Abstract
The lectin-like oxidised low-density lipoprotein receptor-1 (LOX-1) is a vascular scavenger receptor that plays a central role in the pathogenesis of atherothrombotic disease, which remains the main cause of mortality in the Western population. Recent evidence indicates that targeting LOX-1 represents a credible strategy for the management vascular disease and the current review explores the role of this molecule in the diagnosis and treatment of atherosclerosis. LOX-1-mediated pro-atherogenic effects can be inhibited by anti-LOX-1 monoclonal antibodies and procyanidins, whereas downregulation of LOX-1 expression has been achieved by antisense oligonucleotides and a specific pyrrole–imidazole polyamide. Furthermore, LOX-1 can be utilised for plaque imaging using monoclonal antibodies and even the selective delivery of anti-atherosclerotic agents employing immunoliposome techniques. Also, plasma levels of the circulating soluble form of LOX-1 levels are elevated in atherosclerosis and therefore may constitute an additional diagnostic biomarker of vascular pathology.
Introduction
Atherosclerosis is a complex vascular disease that is associated with extensive global morbidity and mortality. 1 Our understanding of atherosclerosis has evolved dramatically in the past few decades, and there have been significant advances in the treatment of this condition. 2 However, despite these improvements in management, atherosclerosis remains the leading cause of death worldwide 1 highlighting the need for new, alternative treatment strategies.
As our knowledge of atherosclerosis continues to expand, recent evidence implicates lectin-like oxidised low-density lipoprotein receptor-1 (LOX-1) as a key molecule involved in the pathogenesis of atherosclerosis. 3 This scavenger receptor, through binding and internalisation of oxidised low-density lipoprotein (ox-LDL) cholesterol particles, plays an essential role in pro-atherogenic mechanisms including endothelial dysfunction and foam cell formation. This review describes the involvement of LOX-1 in the pathogenesis of atherosclerosis and explores the use of this molecule as a diagnostic and therapeutic target in atherothrombotic disease.
LOX-1: an overview
Structure
LOX-1 is a class E scavenger receptor that was first cloned and identified relatively recently, as a vascular endothelial ox-LDL receptor. 4 LOX-1 is encoded by the OLR1 gene on human chromosome 12 5 and the gene product consists of a 50-kDa homodimer 6 consisting of four domains: an extracellular C-terminal C-type lectin domain, a connecting neck domain, a single transcellular domain and a short N-terminal cytoplasmic tail 7 (Figure 1). A soluble form of LOX-1 (sLOX-1) also exists and is formed through the proteolytic cleavage of its extracellular domain, 8 further discussed in detail below.
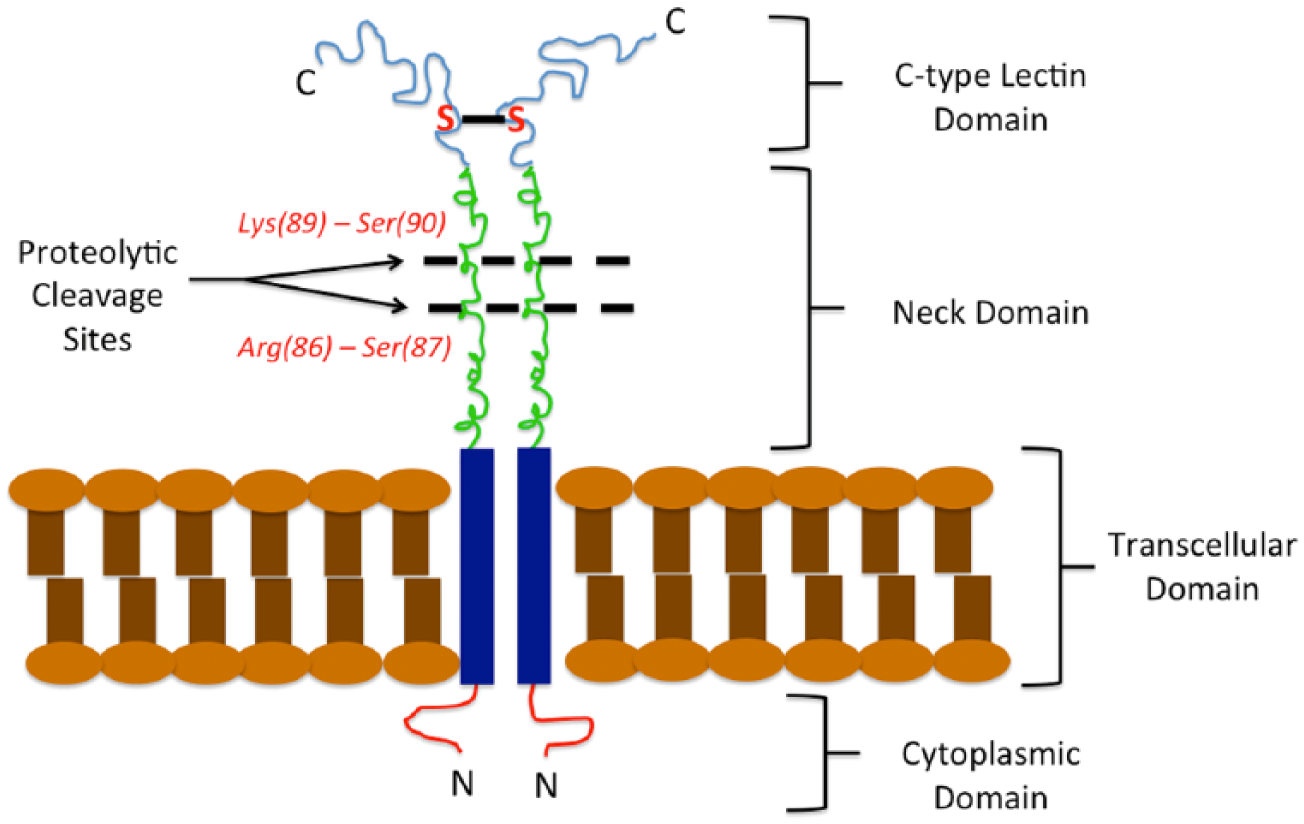
Structure of LOX-1. The molecule exists as a homodimer and has four domains: an extracellular C-terminal C-type lectin domain, a connecting neck domain, a single transcellular domain and a short N-terminal cytoplasmic tail. A disulphide bond exists between cysteine 140 in each of the C-type lectin domains. Cleavage of the extracellular portion occurs at two sites in the neck domain to form soluble LOX-1.
Expression and function of LOX-1
LOX-1 is expressed primarily in endothelial cells and also in numerous other cells including platelets, macrophages and smooth muscle cells.9,10 LOX-1 binds a diverse array of ligands including modified lipoproteins, C-reactive protein (CRP) and fibronectin. 3 Upon binding of ox-LDL to LOX-1, membrane-bound nicotinamide adenine dinucleotide phosphate (NADPH) oxidase is activated, which generates reactive oxygen species (ROS) that activate nuclear factor-κB. Nuclear factor-κB subsequently binds to promoter regions on the OLR1 gene, increasing LOX-1 transcription. In addition, persistently increased ROS concentrations trigger multiple other intracellular signalling pathways that activate nuclear factor-κB, further increasing LOX-1 expression. 3 Basal expression of LOX-1 is low, but significant upregulation occurs during several pathological states including diabetes, hypertension and atherosclerosis due to the presence of inflammatory and oxidative stimuli. 3 In the presence of these stimuli, numerous intracellular pathways can become induced, which activate nuclear factor-κB as well as other OLR1 transcription factors such as peroxisome proliferator-activated receptor (PPAR)-γ, Oct1 and activator protein-1 (AP-1), leading to increased LOX-1 expression. 3
In the past decade, multiple studies have indicated that LOX-1 has important role in atherogenesis, particularly in early stages of disease pathogenesis, through its binding and internalisation of circulating ox-LDLs. 11 Studies have also implicated LOX-1 in angiogenesis and as an immune receptor,12,13 but these roles are less clearly understood.
Although research has focused primarily on LOX-1 in atherogenesis pathology, it is likely that this receptor also has some degree of physiological function since it binds to and takes up aged red blood cells and apoptotic cells. 14 Furthermore, LOX-1 may be involved in the transcytosis of pancreatic enzymes in intestinal cells. 15 However, it is likely that LOX-1 plays a more important role in pathology rather than physiology since its basal expression is very low and is upregulated significantly in a number of pathological states.16,17 Thus, although our knowledge of LOX-1 has developed immensely, it is likely that this receptor has numerous other functions that still remain to be uncovered.
LOX-1 in the pathogenesis of atherosclerosis
Atherosclerosis is a complex, chronic inflammatory process that affects large- and medium-sized arteries2,11 and may start as early as childhood. The primary event in atherogenesis is endothelial dysfunction, characterised by failure of these cells to maintain normal homeostatic function. This is a complex process that continues throughout atherogenesis and leads to a shift in function of the endothelium to a pro-inflammatory and prothrombotic state.
Another important early event of atherosclerosis is the oxidative modification of circulating LDLs, which occurs following their entrapment in pockets within the subendothelial space, 18 secondary to production of oxidants coupled with the relatively low concentrations of antioxidants. 18 These ox-LDL particles bind to LOX-1 on endothelial cells leading to secretion of chemokines and pro-inflammatory molecules 19 as well as the upregulation of adhesion molecules. 20 Secreted chemokines attract circulating monocytes, which subsequently adhere to the vessel wall. The monocytes migrate across into the intimal space and form macrophages, which begin to express LOX-1. 9 This uptake of ox-LDL by the endothelial cells into the intimal space is carried out mainly through LOX-1 21 and also, to a lesser extent, by other scavenger and non-scavenger receptor–mediated pathways. 22 In addition, the binding of ox-LDL to LOX-1 upregulates LOX-1 expression, 17 inducing a vicious pro-inflammatory cycle.
Within the intimal space, macrophages bind and take up ox-LDL through LOX-1 leading to activation and secretion of pro-inflammatory cytokines and ROS, further enhancing local inflammation. Accumulation of cholesterol in macrophages leads to the formation of foam cells, which constitute an important component of the atherosclerotic plaques. A collection of foam cells forms the fatty streak, which occurs in the early stages of the atherosclerotic process and transforms over decades into mature plaques through a continuing inflammatory process and the deposition of collagen. 11
LOX-1 is also involved in the latter stages of the atherothrombotic process. LOX-1 is expressed on platelets and appears to augment adenosine diphosphate (ADP)-induced platelet aggregation. 23 This indicates that LOX-1 is involved in early atheroma formation as well as intravascular thrombus formation through the control of platelet activation. Figure 2 summarises the pathogenesis of atherosclerosis and the role of LOX-1.
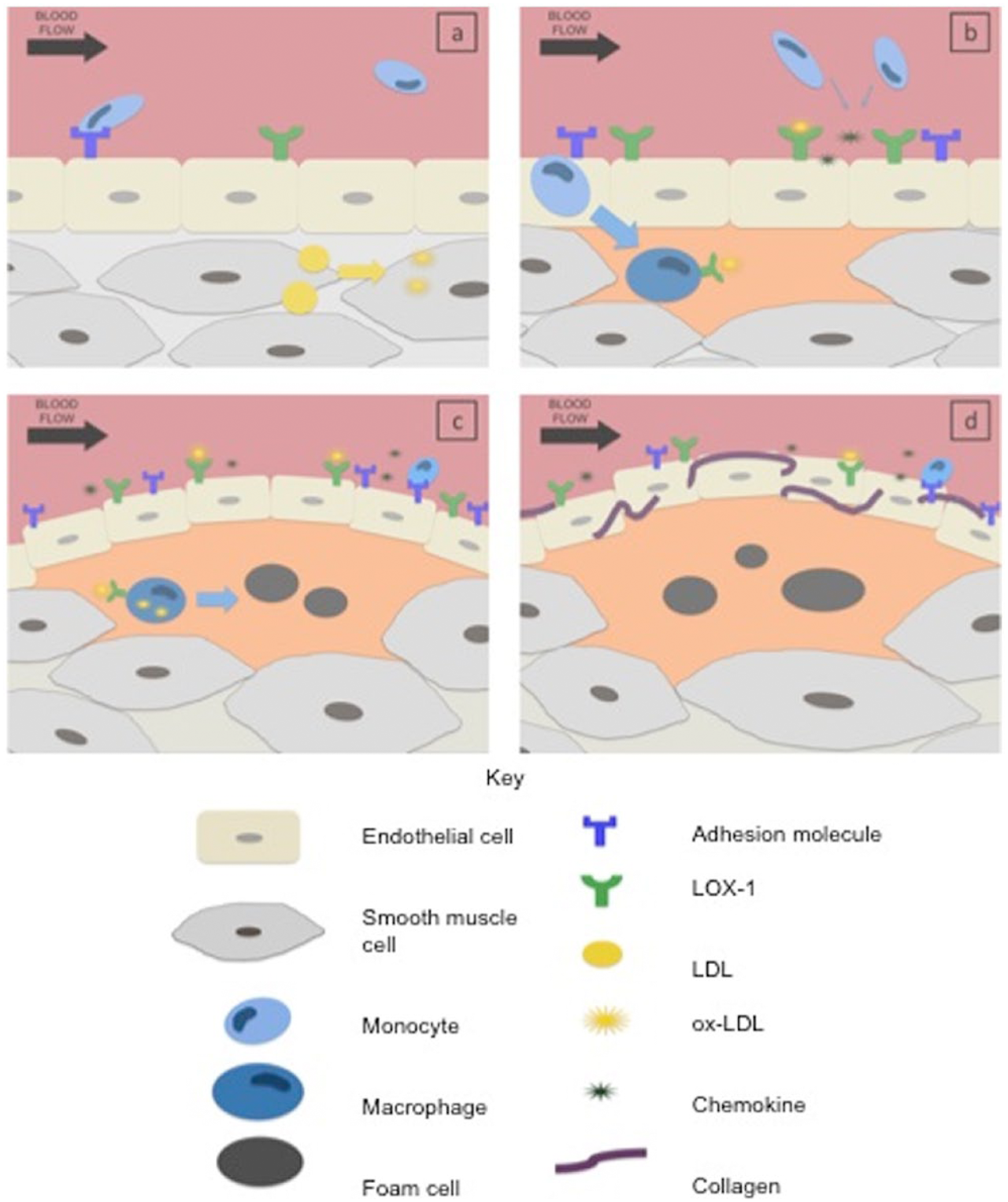
Pathogenesis of atherosclerosis and the role of LOX-1. (a) Endothelial dysfunction characterised by increased adhesion capability of these cells and increased intercellular permeability. This results in adhesion of monocytes and ‘leakage’ of LDL into the subendothelial space, where it is converted to ox-LDL. (b) Monocytes migrate across into the intimal space and differentiate into macrophages, which begin to express LOX-1. This receptor, expressed on endothelial cells and macrophages, binds ox-LDL triggering the release of chemokines and increasing the inflammatory reaction. Also, this upregulates the expression of LOX-1 and adhesion molecules on the endothelium. (c) Macrophages that have taken up ox-LDL are converted to foam cells, which group together and form the fatty streak, the earliest visible abnormality in the atherosclerotic process. (d) Subsequent deposition of collagen in the fatty streaks converts these lesions into atheromatous plaques, which can increase in size and result in narrowing of the vessel lumen.
Modulation of LOX-1 expression and function
Given the importance of LOX-1 in the pathogenesis of atherothrombosis, attempts have been made to modulate protein expression or function, and the main strategies used are briefly discussed below.
Genetic manipulation
An important aspect of LOX-1 biology is the discovery that certain allelic polymorphisms in the human LOX-1 (OLR1) gene modulate cardiovascular disease risk. 24 Of particular interest are single nucleotide polymorphisms (SNPs) in introns 4, 5 and 3′ untranslated region (UTR) embedded in a linkage disequilibrium block of the OLR1 gene, which each have a strong positive correlation with acute myocardial infarction. 25 Other polymorphisms, which have shown some degree of association with increased cardiovascular disease risk, include a lysine-to-asparagine substitution at position 167 (K167N)26,27 and three substitutions in introns 4, 5 and 3′ UTR. 28 However, a general criticism of these studies is the lack of clear mechanistic explanations for the protective or harmful effects of LOX-1 genetic variants, in particular given the location of these polymorphisms in intronic regions. Using a minigene approach, Mango and colleagues have shown that intronic LOX-1 SNPs regulate the expression of a splice variant of the OLR1 gene, termed LOXIN. It subsequently became apparent that LOXIN, which lacks exon 5 and part of the functional C-type lectin domain, has a functional role as it blocks LOX-1 activation through the formation of a hetero-oligomer with the protein. 25 In vitro evidence indicates that LOXIN reduces uptake of ox-LDL by interfering with binding or possibly through reduction of cellular trafficking resulting in lower levels of LOX-1 at cell surface. A direct anti-apoptotic role for LOXIN has also been proposed.29,30 Targeting LOXIN, therefore, could offer a novel route of preventing and/or treating atherothrombotic disease.
In addition to studies investigating LOX-1 polymorphisms, there has also been a growing body of research using transgenic mouse models with altered LOX-1 loci. Mice overexpressing LOX-1 crossed with apoE knockout mice fed a high-fat diet, exhibited increased plaque ox-LDL and macrophage accumulations in the blood vessels compared with wild-type mice. 31 Furthermore, Mehta and colleagues32,33 found that atherosclerotic lesion development, collagen deposition and matrix metalloproteinase expression were significantly lower in LOX-1 knockout mice crossed with LDL receptor knockout mice compared with wild-type LOX-1 mice. The mouse studies support an important role of LOX-1 in atherosclerotic disease and further highlight its potential as a therapeutic target.
Antisense oligonucleotides
Antisense oligonucleotides are short, single strands of DNA or RNA molecules that reduce the production of a specific protein through binding and inhibition of messenger ribonucleic acid (mRNA) expression. 34 The use of a LOX-1 antisense was first documented in 2000 when Li and Mehta 21 demonstrated the ability of this approach to suppress LOX-1-mediated overexpression of monocyte chemoattractant protein-1 (MCP-1) in vitro. Since then, this antisense molecule has been used in numerous studies examining the involvement of LOX-1 in the pathogenesis of atherosclerosis.17,35 In 2012, Amati et al. studied, for the first time, the therapeutic potential of an oligonucleotide antisense targeted against LOX-1 in vivo. This antisense oligonucleotide was created in the laboratory and targeted mRNA of the gene coding for LOX-1, OLR1. 5 The antisense oligonucleotide was infused into double apolipoprotein E knockout mice fed a high-cholesterol diet and a significant decrease in LOX-1 mRNA and protein expression was detected compared with the control group, without a change in pro-inflammatory markers. 36 However, this work did not investigate the role of this approach on prevention of atheroma formation. These findings are encouraging and further work should be carried out examining the effects of this antisense molecule on the development and progression of the atherosclerotic process.
Pyrrole–imidazole polyamides
Pyrrole–imidazole polyamides (P–I polyamides) are a novel class of synthetic compounds that bind to specific sequences of DNA with high affinity. Pyrrole and imidazole groups are combined in an antiparallel side-by-side dimeric complex and competitively bind to the minor groove of regulatory sequences on DNA. 37 This disrupts binding of transcription factors leading to a subsequent reduction in target gene expression. Multiple in vitro studies have demonstrated the ability of P–I polyamides to inhibit the expression of their target gene, and in 2009 a specific P–I polyamide targeted against LOX-1 was developed by Ueno et al. 38 A DNA sequence proximal to the binding site of AP-1, a transcription factor involved in the angiotensin-II-mediated signalling pathway, was targeted on the promoter region of the OLR1 gene. This specific P–I polyamide effectively inhibited promoter activity in human embryonic kidney (HEK) 293 cells and reduced LOX-1 mRNA levels in human umbilical vein endothelial cell (HUVEC) through a dose-dependent manner. A reduction in apoptosis was seen in the HUVECs. Since apoptosis of endothelial cells is an important process in the rupture of atherosclerotic plaques, use of this specific P–I polyamide could be particularly effective in targeting vulnerable plaques. Interestingly, the specific P–I polyamide used retained the basic expression of LOX-1, inhibiting only the AP-1 pathological enhancement of its expression, a feature that distinguishes P–I polyamides from antisense oligonucleotides. As a result, this specific polyamide targeted against LOX-1 is less likely to disrupt any physiological function this receptor may have. Clinical use of this P–I polyamide is therefore less likely to cause adverse events and could be advantageous compared with other methods of modulating LOX-1 expression. In animal models, P–I polyamides have had significant success due to their ability to permeate cells without cytotoxicity, indicating such an approach is a possibility in man.
Small-molecule inhibition of LOX-1
It is well documented that certain diets are protective against atherosclerosis and vascular disease, but little is known about the mechanisms involved. 39 In 2011, Nishizuka et al. 40 conducted a study investigating which food extracts inhibit ox-LDL binding to LOX-1 and procyanidins, a flavonoid found in various fruits, was found to be a potent inhibitor of this interaction. The researchers also found that prolonged administration of purified short-chained procyanidins inhibited lipid deposition in the vessel walls of rats. These findings highlight the potential use of procyanidins as dietary supplements in humans to inhibit plaque formation via the LOX-1-mediated pathway. Previous studies have reported anti-hypertensive, anti-oxidative and anti-hyperlipidaemic properties of procyanidins in vivo, 41 and so its use could benefit patients with atherosclerosis via multiple mechanisms. As procyanidins are naturally occurring, they would be relatively safe in human studies and would be less likely to cause side effects compared with synthesised LOX-1 antagonists. Aside from procyanidins, no other small-molecule inhibitors of LOX-1 have currently been identified or created. Small-molecule inhibitors have, however, been developed against the macrophage scavenger receptor and scavenger receptor class B member I (BI) 42 suggesting that this may be a feasible strategy for LOX-1 inhibition. Interestingly, statins also seem to modulate LOX-1 gene expression, suggesting that this contributes to a reduction in atherosclerosis. 43
Anti-LOX-1 antibodies
In 1997, the first mouse monoclonal antibody that targets LOX-1 was developed by Sawamura et al. 4 in Japan. Since then, a number of other animal monoclonal antibodies have been created and these have been used in multiple studies to further our understanding of LOX-1 both in vitro and in vivo. 3 Human monoclonal antibody was developed at a later date and was used to demonstrate the upregulation of transforming growth factor-β1 receptors in human coronary artery endothelial cells following ox-LDL–LOX-1 interaction. 44 Following this, it has been observed that this antibody inhibits ox-LDL induction of ROS production, MCP-1 expression and the triggering of Ras/Rho1-dependent signalling pathways. 45 Therefore, this represents a promising technique for clinically targeting LOX-1 in the future. Targeting strategies to modulate LOX-1 production or function are summarised in Figure 3.
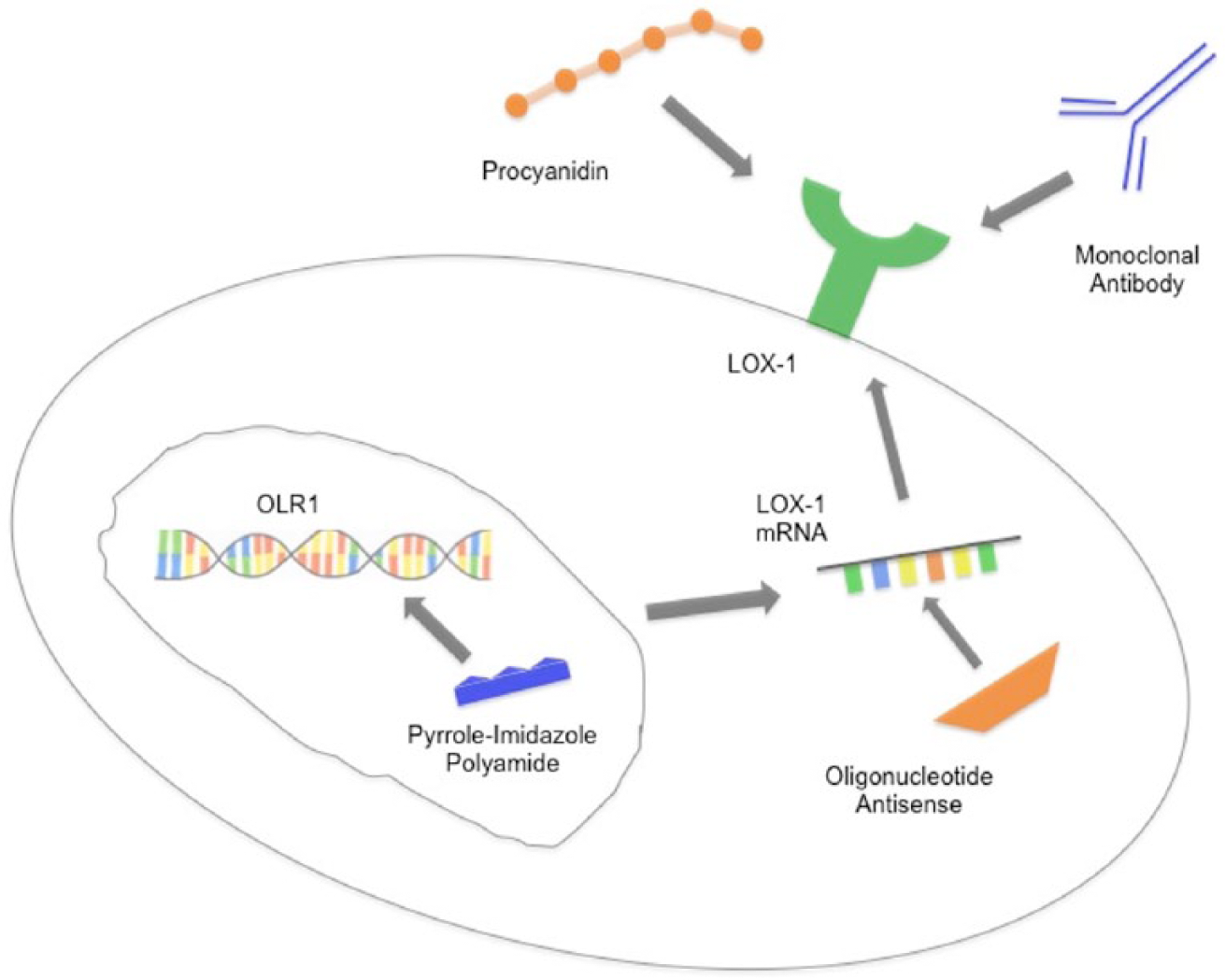
Schematic representation of the novel targeting strategies against LOX-1. Direct protein blockade can be carried out using either monoclonal antibodies or procyanidins that bind to LOX-1 and inhibit ox-LDL uptake. Genetic modulation can be carried out using oligonucleotide antisense molecules or pyrrole–imidazole polyamides. Oligonucleotide antisense molecules bind and inhibit LOX-1 mRNA resulting in downregulation of LOX-1 expression. Pyrrole–imidazole polyamides bind to the minor groove in regulatory sequences of the OLR1 gene, competitively inhibiting specific transcription factors that upregulate LOX-1 expression, which leads to decreased LOX-1 expression.
Potential clinical uses of LOX-1 in diagnosis and therapy
Monoclonal antibodies in plaque imaging
In 2008, Ishino and colleagues used monoclonal antibodies labelled with 99mTc to detect atherosclerotic plaques in Watanabe hyperlipidaemic rabbits. There was significantly more binding to large, advanced atheromas compared with more stable lesions, highlighting the potential use of this antibody in distinguishing plaques at high risk of rupture. 46 However, the mean immunofluorescence intensity detected was low, thereby limiting the potential use of this antibody in molecular imaging.
In 2010, another in vivo study investigated the use of molecular probes targeted against LOX-1 in atherogenic mice. 47 Single-photon emission computed tomography (SPECT) and magnetic resonance imaging (MRI) allowed clear visualisation of the probe binding to LOX-1 in plaques and interestingly, stronger LOX-1 signals were detected in plaques with higher levels of inflammatory cells and increased matrix metalloproteinase-9 (MMP-9) expression. Increased presence of these cells and upregulation of MMP-9 are associated with plaque rupture vulnerability, 48 and therefore, this approach also provides a tool to detect high-risk plaques.
Currently, there are no reliable imaging techniques to detect plaque vulnerability to rupture, 48 and therefore, the above studies, combined with the discovery of higher LOX-1 concentrations in both advanced 49 and unstable plaques, 50 make LOX-1 a possible diagnostic tool for the identification of high-risk lesions.
sLOX-1 plasma levels as a diagnostic biomarker
sLOX-1 was first discovered in 2000 by Murase et al. 8 in cultured bovine endothelial cells and transfected Chinese Hamster Ovarian (CHO) cells. This molecule is formed following the proteolytic cleavage of the extracellular domain of LOX-1 (Figure 1). Two cleavage sites have been identified, and therefore, two forms of this molecule exist. 51 However, clinical studies have not differentiated between these two forms. Currently, the pathophysiological role of sLOX-1 remains unclear and no signalling or scavenging capabilities have been identified; however, studies over the past decade have uncovered its potential as a diagnostic marker in several atherosclerosis-related diseases. 52 Almost a decade ago, Hayashida et al. 53 showed elevated plasma sLOX-1 levels in patients with acute coronary syndrome (ACS) and it was concluded that raised sLOX-1 levels were associated with increased plaque instability. This is consistent with the finding of increased expression of LOX-1 in unstable plaques. 50 Furthermore, LOX-1-mediated ox-LDL activation of endothelial cells is associated with enhanced metalloproteinase expression 54 and these proteases may cleave sLOX-1 from the cellular surface. Another important finding from this study was that peak sLOX-1 levels were detected in patients when they were admitted, suggesting that protein levels were raised prior to onset of symptoms. Moreover, another study found that sLOX-1 levels can differentiate between ACS with and without plaque rupture and ACS with thin-capped fibroatheromas from those without. 55
These findings highlight the potential of sLOX-1 as a biomarker to detect ACS in its earliest stages or even to predict ACS in high-risk patients. Currently, there are no biomarkers that predict ACS, and therefore, sLOX-1 could be particularly useful in the future. Further studies are required to determine the exciting prospect of early detection ACS through measuring sLOX-1 levels, although this will require large studies with careful phenotyping of patients.
LOX-1 as a target for the selective delivery of drugs
Targeting receptors for the selective delivery of drugs is pharmacologically desirable as it minimises unwanted side effects leading to better therapeutic outcomes. LOX-1 is expressed on the main cells involved in atherogenesis as well as on the plaques themselves. 56 Although LOX-1 is located on a number of non-vascular cells such as chondrocytes and intestinal cells,15,57 it is not as widely expressed as other scavenger receptors such as Cluster of Differentiation 36 (CD36). It is therefore an attractive target for the development of selective drug delivery systems.
In 2011, Saito et al. developed a liposome conjugated with an antibody fragment to deliver the drug fasudil, a Rho-kinase inhibitor, into the carotid plaques of rats. It was found that there was a significant increase in the accumulation of fasudil in the plaques when targeted liposomes were used compared with non-targeted liposomes. Although the results of this study cannot confirm that the increased fasudil accumulation was a result of the conjugated LOX-1 antibody, it is the likely cause.
This study suggests that LOX-1 may be feasible for the specific targeting of atherosclerotic plaques and multiple drugs could potentially be delivered to plaques via this mechanism in the future.
Conclusion
Targeting LOX-1 is becoming an attractive strategy for modulating the development and/or progression of atherosclerosis, which may translate into clinical applications. However, this strategy remains in its infancy and we should be cautious with this approach.
LOX-1 is likely to have some degree of physiological function since it is widely expressed and binds a broad range of ligands, including apoptotic and aged red blood cells. Clinically, targeting LOX-1 could, therefore, disrupt physiological activity and further work studying LOX-1 in non-vascular cells and examining the specific ligand-binding effects is needed to establish these alternate functions. Studying the specific cellular effects that each individual ligand exerts could also highlight LOX-1–ligand interactions that are desirable to inhibit.
Currently, a number of different targeting strategies exist that inhibit LOX-1 either through direct blockade of protein function, using monoclonal antibodies, or modulation of production, employing antisense oligonucleotides. These strategies are currently in clinical use, indicating that this is a viable option of targeting LOX-1 in humans. The use of P–I polyamides is an attractive alternative since these molecules inhibit solely the pathological upregulation of LOX-1; however, this technique has not yet been used clinically and it is currently unclear whether this is effective in man.
The potential of LOX-1 extends to the diagnosis of atherosclerosis. Plaque imaging using labelled monoclonal antibodies targeted against LOX-1 has been successful in animal models indicating that this could be achievable in humans. A more simple approach is the use of sLOX-1 plasma levels as a diagnostic or predictive biomarker of vascular pathology, although large-scale prospective studies are needed to fully establish the role sLOX-1 as a diagnostic marker.
In summary, targeting strategies against LOX-1 in the diagnosis and treatment of atherothrombotic disease are beginning to emerge. These strategies are in the early stages of development and have significant potential, but the exact functions of LOX-1 need to be uncovered in order to establish whether clinically targeting this receptor would be effective and safe in the future.
Key messages
LOX-1 was first cloned and identified as a vascular endothelial ox-LDL receptor.
LOX-1 appears to play a key role in atherogenesis through binding and internalisation of circulating ox-LDLs.
Targeting LOX-1 can be carried out using antisense oligonucleotides, pyrrole–imidazole polyamides, anti-LOX-1 antibodies and procyanidins, which represent potential future treatment strategies for the management of atherosclerotic disease.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
