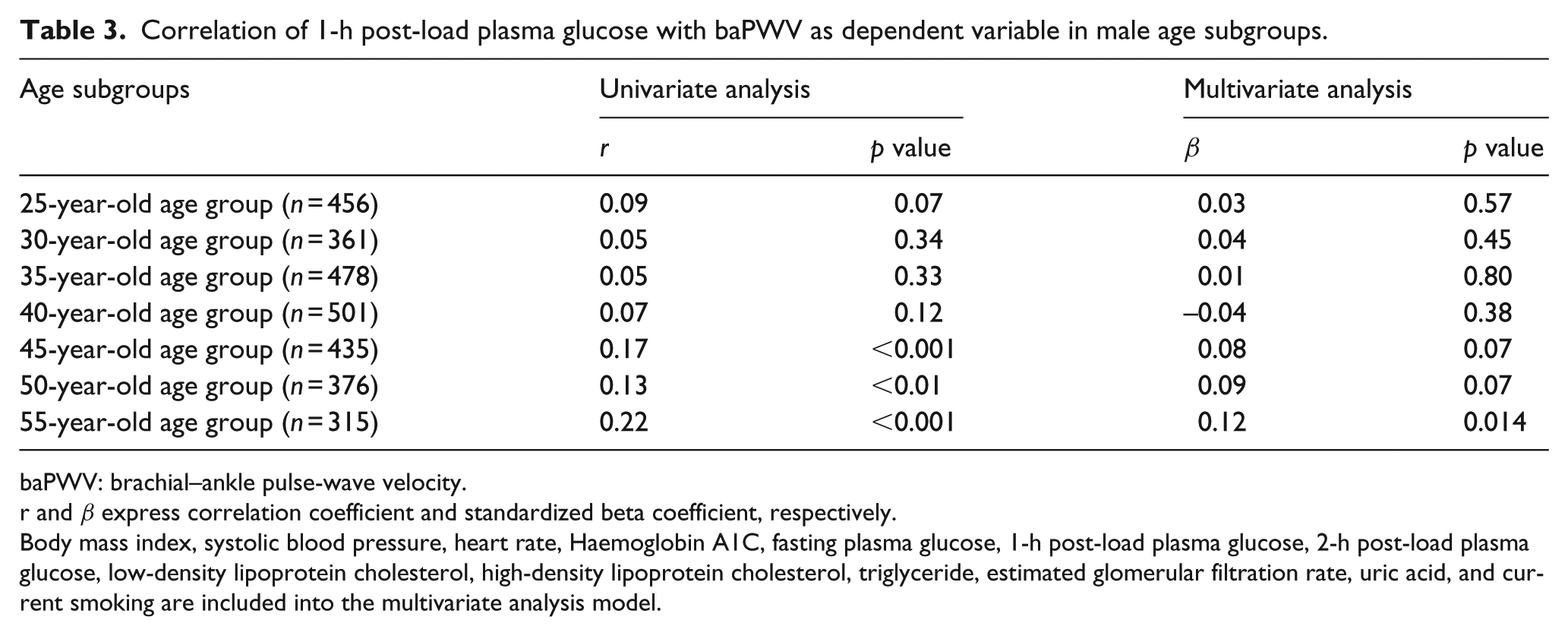
baPWV: brachial–ankle pulse-wave velocity.
r and β express correlation coefficient and standardized beta coefficient, respectively.
Body mass index, systolic blood pressure, heart rate, Haemoglobin A1C, fasting plasma glucose, 1-h post-load plasma glucose, 2-h post-load plasma glucose, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglyceride, estimated glomerular filtration rate, uric acid, and current smoking are included into the multivariate analysis model.
baPWV values according to a cut-off point of 155 mg/dL for 1h-PG
Finally, we examined the difference between the baPWV values of subjects stratified into low and high 1h-PG value groups using an established cut-off point of 155 mg/dL. The prevalence of NGT ⩾ 155 increased with age in male subjects (p < 0.001; Table 4), but not in female subjects. In men, subjects with 1h-PG value ⩾155 mg/dL had higher baPWV values than subjects with 1h-PG < 155 mg/dL in 45-, 50- and 55-year-old age groups (Table 4). After adjustment for confounders, only men with 1h-PG value ⩾155 mg/dL in 55-year-old age groups had significantly higher baPWV value than subjects with 1h-PG value <155 mg/dL (45-year-old: 1257 ± 121 vs 1266 ± 301, p = 0.59; 50-year-old: 1288 ± 149 vs 1305 ± 299, p = 0.33; 55-year-old: 1363 ± 166 vs 1420 ± 354, p = 0.01). Among women with a 1h-PG value ⩾155 mg/dL, there were no significant differences between age groups in terms of baPWV values.
Brachial–ankle PWV values of the study population according to a cut-off point of 155 mg/dL for 1-h post-load plasma glucose in male age subgroups.
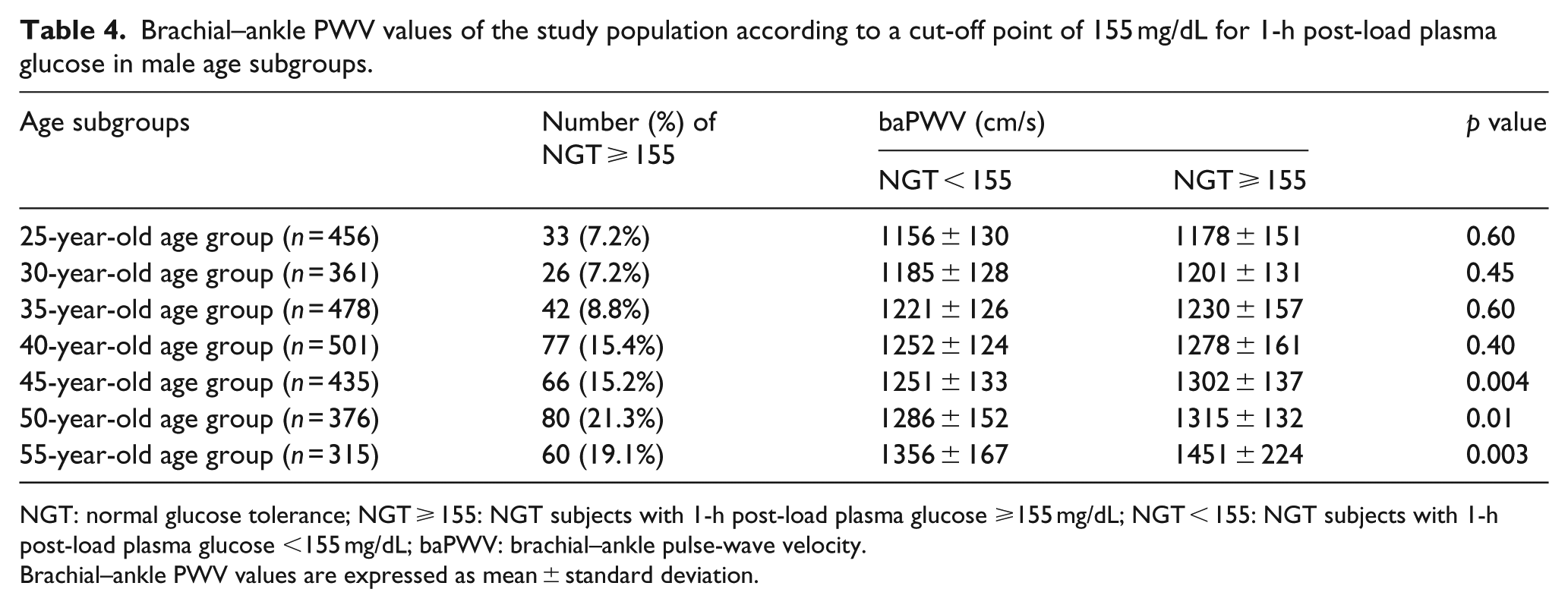
NGT: normal glucose tolerance; NGT ⩾ 155: NGT subjects with 1-h post-load plasma glucose ⩾155 mg/dL; NGT < 155: NGT subjects with 1-h post-load plasma glucose <155 mg/dL; baPWV: brachial–ankle pulse-wave velocity.
Brachial–ankle PWV values are expressed as mean ± standard deviation.
Discussion
In this study, we found that 1h-PG levels correlated with baPWV independent of conventional risk factors in men, but not in women. Moreover, we found a significant interaction between 1h-PG level and age in relation to baPWV; the correlation between baPWV and 1h-PG level was significant only in the 55-year-old age group and neared significance in the 45- and 50-year-old age groups in men. The additional analysis performed in wider age groups also showed that the association was significant only in the 45- to 55-year-old age group, but not in the 25- to 40-year-old age group. In the 55-year-old male group, the baPWV values of NGT ⩾ 155 subjects were significantly higher than those of NGT < 155 subjects.
Glucose metabolism impairment is associated with progression of arterial stiffness.3–5 Sciacqua et al. 16 reported the relationship between arterial stiffness and 1h-PG value in NGT subjects. However, that study included only hypertensive subjects. This is important as PWV measurements are strongly influenced by BP.17,18 With regard to normotensive and healthy NGT subjects, a simple correlation between baPWV and 1h-PG level was reported, 19 but this correlation was not adjusted for possible confounders, specifically age and sex, although baPWV and glucose metabolism are closely related to these two factors.20,22,23 We eliminated the effects of age and sex on the relation between baPWV and 1h-PG value by stratifying subjects by age (5-year age groups) and sex. Therefore, the most relevant finding of this study was the robust association between arterial stiffness and early impairment of glucose metabolism determined by 1h-PG in normotensive NGT subjects. Although NGT subjects are considered to have very low cardiovascular risk, vascular damage may occur from a very early stage of glucose metabolism impairment, earlier than is traditionally thought.
It is noteworthy that age and sex modify the relationship between arterial stiffness and 1h-PG level. An explanation for this modification is that subjects with moderately stiffened arteries are more susceptible to postprandial hyperglycaemia–related vascular damage perhaps because of the effect of other factors enhancing arterial stiffness. For middle-aged male subjects, in addition to ageing, other factors enhancing arterial stiffness such as slightly high BP, worse lipid profiles and high smoking frequency compared with women might make arteries more susceptible to postprandial hyperglycaemia, resulting in the significant association between baPWV and 1h-PG level observed in this study. In female subjects, a beneficial effect of oestrogen on atherosclerosis and vessel wall properties 24 might have a substantial influence on our results because we included young and middle-aged subjects (⩽55 years of age). Female subjects in this study might have been resistant to postprandial hyperglycaemia because of potentially soft arteries or direct protective effects of oestrogen. If this hypothesis holds, there should be a significant association between arterial stiffness and 1h-PG level in aged post-menopausal women as they have increased arterial stiffness due to the augmented, age-related increase in arterial stiffness. 25 Tsuboi et al. 26 reported that 1-h postprandial plasma glucose levels determined after a 600-kcal Japanese meal are associated with the cardio-ankle vascular index in women aged 50–85 years, but not in women <50 years of age. Although postprandial plasma glucose levels must be distinguished from post-load plasma glucose levels, 27 we speculate that these data support our hypothesis.
Heterogeneity among NGT subjects with elevated 1h-PG among groups may be another explanation for the observed age and sex interactions. For example, the degree of glucose metabolism impairment is not homogeneous even among NGT ⩾ 155 subjects. Several studies have indicated that insulin resistance is a common pathology in 1h-PG elevation, but some individuals appear to have impaired insulin secretion,28,29 whereas others have increased insulin secretion. 30 In the early phase of glucose impairment, such as elevated 1h-PG values in young subjects, insulin resistance may occur, but the insulin secretion function may remain preserved. Unfortunately, we could not examine insulin levels, because the health check-up programme did not include insulin measurement: this is a limitation of this study. Detailed assessment based on metabolic profiles, including insulin concentration, may be helpful to assess the cardiovascular risk of subjects with high 1h-PG values.
Our study has potential limitations. First, the cross-sectional nature is a major limitation of our study. Our findings might be due to reverse causation mechanism: reduced perfusion of insulin-sensitive tissues might impair glucose disposal resulting in high 1h-PG. Second, as mentioned above, insulin level was not measured in this study. Third, although the large sample size, comprising both sexes and young to middle-aged subjects, was a major strength of this study, the association that approached (but did not reach) significance in the 45- and 50-year age groups might be due to the limited number of subjects in each of these groups. Fourth, we did not include older aged subjects. Finally, our findings may be applicable only to Asians and it remains unknown whether this can be extended to other ethnic groups.
In conclusion, we investigated the association between 1h-PG level and baPWV values in normotensive subjects with NGT. Although the risk of cardiovascular disease is traditionally considered to be very low in such individuals, we found increased arterial stiffness, associated with elevated 1h-PG values, in middle-aged men. However, the relation was not found in young men or young and middle-aged women. A prospective study is required to determine whether a true sex difference exists and to determine the threshold effect of age on 1h-PG level and its relationship with arterial stiffness and cardiovascular disease.
Footnotes
Declaration of conflicting interests
S.O. received lecture fees from Otsuka Pharmaceutical (Tokyo, Japan) and MSD (Tokyo, Japan). S.O. received a research grant from Kashiwado Memorial Foundation (Chiba, Japan). Y.K. received lecture fees from Daiichi-Sankyo (Tokyo, Japan), Takeda Pharmaceutical (Osaka, Japan), Bayer Yakuhin (Osaka, Japan) and Boehringer Ingelheim (Ingelheim, Germany). Y.K. received research grants from Boehringer Ingelheim (Ingelheim, Germany), Pfizer (New York, USA), Otsuka Pharmaceutical (Tokyo, Japan), Takeda Pharmaceutical (Osaka, Japan), Mitsubishi Tanabe Pharma (Osaka, Japan), Sumitomo Dainippon Pharma (Osaka, Japan), Astellas Pharma (Tokyo, Japan), St. Jude Medical (St. Paul, USA), Abbott Vascular Japan (Tokyo, Japan) and Daiichi-Sankyo (Tokyo, Japan). The other authors have no conflicts to report.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
