Abstract
Background:
Women with a history of gestational diabetes mellitus and preeclampsia are at increased risk of cardiovascular disease later in life, but the mechanism remains unclear. The aim of the study was to evaluate the association between CXC chemokine ligand 16 and indices of glucose metabolism, dyslipidemia and systemic inflammation in gestational diabetes mellitus and preeclampsia.
Methods:
This sub-study of the population-based prospective cohort included 310 women. Oral glucose tolerance test was performed during pregnancy and 5 years later along with lipid analysis. CXC chemokine ligand 16 was measured in plasma (protein) and peripheral blood mononuclear cells (messenger RNA) during pregnancy and at follow-up.
Results:
Circulating CXC chemokine ligand 16 was higher in gestational diabetes mellitus women early in pregnancy and at follow-up, while higher in preeclampsia women late in pregnancy compared to control women. Messenger RNA of CXC chemokine ligand 16 in peripheral blood mononuclear cells were lower in gestational diabetes mellitus and preeclampsia women compared to control women. Increased circulating CXC chemokine ligand 16 level was associated with a higher apolipoprotein B and low-density lipoprotein cholesterol in gestational diabetes mellitus women but not in normal pregnancy at follow-up.
Conclusion:
Our study shows that women with gestational diabetes mellitus and preeclampsia had a dysregulated CXC chemokine ligand 16 during pregnancy, and in gestational diabetes mellitus, the increase in CXC chemokine ligand 16 early in pregnancy and after 5 years was strongly associated with their lipid profile.
Introduction
Women with a history of gestational diabetes mellitus (GDM) and preeclampsia (PE) are at increased risk of cardiovascular disease (CVD) later in life, potentially involving inflammatory mechanisms and an atherogenic lipid profile,1–3 although the precise mechanisms are not well understood. Influx, retention and oxidation of apolipoprotein B (apoB) containing lipoproteins in the artery wall (mainly oxidized low-density lipoprotein [oxLDL]), which again promotes foam cells, facilitated by scavenger receptors on the surface of activated macrophages, are considered initial events that enhance the development of atherosclerosis.
The CXC chemokine ligand 16 (CXCL16) is a multifunctional protein that may contribute to atherosclerosis through several mechanisms including its function as a scavenger receptor and an adhesion molecule. 4 It is expressed and released as a membrane-bound and soluble form, respectively, by numerous cell types such as macrophages, B and T cells, dendritic cells, epithelial cells, fibroblasts, platelets, vascular smooth muscle cells and endothelial cells. 5 Membrane-bound CXCL16 promotes adhesion of T cells expressing its cognate receptor, CXCR6, to the endothelium, and also binds and internalizes oxLDL. 4 Cleavage of membrane-bound CXCL16 results in the release of soluble CXCL16, which act as a chemoattractant for CXCR6 cells, promoting cell migration to sites of inflammation 4 and matrix degradation through induction of matrix metalloproteinases (MMPs). 6 Enhanced expression of CXCL16 and CXCR6 has been found in atherosclerotic lesions in humans 7 and in apolipoprotein E (apoE) deficient mice. 8 Increased circulating CXCL16 is associated with disease severity in acute coronary syndrome and stroke9–11 and may provide information on CVD risk in the general population.12,13
To our knowledge, CXCL16 has not been prospectively evaluated during pregnancy. Based on the multifunctional role of CXCL16 in atherosclerosis acting both as a scavenger receptor and inflammatory mediator, we hypothesized that women with GDM and PE would be characterized by increased circulating CXCL16 levels. We measured plasma protein and peripheral blood mononuclear cells (PBMC) messenger RNA (mRNA) expression of CXCL16 in 310 women from a prospective cohort study at multiple times during pregnancy and at 5-year follow-up to compare CXCL16 between GDM, PE and control women to determine the association between CXCL16 and indices of glucose metabolism, dyslipidemia and systemic inflammation as reflected by C-reactive protein (CRP) levels.
Materials and methods
Study population
The STORK study (Large babies and complications) was a prospective cohort study with a longitudinal design in which 1031 women of Scandinavian ethnicity who gave birth at Oslo University Hospital Rikshospitalet between 2002 and 2008 were followed throughout their pregnancy as previously described. 14 Briefly, each pregnant woman had four antenatal visits at gestational age weeks 14–16, 22–24, 30–32 and 36–38. All 1031 participants from the STORK study got an invitation letter. Exclusion criteria were pregnancy, and last birth had to be at least 1 year ago. A total of 300 women agreed to participate in the follow-up study 5 years after the index pregnancy. 15 This study includes subjects from the follow-up study (including 10 PE), in addition to 28 PE. Women with preterm birth and hypertension without GDM were excluded in this particular study.
Biochemical analysis
Peripheral venous blood was drawn into tubes with ethylenediaminetetraacetic acid (EDTA) additives centrifuged for 25 min at 3000 g at 4 C, separated, and stored at −80 C until analysed. Fasting plasma levels of CXCL16 and high-sensitivity C-reactive protein (hsCRP) (previously published 16 ) were measured in duplicate by enzyme immunoassay with antibodies obtained from R&D Systems (Minneapolis, MN, USA). The intra- and inter-individual coefficients of variation (CV) for these assays in our hands are 3.9% and 9.0% for CXCL16 and 2.6% and 9.1% for hsCRP. All 75-g oral glucose tolerance tests (OGTTs) were performed in the morning after an overnight fast and glucose measured as previously reported. 15 Levels of apolipoprotein A (apoA), apoB, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C) (direct measurements) and triglycerides (TG) were measured from frozen serum samples at follow-up at the clinical chemistry laboratory at Oslo University Hospital Rikshospitalet as previously reported.16,17 LDL-C at weeks 14–16 in pregnancy was determined by Friedewald’s formula, as previously reported in a subset of the women. 18
Diagnosis of GDM and PE
GDM was diagnosed on a 75-g OGTT using the World Health Organization (WHO) criteria from 1999: 2-h plasma glucose ≥ 7.8 mmol/L. 19 Insulin sensitivity, insulin resistance and β-cell function was calculated as previously reported. 15 PE was diagnosed by new-onset blood pressure (BP) ≥140/90 mm Hg and significant proteinuria (urinary total protein/creatinine ratio >30 or +1 on urine dipstick). All cases were diagnosed after 34 weeks of gestation (late-onset PE).
Collection, storage and RNA extraction of PBMC
PBMC was isolated from venous blood using BD Vacutainer CPT Tubes (BD, NJ, USA) at weeks 22–24, 36–38 and at follow-up and stored at −70°C until extraction. RNA was extracted using MagNA Pure Isolation Kit and Instrument (Roche Life Science, Penzberg, Germany) at weeks 22–24 and with MagMAX Isolation Kit and Instrument (Applied Biosystems, Carlsbad, CA, USA) at weeks 36–38 and at follow-up, due to change in instruments at the laboratory over the years.
Quantitative reverse transcription polymerase chain reaction
Reverse transcription was performed using a High-Capacity cDNA Archive Kit (Applied Biosystems, Foster City, CA, USA). mRNA quantification was performed using Power SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA) and the standard curve method on an ABI Prism 7900 (Applied Biosystems). Sequence-specific intron spanning oligonucleotide primers were designed using the Primer Express software version 2.0 (Applied Biosystems) for CXCL16 (accession no. NM_001100812.1, NM_022059.3) TATACTACACGAGGTTCCAGCTCCTT (forward), CAAGACAGCTCATCAATTCCTGAA (reverse) and β-actin (accession no. NM_001101.3) AGGCACCAGGGCGTGAT (forward), TCGTCCCAGTTGGTGACGAT (reverse). Transcript expression levels were normalized to β-actin and expressed as relative mRNA levels.
Statistical analysis
Statistical analyses were conducted using SPSS for Windows, version 21.0 (Chicago, IL, USA). Data are expressed as mean ± standard deviation (SD) when normally distributed and median (25th, 75th percentiles) when skewed. Comparison between women with and without a history of GDM and PE was performed using t-test or Mann–Whitney U depending on distribution and Chi-square test for categorical variables. We performed a stepwise linear regression to identify the strongest predictors at 5 years follow-up of apoB and LDL-C. Univariate and stepwise (probability of F to-enter 0.1 to-remove 0.15) linear regression analyses were carried out on log transformed variables (if skewed) and results given as standardized regression coefficients. Only variables below p < 0.2 were included in the stepwise multivariable models (the variables marked in bold). Interaction analysis was performed including circulating CXCL16 and GDM and their interaction term in a forced univariate model. Two-tailed p values <0.05 were considered significant.
Results
Tables 1 and 2 show the characteristics of the study population during pregnancy and at the time of the 5-year follow-up visit, respectively, stratified into those women who did and did not have GDM or PE in the index pregnancy using WHO diagnostic criteria for GDM. 19 There were only 10 women with PE who participated in the follow-up study, and these were excluded from the extended analysis in the follow-up study to prevent interpretation with a low sample size. There were no difference in the women who later participated in the follow-up study and the women who did not, except for a little older women in the follow-up study [mean (SD) (30.8 (3.9) vs 32.2 (3.8)); Supplemental Table].
Characteristics of the study population of GDM (WHO criteria), preeclampsia and control women during the index pregnancy.
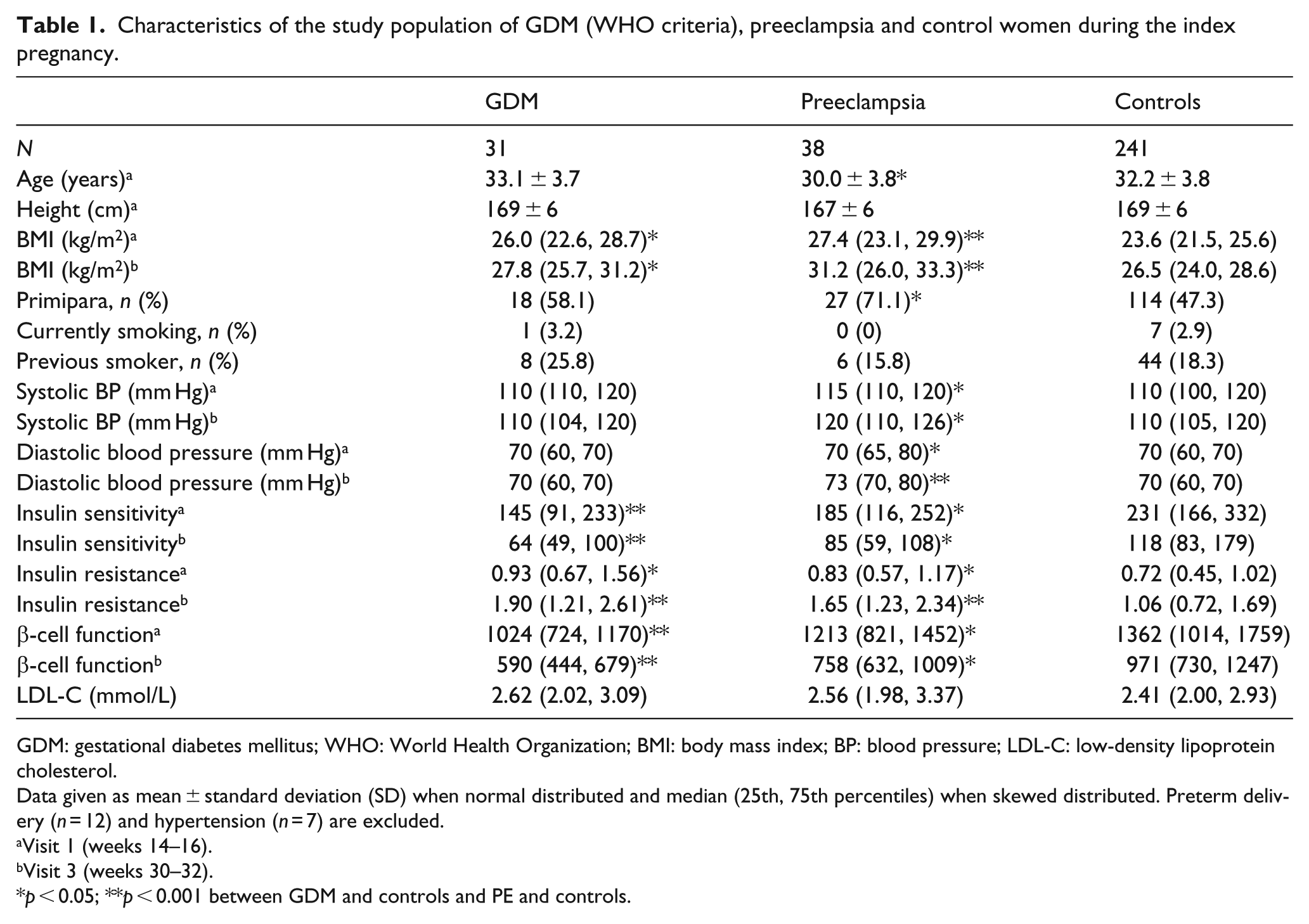
GDM: gestational diabetes mellitus; WHO: World Health Organization; BMI: body mass index; BP: blood pressure; LDL-C: low-density lipoprotein cholesterol.
Data given as mean ± standard deviation (SD) when normal distributed and median (25th, 75th percentiles) when skewed distributed. Preterm delivery (n = 12) and hypertension (n = 7) are excluded.
Visit 1 (weeks 14–16).
Visit 3 (weeks 30–32).
p < 0.05; **p < 0.001 between GDM and controls and PE and controls.
Characteristics of the study population of GDM (WHO criteria), preeclampsia and control women at 5-year follow-up.
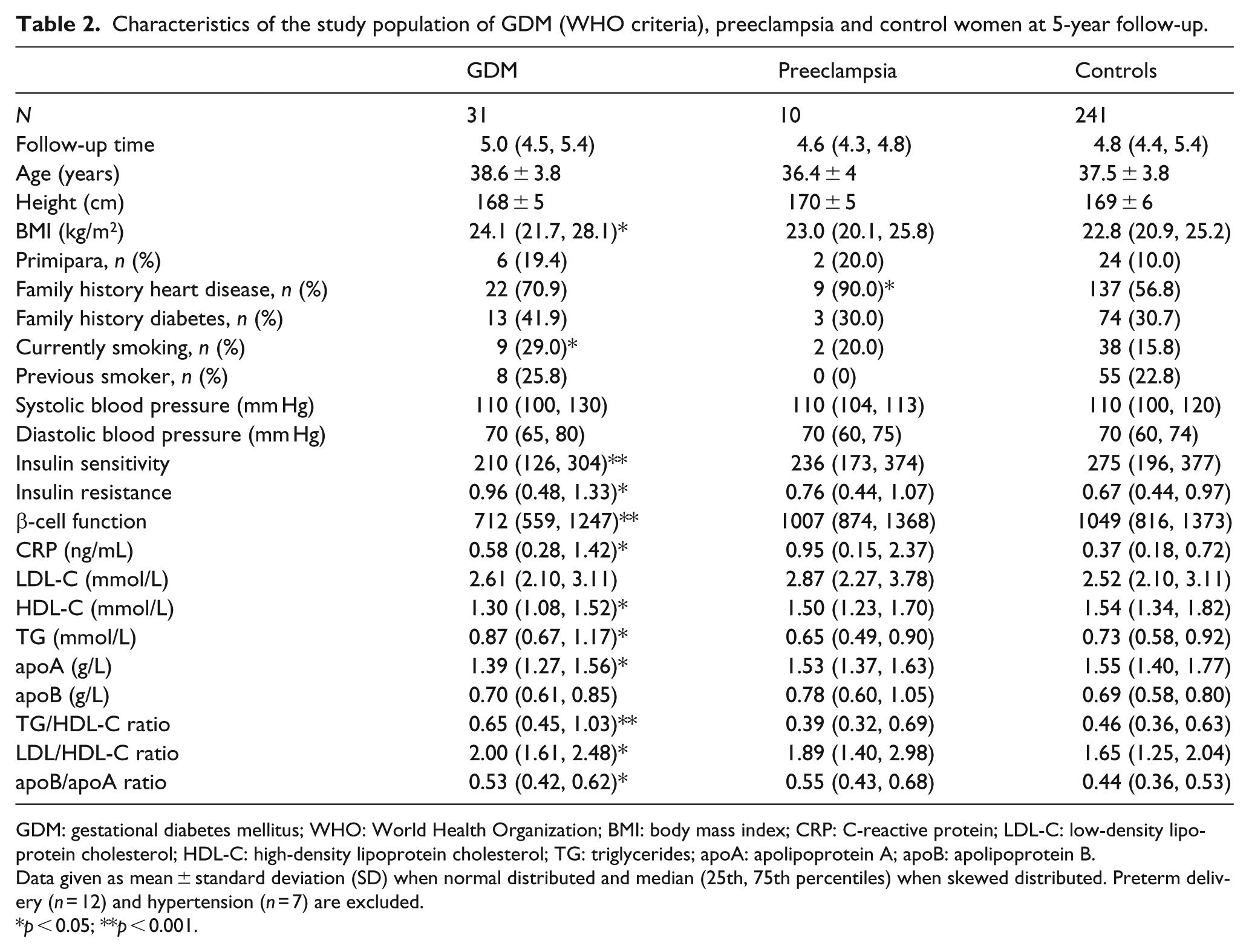
GDM: gestational diabetes mellitus; WHO: World Health Organization; BMI: body mass index; CRP: C-reactive protein; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; TG: triglycerides; apoA: apolipoprotein A; apoB: apolipoprotein B.
Data given as mean ± standard deviation (SD) when normal distributed and median (25th, 75th percentiles) when skewed distributed. Preterm delivery (n = 12) and hypertension (n = 7) are excluded.
p < 0.05; **p < 0.001.
Increased circulating CXCL16 in pregnancy and in women with GDM and PE
CXCL16 showed, in general, increasing levels during pregnancy (p < 0.001, analysis of variance [ANOVA]) and with lower levels at 5 years follow-up (Figure 1(a)). In GDM, CXCL16 was increased at weeks 14–16, 22–24 and at follow-up compared to control women, while in PE women CXCL16 was increased at weeks 22–24, 30–32 and 36–38. When dividing inclusion time to the study in quartiles and assessing CXCL16 levels, we observed higher median CXCL16 levels in samples stored the longest as shown in Supplemental Figure 1, while no differences were observed in the other quartiles. The pattern of CXCL16 throughout pregnancy and at 5 years follow-up remained similar for all storage time groups. Based on the number of individuals in the follow-up study (n = 310) and SDs for all groups (i.e. controls, GDM and PE) at all time points was between 20%–30% of the average CXCL16 levels, we had with a SD of 25% of the average and a frequency of 10% for PE and GDM, a 80% power to detect a 15% difference.
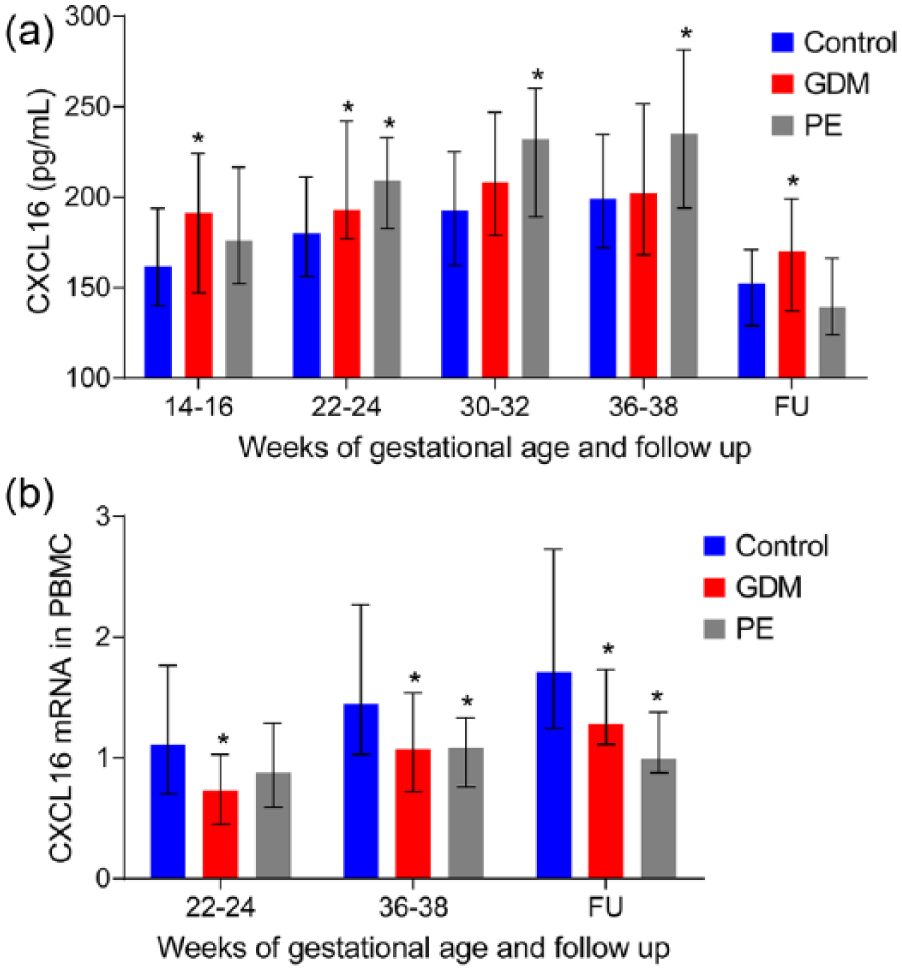
CXCL16: (a) circulating protein levels and (b) mRNA expression from PBMC during pregnancy and 5 years follow-up between Control (blue), GDM (red) and PE (grey) women. Data are given as median and 25th/75th percentile. *p < 0.05, compared with the control group.
Decreased mRNA CXCL16 in PBMC from GDM and PE women
In contrast to plasma levels, PBMC from GDM and PE showed decreased mRNA expression of CXCL16 at weeks 22–24 and 36–38 in pregnancy and at follow-up (GDM) and at weeks 36–38 in pregnancy and at follow-up (PE) (Figure 1(b)).
Few associations between CXCL16 and metabolic risk factors
As shown in Table 3, circulating CXCL16 was positively correlated with systolic BP at weeks 14–16 and 22–24 as well as weeks 30–32 for diastolic BP. No associations between CXCL16 and measures of glucose metabolism were observed during pregnancy except a positive correlation with insulin resistance at 30–32 weeks and a negative correlation between fasting glucose and CXCL16 at 14–16 weeks. However, at 5 years follow-up, CXCL16 was negatively correlated with insulin sensitivity and β-cell function, and positively with insulin resistance (Table 3). However, this association was only present in controls (r = 0.27, p < 0.001) and not present in GDM (r = 0.05, p = 0.84). No associations were found between CXCL16 and body mass index (BMI) or CRP, as a measure of systemic inflammation, at any visits in pregnancy or follow-up. We found no associations between CXCL16 mRNA in PBMC and the same variables.
Associations of circulating CXCL16 and clinical markers.
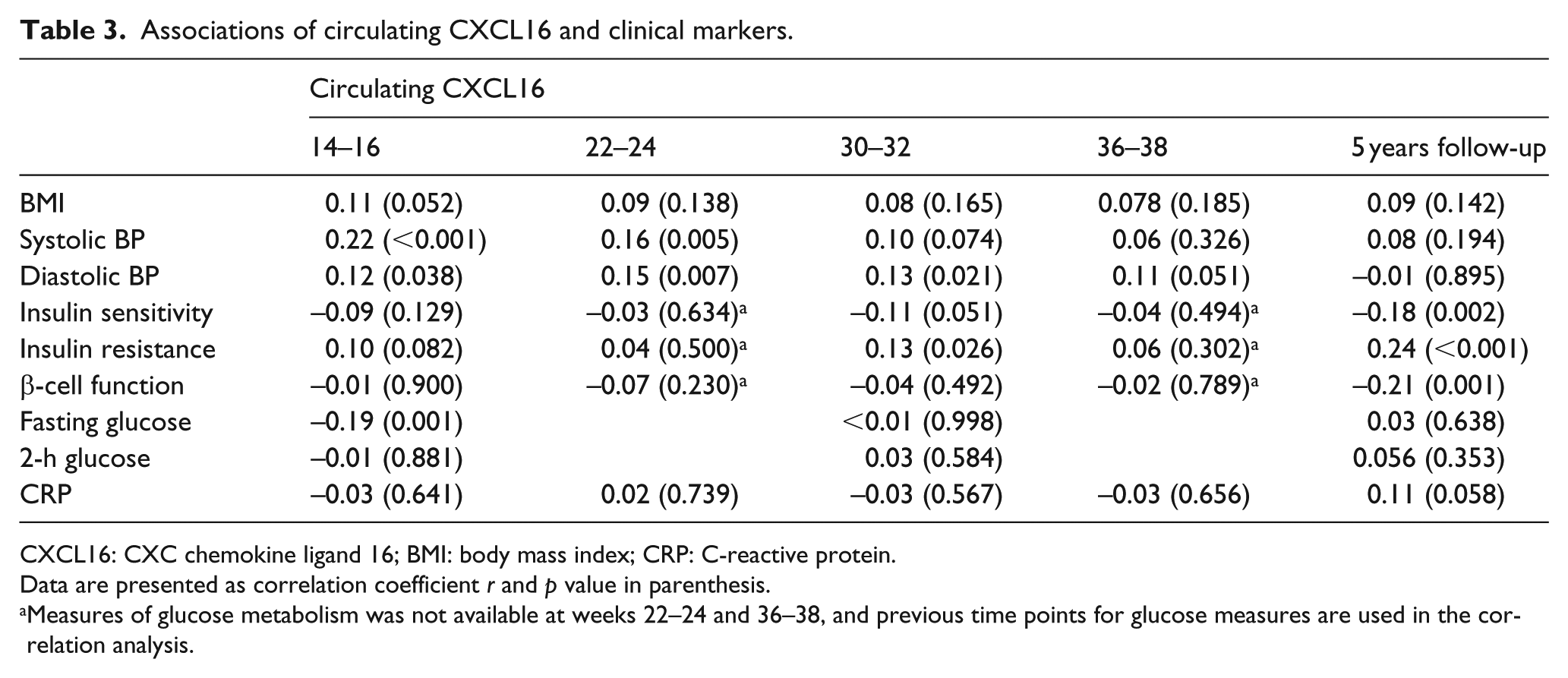
CXCL16: CXC chemokine ligand 16; BMI: body mass index; CRP: C-reactive protein.
Data are presented as correlation coefficient r and p value in parenthesis.
Measures of glucose metabolism was not available at weeks 22–24 and 36–38, and previous time points for glucose measures are used in the correlation analysis.
Associations and interaction between circulating CXCL16 and apoB containing lipoproteins
A modest association was found between circulating CXCL16 and apoB (r = 0.14, p = 0.022), LDL-C (r = 0.12, p = 0.037) and TG (r = 0.13, p = 0.028) in the whole cohort at 5 years follow-up but not between CXCL16 and LDL-C at gestational weeks 14–16 (r = 0.02, p = 0.706). No associations were found between CXCL16 and apoA and HDL-C at 5 years follow-up. When assessing the association between CXCL16 and metabolic parameters in GDM and non-GDM women separately by univariate and multivariate analyses, we found that in GDM women, but not in non-GDM women, CXCL16 was strongly positively correlated with both apoB (r = 0.56, p < 0.001) and LDL-C (r = 0.66, p < 0.001) at 5 years follow-up and with LDL-C at weeks 14–16 during pregnancy (r = 0.47, p = 0.008). As shown in Figure 2(a) and (b), interaction analysis revealed significant interactions between GDM and the CXCL16 at 5-year follow-up for LDL-C (p < 0.001), apoB (p = 0.005) and also for LDL-C at the first visit (14–16 weeks) during pregnancy (p = 0.012). Finally, when evaluated in a multivariable model, CXCL16 remained a significant predictor of both apoB and LDL-C together with indices of dysregulated glucose metabolism at 14–16 weeks of pregnancy and at 5 years follow-up (Table 4).
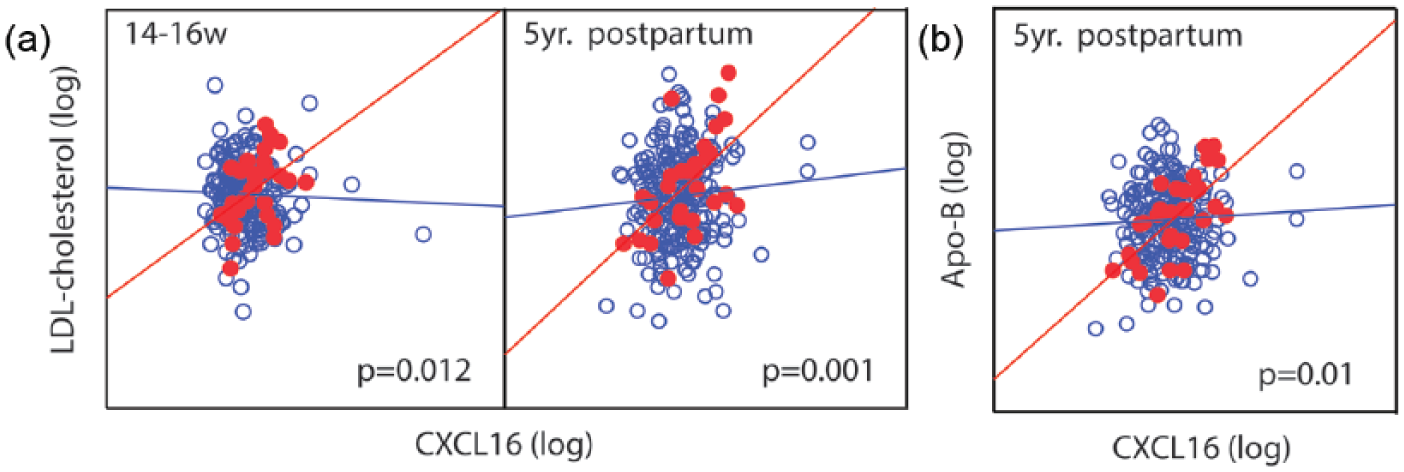
Interaction analysis between circulating CXCL16 and (a) LDL-C at 14–16 weeks and 5-year follow-up and (b) apoB at 5 years follow-up in GDM and non-GDM women. p values are given for the interaction term.
Predictors of apoB and LDL in GDM and controls.
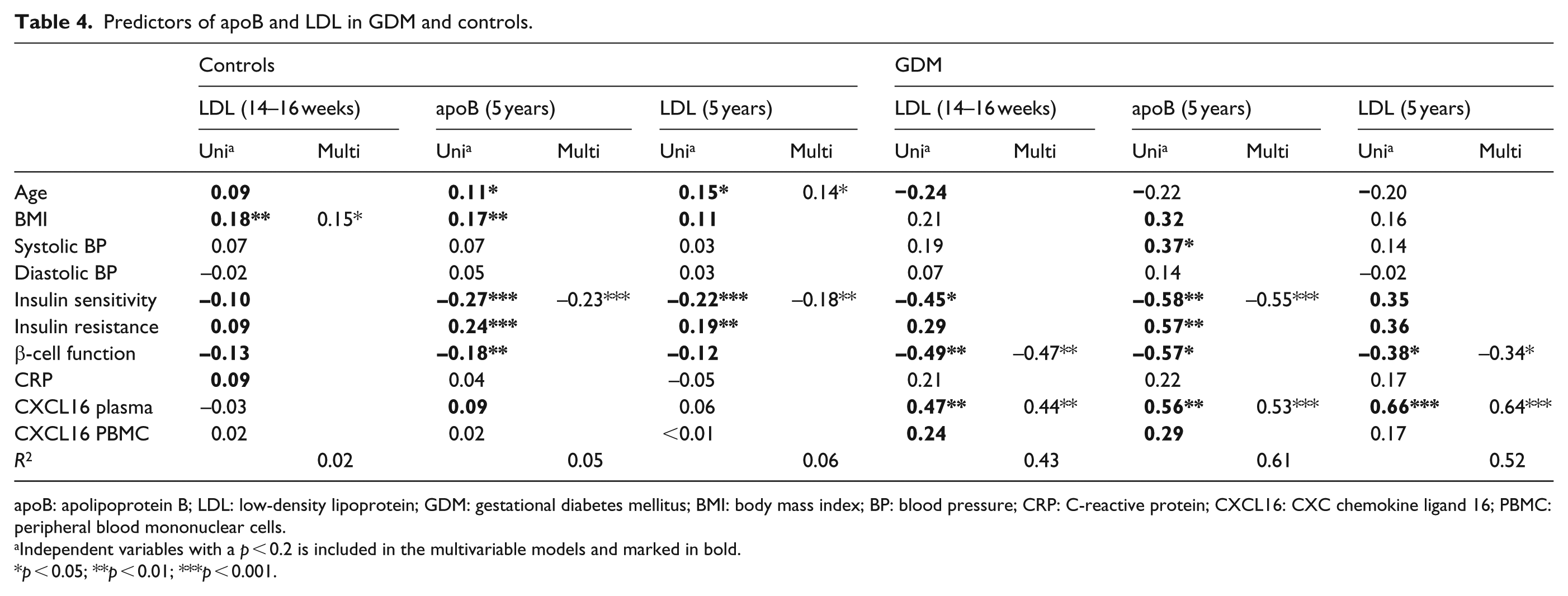
apoB: apolipoprotein B; LDL: low-density lipoprotein; GDM: gestational diabetes mellitus; BMI: body mass index; BP: blood pressure; CRP: C-reactive protein; CXCL16: CXC chemokine ligand 16; PBMC: peripheral blood mononuclear cells.
Independent variables with a p < 0.2 is included in the multivariable models and marked in bold.
p < 0.05; **p < 0.01; ***p < 0.001.
Discussion
This study demonstrates that women with complicated pregnancy due to GDM or PE are characterized by a different temporal profile of circulating CXCL16 with higher levels early in pregnancy in GDM and late in PE. CXCL16 levels were higher and strongly correlated with apoB containing lipoproteins at 5 years follow-up in GDM. Our findings suggest an interaction between high plasma levels of CXCL16 and lipid profile in GDM, potentially predisposing to CVD later in life.
Our finding that both women with GDM and PE were characterized by increased CXCL16 levels and decreased CXCL16 mRNA in PBMC at some time point compared to normal pregnancy could suggest they share some common risk factors. However, CXCL16 was not correlated with a poor metabolic profile, which both GDM and PE displayed throughout pregnancy, and the temporal profile of plasma CXCL16 during pregnancy was quite different, with higher levels early in GDM and higher levels late in pregnancy in PE. This supports the notion that GDM may be predisposed and pregnancy may mask or inhibit the increased levels of inflammation markers and contribution from the placenta to systemic inflammation in late gestation, 20 while in PE, increased levels late in pregnancy could be caused by severely increased inflammation, 11 and/or placental oxidative stress and interactions between released syncytiotrophoblast debris antagonizing the maternal vasculature or circulating immune cells. 21 However, the decreased CXCL16 mRNA in PBMC suggests that circulating immune cells are an unlikely source. Other potential sources are endothelial or smooth muscle cells. Nonetheless, the increase in CXCL16 in PE during pregnancy and normalization post-partum supports our recent report in a different PE population, where CXCL16 normalized already 6 months after pregnancy. 22
The oxidative changes of LDL and internalization are facilitated by scavenger receptors, including CXCL16, on the surface of activated macrophages, transforming them into foam cells and fatty streaks in atherosclerotic lesions. We have previously demonstrated that these GDM women present with dyslipidemia and lipid CV risk ratios at 5 years follow-up.16,17 A major finding in our study was that increased plasma CXCL16 at 5 years follow-up in GDM was closely associated with apoB and LDL-C. Furthermore, the association between CXCL16 and LDL within the GDM group was also present early in pregnancy and not present in controls or PE patients. CXCL16 has been found increased in the circulation of type 2 diabetes/coronary artery disease (CAD) patients 23 and suggested to be involved in the pathogenesis of diabetic nephropathy. 24 However, although we found some modest correlations between CXCL16 and indices of glucose metabolism in our study, this association was not present within the GDM group. Nonetheless, decreased beta-cell function was, together with CXCL16, a predictor of LDL levels both early in pregnancy and at 5 years follow-up suggesting independent interactions between glucose metabolism, CXCL16 and dyslipidemia in GDM. Both apoB and LDL-C are closely correlated with oxLDL25,26 which is elevated in GDM 27 but difficult to determine accurately due to the heterogeneous nature of the chemistry of LDL composition and oxidation. 28 Furthermore, apoB is more predictive of CVD progression than oxLDL. 26 We have recently demonstrated that increased CXCL16 is associated with the risk of developing CVD in healthy, although markedly older, individuals. 13 However, the key process of atherogenesis starts early in life as demonstrated in numerous studies (summarized in the study of Hong 29 ) showing raised fatty streaks and fibrous-plaque lesions present in 40%–85% around the age of 30–39 years. It is tempting to hypothesize that the strong correlation and interaction between CXCL16 and LDL and apoB could be related to the clearance of oxLDL, either directly by influencing the scavenger function of membrane-bound CXCL16 or by reflecting the inflammatory response due to lipid accumulation as CXCL16 is upregulated in macrophages by atherogenic lipids. 30
Experimental studies in mice deficient in both CXCL16 and Ldlr revealed accelerated atherosclerosis indicating that the scavenger receptor activity mediated by membrane-bound CXCL16 in vivo is atheroprotective. 31 However, the membrane-bound and soluble CXCL16 may have different biological functions, and in vitro experiments in rheumatoid arthritis (RA) patients do not support a stable binding between soluble CXCL16 and oxLDL. 32 In contrast, Shimaoka et al. 33 demonstrated that CXCL16 has scavenger receptor–like activity through the N-terminal chemokine domain. Thus, a combinations of decreased CXCL16 on circulating PBMC (assuming decreased CXCL16 mRNA indicates a lower level of membrane-bound CXCL16 protein) and higher circulating CXCL16 levels that could interfere with the membrane-bound scavenger function could potentially attenuate lipoprotein removal and accelerate atherogenesis in GDM women. The increase in plasma CXCL16 early in pregnancy in GDM women displayed similar interactions with LDL-C and apoB as at 5 years suggesting that CXCL16 could be an early marker of increased dyslipidemia in these women.
In contrast to CXCL16 transgenic mice, CXCR6-deficient (i.e. lacking the CXCL16 receptor) apoE mice showed attenuated atherosclerosis, indicating a proatherogenic role of CXCL16/CXCR6 interaction. Soluble CXCL16 may promote the migration of T cells and augment a proatherogenic inflammatory response. CXCL16 was poorly correlated with CRP in our study. However, given the link between inflammatory mediators such as interleukin-1β, tumour necrosis factor and interferon γ and the cleavage of the membrane-bound CXCL16 to the soluble form of CXCL16, it has been suggested that soluble CXCL16 could serve as a reliable and stable marker of these inflammatory pathways. 34 Thus, activity in other pathways such as CRP/IL-6 may not necessarily be strongly associated with CXCL16. Nonetheless, how the increased circulating levels of CXCL16 relate to the membrane-bound CXCL16 on immune cells and if this affects the scavenger potential of these cells or is related to fatty streak formation is unknown and will have to be further investigated in future studies.
Limitations
Despite performing quite a few correlations in this study with the chance of spurious findings, we did not perform Bonferroni adjustments. We think this is justified since the associations were evaluated at multiple time points using several measures of disturbed glucose metabolism, and for some analysis, stepwise regression was used to identify the strongest predictors. Thus, random findings are unlikely to impact our results. Of note, the GDM group had a very strong history of diabetes (41.9%) compared to controls (30.7%) possibly due detailed inquiry including grandparents in addition to parental information as has been observed in other studies. 35 The samples in this study were stored for many years prior to analysis. In a previous study with comparable inclusion and storage time, we found no effect of storage time on CXCL16 levels. The higher levels in samples stored the longest (i.e. first quartile of inclusion) in this study is most likely by chance as storage time had little impact on CXCL16 in the remaining samples. Furthermore, the pattern of CXCL16 in pregnancy and at 5 years follow-up was similar regardless of storage time. Finally, we have previously demonstrated in a case control study nested within a population-based cohort that CXCL16 was a predictor of myocardial infarction (MI) also in adjusted analysis. 13 In that study, there was only a ~5% difference in CXCL16 levels between individuals who developed MI and those who did not. Thus, only minor elevations in CXCL16, perhaps in combination with other CVD risk factors, such as LDL in this study, may translate into increased CVD risk, and our study was somewhat underpowered to detect these small differences.
Our study found that women with GDM and PE had a dysregulated CXCL16 during pregnancy. In GDM, CXCL16 was increased early in pregnancy and after 5 years, and was strongly associated with LDL-C and apoB. Further studies evaluating the interaction between CXCL16 and dyslipidemia and CVD risk in these women are warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a postdoctoral grant from Norwegian Health South-East (Helse Sør-Øst) Regional Health Authority.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
