Abstract
Hemophagocytic lymphohistiocytosis (HLH) is an aggressive and life-threatening syndrome of excessive immune activation. Mesenchymal stem cells (MSCs) generate an immunosuppressive microenvironment by secreting cytokines and have been used to treat autoimmune diseases. We report the first case of refractory secondary HLH treated with umbilical cord MSCs. A 52-year-old Chinese female patient with a history of type 2 diabetes was diagnosed with refractory secondary HLH based upon the HLH-2004 protocol and was treated by infusion of third-party umbilical cord MSCs (1.4 × 106 cells/kg of body weight, 70 × 106 cells in total) from the stem cell bank of Hainan Province. Body temperature recovered to normal on the sixth day after infusion with umbilical cord MSCs, and the levels of inflammatory factors macrophage inflammatory protein (MIP)-1α, interleukin (IL)-12p70, stromal cell-derived factor (SDF)-1α, and IL-7 decreased significantly. Blood glucose levels were significantly lower than before treatment, and the amount of insulin needed was significantly reduced. Umbilical cord MSCs can relieve the symptoms of refractory secondary HLH and have a therapeutic effect on insulin resistance in type 2 diabetes mellitus.
Keywords
Introduction
Hemophagocytic lymphohistiocytosis (HLH) is a rapidly progressive, life-threatening syndrome of excessive immune activation. If left untreated, patients with HLH survive for only a few months because of progressive multiorgan failure. Although the advent of the HLH-94 protocol 1 has dramatically increased the survival rate, the mortality associated with HLH remains high. In particular, patients with refractory secondary HLH of unknown origin cannot undergo hematopoietic stem cell transplantation, and optimal treatments remain under debate. 2 Familial HLH has been treated by using third-party mesenchymal stem cells (MSCs), although it is unknown whether refractory secondary HLH of unknown origin can be treated with MSCs. 2 There are currently no reports of this approach in the literature. Herein, we share a report of the treatment of refractory secondary HLH of unknown origin with third-party umbilical cord MSCs. Moreover, treatment with MSCs was found to have a therapeutic effect on insulin resistance in this patient with type 2 diabetes.
Case presentation
Consent for publication of patient information and images was given by the patient’s legally authorized representative.
A 52-year-old Chinese female was admitted because of high fever (40°C) and fatigue that had lasted for 7 days in June 2017. Before admittance, she had been treated with antipyretic drugs and cephalosporin, the details of which are unknown. There were no obvious abnormalities in the examination. A complete blood count showed a white blood cell count of 3.87 × 109/L, a hemoglobin level of 9 g/dL, and a platelet count of 90 × 109/L. Splenomegaly was found by color Doppler ultrasound. Hypertriglyceridemia was noted (triglycerides: 7.49 mmol/L), ferritin was >2000 ng/mL, blood glucose was 8.37 mmol/L, glycosylated hemoglobin was 6.9% (reference value 4.2%–6.5%), and there was low natural killer (NK) cell activity (35.6%, reference value 47.6%–76.8%). Bone marrow biopsy found hemophagocytosis, and flow cytometry analysis did not detect blasts. The blood clotting test was normal. The expression of perforin 1 (PRF1), Unc13 homolog D (UNC13D), syntaxin 11 (STX11), and syntaxin-binding protein 2 (STXBP2) genes was negative. The following tests were all in the normal range: α-fetoprotein, carcinoembryonic antigen, carbohydrate antigen 125, carbohydrate antigen 242, keratin 19 fragment, human chorionic gonadotropin, and carbohydrate antigen 199; antinuclear antibody and extractable nuclear antigen; complement C3 and C4; Coombs test; bilirubin; CD55+ and CD59; Epstein-Barr and cytomegalovirus DNA; and herpes simplex virus (HSV)-1 immunoglobulin (Ig)M, HSV-2-IgM, rubella-IgM, toxoplasmosis (TOX)-IgM, TOX-IgG, and T-SPOT.TB (interferon-γ release assay). The serum galactomannan or β-glucan test was negative. Blood culture was negative, and treatment with doxycycline, imipenem and cilastatin sodium, and vancomycin did not relieve the patient’s fever. Pulmonary infection was not found by lung computed tomography, and enlarged lymph nodes were not detected by positron emission tomography–computed tomography. The patient was diagnosed with secondary HLH of unknown origin and type 2 diabetes mellitus based on the HLH-2004 protocol on the fifth day after admittance. No similar history existed in the family.
The patient received treatment based on the HLH-94 protocol. 1 After 15 to 16 weeks of chemotherapy, the patient became febrile. The anti-infection drug was ineffective; thus, we considered disease relapse. The patient was then treated again with the HLH-94 protocol and hematopoietic stem cell transplantation was suggested. However, the patient declined the stem cell transplant for financial reasons. The patient relapsed with HLH after the second HLH-94 treatment in the seventh week. The patient received CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) chemotherapy after the second relapse, but the noninfectious high fever returned with prednisone withdrawal, indicating that CHOP chemotherapy was not effective. She had continuous fever, high blood sugar, high-dose insulin treatment, intractable hypoproteinemia, and whole-body edema. She refused chemotherapy and her ECOG/WHO/Zubrod score was 4. After discussion, the patient agreed to an experimental MSC treatment. The patient gave informed consent in accordance with the Declaration of Helsinki, and the treatment was approved by the Ethics Committee of the Affiliated Haikou Hospital Xiangya School Central South University. MSCs were donated by the stem cell bank of Hainan Province. Frozen cells (1.4 × 106 cells/kg of body weight; 70 × 106 cells in total) were thawed before infusion. Five milligrams of dexamethasone was used to prevent adverse reactions before MSC infusion. Because the first patient who received MSCs for HLH treatment died of fungal infection, 3 we administered voriconazole to prevent fungal infection on the day of MSC infusion. The day of infusion of MSCs was considered day (d) 0.
The data were analyzed using SAS software, version 8.1 (SAS Institute Inc., Cary, NC, USA), and p-values < 0.05 were considered statistically significant. The analysis method was one-way ANOVA, and the homogeneity of variance was tested by the Bartlett, Brown-Forsythe, and Levene tests.
Three days before infusion of MSCs, the patient’s average maximum temperature was 39.3°C, and the average maximum temperature during d 1 to 6 decreased to 38.5°C (p < 0.05). The patient’s temperature returned to normal on the sixth day after infusion of umbilical cord MSCs. The average maximum temperature on d 7 to 12 further decreased to 36.8°C (p < 0.05), and the highest temperature after d 13 was 37.9°C, which was slightly higher than the temperature from d 7 to 12 (p < 0.05) but lower than the temperature before treatment (p < 0.05) (Figure 1a and 1b).

The patient’s maximum body temperature (°C) on different days of mesenchymal stem cell (MSC) infusion. (a) Scatter plot of different days and body temperature before and after infusion of MSCs. Body temperature could be divided into four time groups relative to infusion of MSCs on d 0: d −3, −2, −1 (group 1), d 1 to 6 (group 2), d 7 to 12 (group 3), and d 13 to 19 (group 4) of infusion MSCs. (b) The average highest body temperature was 39.3°C, 38.5°C, 36.8°C, and 37.9°C, respectively, in the 4 groups. The box plot indicates the interquartile range (box), the median (line), and the range of values (whiskers). After MSCs infusion, body temperature decreased and the differences in body temperature between each group were significant (p < 0.05).
In total, 34 inflammatory factors were detected on d 0, d 7, and d 14 by Luminex multiplex assay (Thermo Fisher Scientific, Waltham, MA, USA). Of these, macrophage inflammatory protein (MIP)-1α and interleukin (IL)-12p70 were normal before infusion and decreased after infusion (Figure 2a), whereas stromal cell-derived factor (SDF)-1α and IL-7 were increased before infusion and decreased after infusion (Figure 2b and 2c). Therefore, MSCs might regulate the effect of immunotherapy on HLH by decreasing levels of MIP-1α, IL-12p70, SDF-1α, and IL-7. Levels of interferon (IFN)-γ-induced protein (IP)-10, RANTES (regulated on activation, normal T cell expressed and secreted), and monocyte chemoattractant protein (MCP)-1 increased before infusion, decreased after infusion, and then increased at d 14 (Figure 2d). These results suggested that MSCs might regulate IP-10, RANTES, and MCP-1 in HLH treatment and insulin insensitivity because changes in these factors were consistent with fluctuations in the patient’s blood glucose. IL-10 was increased before infusion and higher after infusion (Figure 2e), indicating that IL-10 was involved in the pathogenesis of HLH, and that MSC did not exert an effect on HLH through IL-10. Eotaxin was normal during treatment, indicating that eotaxin was not involved in the pathogenesis of HLH. Because of the small sample size, the mechanism of individual inflammatory factors in this patient could not be determined. Levels of IL-27, IL-4, IL-5, IL-6, IL-8, IL-13, IL-17A, IL-31, granulocyte-macrophage colony-stimulating factor (GM-CSF), tumor necrosis factor (TNF)-α, MIP-1α, MIP-1β, IFN-α, IL-9, TNF-β, IL-1α, IL-23, IL-15, IL-21, and IL-22 were lower than normal. IL-1 receptor agonist decreased before infusion, was normal after infusion, and then increased. Levels of IFN-γ and IL-18 were higher than normal before infusion and remained higher after infusion. Before transfusion, growth-regulated oncogene (GRO)-α was normal; after infusion, it decreased and then increased again.

Changes in inflammatory factors (pg/mL) at different time periods. (a) MIP-1α (red) and IL-12P70 (pink); (b) SDF-1α; (c) IL-7; (d) RANTES (pink), IP-10 (red), and MCP-1 (orange); and (e) IL-10. Time periods: group 1 = 1 week before infusion of mesenchymal stem cells (on d 0), group 2 = d 2 to 9 of infusion, group 3 = d 10 to 14 of infusion, group 4 = d 15 to 19 of infusion.
The total amount of insulin needed to control the patient’s blood sugar decreased from d 2 to 9 (mean ± SD; from 88 ± 2 to 80 ± 3, p < 0.05). The decrease in amount of insulin required from d 10 to 14 was more obvious (from 80 ± 3 to 46 ± 7, p < 0.05) (Figure 3). Blood glucose decreased significantly from d 2 to d 9 (from 70.5 ± 6.5 to 43.4 ± 8.9, p < 0.05), but decreases in blood glucose from d 2–d 9 to that from d 10–d 14 did not differ (43.4 ± 8.9 to 32.3 ± 9.3, p > 0.05) (Figure 4). However, given that the amount of insulin required from d 10 to d 14 was significantly less than before and the blood glucose level of the patient was no higher than before, MSCs might regulate insulin resistance better at d 10 to 14. Blood sugar after d 15 was higher than that in the previous time interval (76.9 ± 5.1 vs 32.3 ± 9.3, p < 0.05), and the amount of insulin needed was significantly reduced at this time (63 ± 0 vs 88 ± 2, p < 0.05). Thus, MSC might have a beneficial effect on insulin resistance but the optimal time for this effect was d 2 to d 14 (Figure 4b). Therefore, the infusion of MSCs did not cure this case of type 2 diabetes and the treatment of type 2 diabetes was time-dependent.
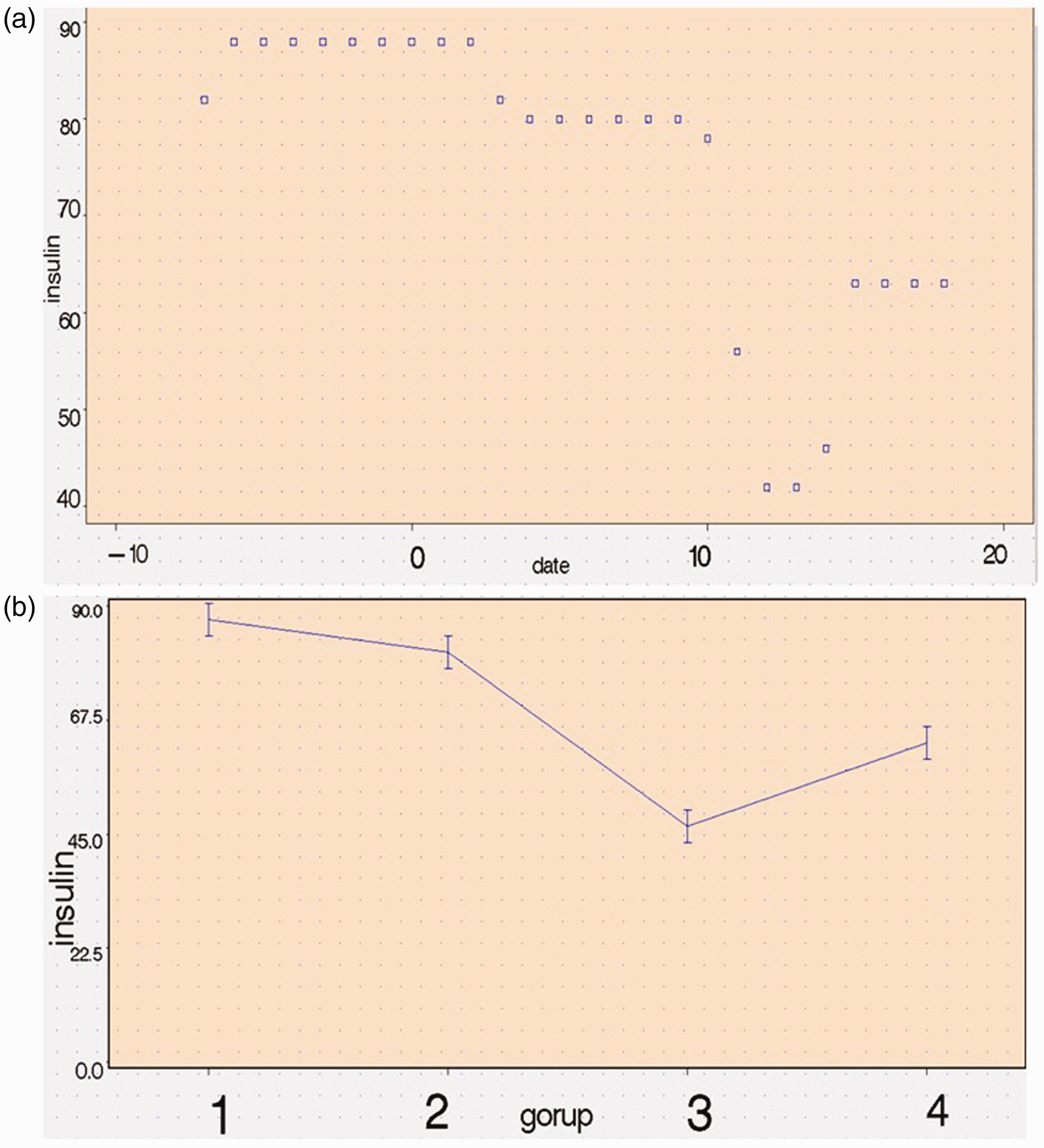
Changes in the total amount of insulin required on different days of mesenchymal stem cell (MSC) infusion. (a) Scatter plot of insulin levels on different days, and (b) total amount of insulin required (means ± standard deviations) before and after infusion of MSCs (on d 0); group 1 = 1 week before infusion of MSCs, group 2 = d 2 to 9 of infusion, group 3 = d 10 to 14 of infusion, group 4 = d 15 to 19 of infusion.

Changes of the total amount of blood glucose on different days of mesenchymal stem cell (MSC) infusion. (a) Scatter plot of blood sugar levels on different days, and (b) blood sugar levels (means ± standard deviations) before and after infusion of MSCs (on d 0); group 1 = 1 week before infusion of MSCs, group 2 = d 2 to 9 of infusion, group 3 = d 10 to 14 of infusion, group 4 = d 15 to 19 of infusion.
During this period (from the day of infusion of MSC), albumin was not elevated and the complete blood count was not increased. Albumin was approximately 19.44 to 24.53 g/L, and the complete blood count showed that the white blood cell count was 1.19 to 2.94 × 109/L, hemoglobin was 6.2 to 6.5 g/dL, and the platelet count was 3 to 11 × 109/L. The patient’s body was swollen, her legs were weak, and leg muscular strength (myodynamic examination) was 3 at 10 d after infusion of MSCs. Whether these symptoms were due to HLH central invasion or the adverse effects of MSC infusion is not clear. The patient was given myeloid growth factors, albumin infusion, component blood infusion, and other symptomatic support treatments. Nineteen days after MSC infusion, treatment was discontinued, and the patient returned home. She died on the second day after returning home due to cerebral hemorrhage caused by low platelets.
Discussion
HLH most frequently affects infants and children but the disease is also observed in adults, including those as old as 70 years of age. 4 Adult presentation of HLH is increasingly recognized. HLH can occur as a familial or secondary disorder and can be triggered by a variety of events that disrupt immune homeostasis. Secondary (sporadic or acquired) HLH is generally used to describe the disease in individuals without a known familial mutation; 5 secondary HLH may be triggered by viral illness, autoimmune disease, or lymphoma, or it can be of unknown origin. 6
HLH is a syndrome of inflammation and tissue destruction caused by excessive immune activation and inflammation. 7 In general, excessive inflammation is thought to be caused by the absence of normal downregulation of activated macrophages and lymphocytes. 8 Most patients with HLH exhibit impaired cytotoxic function of NK cells and cytotoxic T lymphocytes (CTLs), coupled with excessive activation of macrophages.9,10 Excessive macrophage activity and highly elevated levels of IFN-γ and other cytokines, which are produced by macrophages, NK cells, and CTLs, are thought to be the primary mediators of tissue damage. 8 Other lymphocyte abnormalities include altered numbers of CD4 and CD8 lymphocyte subsets. 11
Most patients relapse within a year of the initial acute illness. All HLH treatments are experimental at present, 12 and more than 10 years of clinical trial data have shown that there is no significant reduction in mortality or reactivation using the HLH-2004 protocol compared with HLH-94. 1 Thus, there is no strong statistical evidence to indicate the use of HLH-2004 instead of HLH-94. The treatment of HLH remains difficult, 13 and more effective and less toxic treatment options should be explored.
MSCs are multipotent stromal cells that can differentiate into a variety of cell types. 14 Because of their characteristics of hematopoietic support, 15 such as regulating inflammation and the immune response16–18 and repair of organ damage, 19 they have attracted increasing attention. A case study confirmed that MSCs could play a part in treating familial HLH. 3 Because umbilical cord MSCs are easy to obtain, we first treated the refractory secondary HLH of unknown origin with third-party umbilical cord MSCs. Our patient showed some improvement after MSC treatment, especially in body temperature and blood sugar levels but not in complete blood count, and the patient’s general condition, especially the hypoproteinemia, did not improve. This may have been related to her poor general condition (as indicated by the ECOG score of 4) before the MSC treatment was initiated. If the patient’s ECOG score were lower (i.e., indicating better condition) or if treatment had been combined with other immunosuppressive therapies, the outcome might have been better. The MSC treatment was found to have a therapeutic effect on insulin resistance in this patient (who had type 2 diabetes), which is consistent with previous findings.20,21
We conclude that treatment with MSCs may be beneficial for certain patients with refractory secondary HLH of unknown origin. We can confirm transient effects of MSC infusion in HLH and type 2 diabetes mellitus. However, complete remission of HLH could not be achieved with MSCs, and the function of MSCs in HLH may be as a bridge therapy before transplantation. However, the efficacy of MSCs and the duration of treatment need further study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
The study was sponsored by the major scientific and technological projects in Hainan (ZDYF2016124 & ZDXM2014075) and the hospital subject and new technology and new project of Affiliated Haikou Hospital Xiangya School Central South University.
