Abstract
Purpose:
Insulin sensitivity changes in response to exercise training demonstrate a large variation. Vascular endothelial growth factor A could promote increased insulin sensitivity through angiogenesis. We investigated associations between changes in expression of key genes and insulin sensitivity, aerobic capacity and glycaemic control following exercise training in diabetes mellitus type 2.
Methods:
Subjects with diabetes mellitus type 2 underwent 12 weeks of structured exercise. Euglycaemic clamp, exercise test and HbA1c were performed. Muscle biopsies were obtained for mRNA expression.
Results:
A total of 16 subjects completed the study. Change in vascular endothelial growth factor A expression was positively associated with an increase in insulin sensitivity (
Conclusion:
The variation in physical adaptation to exercise training in diabetes mellitus type 2 was associated with changes in expression of vascular endothelial growth factor A in muscle. This difference in induced gene expression could contribute to the variation in exercise training effects on insulin sensitivity. Measures of capillary blood flow need to be assessed in future studies.
Keywords
Introduction
The physiological response to exercise demonstrates a large inter-individual variation in healthy individuals. This is seen both when measured as maximal aerobic capacity (VO2max) 1 and insulin sensitivity. 2 It has been postulated that the individual adaptation to exercise training must be considered when studying exercise-induced gene expression. 1 A recent study suggested that muscle adaptation to exercise was impaired in healthy middle-aged men with heredity of type 2 diabetes mellitus (T2DM) compared to those without heredity for the disease. 3
The mechanisms contributing to improved insulin sensitivity by exercise in T2DM are not fully known but presumably involve both metabolic and structural adaptations. A central event in the remodelling process of skeletal muscle is increased number of capillaries. This leads to a larger endothelial surface area and enhanced nutritive blood flow to skeletal muscle cells, which presumably enhance glucose uptake, lipoprotein metabolism and augmentation of insulin sensitivity.4–6 Moreover, insulin-mediated capillary recruitment and endothelium-dependent vasodilatation are impaired in T2DM.7,8 Among numerous pro-angiogenic factors, vascular endothelial growth factor A (VEGFA) and its receptors, as well as the angiopoietin signalling pathway (Ang1 and 2), are regarded as major regulatory growth factors. 9 Expression of these growth factors change with exercise towards a state known to stimulate vascular growth.10–12
Another exercise-induced change is activation of peroxisome proliferator–activated receptor γ coactivator-1α (PGC-1α). This factor is associated with an increased risk of developing T2DM and suggested to be down regulated in T2DM. 13 Initially, PGC-1α was described to regulate mitochondrial biogenesis 14 but has also been shown to regulate substrate metabolism, angiogenesis and fibre hypertrophy in skeletal muscle.15,16
We studied if associations exist between changes in expression of these key genes and improved insulin sensitivity, VO2max and glycaemic control following exercise training in T2DM, in the context of physiological adaptation.
Patients and methods
Study population
The subjects were recruited from referrals to our diabetes outpatient day care unit and by advertising in local newspapers. They should have a diagnosis of T2DM established at least 3 months prior to inclusion. Subjects who, at the start of the wash-out period, had HbA1c <7.5% (59 mmol/mol) aged 45–60 years and body mass index (BMI) 25–30 kg/m2 were eligible for the study. The subjects were treated with diet and maximum one oral glucose lowering agent. None of the study subjects had previously participated in any regular exercise programme with more than one training session per week.
Study design
This study was performed within a previously reported exercise study.
17
Following a wash-out period of antidiabetic medication for 6 weeks, 19 individuals within the larger exercise study were randomised to undergo muscle biopsies for this study. The initial study protocol included randomisation to either exercise alone (
Intervention
The exercise intervention has been described in detail. 17 In short, the participants performed a 50 min combined aerobic and resistance training group programme, three times per week. A specialised physiotherapist led and supervised each training session. Two exercise targets were used: (1) an individual exercise intensity of ⩾50% of maximal exercise capacity, based on the maximal heart rate reached during the baseline exercise test, during at least 40 min and (2) ⩾80% during three heavier periods of 3–4 min, engaging large muscle groups for training of central circulation. To ascertain that the targeted individual exercise intensity was reached, a computed online heart rate recording system was used (Activio AB, Stockholm, Sweden) on three different occasions during the study.
The target dose of acarbose (Bayer AG) was 100 mg three times daily to be taken with major meals. The dose was up-titrated during the first 4 weeks of intervention. A total of 50 mg was added every week, reaching the dose 50 mg t.i.d. the third week. The fourth week the dose was increased to 100 mg t.i.d.
Assessments
The hyperinsulinemic euglycaemic clamp investigation has been described previously. 17 The target blood glucose was 5.0 mmol/L. 18 The glucose infusion rate at steady state assessed insulin sensitivity (M) and was corrected for lean body mass. Insulin sensitivity index (M/I) was calculated by dividing M by the mean insulin levels during steady state. Dual X-ray absorptiometry (DXA) was used to determine total fat mass and lean body mass. VO2max was assessed during an exercise test performed on a bicycle ergometer and corrected for lean body mass. Glucose was determined by a glucose oxidase method. HbA1c was analysed using the high-performance liquid chromatography (HPLC) Mono-S method and recalculated to International Federation of Clinical Chemistry (IFCC) and National Glycohemoglobin Standardization Program (NGSP) values.
Gene expression
Muscle biopsies were obtained from the vastus lateralis in resting state. In the end of the study, the biopsies were taken 24–48 h after the last bout of exercise. Quantification of mRNA expression of selected genes included VEGFA and its receptors 1 and 2, Ang1 and Ang2, and also PGC-1α. Detection of mRNA was performed on an ABI-PRISM® 7700 Sequence Detector (Applied Biosystems Inc, Foster City, CA, USA). Oligonucleotide primers and TaqMan® probes were designed as described earlier.12,19 18S rRNA was selected as an endogenous control to correct for potential variations in RNA loading (4310893E, Applied Biosystems Inc.). All reactions were performed in 96-well MicroAmp Optical plates, using the ABI-PRISM® 7700. For every gene, all samples were amplified simultaneously in duplicate in one assay run. Relative quantification of the samples was carried out using dilution curves for each target gene analogous to a standard curve. The relative distribution of the targets was calculated for each individual. A threshold cycle (ΔCt) value was obtained by subtracting 18s Ct values from respective target gene Ct values. The relative expression of each isoform was then calculated by 2−Δ–ΔCt and presented as arbitrary units.
Statistical analysis
All continuous variables are presented as median (interquartile range). The Wilcoxon signed-rank test was used to assess pre- and post-intervention differences within the intervention groups, and the Mann–Whitney
Results
In total, 16 subjects, 4 women and 12 men, completed the study per-protocol. Three men in the acarbose-treated group were excluded from the analyses: two due to non-compliance and one that withdrew at will. The basic characteristics and pre- and post-intervention findings are presented in Table 1.
Basic characteristics and pre- and post-intervention findings.
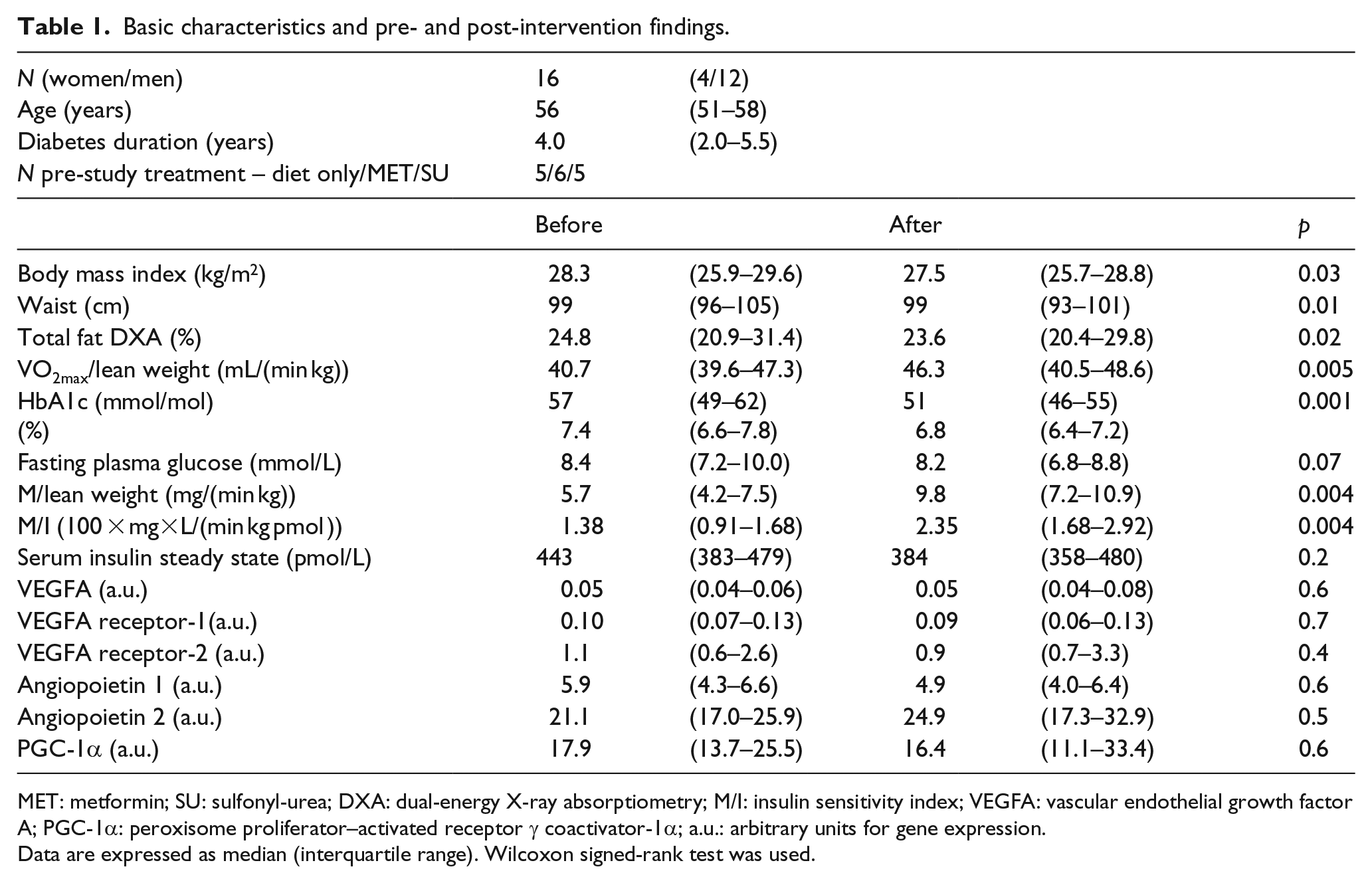
MET: metformin; SU: sulfonyl-urea; DXA: dual-energy X-ray absorptiometry; M/I: insulin sensitivity index; VEGFA: vascular endothelial growth factor A; PGC-1α: peroxisome proliferator–activated receptor γ coactivator-1α; a.u.: arbitrary units for gene expression.
Data are expressed as median (interquartile range). Wilcoxon signed-rank test was used.
Most clinical variables improved significantly by the intervention, but no significant changes were seen in gene expression. The two different groups (exercise vs exercise + acarbose treatment) did not differ significantly at baseline or in relative change at the end of the study, except for a lower VO2max at baseline in the acarbose-treated group (
Adjusted results by multiple regression showed that the relative change in VEGFA expression was positively associated with a relative increase in M/I (
Adjusted results by multiple regression analyses.
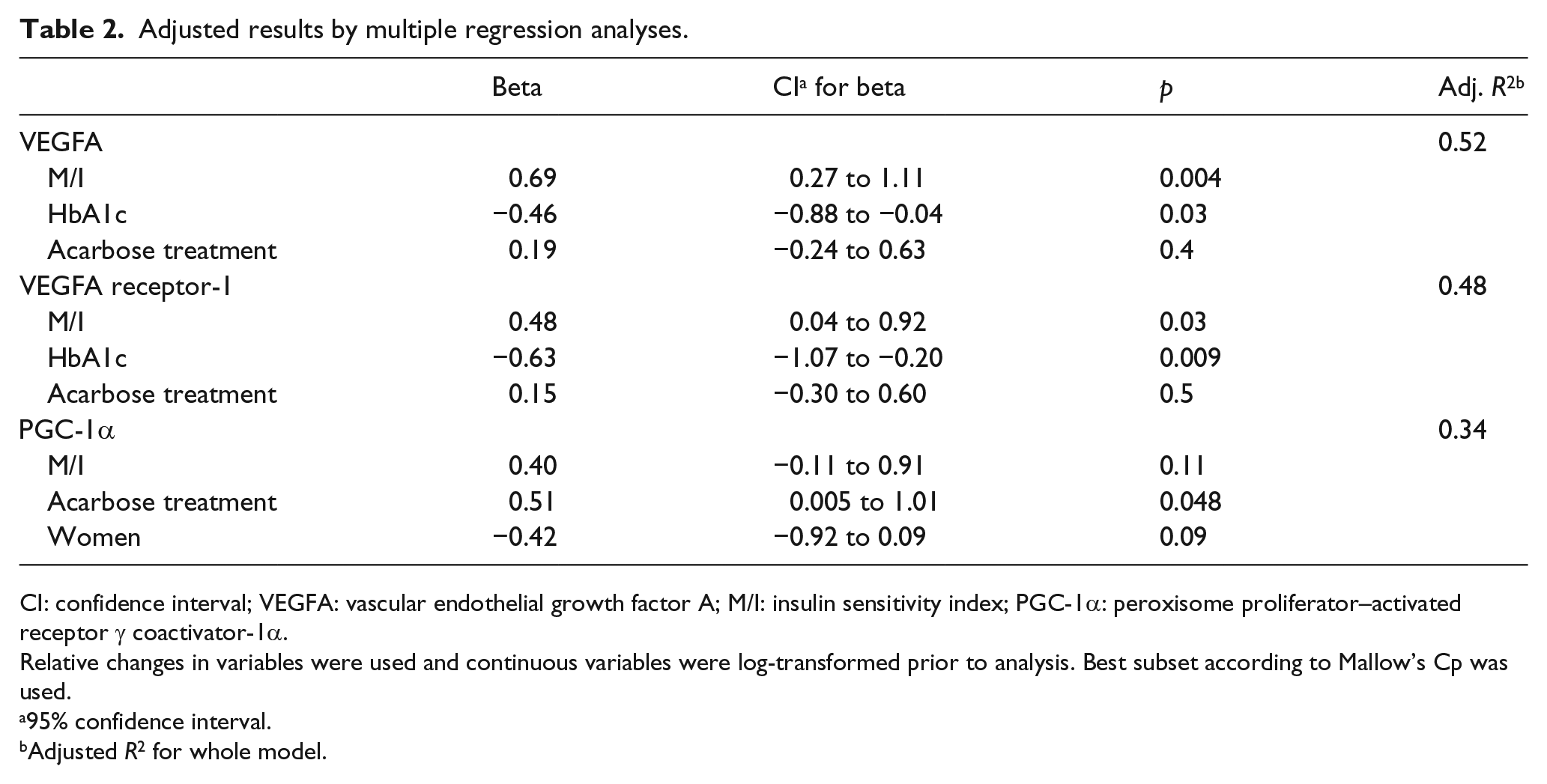
CI: confidence interval; VEGFA: vascular endothelial growth factor A; M/I: insulin sensitivity index; PGC-1α: peroxisome proliferator–activated receptor γ coactivator-1α.
Relative changes in variables were used and continuous variables were log-transformed prior to analysis. Best subset according to Mallow’s Cp was used.
95% confidence interval.
Adjusted
Change in VEGFA receptor-1 expression was also positively correlated with an increase in M/I (
No associations between gene expressions and VO2max were found. Furthermore, relative change in VEGFA receptor-2, Ang1 and Ang2 expression did not show any significant associations with the outcome variables (data not shown). The acarbose therapy differed to exercise alone only in respect to a larger relative increase in PGC-1α (
Discussion
The mechanisms underlying insulin resistance has been extensively investigated during the last decades. Defects in insulin signalling have been shown in muscle and other tissues. The relative contribution of vascular blood flow has been more controversial. 20 Insulin increases vascular flow by capillary recruitment. The vascular flow per se augments the delivery of insulin and glucose to the myocytes, resulting in an increased insulin-mediated muscle glucose uptake.21,22 Exercise has been shown to enhance insulin-mediated capillary recruitment and glucose uptake. 22 The number of capillaries in muscle is positively correlated to peripheral insulin action. 23 Recently, this was shown to be a primary phenomenon in a study on mice with a muscle-specific genetic deletion of VEGFA. A 60% reduction in capillary density resulted in a 40%–45% decrease in insulin-stimulated glucose uptake in muscle. 24
Our current observations of VEGFA expression add a possible regulatory mechanism behind the reported inter-individual differences in response seen with exercise training. VEGFA has been demonstrated to be crucial in exercise-induced angiogenesis. 25 The effect of VEGFA on pro-angiogenesis could involve (1) an increased endothelial cell surface and/or (2) increased local perfusion with reduced diffusion distance between microcirculation and skeletal muscle cells. Since VEGFA is a major angiogenic growth factor and increased capillary supply of the skeletal muscle enhances substrate and metabolite exchange, 9 this would be the most likely mechanism behind the variation in insulin sensitivity.
A further support in this study for an activation of the VEGFA pathways was the positive association between VEGFA receptor-1 expression and insulin sensitivity. VEGFA receptor-1 is mainly expressed in endothelial cells and is one of the receptors ensuring the action of VEGFA in muscle tissue. 26
The VEGFA findings were also associated with improved glycaemic control, statistically independent of the improvement in insulin sensitivity. Some data suggest that exercise may improve beta cell function in sedentary overweight subjects. 27
The degree of PGC-1α mRNA change showed an association with improved insulin sensitivity when measured as M, but this was not significant when the more accurate M/I was used. The involvement of PGC-1α in T2DM has been suggested from reports of decreased expression of PGC-1α in subjects with T2D as well as in those with heredity for the disease. 28 PGC-1α has mainly been associated with mitochondrial biogenesis but also plays a crucial role in numerous of other remodelling processes.15,16 In mouse skeletal muscle, PGC-1α has evolved as a principal regulator of exercise-induced expression of VEGFA through co-activation of the nuclear receptor ERRα (oestrogen-related receptor-α).15,29,30 However, our clinical study in humans could not support this association between VEGFA and PGC-1α.
We demonstrate that the variation in physical adaptation to exercise in T2DM is associated with changes in VEGFA mRNA expression in muscle. Endpoint effects like capillary density or microvascular blood flow were not measured, which would be desirable in future studies to confirm the impact of our VEGFA expression findings. The difference in training-induced expression of this factor could participate in the known variation in the exercise training–generated changes in insulin sensitivity.
Footnotes
Acknowledgements
H.W. researched data and wrote the manuscript, H.F. researched data and reviewed the manuscript, M.D. collected data and reviewed/edited the manuscript, M.A. interpreted data and reviewed the manuscript and T.G. researched data and wrote the manuscript. Primary data are uploaded as supplementary material. The authors thank P.N. Båvenholm and S. Efendic for planning and supervising the original exercise study. The authors also wish to express their appreciation to research nurses K. Sundquist and B. Ljungström at Karolinska University Hospital and A.-S. Andersson, A. Bylund and N. Blommé at Ersta Hospital; to surgeons J. Nygren and A. Thorell at Ersta Hospital; to the physiotherapists supervising the training; and finally to all volunteers for their participation in the study. Preliminary results of this study was presented as an abstract at the 12th European Congress of Endocrinology, 24–28 April 2010 in Prague, Czech Republic: Wagner H, et al. Variation in response to exercise in type 2 diabetes associated to VEGFA and PGC1-α expression. Endocrine Abstracts (2010) 22 OC1.6.
ClinicalTrials.gov identifier: NCT01244971; URL: ![]() .
.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Swedish Medical Research Council and by an unrestrictive grant from Bayer HealthCare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
