Abstract
Patients with coronary artery disease and concomitant diabetes mellitus tend to have more extensive vessel disease than non-diabetes mellitus coronary artery disease patients, are at high risk of adverse cardiovascular events and suffer from a great anginal burden. Very few trials have specifically addressed the issue of optimal anti-anginal therapy in coronary artery disease patients who also have diabetes mellitus. Among ‘classical’ anti-anginal agents, recent guidelines do not specifically recommend any molecule over others; however, European Society of Cardiology guidelines acknowledge that favourable data in patients with concomitant diabetes mellitus and coronary artery disease are available for trimetazidine and ranolazine, two anti-anginal agents with a non-haemodynamic mechanism of action. The aim of this article is to review available evidence supporting the anti-anginal efficacy of these two drugs in the difficult-to-treat population of diabetes mellitus patients, including their effects on glycated haemoglobin (HbA1c), a measure of medium-term glycaemic control. Although direct head-to-head comparisons have not been performed, available evidence favours ranolazine as an effective anti-anginal agent over trimetazidine in this population. In addition, ranolazine lowers HbA1c, indicating that it may improve glycaemic control in patients with diabetes mellitus. Conversely, scanty data are available on the metabolic effects of trimetazidine in this cohort of patients. Thus, ranolazine may represent a valuable therapeutic option in stable coronary artery disease patients with diabetes mellitus.
Introduction
Stable coronary artery disease (SCAD) is the leading cause of death in Western Countries, with chronic stable angina pectoris being the most common manifestation of the disease, observed in up to 70% of patients. 1 Recent guidelines issued by the European Society of Cardiology (ESC) state that treatment of patients with SCAD has two main goals: first, to improve prognosis by preventing myocardial infarction (MI) and death and, second, to minimize or abolish symptoms. 1
It is anticipated that the incidence and prevalence of SCAD will increase in the next decade as a result of population ageing, better management of MI, increasing burden of atherosclerosis and obesity and greater use of life-prolonging therapies. 1 Ideally, anti-anginal drug treatment should be tailored to the needs of each individual, taking into consideration the presence of co-morbidities such as diabetes mellitus (DM), which affects approximately one-third of patients with stable angina.2,3 In fact, CAD often appears as a complication of DM, and adverse cardiovascular (CV) outcomes in this population are well recognized. 1
Patients with CAD and concomitant DM usually have more extensive vessel disease than CAD patients without DM and are at higher risk of adverse CV events with a poorer prognosis.4–10 Of note, the recent ESC guidelines on the management of SCAD acknowledge that clinical manifestations of CV disease such as angina, MI and heart failure (HF) tend to occur at an earlier age in DM. 1 Silent ischaemia is more frequent in DM due to diabetic autonomic neuropathy,11,12 and these patients suffer a greater anginal burden compared with non-DM patients. In particular, DM appears to be associated with higher angina frequency,13,14 worse physical limitation and worse health-related quality of life. 14 Indeed, effective management of patients with SCAD and DM represents a major clinical need 15 since regardless of symptoms, patients with SCAD and DM carry a high risk of CV events and death. 16
Effective pharmacological anti-anginal interventions are currently available, but the management of patients with SCAD and DM remains a clinical challenge partly because the presence of DM may limit therapeutic choices. As an example, some beta-blockers (particularly non-selective molecules) tend to exert negative glucometabolic effects, worsening glycaemic control in DM. Coronary revascularization by either percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG) has been shown to reduce angina, with the latter being generally favoured over PCI 17 but to a lesser degree in DM than in non-DM patients. 18 Optimal medical therapy remains central to therapeutic approaches in patients with SCAD and DM, especially in those who remain symptomatic after revascularization. Furthermore, as outlined in recent guidelines, optimal medical therapy should be considered as the preferred treatment in patients with SCAD and DM unless there are large areas of ischaemia or significant lesions of the left main or proximal left anterior descending coronary artery. 1 However, despite the relevance of medical management, very few trials have specifically addressed the issue of optimal anti-anginal therapy in SCAD patients with DM, and most information on treatment efficacy is derived from subgroup analyses of existing trials.
Recent ESC guidelines on the management of SCAD do not specifically elaborate on the medical treatment of angina in DM patients. Tight glycaemic control by pharmacological and non-pharmacological interventions is recommended by the ESC guidelines on DM and pre-diabetes based on individual considerations, depending on patient’s characteristics including age, presence of complications and diabetes duration. 17 As for non-DM patients, the aims of SCAD treatment are symptom relief and prevention of CV events. 1 Calcium channel blockers or beta-blockers are indicated as first-line treatments for angina relief, while second-line options include long-acting nitrates, ivabradine, nicorandil, ranolazine or trimetazidine (Figure 1). 1 These second-line agents may also be used as first-line treatment in selected cases, depending on co-morbidities and/or poor tolerance to first-line agents. With regard to the use of specific anti-anginal agents in patients with DM, ESC guidelines acknowledge that specific data for DM patients are available for ranolazine and trimetazidine, two anti-anginal agents with a non-haemodynamic mechanism of action. 1
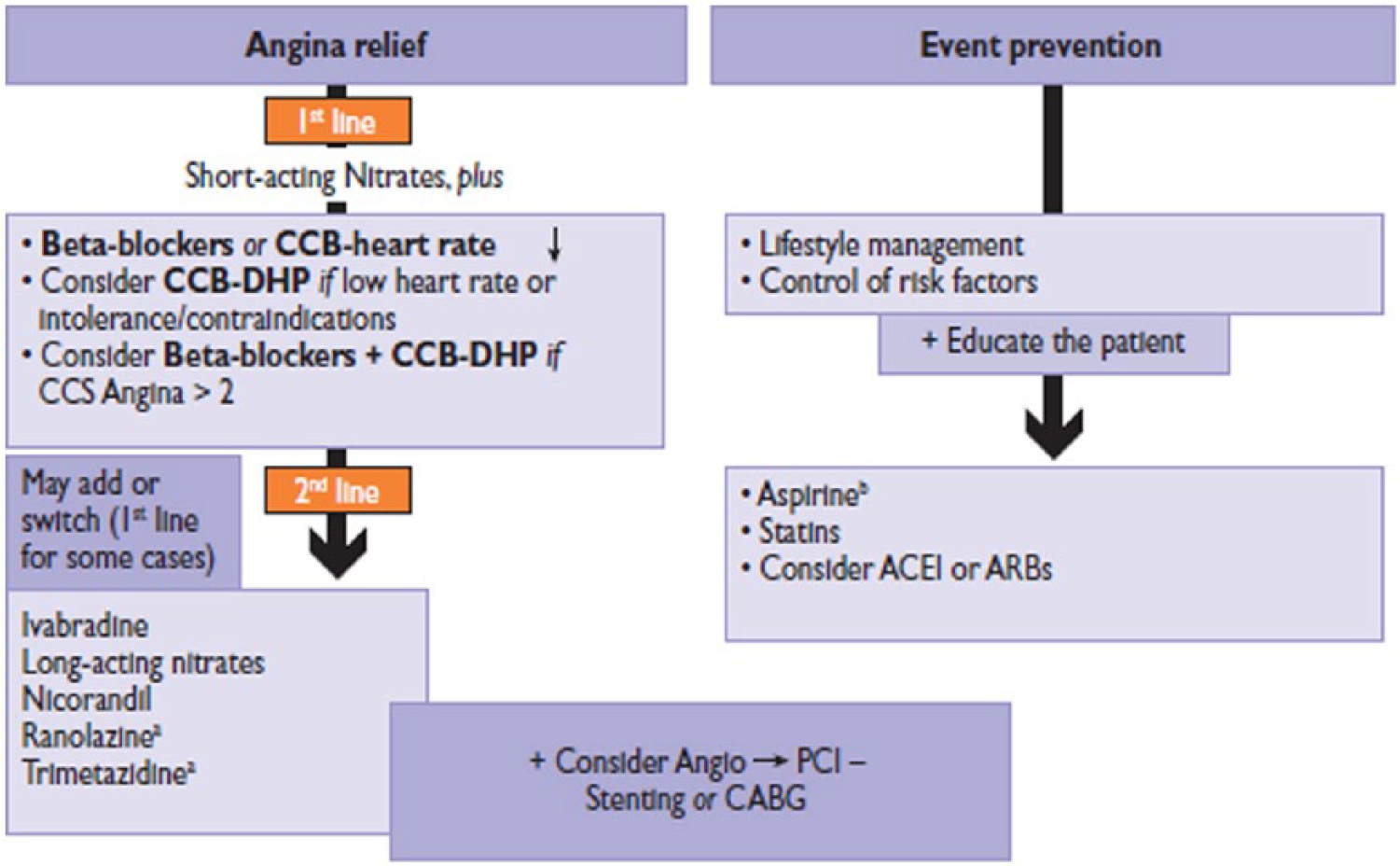
Medical management of patients with SCAD according to the ESC guidelines. 1
The aim of this article is to review available evidence on the use of trimetazidine and ranolazine in patients with DM who also have stable CAD. The effects of each drug on anginal symptoms and HbA1c will be discussed in separate sections. Only studies including patients with both SCAD and DM were considered for this review.
Trimetazidine
Mechanism of action
Under normal conditions, the human heart derives ~60%–90% of its energy from free fatty acid (FFA) oxidation, while glycolysis makes only a small contribution towards adenosine triphosphate (ATP) production and the remainder forms glucose and lactic acid.19,20 However, under conditions of impaired oxygen availability, high FFA oxidation rate is detrimental as it requires approximately 10%–15% more oxygen to generate the same amount of ATP compared with glucose oxidation. 20 In addition, FFA and their metabolites inhibit pyruvate dehydrogenase, which is tightly coupled to cardiac work, as well as glucose oxidation, which in turn increases lactate and proton accumulation (causing intracellular acidosis); impair Ca2+ handling and left ventricular (LV) contractility; and increase the risk of arrhythmias.19–22 Thus, in SCAD, it would be of interest to shift ATP production away from fatty acid oxidation towards carbohydrate oxidation. Metabolic modulators shift myocardial substrate metabolism from FFA to glucose oxidation and, therefore, optimize metabolic efficiency, reversing cellular abnormalities and improving LV function in SCAD patients.19,20,23
Trimetazidine [1-(2,3,4 trimethoxybenzyl) piperazine dihydrochloride] has been available for over 30 years and is currently available in several European countries as add-on therapy for the symptomatic treatment of patients with stable angina pectoris who are inadequately controlled by, or intolerant to, first-line anti-anginal therapies (i.e. beta-blockers and calcium channel blockers). Trimetazidine exerts its anti-anginal and anti-ischaemic effects via the inhibition of mitochondrial long-chain 3-ketoacyl coenzyme A thiolase (3-KAT), that is, the last enzyme involved in mitochondrial fatty acid β-oxidation.24–26 As a result, trimetazidine shifts cardiac metabolism from fatty acid oxidation to more oxygen-efficient glucose oxidation. 24 Inhibition of fatty acid oxidation by trimetazidine is believed to improve the coupling of glycolysis to glucose oxidation,24,25 thus decreasing proton production. This latter action is expected to result in decreased tissue acidosis and improved cardiac efficiency, thus protecting the heart from the deleterious consequences of ischaemia.24,25,27,28 Accordingly, in SCAD patients, trimetazidine can be considered a metabolic modulator, and unlike conventional anti-anginal agents, its anti-anginal and anti-ischaemic effects are not associated with changes in haemodynamic determinants of myocardial oxygen consumption, such as heart rate, systolic blood pressure, and rate–pressure product, nor myocardial blood flow.29–31 It has also been speculated, but not proved, that a shift from fatty acid to glucose oxidation could improve peripheral glucose extraction and utilization, thus ameliorating the deleterious glucometabolic effects of DM. 32 ESC guidelines mention that ‘trimetazidine improved HbA1c and glycaemia, while increasing forearm glucose uptake’, although no reference is made to the anti-anginal efficacy of trimetazidine in this patient population. 1
However, there is some controversy over the hypothesis that trimetazidine might exert its anti-anginal benefit via inhibition of mitochondrial 3-KAT, 33 indicating that the mechanism of action remains ill-defined, and other possible effects have been proposed to explain its anti-anginal benefits, including (a) a direct effect on cardiac fast inward Na+ current; 34 (b) a decrease in the production of superoxide free radicals; 35 (c) an increase in glycerol uptake from plasma which incorporates all four main phospholipid classes (phosphatidylcholine, phosphatidylinositol, phosphatidylethanolamine and cardiolipid) of the cardiac membranes; 36 (d) a protective effect against the damage induced on cardiac energetics and oxidative injuries by moderate ischaemia and reperfusion injury, decreasing the cardiac levels of malondialdehyde (a biomarker for oxidative stress); 37 (e) inhibition of myocardial neutrophil activation and accumulation induced by post-ischaemic reperfusion; 38 and (f) an effect related to binding of trimetazidine to target sites located on both the outer and the inner mitochondrial membranes implicated in closure of the mitochondrial permeability transition pore. 39
Until recently, trimetazidine-containing medicinal products were prescribed in the European Union for the treatment of SCAD, and also for the symptomatic treatment of vertigo and tinnitus, and vascular-related visual acuity loss and visual field disturbances. 40 However, because of concerns over the effectiveness of trimetazidine in those latter conditions and reports of movement disorders (including parkinsonism, ‘restless leg’ syndrome, tremors and gait instability), the European Medicines Agency (EMA) 40 recently reviewed the safety and effectiveness of this drug and concluded that its use should be restricted solely to the therapeutic indication of stable angina pectoris. The EMA 40 recommended trimetazidine should not be used in Parkinson’s disease, parkinsonism, tremors, ‘restless leg’ syndrome or other related movement disorders, or in severe renal impairment. The EMA also recommended caution when prescribing trimetazidine to patients with moderate renal impairment or elderly patients and to consider dose reduction in these subjects. This can be a limitation for the drug use in patients with angina and diabetic nephropathy.
Anti-ischaemic efficacy in patients with CAD and DM
Several studies have demonstrated the anti-anginal efficacy of trimetazidine, as monotherapy and in combination with other agents (mainly beta-blockers; Table 1).41–54 A meta-analysis by the Cochrane Collaboration analysed 23 randomized trials of trimetazidine versus placebo or other anti-anginal agents in 1378 patients with chronic stable angina. 41 The analysis showed that compared with placebo, trimetazidine significantly reduced the number of weekly angina attacks and weekly nitroglycerin tablet consumption and increased the time to onset of significant ST-segment depression. However, the analysis concluded that there was too little information about mortality, CV events and quality of life to propose the use of trimetazidine in angina pectoris, although fewer people seemed to discontinue treatment because of side effects than with other anti-anginal drugs. The meta-analysis concluded that ‘large, long-term trials comparing trimetazidine with other anti-anginal drugs assessing clinically relevant important outcomes are required to establish its role in clinical management’.
Studies assessing the anti-ischaemic and anti-anginal efficacy of trimetazidine and ranolazine in patients with stable CAD and diabetes.
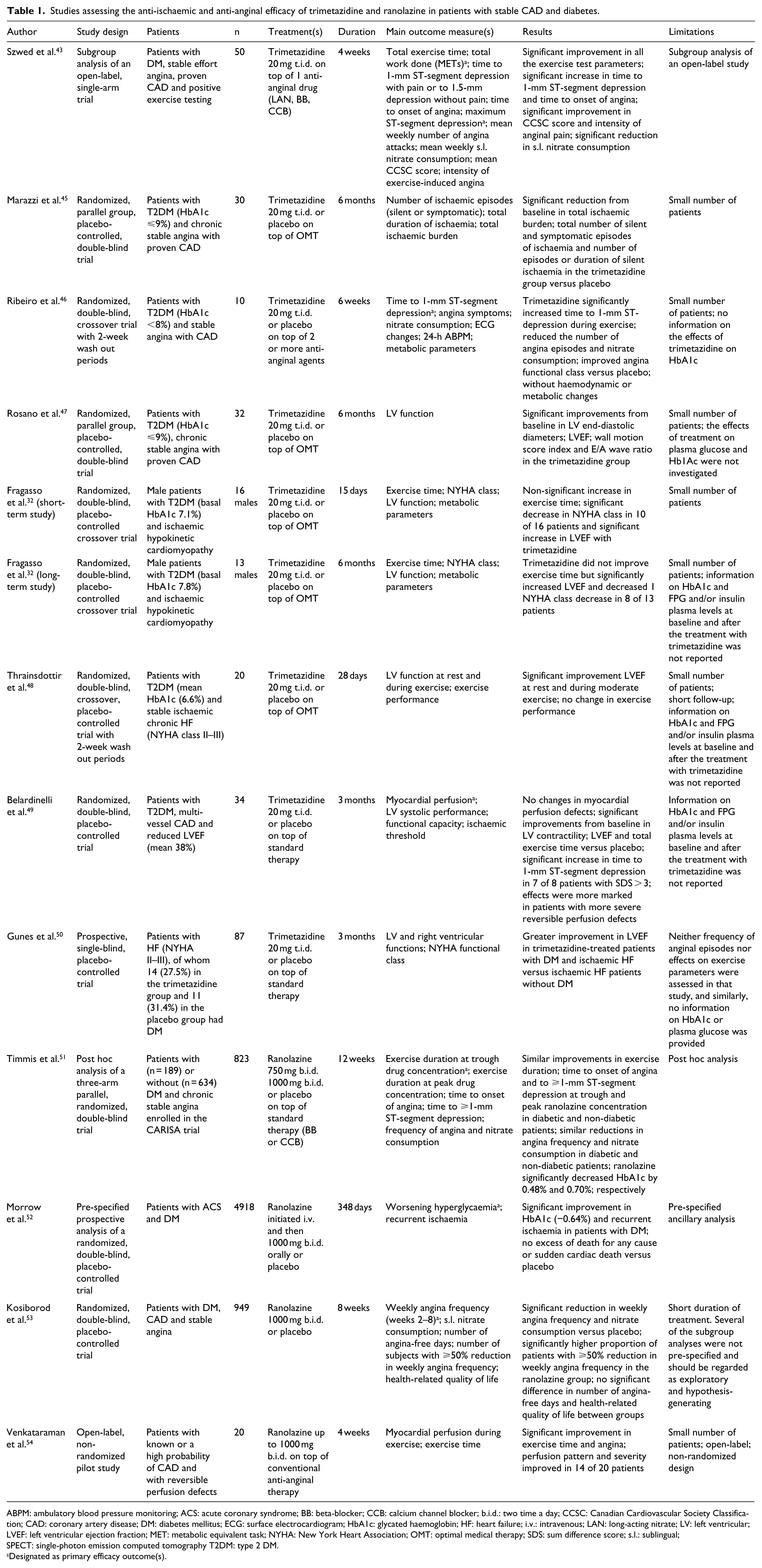
ABPM: ambulatory blood pressure monitoring; ACS: acute coronary syndrome; BB: beta-blocker; CCB: calcium channel blocker; b.i.d.: two time a day; CCSC: Canadian Cardiovascular Society Classification; CAD: coronary artery disease; DM: diabetes mellitus; ECG: surface electrocardiogram; HbA1c: glycated haemoglobin; HF: heart failure; i.v.: intravenous; LAN: long-acting nitrate; LV: left ventricular; LVEF: left ventricular ejection fraction; MET: metabolic equivalent task; NYHA: New York Heart Association; OMT: optimal medical therapy; SDS: sum difference score; s.l.: sublingual; SPECT: single-photon emission computed tomography T2DM: type 2 DM.
Designated as primary efficacy outcome(s).
Although trimetazidine was mentioned in the recent ESC guidelines as an anti-anginal drug suitable for patients with SCAD and concomitant DM, it should be recognized that it has not been evaluated in large trials in this specific setting. The anti-ischaemic efficacy of trimetazidine in patients with diabetes was initially assessed by Szwed et al.43,44 in 1999 in a substudy of the TRIMetazidine in POLand-1 (TRIMPOL-1) trial. TRIMPOL was an open-label, single-arm, multicentre trial of 700 patients with stable angina, documented CAD or previous MI and positive exercise test, treated with one anti-anginal agent (long-acting nitrate, beta-blocker or calcium channel blocker) as background therapy. Patients received trimetazidine 20 mg three times a day (t.i.d.) in addition to background treatment for 4 weeks. The analysis of the subgroup of patients with DM included in the TRIMPOL-1 study (n = 50) showed that trimetazidine induced significant improvements in all exercise parameters evaluated, 43 including exercise-induced ischaemia and time to onset of angina. Severity of anginal pain [as assessed by the Canadian Cardiovascular Society Classification (CCSC) score] and intensity of anginal pain during exercise (Borg scale) were also significantly decreased at the end of the 4-week treatment period in DM patients. 43 Trimetazidine also reduced the mean number of weekly anginal attacks (from 4.8 to 3.1, p < 0.01) and nitrate consumption (from 4.2 to 2.3 doses; Figure 2), and it was well tolerated in this population. 43 However, this trial has a number of limitations, most notably the small number of patients with DM and the open-label design.
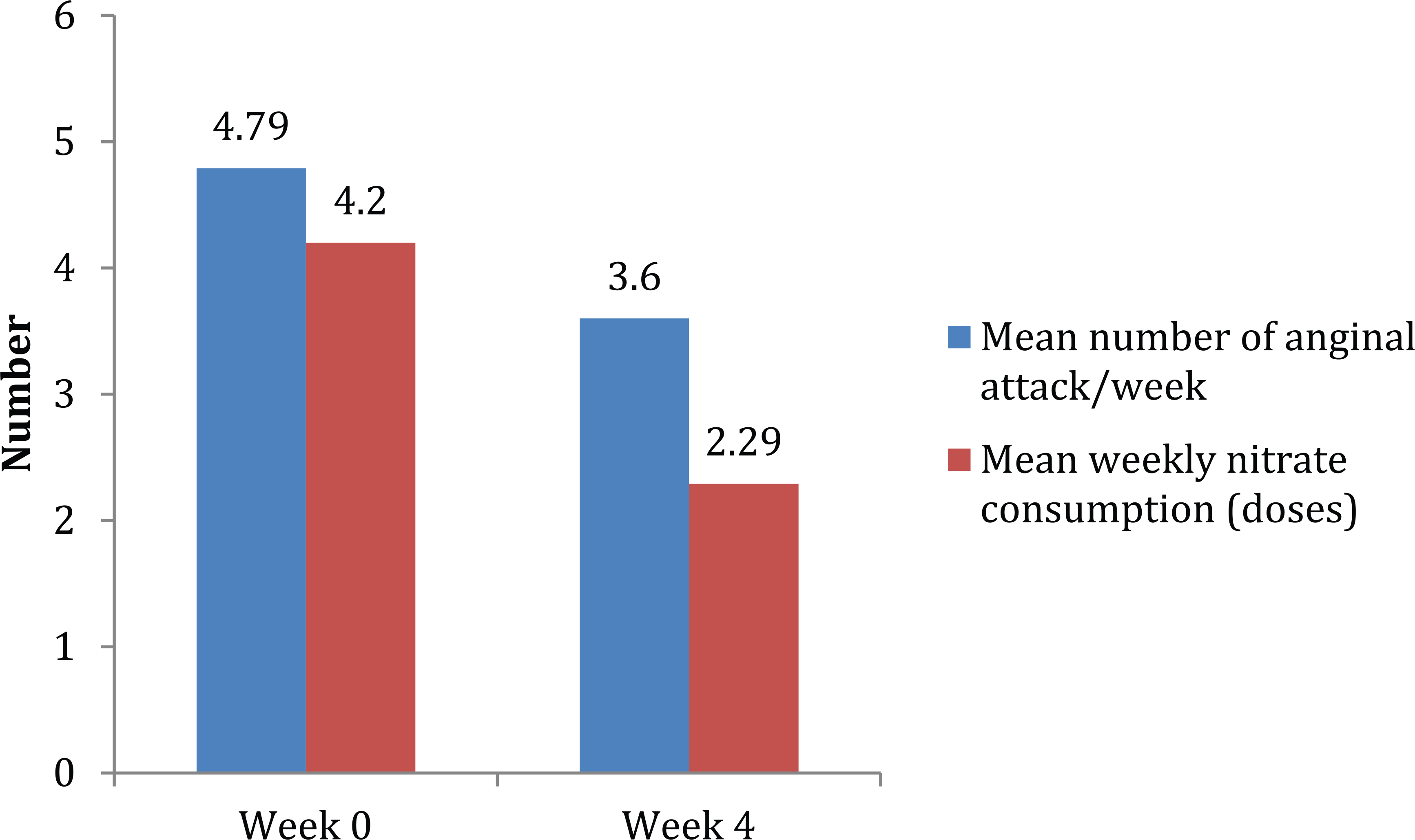
Mean number of anginal attack and weekly nitrate consumption with trimetazidine in the TRIMPOL-I trial (substudy). 43 p < 0.01 for both comparisons.
In a small randomized, placebo-controlled trial including 30 patients with T2DM (HbA1c 7.2% and 7.5% in the trimetazidine and control group, respectively) and chronic stable angina treated with trimetazidine 20 mg t.i.d. or placebo in addition to optimal medical therapy (OMT) for 6 months, a significantly lower incidence of total ischaemic burden [defined as the sum of the duration of all episodes of significant ST-segment depression occurring during 24-h ambulatory electrocardiogram (ECG) monitoring] and silent and symptomatic episodes of myocardial ischaemia was observed in the trimetazidine group. 45 Of note, no information was provided on the effect of trimetazidine on HbA1c.
Ribeiro et al. 46 conducted a small randomized, double-blind, crossover trial in 10 patients with type 2 diabetes mellitus (T2DM; HbA1c < 8%) and stable angina who received trimetazidine 20 mg t.i.d. or placebo for 6 weeks on top of standard anti-anginal therapy with at least two agents. After 6 weeks, trimetazidine significantly increased time to 1-mm ST-segment depression and exercise duration and decreased the number of weekly angina episodes (from 2.2 to 0.4, p < 0.01) and sublingual nitrate doses use (from 1.5 to 0.1, p < 0.001) compared with placebo. Furthermore, a significant improvement in CCSC functional class was observed in patients treated with trimetazidine; 46 however, trimetazidine was not associated with changes in haemodynamic, lipid or glycaemic profiles.
Three studies investigated the effect of trimetazidine on LV function and exercise tolerance in patients with T2DM and CAD. Fragasso et al. 32 reported the short- and long-term effects of trimetazidine 20 mg t.i.d. added to standard treatment on symptoms of HF [as assessed by the New York Heart Association (NYHA) classification], exercise tolerance and LV function in 16 male patients with T2DM and ischaemic cardiomyopathy (NYHA class <IV) in a double-blind, crossover, randomized placebo-controlled trial. Trimetazidine improved symptoms of HF and LV ejection fraction (LVEF) in both short- (15 days) and long-term (6 months) evaluation and decreased fasting glucose, whereas no significant change in exercise time was observed. 32
The effect of add-on treatment with trimetazidine 20 mg t.i.d. on LV function and exercise tolerance was also assessed in a pilot, crossover, randomized placebo-controlled study (4-week treatment periods) in 20 patients with T2DM and chronic HF of ischaemic aetiology (NYHA class II–III). 48 The changes in metabolic parameters produced by trimetazidine were not described. 48
More recently, a 3-month randomized placebo-controlled trial evaluated the effect of trimetazidine 20 mg t.i.d. added to standard therapy on myocardial perfusion and LV systolic function [assessed by gated single-photon emission computed tomography (SPECT) myocardial scintigraphy with a 2-day stress (Bruce)-rest protocol] in 34 T2DM patients with ischaemic heart disease and LV dysfunction. 49 The effect of trimetazidine on functional capacity and ischaemic threshold was a secondary endpoint. After 3 months of treatment, trimetazidine significantly improved total exercise time, LVEF and systolic wall thickening, and a significant increase in time to 1-mm ST-segment depression was observed in the subgroup of patients treated with trimetazidine with greater severity of perfusion defects (summed difference score >3). However, there were no significant changes in myocardial perfusion defects, 49 and the effects of treatment on plasma glucose and Hb1Ac were not investigated.
Two other studies assessed the effects of trimetazidine in DM patients with ischaemic heart disease, but the specific effects of the drug on anginal symptoms were not addressed. In an earlier randomized, placebo-controlled study, 32 T2DM patients with chronic stable angina and proven CAD received trimetazidine 20 mg t.i.d. or placebo in addition to standard therapy. 47 After 6 months, patients treated with trimetazidine showed significant improvements in LV volumes and ejection fraction compared with placebo. Neither frequency of anginal episodes nor effects on exercise parameters were assessed in that study, and similarly, no information on HbA1c or plasma glucose was provided. 47
Gunes et al. 50 investigated the effect of trimetazidine (20 mg t.i.d. on top of standard therapy) versus placebo on LV and right ventricular functions in 87 patients with HF (NYHA class <IV). CAD was the underlying aetiology of HF in 68.6% and 62.9% of the patients in the trimetazidine group and the placebo group, respectively. Only 14 (27.5%) patients in the trimetazidine group and 11 (31.4%) patients in the placebo group had DM. After 3 months of treatment, trimetazidine significantly improved LVEF. This improvement tended to be greater in patients with both ischaemic HF and DM. 50 Plasma glucose levels and HbA1c following the administration of trimetazidine were not reported.
Finally, a very recent study evaluated the effect of periprocedural administration of trimetazidine on the incidence of PCI-induced myocardial injury and contrast-induced nephropathy (CIN) in 100 DM patients with mild-to-moderate renal dysfunction. 55 Patients were randomized to receive periprocedural trimetazidine or placebo, at a dose slightly higher than that used in clinical practice [35 mg two times a day (b.i.d.)] for 72 h, starting 48 h before PCI, in addition to periprocedural intravenous (IV) infusion of isotonic saline solution and oral N-acetylcysteine. Serum creatinine levels in the control group increased significantly 3 days after PCI, while no changes in serum creatinine were observed in the trimetazidine group. The incidence of CIN was 12% in the trimetazidine group and 28% in the control group (p < 0.05). Cardiac troponin I levels were significantly lower in the trimetazidine group (6 h: 8 ± 0.3 vs 16 ± 0.2 pg/mL, 12 h: 13 ± 0.9 vs 24 ± 0.8 pg/mL and 24 h: 7 ± 0.7 vs 14 ± 0.3 pg/mL, p < 0.001) consistent with myocardial protection against ischaemic injury. However, follow-up of major adverse cardiac events was not included in the study protocol.
Effect on glucose metabolism and glycaemic control
Two studies specifically investigated the effect of trimetazidine on glucose metabolism in patients with T2DM and ischaemic heart disease.32,56 In a small study by Fragasso et al., 32 short- and long-term metabolic effects of trimetazidine 20 mg t.i.d. added to standard treatment were assessed in 16 male patients with T2DM and ischaemic cardiomyopathy. Forearm and whole body glucose uptake were estimated using the hyperinsulinaemic euglycaemic clamp method. At 15 days, both forearm and whole body insulin-stimulated glucose uptake were significantly higher, and fasting plasma glucose (FPG) was significantly lower in the trimetazidine group compared with placebo recipients (11% reduction from 136 ± 40 to 121 ± 30 mg/dL; p < 0.05), whereas plasma FFAs and triglycerides were similar in the two groups. After the 15-day period, patients were randomized again and entered the long-term phase of the study. At 6 months, forearm and whole body insulin-stimulated glucose uptake were significantly higher in the trimetazidine group, and Hb1Ac levels were significantly lower after trimetazidine therapy than after placebo (7.1% ± 1.1% vs 7.8% ± 1.4%; p = 0.05). This was the reflection of the fact that mean Hb1Ac levels remained stable with trimetazidine, while they tended to increase on placebo (p = 0.05). No significant differences in FPG, FFAs or triglycerides were evident between groups. Based on these findings, the authors concluded that trimetazidine improves glucose metabolism. 32 However, these results should be interpreted with caution because baseline values relative to whole body insulin sensitivity and forearm glucose uptake were not provided. In addition, the rationale for a more evident effect of trimetazidine after 2 weeks of treatment – with respect to long-term therapy – remains unclear and might be related to a bias introduced by the limited number of patients and the difference in patients numbers in the two studies (n = 18 in the short-term study and n = 13 in the long-term study).
In a similar, crossover study by the same group, 15 patients with T2DM and ischaemic cardiomyopathy were randomly assigned to trimetazidine 20 mg t.i.d. or placebo for 15 days on top of standard therapy. 56 At the end of the treatment period, FPG and insulin resistance [as estimated by the homeostasis model assessment of insulin resistance (HOMA-IR) index] were significantly lower in the trimetazidine group, with no differences in plasma triglycerides and FFAs. Compared to placebo, trimetazidine decreased basal glucose levels by 12% (126 ± 8 vs 143 ± 11 mg/dL; p < 0.05) and HOMA-IR (3.67 ± 0.04 vs 4.48 ± 0.06, p < 0.05) with a non-significant reduction in insulin levels to suggest improvement in insulin sensitivity, whereas unsurprisingly, given the short duration of the study, HbA1c values remained unchanged (7.1% ± 0.4% vs 7.1% ± 0.3%). In the trimetazidine group, whole body insulin sensitivity, forearm glucose uptake and glucose oxidation measured during the hyperinsulinaemic euglycaemic clamp were significantly higher, and lipid oxidation was almost completely inhibited in comparison with placebo. 56
Despite the use of state-of-the-art approaches, and although these results are consistent with the previous report by the same group, 32 it is difficult to definitively conclude that trimetazidine improves glucose metabolism in patients with T2DM and ischaemic heart disease. In brief, the design and results of the above-mentioned studies were highly heterogeneous, included an overall limited number of patients (n = 314, with sample size in single studies ranging from 10 to 87) and the duration of trimetazidine treatment ranged from 2 weeks to 6 months. More importantly, in many of the studies on trimetazidine in patients with DM and SCAD, HbA1c and FPG and/or insulin plasma levels at baseline and after the treatment with trimetazidine was not reported. Additionally, in the study by Ribeiro et al., 46 trimetazidine 20 mg t.i.d. for 6 weeks did not affect FPG or HbA1c levels.
Because of these reservations, it is not possible to evaluate the long-term effects of trimetazidine on mortality, major adverse CV events or adverse events. Further larger and longer studies are needed to fully evaluate the effects of trimetazidine in patients with T2DM.
Ranolazine
In Europe, ranolazine [(+)N-(2,6-dimethylphenyl)-4(2-hydroxy-3-(2-methoxyphenoxy)-propyl)-1-piperazine acetamide dihydrochloride] is approved by the EMA as add-on therapy, at a maximum dosage of 750 mg b.i.d., for the symptomatic treatment of chronic stable angina in patients inadequately controlled by, or intolerant to, first-line anti-anginal therapies. The drug is also available at a maximum dose of 1000 mg b.i.d. in the United States, where it is also indicated for the treatment of chronic angina as monotherapy. The anti-anginal efficacy of ranolazine, both as a single agent and in combination with other drugs, has been demonstrated in several large, randomized, double-blind controlled trials.57–60
Mechanism of action
Ranolazine was initially believed to have a mechanism of action similar to that described for trimetazidine, 61 that is, stimulation of glucose oxidation as a consequence of reduced fatty acid oxidation. 61 However, this effect only occurs at plasma concentrations much higher (>10-fold) than the therapeutic level (~4–6 µM), which indicated that this was not the mechanism responsible for the anti-anginal effect of ranolazine. In isolated rat hearts perfused with a buffer containing high concentrations of FFAs, ranolazine (20 µM) substantially improved recovery after a period of no-flow ischaemia, but it inhibited fatty acid oxidation by only 12%. 33 In addition, in a study comparing the effects of ranolazine and CVT-4325, a more potent fatty acid oxidation inhibitor, in isolated rat hearts during ischaemia and reperfusion, 62 ranolazine had no effect on carbohydrate or fatty acid oxidation but significantly improved LV functional recovery following ischaemia or reperfusion at the same time. In contrast, CVT-4325 significantly reduced fatty acid oxidation and increased carbohydrate oxidation but had no significant cardioprotective effect. These results confirmed that ranolazine-induced ischaemic protection is not mediated by inhibition of fatty acid oxidation.
It is now established that ranolazine exerts its anti-ischaemic and anti-anginal effects through inhibition of the late inward sodium current (late INa), which contributes to the action potential plateau in cardiomyocytes.63,64 In atrial and ventricular muscle and in Purkinje fibres, Na+ entry during the action potential occurs in two phases: (a) through a peak INa lasting only a few milliseconds which plays a key role in the excitability and conduction velocity of cardiac action potential and (b) a small late INa (INaL) lasting hundreds of milliseconds. Under normal conditions, INaL is generated via a small percentage of Na+ channels which either fail to inactivate close properly or close and then reopen during the plateau phase, when most of the Na+ channels normally remain closed.63–65 The amplitude of the INaL increases under several pathophysiological conditions, including myocardial ischaemia. The increase in INaL during ischaemia plays a critical role in the rise of cytosolic Na+ concentration, which is exchanged for extracellular Ca2+ via activation of the Na+-Ca2+ exchanger (NCX) in the reverse mode, thus causing a ‘sodium-induced Ca2+ overload’ in cardiac myocytes. 65 Higher Na+ and Ca2+ concentrations results in increased actin or myosin filament interaction and LV diastolic tension, which increases diastolic wall tension and myocardial O2 demands. Increased diastolic tension is also thought to promote micro-circulatory resistance further impairing subendocardial coronary blood flow during diastole, to reduce myocardial O2 supply to the ischaemic territory and exacerbate ischaemia.63–65 Furthermore, the increase in cytosolic Na+ may affect the regulation of oxidative phosphorylation, which could result in a mismatch between ATP consumption and production. 64 The increase in INaL prolongs in a heterogeneous manner the duration of the ventricular action potentials (as reflected by a prolongation of the QT interval on the surface ECG), to increase dispersion of the QT interval and induce early after depolarizations that may lead to the development of serious cardiac arrhythmias.65,66 Myocardial ischaemia is, therefore, associated with an abnormal increase in INaL, which appears to drive the further development of mechanical, metabolic, and electrical abnormalities observed in the ischaemic heart. If so, selective inhibitors of the INaL represent a new therapeutic strategy for the management of myocardial ischaemia, contractile dysfunction and cardiac arrhythmias.
By inhibiting the late INa, ranolazine may reduce calcium uptake via the NCX and preserve ionic homeostasis, thus blunting ischaemia-induced diastolic dysfunction. 65 Similar to trimetazidine, ranolazine appears not to affect haemodynamic parameters including blood pressure, heart rate or cardiac contractility.57,60 Data indicating that ranolazine has similar efficacy in both DM and non-DM patients, and that it may also lower HbA1c,51,53 prompted the ESC guidelines on the management of stable CAD to advise that this drug can be added to other well-established anti-anginal agents and may be of particular value in patients with a higher HbA1c. 1 In this respect, preliminary findings indicate that the beneficial effect of ranolazine on glycaemic control may be mediated by promoting glucose-stimulated insulin secretion, increasing pancreatic beta-cell survival 67 and/or by reducing glucagon release through inhibition of Na+ channels in pancreatic alpha-cells. 68
Anti-ischaemic efficacy in patients with CAD and DM
Evidence on the anti-anginal efficacy of ranolazine in patients with stable CAD and T2DM was initially provided by subgroup analyses of two large randomized, placebo-controlled studies.51,52 The Combination Assessment of Ranolazine in Stable Angina (CARISA) trial was a double-blind, randomized, placebo-controlled study of 823 patients with chronic stable angina who were symptomatic despite treatment with standard doses of atenolol, amlodipine or diltiazem and were randomly assigned to placebo or ranolazine (750 or 1000 mg) b.i.d. for 12 weeks. 57 In the main trial, both doses of ranolazine significantly increased exercise duration at trough and peak drug levels, prolonged time to angina and to ECG signs of ischaemia and reduced the mean weekly number of angina attacks and nitroglycerin consumption. 57 Subsequently, CARISA data were analysed to compare the effects of ranolazine in patients with (n = 189) and without (n = 634) DM. 51 Importantly, this analysis revealed that ranolazine improved exercise duration, time to onset of angina and to 1-mm ST-segment depression both in DM and non-DM patients. No difference in the reduction of weekly angina episodes and nitroglycerin consumption were observed in the two subgroups, indicating that the anti-anginal efficacy of ranolazine was comparable in patients with or without DM. 51 Those findings were confirmed by the results of a pre-specified analysis of the Metabolic Efficiency With Ranolazine for Less Ischaemia in Non–ST-Elevation Acute Coronary Syndromes–Thrombolysis in Myocardial Infarction 36 (MERLIN-TIMI 36) trial. 52 MERLIN-TIMI 36 was a randomized, double-blind, placebo-controlled trial which included 6560 patients with acute coronary syndrome who were treated with ranolazine (1000 mg b.i.d.) or placebo within 48 h of ischaemic symptoms, and followed up for a median of 348 days. 59 The primary endpoint of MERLIN-TIMI 36 was a composite of CV death, MI or recurrent ischaemia. In MERLIN-TIMI 36, ranolazine did not meet the primary efficacy composite endpoint. However, the drug significantly reduced the incidence of recurrent ischaemia and worsening angina [13.9% vs 16.1%; hazard ratio (HR), 0.87; 95% confidence interval (CI), 0.76–0.99; p = 0.03], as well as the need for additional anti-anginal therapy (10.6% vs 13.0%; HR, 0.80; 95% CI, 0.69–0.93; p = 0.003). 59 Additionally, ranolazine reduced angina frequency as compared with placebo. Consistent with the main trial, recurrent ischaemia was significantly reduced by ranolazine (HR, 0.75; 95% CI, 0.61– 0.93; p = 0.008) in the large subgroup of patients with DM (n = 2220). 52 The promising results of these subgroup analyses prompted a prospective trial to specifically assess the anti-anginal efficacy of ranolazine in patients with T2DM. The Type 2 Diabetes Evaluation of Ranolazine in Subjects With Chronic Stable Angina (TERISA) study was a randomized, double-blind, placebo-controlled trial in which 949 patients with T2DM, CAD and stable angina treated with up to two anti-anginal agents were randomly allocated to ranolazine 1000 mg or placebo b.i.d. for 8 weeks. 53 The primary efficacy endpoint in TERISA was the mean weekly number of anginal episodes over the last 6 weeks of the study. Secondary endpoints included weekly frequency of sublingual nitroglycerin consumption, number of angina-free days, and the proportion of subjects with a ⩾50% reduction in their mean number of anginal episodes. 66 Approximately two-thirds of patients enrolled in TERISA had a previous MI, and about 50% prior myocardial revascularization. The majority of patients (93%) were treated with glucose-lowering agents, including 19% treated with insulin. In TERISA, ranolazine significantly reduced weekly frequency of angina, as well as consumption of sublingual nitroglycerin (Figure 3). In addition, the proportion of patients who achieved a ⩾50% reduction in the mean number of anginal episodes was greater in the ranolazine group compared with placebo, although this was considered non-significant based on the pre-specified multiple testing procedure. 53 The proportion of angina-free days over the last 6 weeks of the study was similar in the ranolazine and placebo groups. Subgroup analyses revealed that baseline number of weekly angina episodes, number of concomitant anti-anginal medications, age, gender or previous revascularization did not affect the efficacy of ranolazine in reducing the frequency of angina. 53 Interestingly, the superiority of ranolazine over placebo in reducing the primary endpoint was more pronounced in patients with higher baseline HbA1c (p for interaction <0.027). The mechanisms underlying the increased efficacy of ranolazine in the presence of poor glycaemic control remain to be determined. However, ex vivo cardiomyocytes exposed to hyperglycaemia or isolated from hearts of diabetic animals have up-regulated phosphorylated calcium/calmodulin-dependent protein kinase II (CaMKII). This kinase phosphorylates the cardiac sodium channel, which increases late INa and leads to an increase in intracellular Na+ and Ca2+ concentrations. Consistent with the hypothesis, the late INa is increased in the heart in diabetes, and the late INa density was higher in cardiac myocytes from both insulin-deficient (Ins2Akita) and insulin-resistant (db/db) mice as compared with wild-type myocytes. 69 These findings could explain why ranolazine might be expected to exert a greater therapeutic effect at the cardiac level in patients with poorer glycaemic control. 53 Moreover, in an open-label, non-randomized pilot study of 20 patients (50% of whom had DM) with known or a high probability of CAD and reversible perfusion defects by exercise treadmill gated SPECT, ranolazine (up to 1000 mg b.i.d. on top of standard therapy) significantly increased exercise time and ameliorated angina after 4 weeks. 69 In addition, ranolazine significantly improved perfusion pattern and severity in 14 of 20 patients.
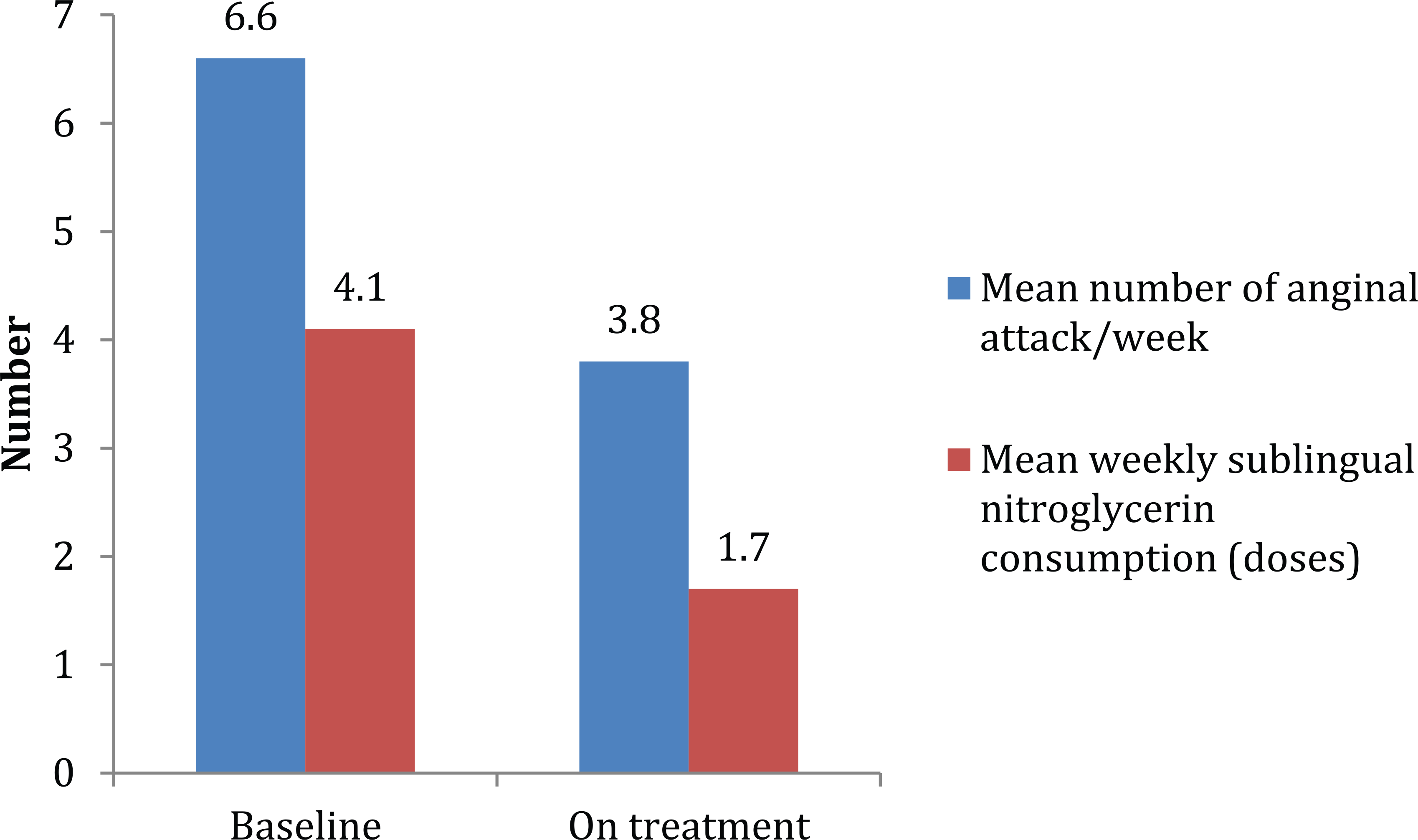
Mean number of anginal attack and weekly consumption of sublingual nitroglycerin with ranolazine in the TERISA. 53 p < 0.01 for both comparisons.
Effect on glucose metabolism and glycaemic control
The effects of ranolazine on glucose homeostasis (primarily assessed by changes in HbA1c) in patients with T2DM and CAD were investigated in post hoc analyses of the CARISA and MERLIN-TIMI 36 trials.51,52,70 The analysis of data from CARISA included 131 DM patients for whom both baseline and 12-week HbA1c values were available. Compared to placebo, ranolazine 750 and 1000 mg b.i.d. significantly reduced HbA1c from baseline (by 0.48%, p < 0.008; and 0.7%, p = 0.0002, respectively). 52 This effect is of particular relevance because baseline HbA1c values of DM patients in CARISA were relatively low (7.65% and 7.92% in the ranolazine 750 and 1000 mg b.i.d. groups, respectively). Stratifying patients by insulin treatment revealed that those on insulin therapy had greater reductions in HbA1c. Furthermore, significantly more patients treated with ranolazine 1000 mg group achieved an HbA1c of <7%, the target recommended by most diabetes guidelines.17,71 After completing the CARISA study, eligible patients entered an open-label extension trial (Ranolazine Open-Label Experience, ROLE). 72 Analysis of data from the first 2 years of this 6-year extension study showed that HbA1c levels remained unchanged beyond 12 weeks of ranolazine in patients treated with ranolazine. 51 The analysis of the effect of ranolazine on HbA1c in MERLIN-TIMI 36 yielded similar results, showing a significant reduction in HbA1c from 7.5% to 6.9% in DM patients who received ranolazine (n = 707). 52 Consistent with the results from CARISA, DM patients treated with ranolazine in the MERLIN-TIMI 36 trial were more likely to achieve an HbA1c of <7% at 4 and 8 months. Notably, in patients without DM at baseline, the new occurrence of fasting glucose >110 mg/dL or HbA1c ⩾6% was reduced by ranolazine (31.8% vs 41.2%; p = 0.003). No significant changes in FPG or weight were seen, 52 and the incidence of hypoglycaemic events was low and similar between groups. The effects of ranolazine on FPG and HbA1c in DM patients with moderate or severe baseline hyperglycaemia were assessed in a recent analysis of data from the MERLIN-TIMI 36. 70 The placebo-corrected reduction in HbA1c with ranolazine was 0.28% (p = 0.045) in DM patients with better glycaemic control (HbA1c 6%–<8%) and 0.59% (p < 0.001) in those with poorer glycaemic control (HbA1c ⩾8%–10%). In patients with severe hyperglycaemia at baseline (FPG ⩾150–400 mg/dL), ranolazine significantly decreased FPG (25.7 mg/dL, p = 0.001), whereas there was no change in patients with baseline levels of <150 mg/dL. 70 These findings indicate that the metabolic effect of ranolazine is greater in patients with more severe hyperglycaemia. Analysis of anti-diabetes treatment revealed that changes in therapy were similar in the ranolazine and placebo groups, suggesting that the effect of ranolazine on glycaemic control was not related to intensification of DM treatment. These findings were corroborated in a very recent post hoc analysis of the TERISA study, conducted on 913 patients. 73
Discussion
Individuals with chronic stable angina and DM are a difficult-to-treat patient population. 15 Therefore, it is crucial to identify effective therapeutic strategies for the medical management of these patients. Trimetazidine and ranolazine are effective anti-anginal agents that have minimal or no haemodynamic effects.31,57,60 Both agents have been studied in patients with ischaemic heart disease and DM, and both have been reported to lower HbA1c. However, the strength of evidence in support of the anti-anginal efficacy of ranolazine appears greater because only a few studies specifically assessed the effect of trimetazidine on patients with CAD and DM, and most were open-label studies which included a small number of subjects or did not have a control group. 44 Frequency of angina episodes or sublingual nitrate consumption was not part of the assessment of trimetazidine32,47–50 and duration of treatment with trimetazidine differed largely in these trials, and it was relatively short, ranging from 15 days 32 to 6 months.32,45,47 Therefore, the long-term safety of trimetazidine cannot be fully evaluated at present according to available evidence. In addition, conflicting findings have been reported on the effect of trimetazidine on exercise tolerance, with some studies reporting an increase46,49 and others no change.32,48 These discrepancies are likely attributable to differences in study design and patient characteristics.
In contrast, the anti-anginal efficacy of ranolazine and its effect on HbA1c have been evaluated in large, randomized placebo-controlled trials that included more than 3000 patients treated for up to 1 year. The initial results of CARISA and MERLIN-TIMI 36,51,52 which suggested a similar efficacy of ranolazine in stable angina patients with or without DM, have been recently confirmed by TERISA, a large prospective, randomized trial specifically designed to assess the efficacy of ranolazine in DM with CAD and stable angina treated with 1–2 anti-anginal agents. 53 In TERISA, ranolazine significantly reduced weekly angina frequency and sublingual nitrate consumption, and these effects were more pronounced in patients with higher HbA1c. 53 Although it is unclear whether the greater efficacy of ranolazine in patients with poorer initial glycaemic control depends on inhibition of the late sodium current or is related to improving glucose metabolism, it is tempting to speculate that ranolazine therapy might have an added value in DM patients with chronic stable angina. Both CARISA and MERLIN-TIMI 36 indicated that ranolazine lowers HbA1c,51,52 particularly in patients with higher HbA1c53,70 to suggest that this metabolic effect is in some way mirrored by the CV responses to ranolazine in conditions of poor glycaemic control. It is worth noting that what appear to be small improvements in HbA1c from a starting value >8% are associated with a significant reduction in the development and progression of microvascular complications in both T1DM and T2DM. In addition, a threshold below which no further benefit is accrued has not been identified, to indicate that all DM patients who lower their HbA1c with ranolazine should obtain some additional microvascular benefit in the longer term. 74 The safety profile of ranolazine is favourable, and it has been assessed up to a 1-year follow-up.
In conclusion, the published data on the effects of trimetazidine and ranolazine on stable angina in DM patients indicate that ranolazine has clinically relevant beneficial effects on both angina symptoms and HbA1c, where less consistent data from smaller trials are available on trimetazidine. The combined anti-ischaemic and HbA1c lowering effects of ranolazine indicate that this agent may become the preferred anti-anginal agent for the management of DM patients with stable CAD. This treatment approach could be relevant since many beta-blockers are associated with adverse metabolic effects – in particular in terms of glucose and insulin resistance.
Footnotes
Acknowledgements
P.J.G. has given a lecture on stable coronary artery disease and DM supported by Menarini. G.A. reports having received research funds, speakers’ bureau fees and consultantships, from Menarini International. Editorial assistance for the preparation of this article was provided by Luca Giacomelli, PhD, and Ambra Corti, on behalf of Content Ed Net; this assistance was funded by Menarini.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
