Abstract
Objective
Modified Shenzhu Guanxin Formula (mSGF) has beneficial effects in coronary artery disease. Previously, we found that mSGF inhibited platelet aggregation in rats. In the present study we further investigated the antiplatelet and antithrombotic activities of mSGF in rats.
Methods
Rats were orally administered mSGF (4.2, 8.4, or 16.8 g crude drug/kg), the adenosine 5′-diphosphate (ADP) receptor antagonist clopidogrel (7.875 mg/kg), or saline once a day for 7 days. The effects of mSGF on platelet aggregation were measured. Levels of cyclic adenosine monophosphate (cAMP) and phosphoinositide 3-kinase (PI3K) signaling were analyzed by ELISA and western blotting, respectively. The antithrombotic effect of mSGF was investigated using a FeCl3-induced carotid arterial thrombosis model and effects on bleeding time were assessed in a rat tail transection model.
Results
mSGF significantly inhibited ADP-induced platelet aggregation in a dose-dependent manner, elevated cAMP levels and inhibited phosphorylation of extracellular signal-regulated kinase (ERK) and PI3K/protein kinase B (Akt). Moreover, mSGF dose-dependently inhibited thrombosis in a FeCl3-induced carotid arterial thrombus model and had a significantly smaller effect on bleeding time compared with clopidogrel.
Conclusions
mSGF represents a potent and safe antithrombotic agent whose antiplatelet activity is probably mediated through blockade of PI3K/Akt signaling and increased cAMP generation.
Keywords
Introduction
Platelets are anucleate cells derived from megakaryocytes and play essential roles in both physiological hemostasis and pathological thrombosis. 1 At sites of vascular injury, platelet activation by exposed subendothelial collagen and von Willebrand factor leads to an increase in intracellular Ca2+ concentration as well as altered platelet shape, secretion, and aggregation. Activated platelets release mediators of aggregation, including adenosine triphosphate, serotonin, adenosine diphosphate (ADP), thromboxane A2, and various proteins, which recruit additional platelets from the blood to the injury site.2,3 Platelet adhesion and subsequent aggregation initiate the development of acute thrombotic occlusions at regions of atherosclerotic plaque rupture, leading to progression of ischemic diseases such as myocardial infarction or stroke. 2 Antiplatelet therapy has been established as a cornerstone of prevention and treatment of atherothrombotic diseases. Currently available antiplatelet drugs are hampered by side effects including increased bleeding risk, and therefore priority has been given to the development of safer and more effective antiplatelet agents.
ADP is an important agonist of platelet activation: it is released from platelet granules following activation and further amplifies platelet activation induced by other agonists. 4 ADP activates signaling of G protein-coupled receptor such as the Gq-coupled P2Y1 receptor and the Gi-coupled P2Y12 receptor. The P2Y1 receptor activates phospholipase C-β whereas the P2Y12 receptor activates phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) and extracellular signal-regulated kinase (ERK) and inhibits adenylyl cyclase activity. 5 P2Y12 is more important than P2Y1 for platelet activation and is a key target of antiplatelet drugs including the thienopyridine compounds ticlopidine, clopidogrel, and prasugrel as well as the direct, reversible antagonists ticagrelor and cangrelor.
Herbal medicine play an important role in modifying cardiovascular disease progression, especially through effects on platelet function. 6 Chinese traditional herb formulae have caught our attention for their potent antiplatelet/antithrombotic properties. We have optimized a three-herb traditional Chinese medicine formula, called modified Shengzhu Guanxin Formula (mSGF), consisting of Panax notoginseng (Buck.) F. H. Chen (Araliaceae), Panax ginseng C. A. Meyer (Chinese and Korean ginseng), and Atractylodes lancea (Thunb.) DC. The traditional SGF, formulated by modifying Deng’s Guanxin formula, was created by the Chinese doctor Prof. Deng Tie-Tao; it has been used clinically for more than 30 years and showed marked effects in treating coronary heart disease and angina pectoris. SGF is composed of P. notoginseng, P. ginseng, A. lancea, Panax quinquefolium, Nelumbo nucifera and Pinellia ternata. Our previous clinical studies demonstrated that SGF had extensive pharmacological effects in patients with coronary artery disease, improving hemodynamic indices, alleviating the clinical symptoms of angina pectoris, reducing and delaying acute exacerbation, and improving heart function and exercise capacity.7–10
According to traditional Chinese medicine formulation theory, P. notoginseng, P. ginseng, and A. lancea are mutually complementary as monarch herbs of the formula and represent components with major therapeutic roles. The major bioactive components of P. notoginseng and P. ginseng are saponins (e.g., notoginsenosides and ginsenosides). Both P. notoginseng and P. ginseng have been shown to have significant effects on the cardiovascular system in improving myocardial relaxation function, protecting the arterial endothelium from injury, and exerting anti-inflammatory effects in atherosclerotic lesions of the aorta. P. notoginseng has also been reported to exert antiplatelet effects. P. notoginseng extract inhibited platelet activation induced by ADP in vivo and in vitro, 11 and inhibited platelet adhesion to injured endothelial cells. 12 P. ginseng has been shown to inhibit platelet aggregation and is a candidate for therapy of platelet aggregation-mediated thrombotic diseases.13,14 The combination of P. notoginseng and P. ginseng is widely used in traditional Chinese medicine to treat cardiovascular and thrombotic diseases. Various pharmacological activities of A. lancea have on the cardiovascular system been demonstrated in vitro and in animal models. 15 In addition, A. lancea extract exerted anti-platelet activity in a collagen-induced platelet aggregation model. 16 Theses findings confirmed that P. notoginseng, P. ginseng, and A. lancea have antiplatelet effects.
Considering the properties of these three herbs, SGF was modified to synergistically enhance its anti-platelet and anti-thrombotic effects. The modified formulation, mSGF, has been used to treat coronary heart disease in the Guangdong Provincial Hospital of Traditional Chinese Medicine, Guangzhou, China, but there have been no reports describing the effects of mSGF on platelet function. Our previous experiments found that mSGF inhibited platelet aggregation in rats. The goal of the present study was to assess the effects of mSGF on platelet aggregation in response to ADP and to investigate the underlying mechanisms. The in vivo antithrombotic effects of mSGF were also investigated using a FeCl3-induced carotid arterial thrombosis model.
Materials and methods
Materials
Roots of P. notoginseng (Burk.) F. H. Chen, roots of P. ginseng C.A. Mey and rhizoma of A. lancea (Thunb.) DC. were purchased from Kangmei Pharmaceutical Co., Ltd. (Guangzhou, China). Species of P. notoginseng, P. ginseng and A. lancea were identified by Prof. Wei Li (College of Chinese Materia Medica, Guangzhou University of Chinese Medicine) and voucher specimens were deposited in our laboratory.
Clopidogrel was obtained from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). ADP was purchased from Sigma-Aldrich (St Louis, MO, USA). Cyclic adenosine monophosphate (cAMP) enzyme immunoassay kits were from Yuanye Biotech Co. (Shanghai, China). The bicinchoninic acid protein assay kit and radioimmunoprecipitation assay lysis buffer were from Beyotime (Jiangsu, China). Antibodies against Akt, phospho-Akt(Ser473), p44/42 MAPK (ERK1/2), phospho-p44/42 MAPK (ERK1/2)(Thr202/Tyr204), phospho-(Ser) protein kinase C (PKC) substrate and β-actin were from Cell Signaling Technology (Beverly, MA, USA). The PI3K p110 beta antibody was from Abcam (Cambridge, UK). Enhanced chemiluminescence western blotting detection reagent and polyvinylidene difluoride membranes were purchased from Amersham (Buckinghamshire, UK).
Animals
Male 6- to 8-week-old Sprague-Dawley (SD) rats (specific pathogen-free, 180–220 g) were obtained from the Experimental Animal Center of Guangdong Province. The rats were housed in climate-controlled cages (22–26°C, 40% to 70% humidity) with a 12-hour light/dark cycle and free access to food and water. All animal experiments were approved by the Animal Care and Use Committee of Guangzhou University of Chinese medicine. All animal procedures conformed to the ‘Guide for the Care and Use of Laboratory Animals’ as promulgated by the National Institutes of Health.
Preparation of mSGF
The formula was composed of three herbs (P. notoginseng, P. ginseng, and A. lancea) in a mass ratio of 3:2:3. Briefly, crude herbs were powdered and then extracted with eight volumes (v/w) of 70% ethanol. Circumfluent extraction was performed twice for 2 hours. The ethanol extracts were combined and concentrated to 18.73 g crude drug/mL using a rotary evaporator. The concentrate was stored at 4°C. The extracts were freshly prepared by diluting in distilled water to an appropriate concentration as herb solution for subsequent experiments.
Drug administration and preparation of platelet-rich plasma (PRP)
Male SD rats were randomly divided into five groups (six rats per group). The rats were orally administered mSGF (4.2, 8.4, or 16.8 g crude drug/kg), clopidogrel (7.875 mg/kg) or saline once a day for 7 days. The doses were determined from a preliminary experiment. SD rats were anesthetized with 1% pentobarbital 60 minutes after the last treatment. Blood was then collected form the ventral aorta and transferred into plastic tubes containing 0.1 volumes of 3.8% trisodium citrate as anticoagulant. Blood was centrifuged at 100 ×g for 10 minutes at room temperature to obtain PRP. Platelet-poor plasma (PPP) was obtained by further centrifugation of the remainder of the sample at 1600 ×g for 10 minutes. PRP was adjusted to 4 × 108 cells/mL with PPP.
Platelet aggregation test
Aggregation experiments were performed using an aggregometer (Model LBY-NJ4, PRECIL Corp., Beijing, China) based on the method of Born. 17 PRP (300 μL) was incubated at 37°C for 5 minutes in the aggregometer before adding 5 μL of ADP (5 μM). The maximum platelet aggregation rate within 5 minutes was determined with continuous stirring.
Platelet cAMP assay
At the end of the platelet aggregation assay, 10 mM ethylenediaminetetraacetic acid (EDTA) was added to terminate the reaction. The solution was centrifuged at 2000 × g for 10 minutes at 4°C and then washed twice with phosphate-buffered saline. After freezing at −80°C and thawing at 37°C five times, the platelet suspension was centrifuged at 2000 ×g for 10 minutes at 4°C. The supernatant was subjected to the cAMP enzyme immunoassay as described in the manufacturer’s protocol.
Western blotting
After the end of the platelet aggregation assay, total proteins were prepared using radioimmunoprecipitation assay lysis buffer. Protein concentrations were determined using a bicinchoninic acid protein assay kit. Twenty micrograms of total protein were separated by 10% SDS-PAGE and then transferred onto polyvinylidene difluoride membranes. The membranes were blocked with 5% bovine serum albumin and incubated at 4°C overnight with primary antibodies against ERK, phospho-ERKThr202/Tyr204, phospho-PKC substrate, Akt, phospho-AktSer473, PI3Kp110β, and β-actin. The membranes were then incubated with secondary antibodies at room temperature for 1 hour. After washing with Tris-buffered saline containing 0.1% Tween three times, protein bands were visualized using enhanced chemiluminescence.
FeCl3-induced carotid arterial thrombus model
To assess the effect of mSGF on thrombogenesis, a FeCl3-induced carotid arterial thrombus model developed by Eckly et al. 18 was used with slight modifications. In brief, SD rats were randomly divided into six groups: sham group, model group, formula groups (4.2, 8.4, or 16.8 g crude drug/kg) and clopidogrel group (7.875 mg/kg). The sham and model rats received an equivalent volume of saline alone. All drugs were administered orally once a day for 7 days. Arterial thrombosis was induced 60 minutes after the last treatment. Rats were anaesthetized with 1% pentobarbital and fixed in a supine position. The left carotid artery was exposed and cleaned, and baseline carotid artery blood flow was measured with a miniature Doppler flow probe (1 mm diameter) and a Transonic Model TS420 flowmeter (Transonic Systems, Ithaca, NY, USA). After stabilization of blood flow, vascular injuries were generated by applying a 1 mm-diameter patch of 1M Whatmann filter paper saturated with 10% (w/v) FeCl3 on top of the left carotid artery for 25 minutes. Blood flow was continuously monitored for 25 minutes. At the end of the experiment, the left carotid artery was dissected. Hematoxylin and eosin staining was performed on the artery.
In vivo tail bleeding time study
Tail bleeding time was evaluated by the tail-transection test as previously described. 19 Male SD rats were randomly divided into three groups (six rats per group). The rats were orally administered mSGF (16.8 g crude drug/kg), clopidogrel (7.875 mg/kg) or saline once a day for 7 days. One hour after the last treatment, rats were anaesthetized with 1% pentobarbital and placed on a hotplate to maintain body temperature at 37°C. The tail was transected at a site 3 mm proximal to the tip. Time to blood flow cessation (defined as no bleeding for 15 s) was measured. When bleeding times lasted longer than 20 minutes, measurement was stopped and the bleeding time was recorded as 20 minutes.
Statistical analysis
Data were presented as means ± standard deviations, and n was used to denote the number of experimental replicates. Differences among groups were assessed using one-way analysis of variance followed by either Fisher’s least significant difference or Dunnett’s T3 post hoc tests. Values of P<0.05 were considered statistically significant. All statistical analyses were performed using the SPSS 17.0 statistical package (SPSS, Inc., Chicago, Illinois).
Ethics and consent
All animal experiments were approved by the Animal Care and Use Committee of Guangzhou University of Chinese medicine. All animal procedures conformed to the ‘Guide for the Care and Use of Laboratory Animals’ as promulgated by the National Institutes of Health. There were no human experiments conducted in our study.
Results
mSGF inhibited ADP-induced platelet aggregation
As shown in Figure 1, mSGF inhibited ADP-induced platelet aggregation in a dose-dependent manner over a range of doses (8.4–16.8 g crude drug/kg). Oral administration of mSGF to rats at doses of 8.4 and 16.8 g crude drug/kg body weight for 7 days significantly reduced ADP-induced platelet aggregation from 51.89 ± 2.27% to 45.23 ± 1.71% and 34.89 ± 2.43%, respectively. However, mGSF inhibited ADP-induced platelet aggregation to a lesser degree than clopidogrel (20.99 ± 6.50%). Moreover, mSGF at 4.2 g crude drug/kg had no effect on ADP-induced platelet aggregation.
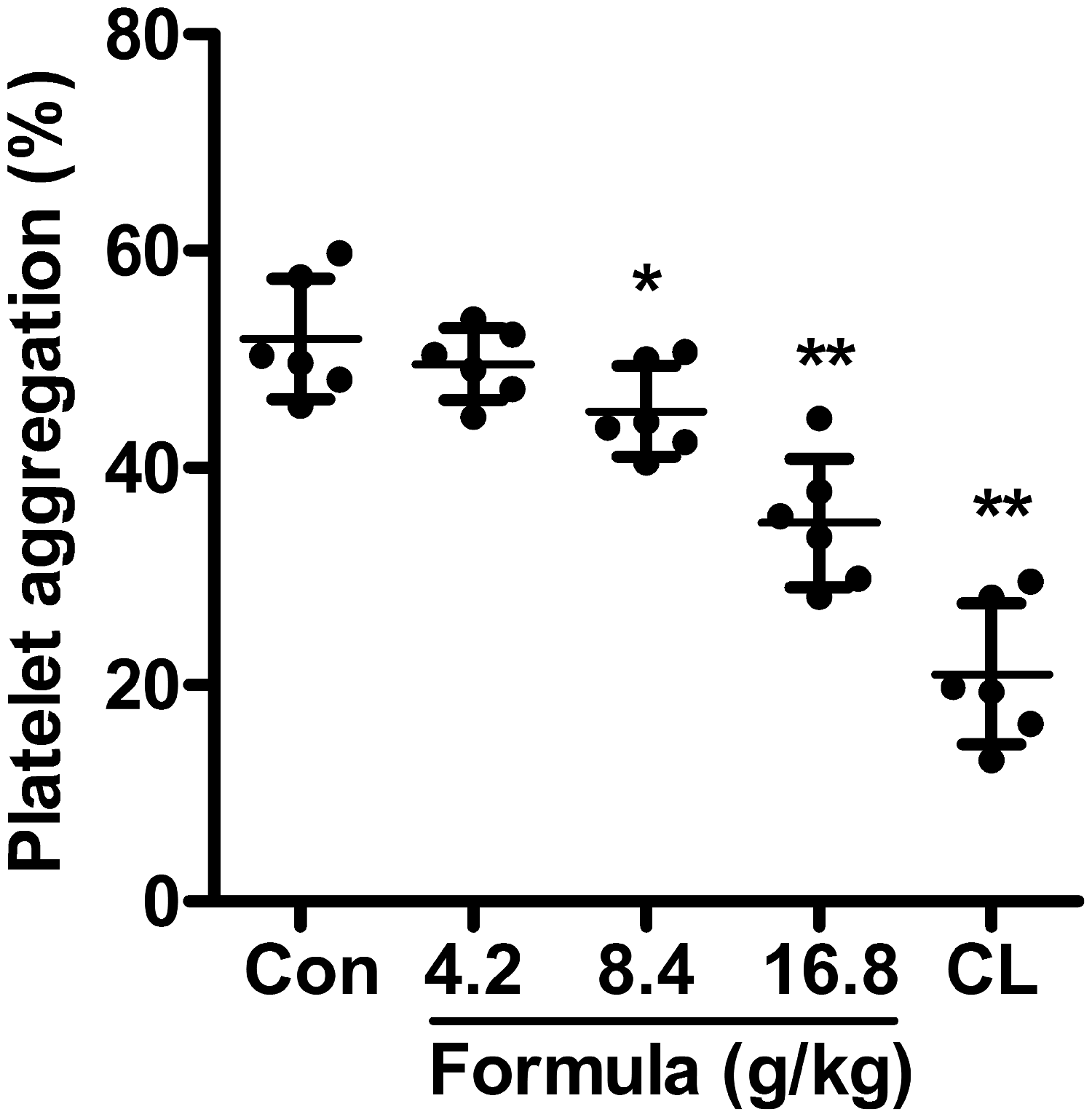
mSGF inhibited ADP-induced rat platelet aggregation. Sixty minutes after the last administration of mSGF (4.2, 8.4 and 16.8 g crude drug/kg), clopidogrel (7.875 mg/kg) or vehicle, blood was collected and platelet aggregation was induced with ADP (5 μM). CL represents the clopidogrel group. Data were expressed as means ± standard deviations, n = 6. *P<0.05, **P<0.01 compared with the control group (Con).
mSGF elevated platelet cAMP levels
Because cAMP formation and resulting protein kinase activation play key roles in inhibiting platelet activation, 20 we next investigated whether mSGF affected cAMP levels in ADP-stimulated platelets. As shown in Figure 2, both mSGF (16.8 g crude drug/kg) and clopidogrel (7.875 mg/kg) significantly elevated cAMP concentrations in ADP-activated platelets. However, lower doses of mSGF (4.2 or 8.4 g crude drug/kg) did not significantly alter cAMP generation. These data indicate that mSGF may suppress platelet activation by promoting cAMP generation.

mSGF elevated cAMP levels in ADP-stimulated rat platelets. PRP was preincubated for 5 minutes and then stimulated with ADP (5 μM) for 5 minutes at 37°C. The reactions were terminated by adding 10 mM EDTA. The cAMP content was measured by enzyme immunoassay. R represents the level of cAMP in resting platelets. CL represents the clopidogrel group. Data were expressed as means ± standard deviations, n = 6. **P<0.01 compared with the control group (Con).
Influence of mSGF on platelet intracellular signaling
To further investigate the mechanisms underlying mSGF inhibition platelet aggregation, levels of signaling molecules such as PI3Kp110β, Akt, ERK and PKC were assessed. The PI3K/Akt pathway is essential for platelet activation and aggregation. In accordance with its inhibition of ADP-induced platelet aggregation, we found that mSGF decreased both PI3Kp110β expression and AktSer473 phosphorylation following platelet aggregation. Moreover, we found that higher doses of mSGF (16.8 g crude drug/kg) administered to rats significantly decreased PI3Kp110β expression and AktSer473 phosphorylation, while lower doses (4.2 or 8.4 g crude drug/kg) produced less inhibition (Figure 3b and c). These results demonstrated that concentration-dependent inhibition of platelet aggregation by mSGF might be mediated through effects on the PI3K/Akt signaling pathway. In addition, ADP-induced ERK phosphorylation was significantly suppressed by mSGF at a dose of 16.8 g crude drug/kg (Figure 3e). Clopidogrel at 7.875 mg/kg showed similar effects on both PI3Kp110β expression and phosphorylation of AktSer473 and ERK as mSGF (16.8 g crude drug/kg). Neither mSGF nor clopidogrel influenced PKC phosphorylation (Figure 3d). Taken together, these data indicated that mSGF affects PI3K-mediated platelet signaling in ADP-activated platelets.

Influence of mSGF on platelet intracellular signaling. Platelets (4 × 108 cells/mL) were preincubated at 37°C for 5 minutes. ADP (5 μM) stimulation was terminated after 5 minutes. Platelet proteins were then extracted and probed with antibodies against PI3Kp110β, Akt, phospho-AktSer473, ERK, phospho-ERKThr202/Tyr204, phospho-PKC and β-actin. (a) Images are representative of three separate experiments. (b–e) The relative protein expression levels were quantified using ImageJ 1.47v software. Data were expressed as means ± standard deviations of at least three independent experiments performed in triplicate. *P<0.05, **P<0.01 compared with the control group (Con).
Effects of mSGF on arterial thrombosis
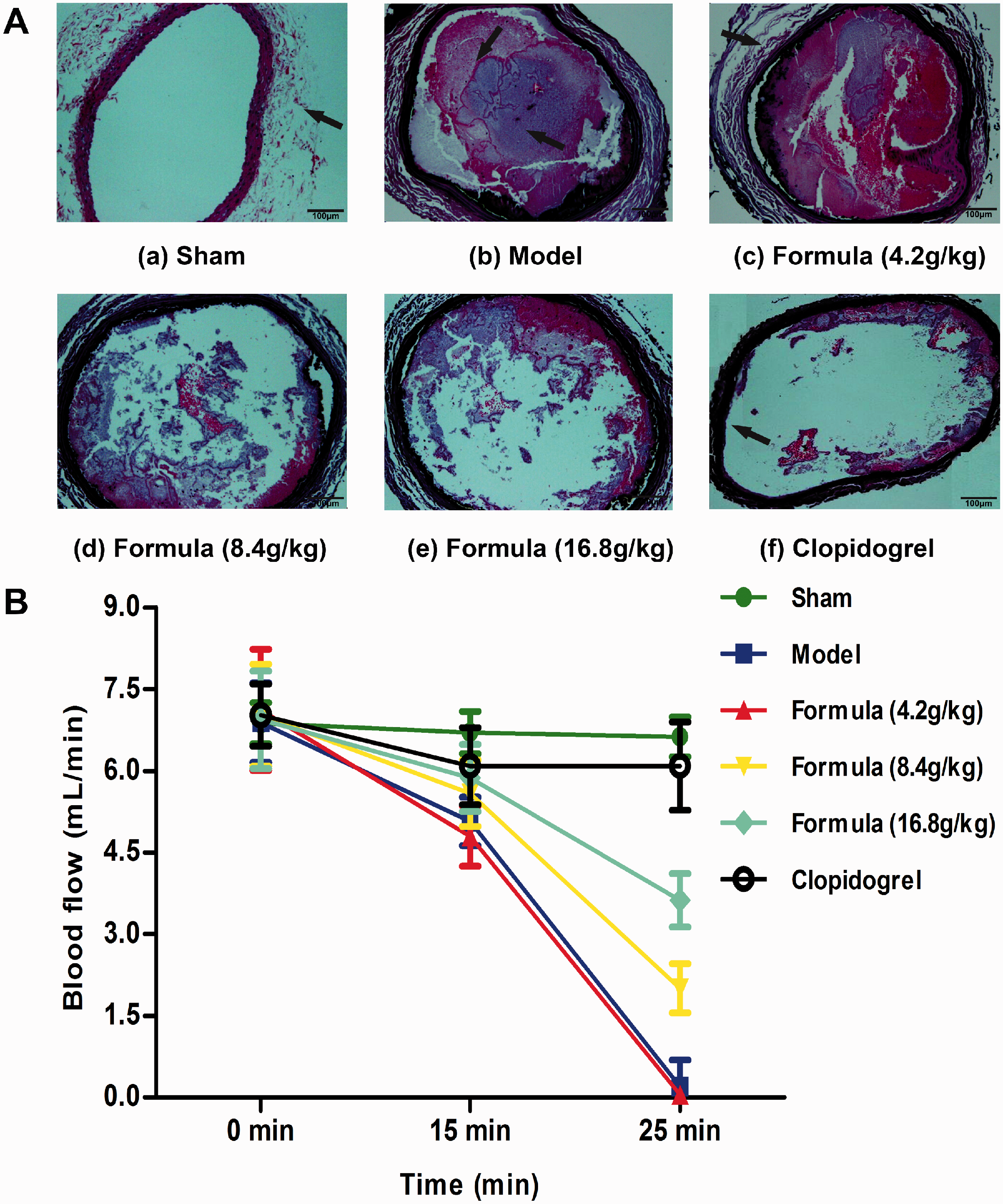
Because platelet aggregation was reduced by mSGF, we speculated that the formula might influence thrombus formation. To investigate the antithrombotic effect of mSGF, arterial thrombosis was induced in rats using 10% FeCl3 in the common carotid artery. Compared with saline-treated rats, arterial blood flow, which indicated the degree of vascular occlusion, was markedly elevated in mSGF- or clopidogrel-treated rats (Figure 4b). Clopidogrel elevated blood flow to a greater degree than mSGF. Complete vascular occlusion in the carotid artery was observed in saline-treated rats following treatment with 10% FeCl3. By contrast, occlusion induced by 10% FeCl3 was markedly and dose-dependently diminished in rats treated with mSGF (8.4 and 16.8 g crude drug/kg) (Figure 4a). Vascular occlusion was also significantly inhibited in rats treated with 7.875 mg/kg clopidogrel (Figure 4a). Taken together, these data suggested that mSGF inhibits thrombus formation through its inhibitory effects on platelet aggregation and/or thrombolysis.
Figure 4. In vivo antithrombotic effect of mSGF in the FeCl3-induced carotid arterial thrombus model. Representative cross sections of carotid arteries are shown from the sham group (a), FeCl3+ saline group (b), FeCl3+ formula (4.2 g crude drug/kg) group (c), FeCl3+ formula (8.4 g crude drug/kg) group (d), FeCl3+ formula (16.8 g crude drug/kg) group (e) or FeCl3+ clopidogrel (7.875 mg/kg) group (f). All sections were stained with hematoxylin and eosin (×100).
Effects of mSGF on bleeding time
The bleeding time was determined in a rat tail cutting model to evaluate potential side effects of mSGF. As shown in Figure 5, mSGF incurred little bleeding risk even at the highest dose (16.8 g crude drug/kg) showing potent antithrombotic effects. The bleeding risk associated with mSGF was significantly lower than that associated with clopidogrel. These results of showed that mSGF may represent an effective and safe antithrombotic agent.
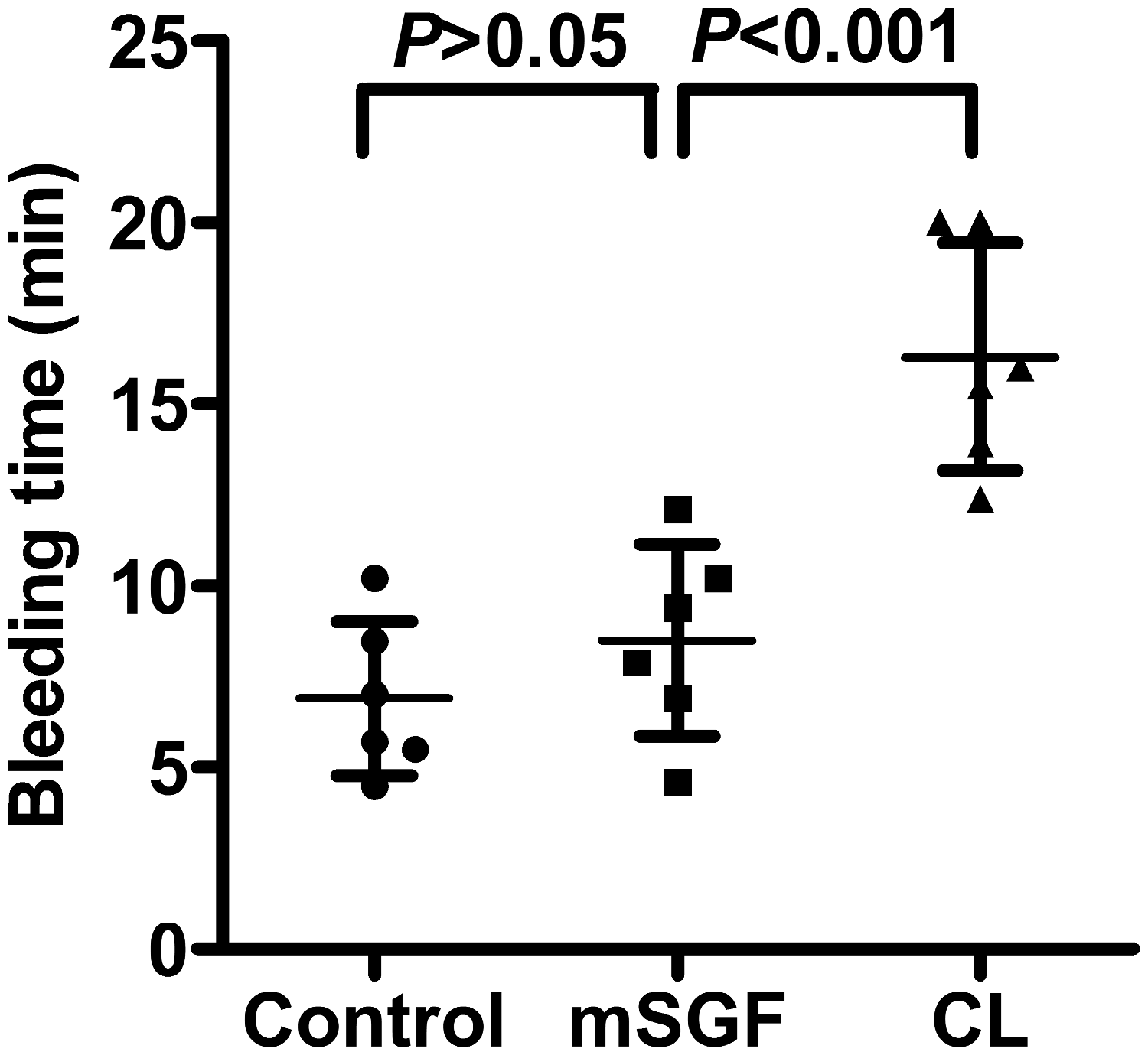
Effect of mSGF on bleeding time. Rats were orally administered mSGF (16.8 g crude drug/kg), clopidogrel (7.875 mg/kg) or saline for 7 days. Sixty minutes after the last administration, the bleeding time was determined in a rat tail transection model. CL represents the clopidogrel group. Data were expressed as means ± standard deviations, n=6.
Discussion
In this study, we found that mSGF suppressed ADP-activated platelet aggregation in a dose-dependent manner over a range of doses (8.4–16.8 g crude drug/kg). The inhibitory effects of mSGF might be attributable to increased cAMP production and marked inhibition of PI3K-mediated platelet signaling, as evidenced by downregulation of PI3K expression along with impaired phosphorylation of Akt and ERK. In accordance with its antiplatelet activities, mSGF prolonged the occlusion time upon vascular injury without prolonging bleeding. Thus, mSGF may be developed as a safe antiplatelet agent for prevention or treatment of thrombotic disorders.
cAMP, as an antiplatelet regulator, plays a critical role in modulating platelet reactivity. Increased intracellular cAMP concentrations inhibit platelet cytoskeleton rearrangement, fibrinogen receptor activation, secretion processes and procoagulant activity. 21 Elevated intracellular cAMP levels inhibit most platelet responses, and reduce intracellular Ca2+ levels by promoting Ca2+ uptake into the dense tubular system, thereby suppressing the activation of PLC and PKC. 21 In this study, we observed that mSGF significantly impaired ADP-induced decreases in cAMP levels within platelets. Therefore, mSGF-mediated inhibition of platelet aggregation is dependent on cAMP generation. The antiplatelet mechanism of mSGF may include blockade of PI3K/Akt signaling with a subsequent elevation of cAMP concentrations and a decrease in Ca2+ mobilization. cAMP formation can be suppressed by Akt activation via enhanced phosphodiesterase activity. 22 However, PI3K/Akt signaling was reported to be suppressed in collagen-activated platelets in the presence of cAMP-elevating agents such as forskolin (an adenylyl cyclase activator) or cilostamide and cilostazol (phosphodiesterase 3 inhibitors). 23 However, our study did not elucidate whether PI3K/Akt or the cyclic nucleotide pathway were involved in upstream signaling, or if the latter pathway involves a negative feedback mechanism. Therefore, exploring the precise mechanisms of interaction between these two signaling pathways in the presence of mSGF should be a goal of further investigations.
PI3Ks, a family of intracellular signaling transducer enzymes, play a crucial role in platelet functional responses and thrombus formation. 24 PI3K isoforms can be divided into three classes based on their primary structures, modes of regulation and substrate specificities. All class I PI3K isoforms (PI3Kα, PI3Kβ, PI3Kδ, and PI3Kγ) can be detected in both human and mouse platelets. 24 Both pharmacological and genetic studies have confirmed that PI3Kβ is critical for regulation of platelet function.25–27 Selective inactivation of PI3Kp110β in mouse models and in human whole-blood thrombosis models leads to defective platelet activation and arterial thrombosis, suggesting the importance of PI3Kp110β in thrombosis.27,28 Knock-out mouse models also indicated the vital importance of PI3Kp110β in platelet function during arterial thrombosis. 25 PI3K activation leads to the phosphorylation of the protein Ser/Thr kinase Akt, a well-established downstream effector of PI3K, which mediates several important platelet responses such as granule secretion, secretion-dependent amplification of platelet aggregation and thrombosis.29–31 Therefore, targeting the PI3K/Akt signaling pathway has become a promising strategy for antithrombotic therapy. In this study, we found that mSGF significantly inhibited both PI3K activation and Akt phosphorylation in parallel with its inhibition of ADP-induced platelet aggregation, an event strongly mediated by P2Y12/PI3K/Akt signaling. However, direct inhibition by mSGF of either PI3K or PI3K and Akt remain to be demonstrated in future studies.
ERK (also called ERK1/2 or p44/p42 MAPK) is expressed in platelets and is phosphorylated by a variety of agonists. ERK plays an important role in the suppression of αIIbβ3 integrin activation and platelet activation. 32 Our current study showed that mSGF significantly inhibited ADP-induced ERK phosphorylation, indicating that the antiplatelet effects of mSGF might be related to regulation of ERK activity. Indeed, the importance of ERK in platelet activation have been firmly established.33,34 Similar to our finding, Garcia et al. 34 reported that ERK was activated downstream of P2Y receptors and was a major regulator of ADP-induced thromboxane A2 (TXA2) generation. Furthermore, the authors demonstrated that PI3Kβ was crucial for Gi plus Gz-mediated platelet aggregation and TXA2 generation through regulation of ERK phosphorylation. 35 It was shown in another recent report that PI3Kβ was important for ADP-induced ERK activation. 36 This implies that mSGF-mediated suppression of ERK phosphorylation might indicate that its actual target is P2Y12/PI3K/Akt signaling. PKC isoform ε has been suggested to negatively regulate ADP-activated platelet functional responses, including Ca2+ mobilization, ERK activation, cytosolic phospholipase A2 activation, and thromboxane generation, to inhibit hemostasis and thrombosis in vivo. 37 However, mSGF had no effects on PKC phosphorylation in ADP-stimulated platelets.
The in vivo antithrombotic effects of mSGF were evaluated in the FeCl3-induced rat carotid arterial thrombus model. We found that mGSF attenuated thrombus formation and vessel occlusion. The FeCl3 injury model is a well-established and widely used experimental system to investigate thrombosis.38,39 FeCl3 induces major oxidative stress accompanied by free radical formation, lipid peroxidation, endothelial cells destruction and platelet accumulation and aggregation, the end result of which is generation of occlusive thrombi. 18 In the present study, mSGF significantly attenuated progression of thrombosis in the FeCl3-induced carotid arterial thrombosis model. The antithrombotic effects of mSGF may be attributed to inhibition of platelet aggregation and/or thrombolysis.
In this study, we found that mSGF showed potent inhibitory effects on both platelet aggregation and thrombosis, although these effects were weaker than those of clopidogrel, a widely used antiplatelet agent. However, all antiplatelet drugs currently available increase bleeding risk at antithrombotic doses, limiting their doses. We also evaluated the bleeding risk of mSGF in a rat tail transection model. Administration of mSGF had no effect on bleeding time up to a high dose (16.8 g crude drug/kg) that showed potent antithrombotic effects. By contrast, clopidogrel markedly prolonged the bleeding time. This demonstrated that mSGF possesses antiplatelet and antithrombotic efficacy without incurring significant bleeding risk. Thus, mGSF represents an effective and safe antiplatelet agent for prevention and treatment of atherothrombotic diseases.
High doses of mSGF (4.2, 8.4 and 16.8 g crude drug/kg) were administrated to the rats, resulting in dose-dependent inhibition of platelet activation. The effective dose of mSGF is about 8.4 g crude drug/kg in rats, and produced significant inhibitory effects on ADP-induced platelet aggregation under these experimental conditions. The human equivalent dose of 8.4 g crude drug/kg in rats is 93.3 g crude drug per person. We will further investigate dosing in in vivo experiments. The aggregation data in this study were obtained in rats. The effective dose of mSGF may differ across species. Therefore, the effects of mSGF on human platelets require further investigation. In addition, the pharmacokinetics and pharmacodynamics of mSGF should be further evaluated to determine whether this dose of mSGF is sufficient and safe.
In conclusion, mSGF inhibits ADP-induced platelet aggregation probably via blockade of the PI3K/Akt pathway in tandem with increased cAMP generation (see Supplementary Material). In addition, mGSF diminishes arterial thrombosis in vivo without incurring significant bleeding risk. Therefore, mSGF may represent a potent and safe therapeutic agent for platelet aggregation-mediated thrombotic diseases. Our findings also indicate that traditional Chinese medicines may provide great promise for screening and development of novel therapeutic agents for thromboembolic diseases.
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520941326 - Supplemental material for Prevention of platelet aggregation and arterial thrombosis using a modified Shenzhu Guanxin Formula
Supplemental material, sj-pdf-1-imr-10.1177_0300060520941326 for Prevention of platelet aggregation and arterial thrombosis using a modified Shenzhu Guanxin Formula by Manting Huang, Huanlin Wu, Jianping Wu, Qiuxiong Chen, Dezhi Zou and Danping Xu in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Guangzhou Science and Technology Plan project (No. 201710010107), the Natural Science Foundation of China (Nos. 81403341 and 81774219), the Guangdong Provincial Science and Technology Plan (No. 2016A020226011), the Joint Innovation Specific Project in Key Areas from the Guangdong Branch Institute of the China Academy of Chinese Medical Sciences (No. ZZ0908065) and the Clinical Research of Lingnan Academic Schools of TCM from Guangdong Provincial Hospital of Chinese Medicine (No. YN2014LN07).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
