Abstract
Our goal was to test the hypothesis that administration of tetrahydrobiopterin (BH4) would improve impaired endothelial nitric oxide synthase–dependent dilation of cerebral arterioles during type 1 diabetes. In addition, we examined the influence of BH4 on levels of superoxide in brain tissue. In vivo diameter of cerebral arterioles in nondiabetic and diabetic rats was measured in response to endothelial nitric oxide synthase–dependent agonists (acetylcholine and adenosine 5′-diphosphate) and an endothelial nitric oxide synthase–independent agonist (nitroglycerine) before and during application of BH4 (1.0 µM). We also measured levels of superoxide from cortex tissue in nondiabetic and diabetic rats under basal states and during BH4. Acetylcholine and adenosine 5′-diphosphate dilated cerebral arterioles in nondiabetic rats, but this vasodilation was significantly impaired in diabetic rats. In contrast, nitroglycerine produced similar vasodilation in nondiabetic and diabetic rats. Application of BH4 did not enhance vasodilation in nondiabetic rats but improved impaired cerebral vasodilation in diabetic rats. Basal superoxide levels were increased in cortex tissue from diabetic rats, and BH4 reduced these levels to that found in nondiabetic rats. Thus, BH4 is an important mediator of endothelial nitric oxide synthase–dependent responses of cerebral arterioles in diabetes and may have therapeutic potential for the treatment of cerebral vascular disease.
Introduction
Type 1 and type 2 diabetes mellitus impair endothelial nitric oxide synthase (eNOS)- and neuronal nitric oxide synthase (nNOS)-dependent relaxation/dilation of large and small cerebral arteries/arterioles.1–10 Many mechanisms have been proposed to account for impaired eNOS- and nNOS-dependent responses of cerebral arteries/arterioles during diabetes, including the production of a cyclooxygenase constrictor substance, 9 activation of protein kinase C11,12 and an increase in oxidative stress via activation of multiple cellular pathways.8,13–17
Activation of nitric oxide synthases (NOS) has the ability to produce nitric oxide and superoxide.18,19 The production of nitric oxide requires arginine as a substrate and several co-factors [nicotinamide adenine dinucleotide phosphate hydrogen (NADPH), calcium and tetrahydrobiopterin (BH4)] for normal activity. 20 Several studies have shown that diabetes leads to a decrease in levels of BH4 within endothelium of peripheral blood vessels.21,22 In addition, it appears that the biosynthesis of BH4 in the brain is reduced in diabetes. 23 This loss of BH4 would then lead to an uncoupling of eNOS and favour the formation of superoxide over that for nitric oxide.
Many previous studies have shown that acute or chronic supplementation with BH4 can improve/restore impaired eNOS-dependent responses of large and small peripheral arteries/arterioles.22,24–29 In contrast to studies of peripheral arteries/arterioles, there is little information regarding the influence of BH4 on reactivity of cerebral blood vessels during diabetes. A study by Katakam et al. 2 found that impaired relaxation of isolated cerebral arteries in Zucker obese rats (a model of insulin resistance) in response to insulin could be partially reversed by pretreatment with BH4, suggesting a reduction in BH4 bioavailability leading to eNOS uncoupling. However, there is no information available regarding the influence of BH4 on responses of cerebral resistance arterioles to eNOS-dependent agonists during type 1 diabetes. Thus, the first goal of this study was to characterize the influence of acute treatment with BH4 on eNOS-dependent and eNOS-independent responses of cerebral arterioles. Given that oxidative stress is an important component in impaired responses of cerebral arterioles during diabetes, our second goal was to determine the influence of BH4 on superoxide levels in cortex tissue obtained from nondiabetic and diabetic rats.
Methods
Induction of diabetes
All procedures involving rats were reviewed and approved by the Institutional Animal Care and Use Committee. Male Sprague-Dawley rats (200–220 g) were divided randomly into nondiabetic and diabetic groups. All rats had access to food and water ad libitum. One group of rats was injected with streptozotocin [50–60 mg/kg intraperitoneally (IP)] to induce diabetes. The second group of rats (nondiabetic) was injected with vehicle (sodium citrate buffer). Blood samples, for measurement of blood glucose concentration, were obtained 3–5 days after injection of streptozotocin or vehicle and on the day of the experiment.
Preparation of animals
Rats were prepared for studies at 2–3 months after injection of streptozotocin or vehicle. Rats were anaesthetized (thiobutabarbital sodium; Inactin; 100 mg/kg IP), and a tracheotomy was performed. The animals were ventilated mechanically with room air and supplemental oxygen. A catheter was inserted into a femoral vein for injection of supplemental anaesthesia (20–30 mg/kg). A femoral artery was cannulated for measurement of arterial pressure and to obtain a blood sample for the measurement of blood glucose concentration and arterial blood gases.
After these initial procedures, a craniotomy was performed over the left parietal cortex as described previously. 30 Briefly, the rat was placed in a head holder, and an incision was made in the skin to expose the skull. The skin was retracted with sutures and served as a ‘well’ for the suffusion fluid. A cranial window was made in the bone, and the dura was incised to expose the cerebral microcirculation. The cranial window was continuously suffused with a bicarbonate buffer that was bubbled with 95% nitrogen and 5% carbon dioxide. The temperature of the suffusate was maintained at 37°C. The cranial window was connected via a three-way valve to a syringe pump, which allowed for infusion of agonists into the suffusate. This method maintained a constant pH, pCO2 and pO2 of the suffusate during the infusion of agents.
Reactivity of cerebral arterioles
The cranial window was suffused for 30–45 min before testing responses to the agonists. In each rat, we examined responses of the largest pial arteriole exposed by the craniotomy to eNOS-dependent agonists [acetylcholine, 1 and 10 µM and adenosine 5′-diphosphate (ADP), 10 and 100 µM] and a NOS-independent agonist (nitroglycerine, 1 and 10 µM). After an initial examination of vascular reactivity to the agonists, the cranial window was suffused with BH4 (1 µM) for 60 min prior to and during a second application of the agonists. The diameter of cerebral arterioles was measured using a video image shearing monitor.31,32 Baseline diameter was measured immediately prior to application of agonists and during application of agonists at 1-min intervals for 5 min. Steady-state responses to agonists were reached within 3 min after exposure to the drug, and the diameter of cerebral arterioles returned to baseline within 5 min after application of agonist was discontinued. All agonists were prepared using the bicarbonate buffer.
Measurement of superoxide levels
Superoxide levels were measured by lucigenin-enhanced chemiluminescence as described previously.15,33 In separate groups of nondiabetic (n = 8) and diabetic (n = 6) rats, the brain was removed and placed in a Krebs/HEPES buffer (pH 7.4). Samples of cortex tissue, cut from brains of nondiabetic and diabetic rats, were placed in polypropylene tubes containing 5 µM lucigenin. The tubes were then read in a Sirius/FB15 luminometer (Berthold Detections Systems), which reports relative light units (RLU) emitted over a 30-s interval for 5 min. Levels of superoxide reported are the value of tissue plus lucigenin-containing buffer minus background (lucigenin-containing buffer without tissue) and are normalized for tissue weight (RLU/min/mg tissue). We measured the levels of superoxide under basal conditions and following a 30-min pretreatment of cortex tissue with BH4.
Statistical analysis
Analysis of variance (ANOVA) with Fisher’s test for significance was used to compare reactivity of cerebral arterioles to the agonists before and during treatment with BH4, and superoxide levels under basal conditions and during treatment with BH4. Unpaired t-tests were used to compare baseline diameter of cerebral arterioles, blood pressure, blood glucose concentration and body weight. A p-value of 0.05 was considered to be significant.
Results
Control conditions
Baseline diameter of cerebral arterioles was 44 ± 3 µm in nondiabetic rats and 51 ± 1 µm in diabetic rats (p < 0.05). Blood pressure was 106 ± 5 mmHg in nondiabetic rats and 104 ± 7 mmHg in diabetic rats (p > 0.05). On the day of the experiment, blood glucose concentration was 105 ± 9 mg/dL in nondiabetic rats and 393 ± 25 mg/dL in diabetic rats (p < 0.05). Body weight was 454 ± 22 g in nondiabetic rats and 245 ± 30 g in diabetic rats (p < 0.05).
Responses in nondiabetic and diabetic rats
Acetylcholine and ADP dilated cerebral arterioles in nondiabetic and diabetic rats (Figure 1). However, the magnitude of vasodilation was significantly less in diabetic rats compared to nondiabetic rats (Figure 1). In contrast, nitroglycerine produced similar dose-related dilation of cerebral arterioles in nondiabetic and diabetic rats (Figure 1). Thus, impaired responses of cerebral arterioles to acetylcholine and ADP in diabetic rats cannot be explained by nonspecific impairment of vasodilation during diabetes.
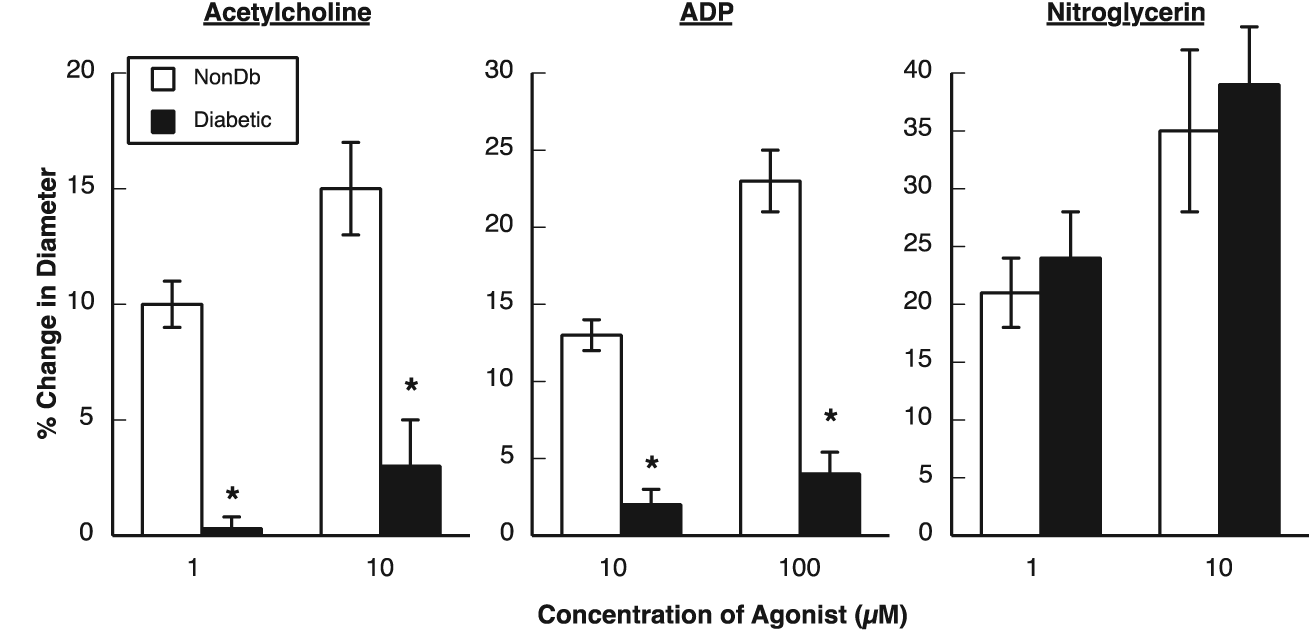
Responses of cerebral arterioles to acetylcholine, ADP and nitroglycerine in nondiabetic (n = 5) and diabetic (n = 6) rats. Values are mean ± SE.
Responses following BH4
Topical application of BH4 (1.0 µM) did not alter baseline diameter cerebral arterioles in nondiabetic rats (42 ± 2 µm before vs 42 ± 3 µm during suffusion with BH4) or diabetic rats (52 ± 2 µm before vs 49 ± 2 µm during suffusion with BH4).
Topical application of BH4 did not alter dilation of cerebral arterioles in response to acetylcholine, ADP or nitroglycerine in nondiabetic rats (Figure 2). In contrast, topical application of BH4 enhanced dilation of cerebral arterioles to acetylcholine and ADP, but not to nitroglycerine, in diabetic rats (Figure 2).
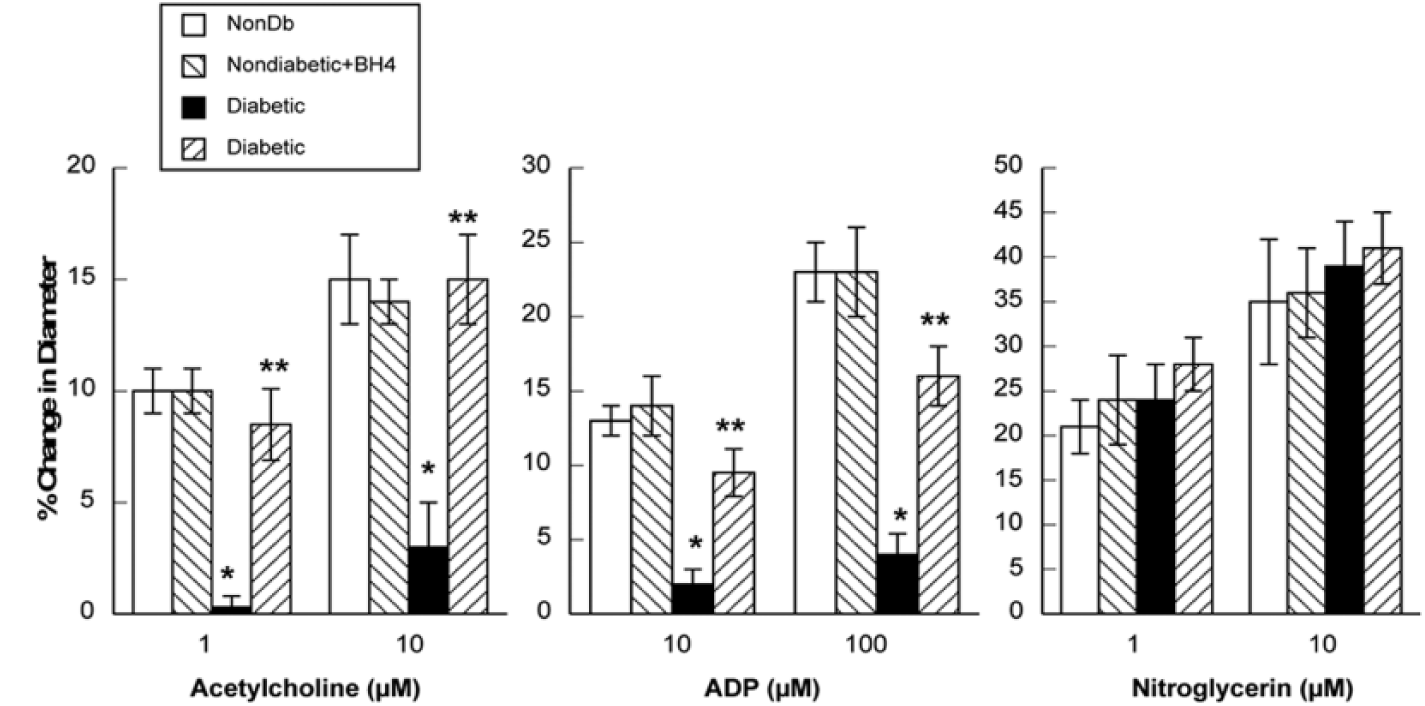
Responses of cerebral arterioles to acetylcholine, ADP and nitroglycerine in nondiabetic and diabetic rats before and during topical application of BH4. Values are mean ± SE.
Superoxide levels
Baseline levels of superoxide were significantly higher in cortex tissue from diabetic than in cortex tissue from nondiabetic rats (Figure 3). Treatment of cortex tissue from nondiabetic rats with BH4 did not alter superoxide levels. In contrast, treatment of cortex tissue from diabetic rats with BH4 produced a significant decrease in superoxide levels (Figure 3).
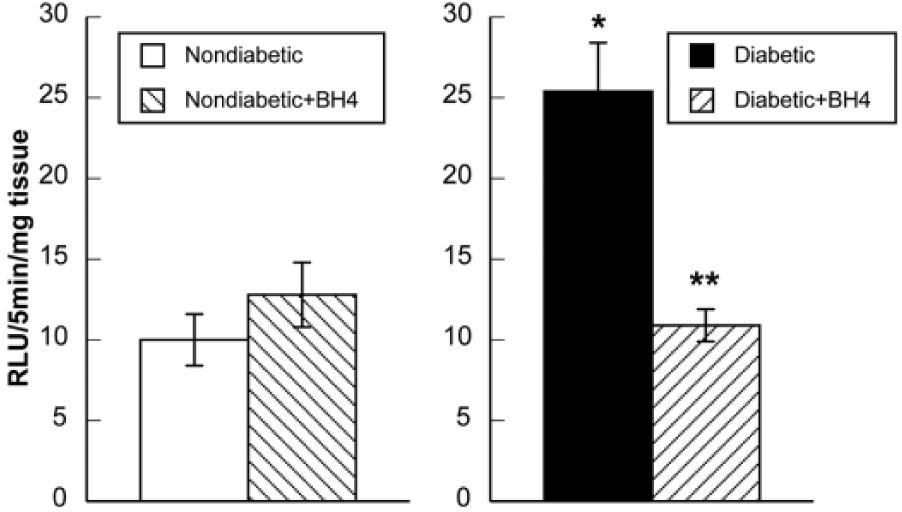
Superoxide levels in brain tissue from nondiabetic (n = 8) and diabetic (n = 6) rats under basal conditions and during exposure to BH4. Values are mean ± SE.
Discussion
There are two major new findings of this study. First, we found that acute treatment with BH4 can improve impaired eNOS-dependent responses of cerebral arterioles during type 1 diabetes. Second, we found that BH4 reduced basal superoxide levels in cortex tissue from type 1 diabetic rats. Thus, it appears that a deficiency of BH4 during type 1 diabetes leads to an uncoupling of eNOS to favour the production of superoxide, which then contributes to impaired eNOS-dependent dilation of cerebral arterioles observed in diabetic rats.
Responses to acetylcholine and ADP
Acetylcholine and ADP were used to examine eNOS-dependent responses of cerebral arterioles. Studies have shown that dilation of large and small cerebral blood vessels in response to acetylcholine is dependent upon the synthesis/release of nitric oxide, presumably via the activation of eNOS.9,34–38 These previous studies found that acetylcholine produced dose-related dilatation of the basilar artery and cerebral (pial) arterioles that was inhibited by enzymatic inhibitors of NOS (L-NMMA and L-NNA). Previous studies also have shown that ADP dilates cerebral arterioles via the synthesis/release of nitric oxide, presumably via activation of eNOS.38–41 Although, others42,43 have suggested that relaxation of the rat middle cerebral artery to purines42,43 and dilation of pial arterioles in mice 44 are related, in part, to the synthesis/release of nitric oxide and to synthesis/release of an endothelium-derived hyperpolarizing factor(s) to activate potassium channels. However, studies by others45–49 have suggested that activation of potassium channels does not play a significant role in dilatation of cerebral arterioles to the agonists used in this study. Based upon these previous findings, we suggest that acetylcholine and ADP are appropriate agonists to evaluate eNOS-dependent dilatation of cerebral arterioles.
Consideration of previous studies
Several studies have examined the influence of exogenous treatment with BH4 or its precursor, sepiapterin, on responses of large and small peripheral blood vessels during type 1 and type 2 diabetes. Pieper 24 reported that relaxation of the aorta from type 1 diabetic rats in response to acetylcholine could be restored to that observed in nondiabetic rats by acute exposure to BH4. Shinozaki et al.22,50 found that impaired eNOS-dependent relaxation of the aorta in insulin-resistant rats (high fructose diet) could be restored by acute treatment with BH4. Pannirselvam et al.27,29 reported that acute and chronic treatment of db/db mice with BH4 or sepiapterin restored eNOS-dependent responses of mesenteric arterioles. Bagi and Koller 51 found impaired flow-mediated dilation of isolated arterioles from the gracilis muscle in type 1 diabetes rats, and that acute treatment with sepiapterin could reduce this impairment. Finally, Alp et al. 52 found that transgenic mice with endothelial-targeted overexpression of guanosine triphosphate-cyclohydrolase I, the rate-limiting enzyme in BH4 synthesis, prevented impaired eNOS-dependent relaxation of the aorta during type 1 diabetes. In addition to studies using animal models of diabetes, studies examining the influence of BH4 have also been conducted using human subjects with diabetes. Heitzer et al. 25 found that flow-mediated dilation was impaired in humans with type 2 diabetes, and acute treatment with BH4 could improve this impairment. In addition, a study by Ihlemann et al. 26 found that an oral glucose challenge in human subjects impaired flow-mediated dilation and this could be reversed by acute treatment with BH4. Thus, it appears that BH4 is an important mediator of eNOS regulation of vascular function in both animals and humans with type 1 and type 2 diabetes.
The mechanisms for the effect of BH4 or sepiapterin on peripheral vascular function during diabetes appear to be related to the effects of BH4 on oxidative stress and nitric oxide production/bioavailability. Previous studies have shown that reduced levels of BH4 during diabetes, due to oxidative degradation, lead to the formation of dihydrobiopterin (BH2).21,22,50 BH2 can then compete for binding to eNOS at the BH4 binding site and thus promotes eNOS uncoupling and a loss of nitric oxide production/bioavailability.20,53,54 Many of the studies discussed above have found increases in oxidative stress and decrease in nitric oxide levels during diabetes that appear to be restored towards that seen in nondiabetic models by treatment with BH4 and sepiapterin.
Consideration of present study
With regard to the cerebral circulation, a very little information exists regarding the influence of BH4 on cerebral vascular function. Katakam et al. 2 have reported that acute treatment of middle or posterior cerebral arteries with sepiapterin improved relaxation in response to insulin in Zucker obese rats. In addition, Hamon et al. 23 have reported reduced levels of BH4 in the brain of type 1 diabetic rats, but no function studies were conducted. This study, to the best of our knowledge, is the first to determine the influence of BH4 on eNOS-dependent responses of cerebral resistance arterioles in vivo during type 1 diabetes. We found that acute treatment with BH4 rescued impaired eNOS-dependent responses of cerebral arterioles observed during type 1 diabetes. Surprisingly, we did not observe an effect of BH4 on baseline diameter of cerebral arterioles. Although we did not expect that BH4 would alter baseline diameter of cerebral arterioles in nondiabetic rats, we did consider the possibility that BH4 would increase baseline diameter in type 1 diabetic rats. However, this was not the case. Our findings are similar to that reported by others for cerebral and peripheral arteries.2,29,51 The precise reason(s) for a lack of an effect of BH4 on baseline diameter in diabetic animals remain unclear.
In addition to our findings regarding cerebral vascular function, we found that basal levels of superoxide were increased in cortex tissue from diabetic rats, and that these levels could be reduced by exogenous treatment with BH4. This finding may suggest that impaired responses of cerebral arterioles during type 1 diabetes may be related to a decrease in the bioavailability of BH4 leading to an increase in the levels of superoxide through the uncoupling of eNOS.
We suggest that the findings from this study may have important therapeutic implications for the treatment of cerebral vascular diseases observed in diabetic patients. Others have reported that BH4, when administered peripherally (oral, intravenous and intraperitoneal) to monkeys, 55 rats 56 and humans, 57 has the ability to cross the blood–brain barrier and enter the central nervous system. Thus, it is conceivable that treatment of type 1 diabetic subjects with BH4, by rescuing cerebral vascular reactivity, could then decrease the risk of cerebral vascular diseases, including stroke and cognitive dysfunction, associated with type 1 diabetes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by the National Heart, Lung, and Blood Institute Grant HL-079587.
