Abstract
Aim:
Insulin resistance and type 2 diabetes are independent risk factors for cardiovascular diseases. Levels of C-peptide are increased in these patients and its role in the atherosclerosis progression was studied in vitro and in vivo over the past years. To evaluate the possible use of C-peptide as cardiovascular biomarkers, we designed an observational study in which we enrolled patients with mono- or poly-vascular atherosclerotic disease.
Methods:
We recruited 431 patients with stable atherosclerosis and performed a yearly follow-up to estimate the cardiovascular and total mortality and cardiovascular events.
Results:
We performed a mean follow-up of 56 months on 268 patients. A multivariate Cox analysis showed that C-peptide significantly increased the risk of cardiovascular mortality [Hazard Ratio: 1.29 (95% confidence interval: 1.02−1.65, p < 0.03513)] after adjustment for age, sex, diabetes treatment, estimated glomerular filtration rate and known diabetes status. Furthermore, levels of C-peptide were significantly correlated with metabolic parameters and atherogenic factors.
Conclusion:
C-peptide was associated with cardiovascular mortality independently of known diabetes status in a cohort of patients with chronic atherosclerotic disease. Future studies using C-peptide into a reclassification approach might be undertaken to consider its potential as a cardiovascular disease biomarker.
Introduction
Insulin resistance is a hallmark and risk factor for cardiovascular atherosclerotic disease and its complications such as myocardial infarction and stroke.1,2 Insulin regulates several anabolic and catabolic processes that influence lipid and glucose metabolism with an impact on cardiovascular homeostasis. The interaction among insulin, its metabolic functions and cardiovascular events is an intensive area of research given the implications on the potential discovery of new biomarkers and therapeutic targets. 3
Among many conventional biomarkers for cardiovascular disease, C-peptide is a potential unifier for insulin resistance and defective insulin secretion with cardiovascular diseases (CVD). Recent studies suggested that C-peptide exhibit biological activity, induces vascular smooth muscle cells migration and infiltration of inflammatory cells in in vitro studies while promoting atherosclerosis progression in a mouse model.4–7 Finally, the LUdwigshafen RIsk and Cardiovascular Health (LURIC) study identified an association between C-peptide and cardiovascular mortality. 8 In this study, we identified C-peptide as an important risk predictor for cardiovascular mortality in subjects with stable atherosclerosis.
Methods
Description of patients
Four hundred and thirty-one patients with mono- or poly-vascular atherosclerosis disease were recruited at the Center for Atherosclerosis of the Policlinico Tor Vergata in Rome, for a clinical and therapeutical assessment. Patients were enrolled in the Tor Vergata Atherosclerosis Registry (TVAR) from 2007, listed on the International Standard Randomised Controlled Trial Number (ISRCTN) registry with study ID ISRCTN42405215. Patients were diagnosed with established and documented atherosclerotic vascular disease at the atherosclerosis ambulatory clinic that includes cardiology, diabetology and vascular surgery specialists. All the patients have had a major CardioVascular (CV) event or undergone a vascular procedure for significant vascular stenosis. Exclusion criteria included liver disease, renal insufficiency, heart failure, coagulopathy or any other severe systemic disease. Subjects were also excluded if they had positive blood tests for HIV, hepatitis B or hepatitis C.
Cerebrovascular disease was defined clinically by documented history of a previous cerebrovascular disease event or by documented carotid plaques (50% luminal obstruction). Coronary artery disease (CAD) was defined by documented history of a previous acute myocardial infarction, presence of clinical signs and coronary angiography. Peripheral vascular disease was defined by a history of intermittent claudication, angiographic documentation of atherosclerotic luminal obstruction of the peripheral arteries or a history of a peripheral arterial intervention for atherosclerotic disease (angioplasty or surgery).
The follow-up was performed every year by phone interview. The primary endpoint was major cardiovascular events (cardiovascular death, nonfatal stroke, nonfatal myocardial infarction and peripheral vascular surgical procedures) and the secondary endpoint was death for any other diseases. Information on nonfatal events was sought yearly from study participants by phone interviews and confirmed by review of hospital records. In case of fatal events, information was obtained from their primary care physicians or from death certificates. In 2014, we performed an analysis on the status of the patients who answered to the follow-up. Prospective data were available for 268 subjects for an average follow-up of 56 months, 80 patients did not answer to phone interview, 32 patients had missing data on C-peptide and 51 patients were excluded because followed for less than 1 year. At recruitment, all patients underwent thorough medical history, clinical examination and anthropometric measurements.
Diabetes treatment
Forty-one of the 268 subjects with available follow-up were under diabetes treatments: 10 were under combined insulin and metformin treatment, 3 of them were on insulin therapy, 13 were on metformin and 15 on secretagogues.
Informed consent and ethics committee approval
An informed written consent was obtained from all participants. The study was approved by the local ethics committee and the reported investigations were carried out in accordance with the principles of the Declaration of Helsinki as revised in 2000.
Laboratory measurements
Fasting glucose, total cholesterol, high-density lipoprotein and low-density lipoprotein cholesterol, triglycerides, creatinine, urea, aminotransferases and gamma-glutamyltransferase were assessed by standard immunoenzymatic methods. Insulin and C-peptide levels were measured by immunochemiluminescence assay (ADVIA Centaur XP, Siemens Healthineers Diagnostics) whereas haemoglobin A1c percentage by high-performance liquid chromatography method. High-sensitivity C-reactive protein was measured by nephelometric immunoassay (Dimension Vista 1500, Siemens Healthineers Diagnostics).
Statistical analysis
Patients’ characteristics were reported as mean and standard deviation (SD) for continuous variables and frequencies and percentages for categorical ones. Variables correlations were carried out using Spearman correlation. Time-to-event data were analysed by means of log-rank test or univariate Cox models for categorical or continuous variables, respectively. Significance of C-peptide was also assessed at multivariate Cox modelling, after adjusting for possible relevant confounders. The final multivariable Cox model was chosen with forward stepwise regression based on Akaike’s information criterion (AIC), which guarantees a final parsimonious model with best prediction performance. Of note, all potential predictors were considered for final inclusion.
Final predictive scores were obtained as linear combinations of the most relevant predictors, where the weights were obtained by rounding the Cox regression coefficients. Predictive performance was assessed by means of time-dependent ROC curves and net reclassification index (NRI).
Results
Clinical and biochemical characteristics of the 431 patients enrolled in the TVAR are shown in Table 1. We performed a follow-up for all-cause and cardiovascular event and mortality for a mean of 56.17 ± 28.52 months on 268 patients. During follow-up, 66 nonfatal cardiovascular events (24.6%) and 44 deaths (16.5%) occurred, 20 of which were due to cardiovascular causes (7.5%). Clinical and biochemical characteristics of the 268 patients are summarized in Table 1. Among the several parameters and biochemical variables available, only age was found independently associated with cardiovascular death with univariate Cox analysis. On the basis of this result, we performed a bivariate Cox analysis, after adjustment for age, and we found that C-peptide and gender were independently associated with cardiovascular death (Table 1).
Clinical characteristics of the patients included in TVAR registry and the patients included in the follow-up analysis.
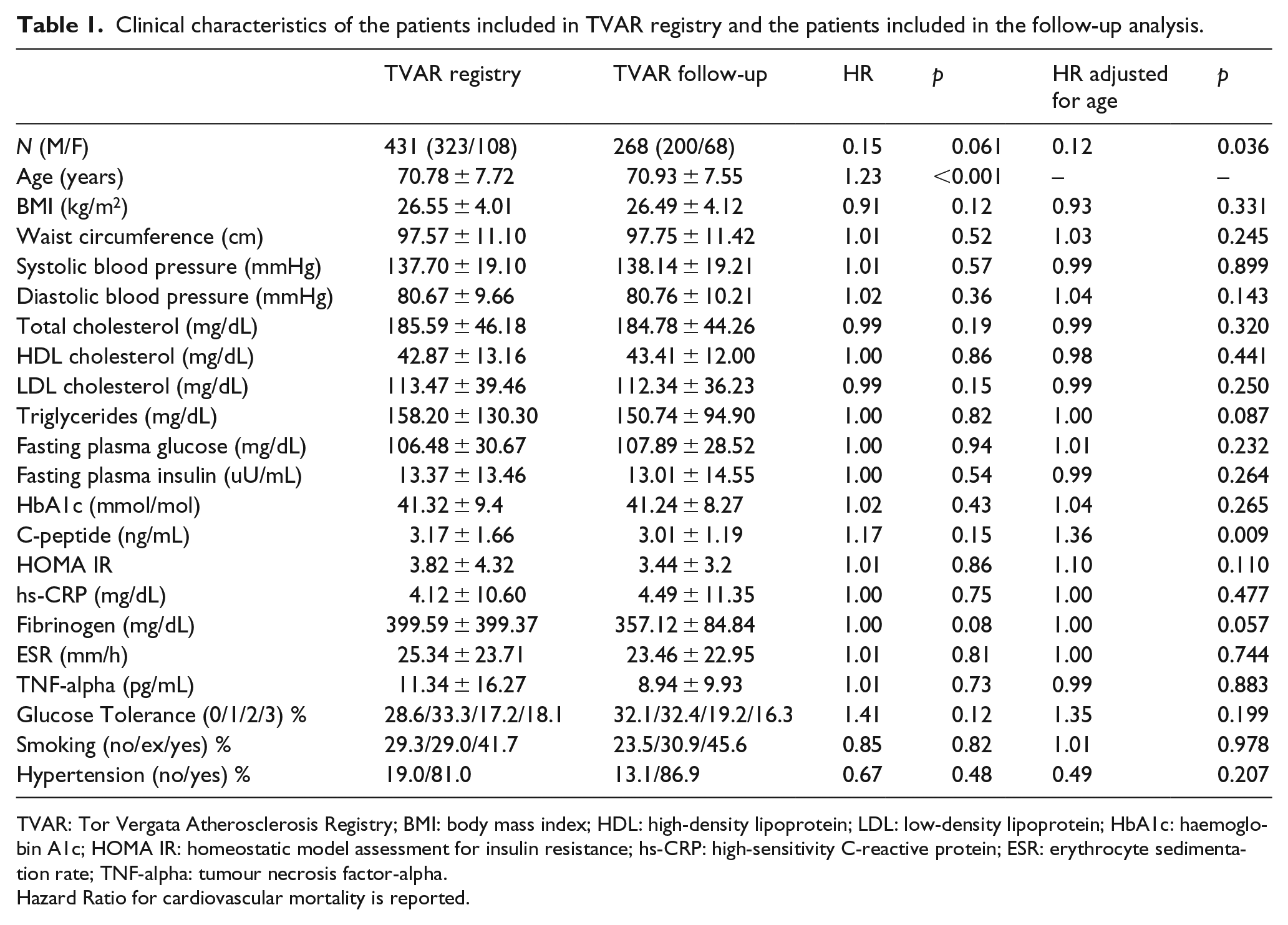
TVAR: Tor Vergata Atherosclerosis Registry; BMI: body mass index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; HbA1c: haemoglobin A1c; HOMA IR: homeostatic model assessment for insulin resistance; hs-CRP: high-sensitivity C-reactive protein; ESR: erythrocyte sedimentation rate; TNF-alpha: tumour necrosis factor-alpha.
Hazard Ratio for cardiovascular mortality is reported.
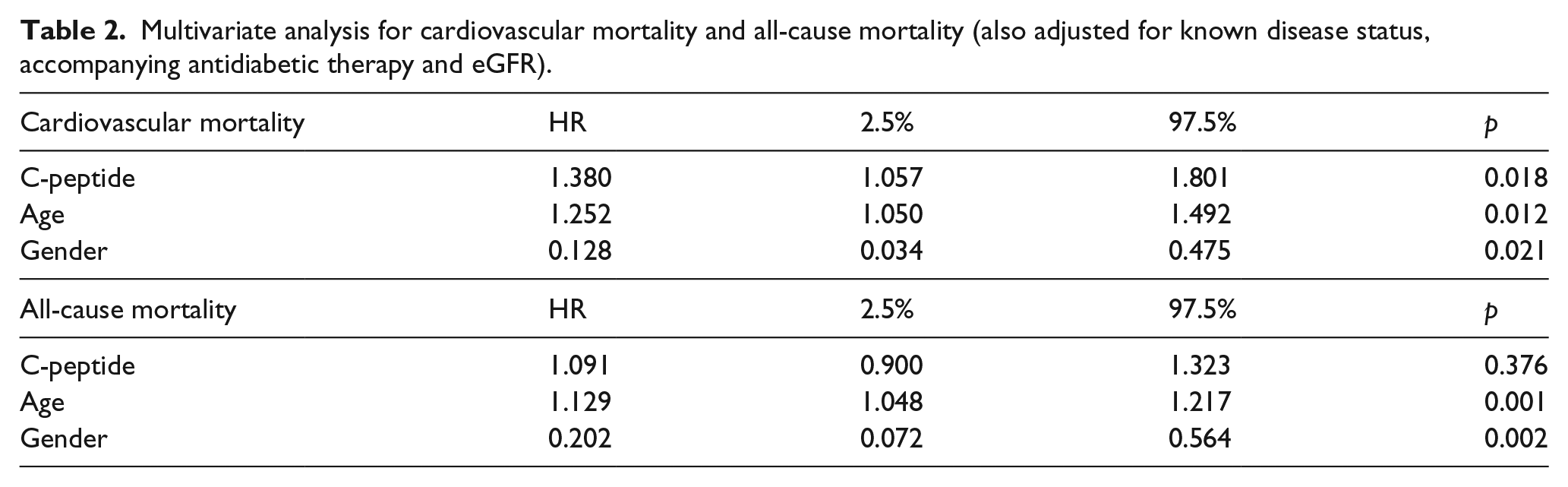
A multivariate Cox analysis showed that C-peptide, included as continuous variable, increased the risk in cardiovascular mortality per SD by [Hazard Ratio: 1.38 (95% confidence interval (CI): 1.057−1.801, p = 0.018)] after adjustment for age, sex, antidiabetic therapy, estimated glomerular filtration rate (eGFR) and known diabetes status, suggesting that C-peptide is a cardiovascular mortality predictor independently from a history of diabetes (Table 2). Effects of C-peptide were not significantly different between men and women (p = 0.208 for cardiovascular mortality and p = 0.71 for total mortality).
Multivariate analysis for cardiovascular mortality and all-cause mortality (also adjusted for known disease status, accompanying antidiabetic therapy and eGFR).
In our cohort, we found that levels of C-peptide were significantly correlated with metabolic parameters such as fasting plasma glucose (R = 0.327, p < 0.0001; adjusted for sex and age R = 0.333, p < 0.0001), homeostatic model assessment for insulin resistance (HOMA IR; R = 0.693, p < 0.0001; adjusted for sex and age R = 0.690, p < 0.0001), body mass index (BMI; R = 0.402, p < 0.0001; adjusted for sex and age R = 0.394, p < 0.0001) and insulin-like growth factor 1 (IGF1; R = 0.206, p < 0.0001; adjusted for sex and age R = 0.195, p = 0.002). Levels of C-peptide were also significantly correlated with atherogenic factors such as high-density lipoprotein (HDL; R = −0.247, p < 0.0001; adjusted for sex and age R = −0.228, p < 0.001), triglycerides (r = 0.302, p < 0.0001; adjusted for sex and age R = 0.302, p < 0.0001) and high-sensitivity C-reactive protein (hs-CRP) (R = 0.246, p < 0.0001; adjusted for sex and age R = 0.191, p < 0.0001).
Finally, we build a predictive score for CVD mortality as follows: we gave −2 points to females, 0.25 points for each year of age, 0.33 points for each unit of C-peptide, −0.1 points for each unit of BMI and 0.002 points for each unit of hs-CRP. The resulting score predicted CVD at 60 months with an AUC of 0.920 (95% CI: 0.778–1.000), p < 0.0001. It shall be noted that a score which did not include C-peptide would yield an Area Under Curve (AUC) of only 0.865 (95% CI: 0.738−0.979). The ½ NRI of C-peptide was 0.281, which was very large despite not being significant (p = 0.153).
When we analysed all-cause mortality, we found that C-peptide did not have a significant effect, as HR of C-peptide was estimated as 1.091 (95% CI: 0.900–1.323, p = 0.376), after adjusting for age, sex, antidiabetic therapy, eGFR and known diabetes status.
Discussion
The major findings from this analysis of the TVAR concern the role of C-peptide as a biomarker for cardiovascular mortality and its potential use to improve cardiovascular prediction. In our study, C-peptide resulted the most significant predictor of cardiovascular mortality in subjects with established atherosclerosis and high cardiovascular risk. Previous studies suggested a role for C-peptide as biomarker for cardiovascular and non-cardiovascular mortality. 8 In our analysis, we did not find an association with non-cardiovascular mortality, a result potentially depending on the different inclusion criteria between TVAR and LURIC study.
The association between cardiovascular mortality and levels of C-peptide might be explained by the direct role of the C-peptide in promoting atherogenesis. Marx and collaborators showed that C-peptide may facilitate the recruitment of inflammatory cells into early lesions 7 and promote lesion progression by inducing smooth muscle cell proliferation 5 in vitro. Furthermore, ApolipoproteinE (ApoE)-deficient mice on a high fat diet treated with C-peptide injections for 12 weeks showed more macrophages and smooth muscle cells in atherosclerotic lesions compared with controls. 4
Our study has some limitations; in fact, our database did not include information on the status of microvascular complications; therefore, we cannot assess whether C-peptide levels increase as a consequence of reduced renal excretion or extrarenal causes. However, adjustment for eGFR did not change our results, although it is intriguing to hypothesize that both decline in renal excretion and increased production may lead to increased C-peptide signalling.
In conclusion, C-peptide was associated with cardiovascular mortality independently of known diabetes status in a cohort of patients with mono- or poly-vascular chronic atherosclerotic disease. Future studies using C-peptide into a reclassification approach might be undertaken to consider its potential as a CVD biomarker.
Footnotes
Acknowledgements
M.C., M.B., M.M., F.D., I.C., G.G., S.R., V.G., O.P., C.P., R.M. and A.I. performed clinical and biochemical experiments. M.C. and A.F performed statistical analysis, contributed to data interpretation, generated tables and wrote the article. M.F. conceived the study and wrote the discussion. All authors discussed the data and commented on the article before submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was in part funded by MIUR PRIN 20123BJ89E, MIUR PRIN 2015MPESJS_004 and Fondazione Roma Non Communicable Diseases 2014 Call to M.F.
