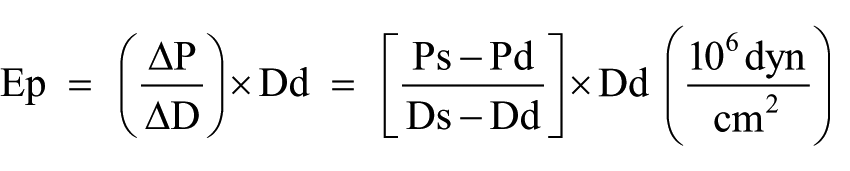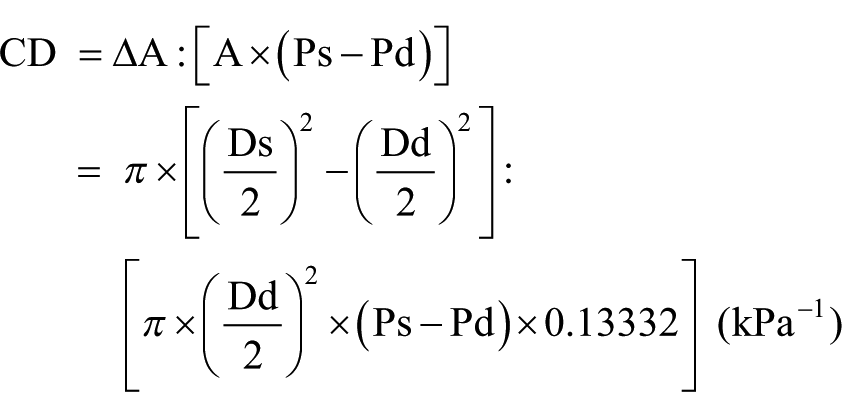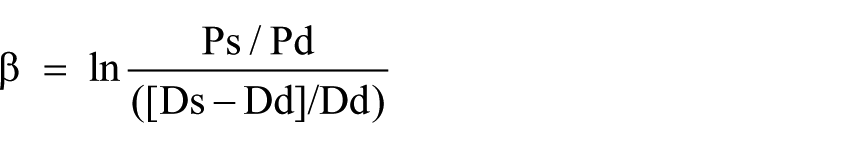Abstract
Tribbles 3, whose expression is up-regulated by insulin resistance, was confirmed to be involved in diabetic cardiomyopathy in our previous study. However, it is not known whether Tribbles 3 has a role on conduit arteries such as the aorta in diabetes. Type 2 diabetic rat model was induced by high-fat diet and low-dose streptozotocin. We evaluated the characteristics of diabetic rats by serial ultrasonography and histopathologic analyses of aortic wall architecture. Diabetic rats displayed increased aortic medial thickness, excessive collagen deposition, diminished elastic fibres and reduced vascular compliance together with Tribbles 3 overexpression. To further investigate the role of Tribbles 3 in aortic remodelling, we used Tribbles 3 gene silencing in vivo 12 weeks after onset of diabetes. Silence of Tribbles 3 significantly reversed pathological aortic remodelling without blood pressure modification. In Tribbles 3-small interfering RNA group, medial thickness and perivascular fibrosis were markedly decreased; moreover, there were prominent reductions in collagen content and collagen/elastin ratio, resulting in an improved arterial compliance. Additionally, with Tribbles 3 silencing, the diminished phosphorylation of PI3K/Akt was restored, and increased activation of MKK4/JNK was decreased. Silence of Tribbles 3 is potent in mediating reversal of aortic remodelling, implicating that Tribbles 3 is proposed to be a potential therapeutic target for vascular complication in diabetes.
Introduction
Diabetes is one among the most costly and burdensome chronic disease of the present day and its incidence is exploding in pandemic proportions, especially in elderly people. More than the disease, the vascular complications associated with the disease are major cause for the increased morbidity and mortality in diabetic patients. 1 The pathogenesis of diabetic cardiovascular disease is multifactorial, including ageing, hyperglycaemia, insulin resistance and inflammation. Among these factors, insulin resistance is one of its main pathological hallmarks. Increasing evidence supports the associations between insulin resistance and vascular disease.2–4
Tribbles 3 (TRB3) is a pseudokinase that links to several pathophysiological conditions, including insulin resistance,5–7 cardiovascular disease 8 and diabetes. 9 TRB3 is a cytosolic protein that is reported to directly associate with Akt and to block phosphorylation at Akt-Thr308. 5 Increased TRB3-Akt binding has been associated with reduced Akt activity and with inhibition of insulin signal transduction. 10 TRB3 also serves as a molecular switch and regulates the relative activation of the three classes of mitogen-activated protein kinase (MAPK). 11
Thus, TRB3 is considered to be an essential regulator for insulin resistance. Previous studies support the notion that both PI3K/Akt 12 and JNK/MAPK13,14 play an important role in vascular remodelling and atherosclerosis. So, we hypothesized that up-regulated TRB3 induced by insulin resistance might participate in the pathophysiological process of aortic remodelling in diabetes. Accordingly, the aims of this study were as follows: (1) to investigate whether TRB3 was implicated in the development of diabetes mellitus (DM)-associated aortic remodelling and (2) to determine the impacts of TRB3 silencing on the aortic remodelling in a type 2 diabetic rat model.
Methods
Induction of type 2 diabetes
All experimental procedures were performed in accordance with animal protocols approved by the Shandong University Animal Care Committee.
Totally, 40 male Sprague–Dawley (SD) rats (120–140 g) were purchased from the experimental animal centre of Shandong University of Traditional Chinese Medicine (Jinan, China). The rats were randomized into four groups: control, chow + streptozotocin (STZ), high-fat (HF) diet and DM. The establishment of the model was referred to our previous experiments. 15
Blood analyses
After rats fasted overnight, we collected jugular blood. Total cholesterol (TC) and triglyceride (TG) levels and fasting blood glucose (FBG) were analysed. Fasting insulin (FINS) level was measured using an enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, USA). Insulin sensitivity index [ISI = ln(FBG × FINS)−1] was calculated.
Blood pressure measurement
Systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP) and heart rate (HR) were measured with a non-invasive tail-cuff system (Softron BP-98A, Tokyo, Japan) as previously described. 16
Assessment of arterial stiffness
Aortic ultrasonography was performed using the Phillips 7500 machines (Phillips Medical Systems, Netherlands). Images were obtained from two-dimensional, M-mode, pulsed wave (PW) Doppler. Minimum (end-diastolic, Dd) and maximum (peak systolic, Ds) diameters were obtained from the M-mode. Two-dimensional ultrasonography was used to calculate Peterson’s elastic modulus (Ep), cross-sectional distensibility (CD) and arterial stiffness index (β). All measurements reported represented the means of three consecutive cardiac cycles and were performed by the same investigator.
Ep, CD and β were estimated automatically by the following formulae17–20
where Ps is the systolic blood pressure, Pd is the diastolic blood pressure, Ds is the systolic diameter, Dd is the diastolic diameter, A is the cross-sectional lumen area of aorta and ΔA is the change in aortic cross-sectional lumen area.
For quantitative analysis of integrated backscatter (IBS) of the aortic wall, we used a commercially available software package (acoustic densitometry; Phillips Medical Systems, Netherlands). The following parameters were measured: standardized integrated backscatter (IB%) and cyclic variation of integrated backscatter (CVIB).
Histology and morphometric analyses
The aorta fixed in paraformaldehyde (4%) was embedded in paraffin, and cut into 4-µm-thick sections for histological analysis. Aortic thickness was determined by haematoxylin–eosin staining. To quantify collagen density, sections were stained with Masson’s trichrome and Picrosirius red. To normalize the area of perivascular collagen around vessels with different sizes, the perivascular collagen content was represented as the perivascular collagen area/luminal area (PVCA/LA) ratio. Elastin deposition was determined by staining with Verhoeff–Van Gieson. Collagen and elastin deposition were expressed as percentage of positively stained area to medial area. Collagen/elastin ratio was calculated. Images were obtained under light microscopy and quantified with a computerized image analysis system (Image-Pro Plus, Version 5.0; Media Cybernatics, Houston, TX, USA).
Average aortic wall architecture score was quantified as previously described. 21 Aortic total collagen content was quantified using the hydroxyproline assay as previously described. 15
Immunohistochemical staining and western blot for collagen I and III
Immunohistochemistry was performed in paraffin sections. The sections were incubated with primary antibodies: rabbit polyclonal antibodies to collagen I and III (Abcam, Cambridge, MA, USA) overnight, then a matching biotinylated secondary antibody for 30 min at 37°C. The results were viewed on a confocal FV 1000 SPD Laser Scanning microscope (Olympus, Japan).
For western blot, an equal amount of protein (30 µg) under non-denatured condition was loaded onto each lane of 6% sodium dodecyl sulphate (SDS) polyacrylamide gel. The levels of collagen I and III were normalized to that of β-actin.
Western blot analysis
For western blotting, we used antibodies against TRB3 (Calbiochem, La Jolla, CA, USA), phospho-PI3K/PI3K (Abcam, Cambridge, MA, USA), phospho-Akt/Akt, phospho-MKK4/MKK4 and phospho-JNK/JNK (Cell Signaling Technology, Beverly, MA, USA). TRB3 protein was corrected by β-actin as an internal control and phospho-specific proteins were normalized by total protein.
Gene silencing of TRB3
Totally, 30 rats were selected and randomized to treatment with TRB3-small interfering RNA (siRNA) or the vehicle. After 12 weeks of diabetes, the animals were injected via the jugular vein with 2.5 × 1010 plaque-forming units of either adenovirus harbouring TRB3 gene (TRB3-siRNA) or control empty virus (vehicle). Adenovirus transfer repeated in 2 weeks. Four weeks after first adenovirus injection, rats were sacrificed.
Statistical analysis
Values were presented as mean ± standard error of the mean (SEM). SPSS 16.0 was used for statistical analysis. Results were compared by one-way analysis of variance (ANOVA). A p value of less than 0.05 was considered statistically significant.
Results
Generation of vascular remodelling model in type 2 diabetes
Biochemical characteristics of diabetic rats
The characteristics of the biochemical indicators are given in Table S1. After 4 weeks of a HF diet, serum TG level was significantly higher in the HF and DM groups than control and chow + STZ groups (p < 0.05). ISI was markedly reduced in HF and DM groups (p < 0.05). Insulin resistance appeared in rats fed with HF diet at week 4. One week after STZ injection, FBG was markedly elevated in DM group, and this continued until the end of the experiment. ISI showed consistent decline in DM group after onset of diabetes. Simultaneously, in DM group, serum TC and TG levels also maintained at higher levels than the control (p < 0.05) at the end of the experiment. The diabetic model induced by a HF diet and low-dose STZ, was characterized by insulin resistance, moderate hyperglycaemia and hyperlipidaemia. It was manifested that the animal model closely resembled type 2 diabetes in humans.
Arterial stiffness assessed by echocardiography
There were no differences in SBP and DBP among the four groups (Figure S1A, S1B). The DM rats showed larger systolic aortic lumen than the control and chow + STZ groups from week 6 after induction of diabetes (p < 0.05) (Figure 1(a)). Ep and stiffness index were significantly increased in DM group, while CD was deceased relative to the control during diabetes (p < 0.05) (Figure 1(b)–(d)).
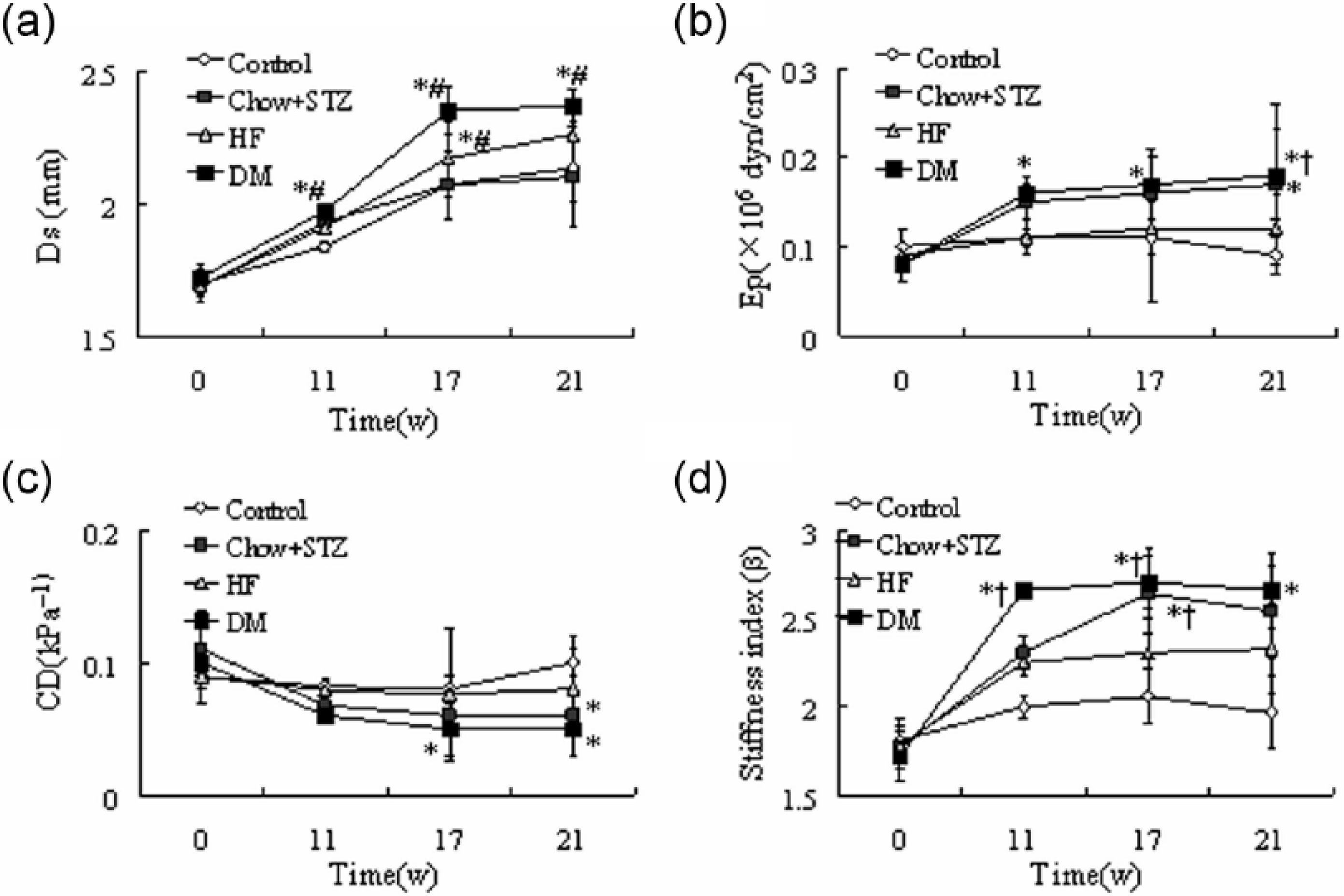
Vascular stiffness is increased in diabetic rats; serial echocardiographic investigations during the process of diabetes. (a) systolic diameter (Ds); (b) Peterson’s elastic modulus (Ep); (c) cross-sectional distensibility (CD) and (d) arterial stiffness index (β). Results are expressed as means ± SEM, n = 7–10 per group. *p < 0.05 versus control; †p < 0.05 versus HF; #p < 0.05 versus chow + STZ.
Pathological aortic remodelling of diabetic rats
The medial thickness was remarkably increased in DM group in comparison with the control and HF groups (p < 0.001, p < 0.05) (Figure 2(a) and (e)). There was a significant increase in perivascular fibrosis as measured by Masson’s trichrome and Picrosirius red staining of arterial sections in DM group (Figure 2(b)–(d)). And the diabetic rats exhibited the highest values of PVCA/LA among the four groups (Figure 2(f)).
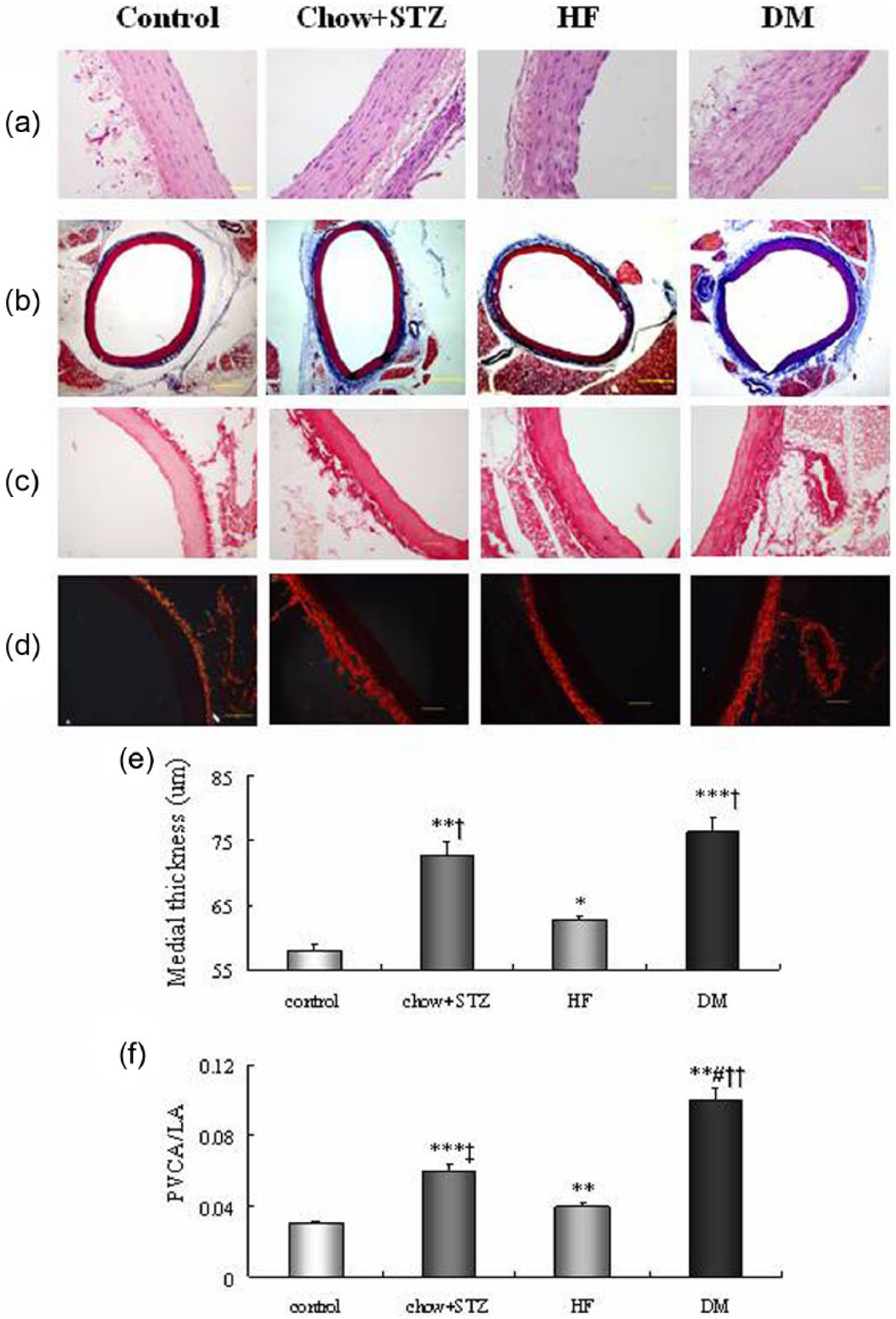
The diabetic rats exhibited increased medial thickness and perivascular fibrosis in aorta. (a) Representative haematoxylin–eosin-stained cross-sectional sections from the aorta. Bar: 50 µm. (b) Micrographs of aortic sections stained with Masson trichrome stain. Bar: 400 µm. (c) Picrosirius red staining (bright-field) of arterial sections. Bar: 100 µm. (d) Picrosirius red staining (dark-field) of arterial sections. Bar: 100 µm. (e) Quantitative analysis of medial thickness. (f) Quantitative analysis of perivascular fibrosis area to lumen area (PVCA/LA). Results are expressed as mean ± SEM, n = 6. **p < 0.01, ***p < 0.001 versus control; †p < 0.05, ††p < 0.01, ‡p < 0.001 versus HF; #p < 0.05 versus chow + STZ.
In addition, histological analyses revealed aberrant collagen deposition with fragmentation and disarray of elastic fibres in DM group. Vascular collagen accumulation was significantly increased in DM group compared to the control and HF groups according to Masson’s trichrome staining (p < 0.001) (Figure 3(a) and (c)). This was reconfirmed by the highest values of collagen content in DM group (p < 0.001) (Figure 3(g)). However, the elastin content was significantly reduced in diabetic rats (Figure 3(d)). The DM rats displayed significantly elevated collagen-to-elastin ratio (p < 0.001) (Figure 3(e)). Histological grading of medial elastin degradation revealed a significant increase in elastin fibre disruption in diabetic rats compared to almost no disruption in control rats (p < 0.001) (Figure 3(f)).
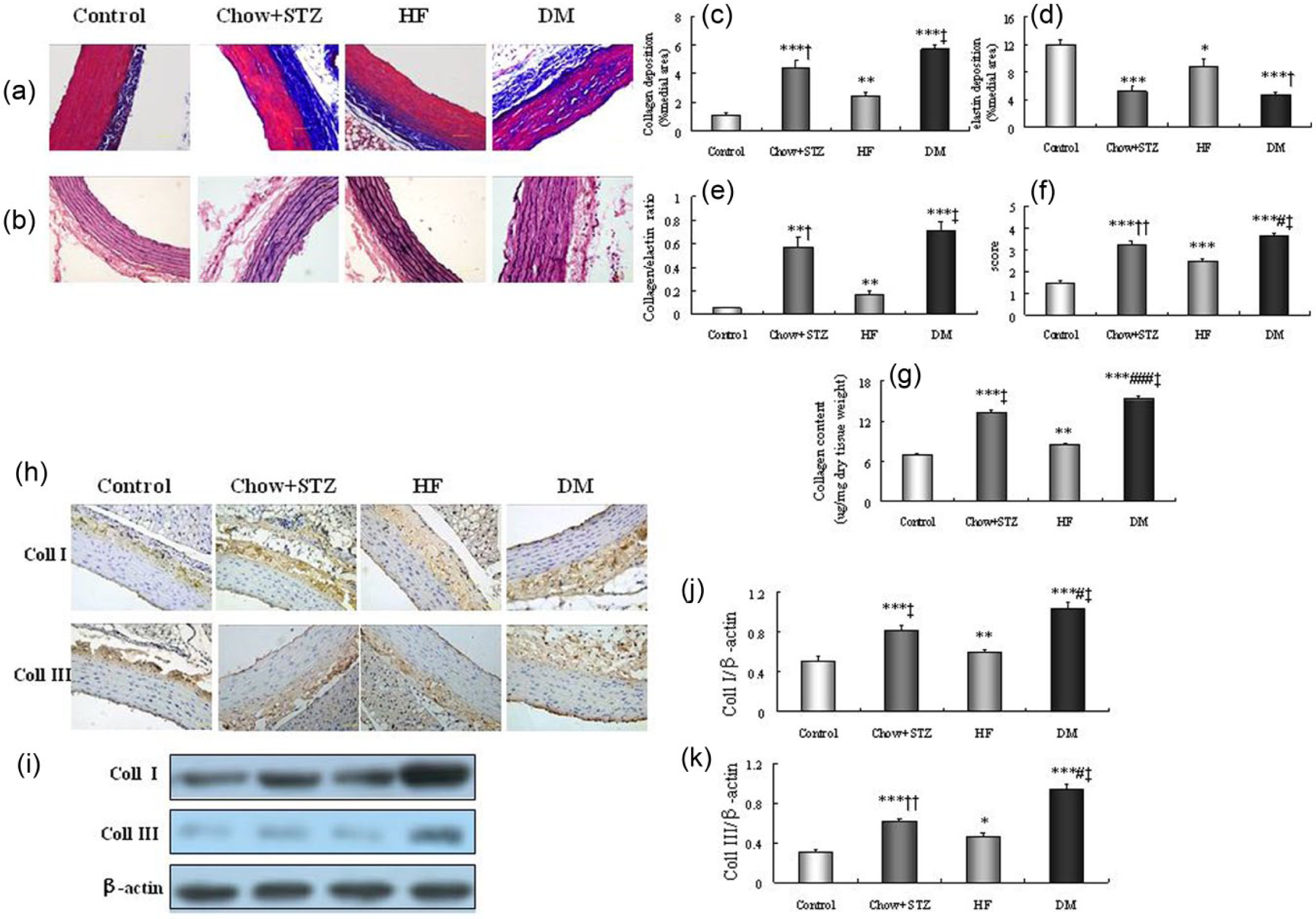
The diabetic rats displayed excessive collagen deposition and diffuse elastic disruption. (a) Collagen deposition in the descending aorta was evaluated by measurement of the blue stained area on the Masson trichrome slides. Bar: 50 µm. (b) Verhoeff’s stain reveals diffuse disruption of elastic fibre architecture. Bar: 50 µm. (c) The percentage of collagen per medial area. (d) The percentage of elastin per medial area. (e) The ratio of collagen to elastin. (f) Average aortic wall architecture score was graded on Verhoeff–Van Gieson-stained sections as following: no disruption (grade 1); disruption of one elastic fibre with intact neighbouring fibres (grade 2); disruption of two or more fibres in direct contact (grade 3) and disruption of all elastic fibres from the internal elastic lamina to the external elastic lamina (grade 4) (six rats of each group measuring five random fields per slide). (g) Collagen content detected by hydroxyproline assay. (h and i) Representative immunohistochemistry (H, brown staining was regarded as positive, bar: 50 µm) and western blot (I) of collagen I, III. (j and k) Western blot analysis of collagen I, collagen III. Bar graph showed mean ± SEM of western blot analysis. coll I, collagen I; coll III, collagen III. *p < 0.05, **p < 0.01, ***p < 0.001 versus control; †p < 0.05, ††p < 0.01, ‡p < 0.001 versus HF; #p < 0.05, ###p < 0.001 versus chow + STZ.
The protein expression of collagen I and III content was increased in DM group through immunohistochemistry and western blot analysis (p < 0.05~p < 0.001) (Figure 3(h)–(k)).
These histological changes were confirmed by echocardiographic results. The diabetic rats displayed increased IB% and decreased CVIB in the aortic wall as compared to the control and HF groups (p < 0.05~p < 0.001) (Table S2).
These above results showed that evident insulin resistance, decreased arterial compliance and pathological vascular remodelling existed in the type 2 diabetic rats.
Impaired PI3K/Akt and activated MKK4/JNK signalling pathways accompanied by TRB3 overexpression observed in DM group
Aortic TRB3 protein expression was significantly increased in DM group (Figure 4(a) and (b)). Accompanied by TRB3 elevation, the phosphorylation of PI3K/Akt was decreased and MKK4/JNK was markedly activated (Figure 4(c) and (e)). These results raised the possibility that PI3K/Akt and JNK signalling pathways participated in mediating TRB3-related vascular damage.
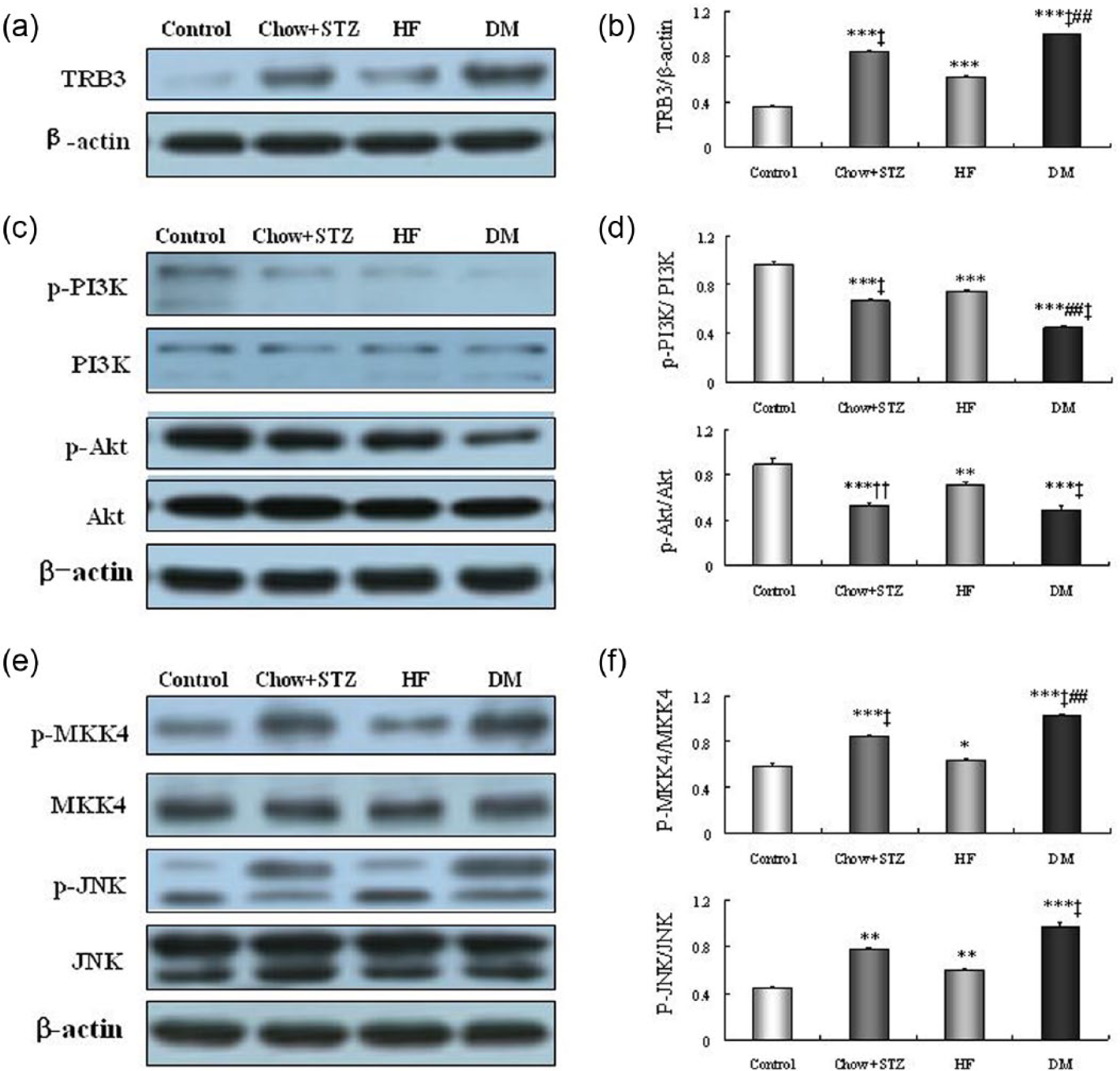
Impaired PI3K/Akt and activated MKK4/JNK, accompanied by TRB3 overexpression, were observed in diabetic rats. (a and b) Protein expression of TRB3. WB shows increased TRB3 protein in DM rats compared with the other three groups. (c) Representative western blots of PI3K/Akt. (d) Western blot analyses of p-PI3K/PI3K, p-Akt/Akt. (e) Representative western blots of MKK4 and JNK. (f) Western blot analyses of p-MKK4/MKK4, p-JNK/JNK. Bar graph showed mean ± SEM of western blot analysis. *p < 0.05, **p < 0.01, ***p < 0.001 versus control; ††p < 0.01, ‡p < 0.001 versus HF; ##p < 0.01 versus chow + STZ.
TRB3 gene silencing reversed aortic remodelling
Detection of aortic TRB3 expression after gene silencing
Compared with vehicle treatment, TRB3-siRNA treatment conferred down-regulated mRNA and protein expression of aortic TRB3 (Figure 5(a) and (b)).
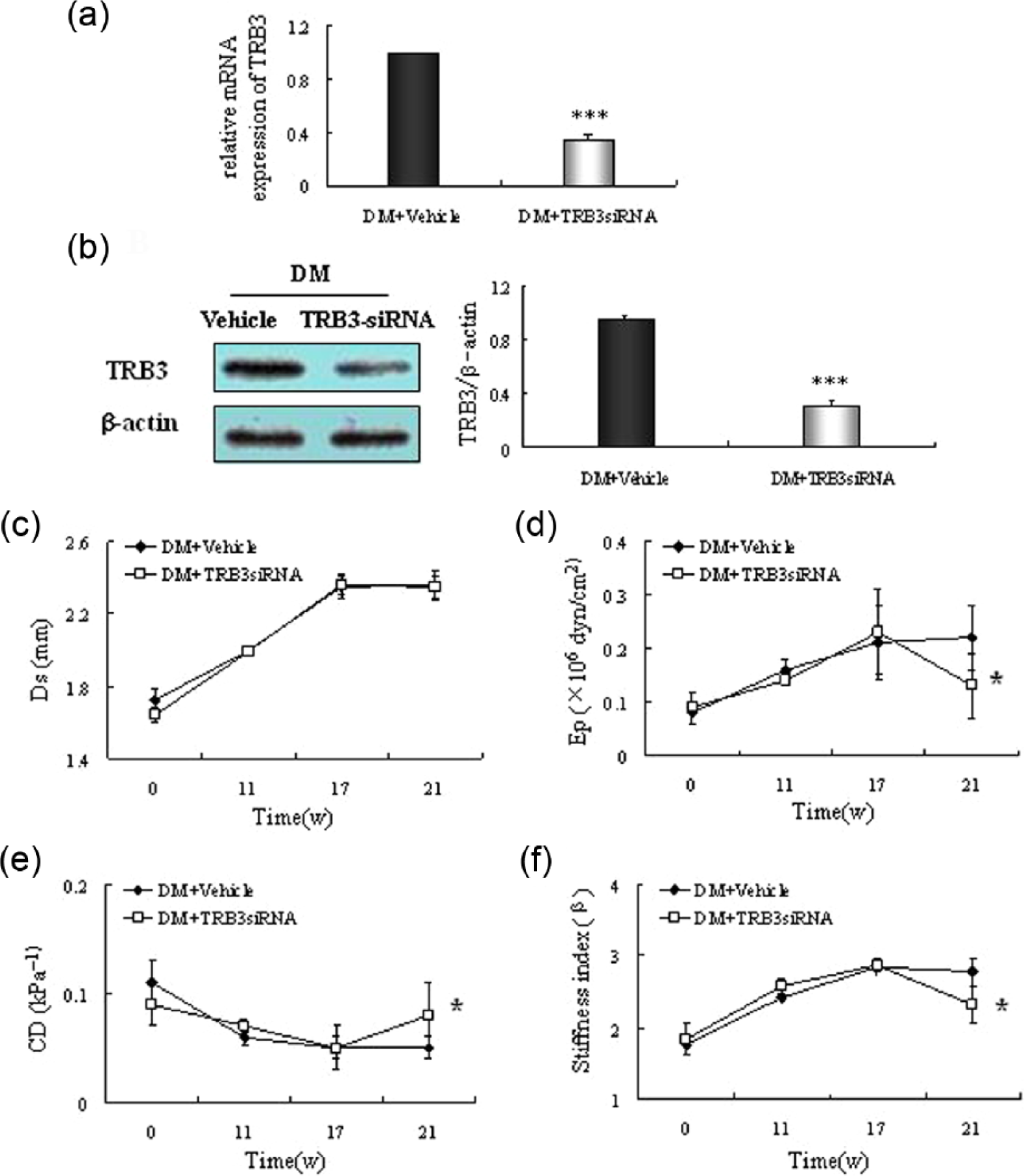
TRB3 gene therapy improves vascular compliance in DM rats. Representative mRNA (a) and Western blot (b) of TRB3. The expression of TRB3 was significantly reduced in TRB3-siRNA group. (c)–(f) Serial echocardiographic investigations. (c) systolic diameter (Ds); (d) Peterson’s elastic modulus (Ep); (e) cross-sectional distensibility (CD); (f) arterial stiffness index (β). *p < 0.05 versus DM + vehicle, ***p < 0.001 versus DM + vehicle.
Recovery of aortic compliance after TRB3 silencing
The elevated Ep and stiffness index were remarkably reduced in TRB3-siRNA group (Ep 0.22 ± 0.06 vs. 0.13 ± 0.06 × 106 dyn/cm2, p < 0.01; stiffness index 2.78 ± 0.20 vs. 2.32 ± 0.26, p < 0.001) (Figure 5(d) and (f)). In contrast, the CD was significantly elevated compared with the vehicle (0.08 ± 0.03 vs. 0.05 ± 0.01 kPa−1, p < 0.01) (Figure 5(e)). However, there were no differences in SBP, DBP and Ds between the TRB3-siRNA and vehicle groups.
TRB3 gene silencing ameliorated metabolic disturbance
Elevated serum TC, TG and FBG levels in diabetic rats were dramatically reduced after 4 weeks of transfection (Table S3). ISI was increased after gene transfer compared with the vehicle (−5.17 ± 0.14 vs. −5.67 ± 0.14, respectively, p < 0.05) (Table S3).
TRB3 gene silencing reverses pathological remodelling
During 4-week follow-up after TRB3-siRNA treatment, the aortic medial thickness and PVCA/LA were decreased in TRB3 silencing group relative to the vehicle (medial thickness 74.88 ± 1.21 vs. 69.38 ± 1.11 µm, p < 0.01; PVCA/LA 0.10 ± 0.005 vs. 0.06 ± 0.006, p < 0.001) (Figure 6(a)–(c)). TRB3 gene therapy markedly reduced the collagen deposition (3.77% ± 0.36% vs. 2.48% ± 0.22%, p < 0.01) (Figure 6(e)) and increased the elastin content (5.29% ± 0.36% vs. 7.79% ± 0.72%, p < 0.01) (Figure 6(f)), leading to a lower collagen/elastin ratio (0.75 ± 0.09 vs. 0.36 ± 0.07, p < 0.01) (Figure 6(g)) in TRB3-siRNA group compared with the vehicle. All these above results pointed to a beneficial regression of large artery structure. Additionally, there was an evident reduction in total collagen content compared to the vehicle (14.16 ± 0.30 vs. 10.70 ± 0.19 µg/mg dry tissue weight, p < 0.001) (Figure 6(i)). Collagen I and collagen III were lower in the TRB3-siRNA group than the vehicle (p < 0.01) (Figure 6(j) and (k)). In keeping with these observations, the profound decreased IB% and increased CVIB of the aortic wall reconfirmed the improved pathological manifestations (p < 0.05~p < 0.001) (Table S4).
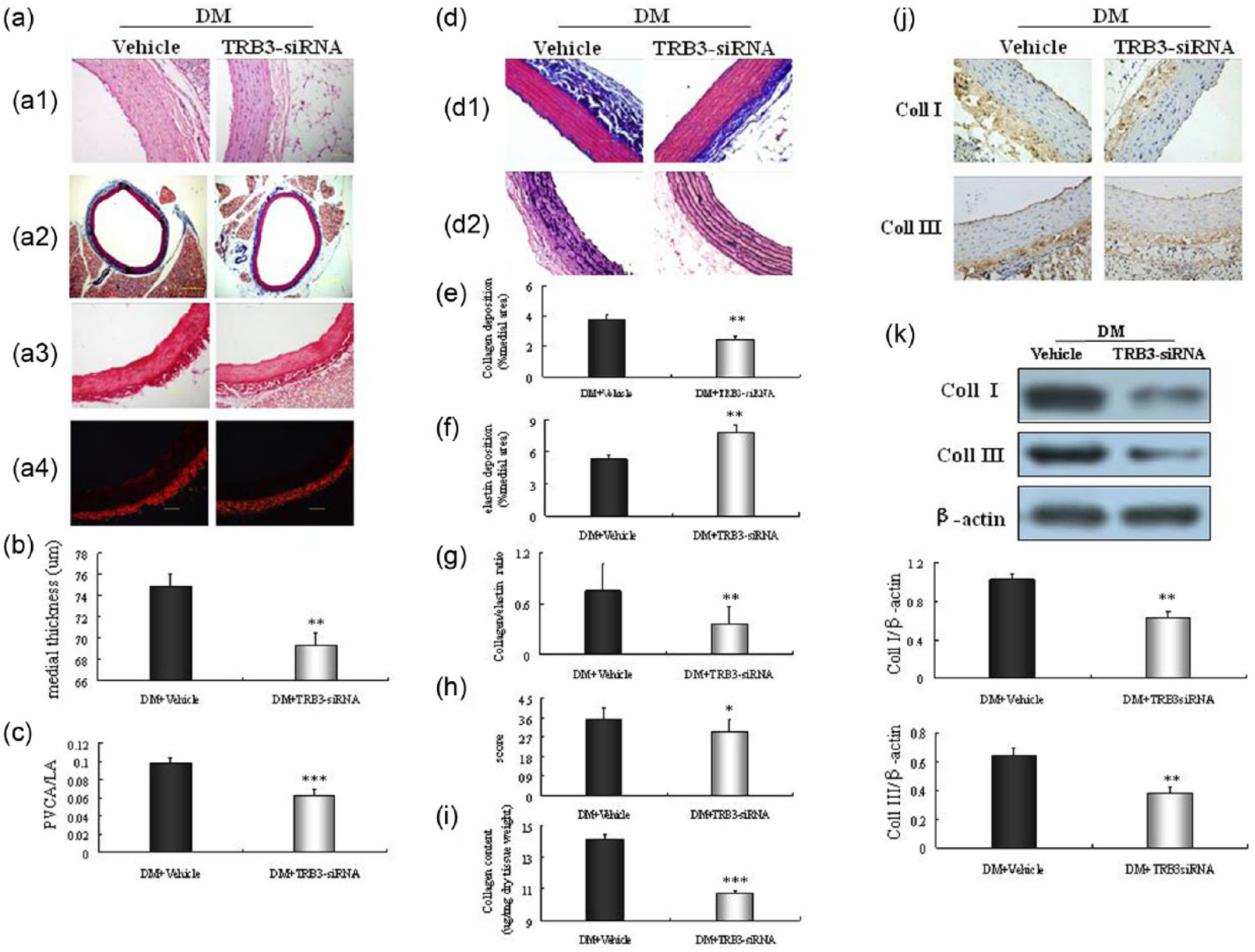
TRB3 gene therapy reverses pathological vascular remodelling. (a) a1: Haematoxylin–eosin staining of arterial sections. Bar: 50 µm. a2: Micrographs of aortic sections stained with Masson trichrome stain. Bar: 400 µm. a3 and a4: Picrosirius red staining (a3: bright-field; a4: dark-field) of arterial sections. Bar: 100 µm. (b) Quantitative analysis of medial thickness. (c) Quantitative analysis of PVCA/LA. The medial thickness and PVCA/LA were remarkably reduced in the TRB3-siRNA group. (d) d1: Masson trichrome slides showing collagen deposition in the aorta. Bar: 50 µm. d2: Verhoeff’s stain showing elastin disruption. Bar: 50 µm. (e–g): Analyses of collagen, elastin and the ratio of collagen to elastin. (h) Average aortic wall architecture score. (i) Collagen content detected by hydroxyproline assay. All these above results pointed to a beneficial regression of aortic structure in TRB3-siRNA group. (j and k) Representative immunohistochemistry (bar: 50 µm) and western blot of collagen I, III. Results are expressed as mean ± SEM, n = 6. *p < 0.05, **p < 0.01, ***p < 0.001 versus DM + vehicle.
TRB3 silencing exerted differential regulations of PI3K/Akt and JNK pathways
With TRB3 silencing, the suppressed phosphorylation of PI3K/Akt was recovered (Figure 7(a) and (b)), and the activation of MKK4 and JNK was abolished by 63% and 34%, respectively, in DM TRB3-siRNA group (Figure 7(c) and (d)). In summary, TRB3 gene silence appeared to have differential effects on PI3K/Akt and MKK4/JNK pathways: restoring phosphorylation of PI3K/Akt but inhibiting MKK4/JNK.
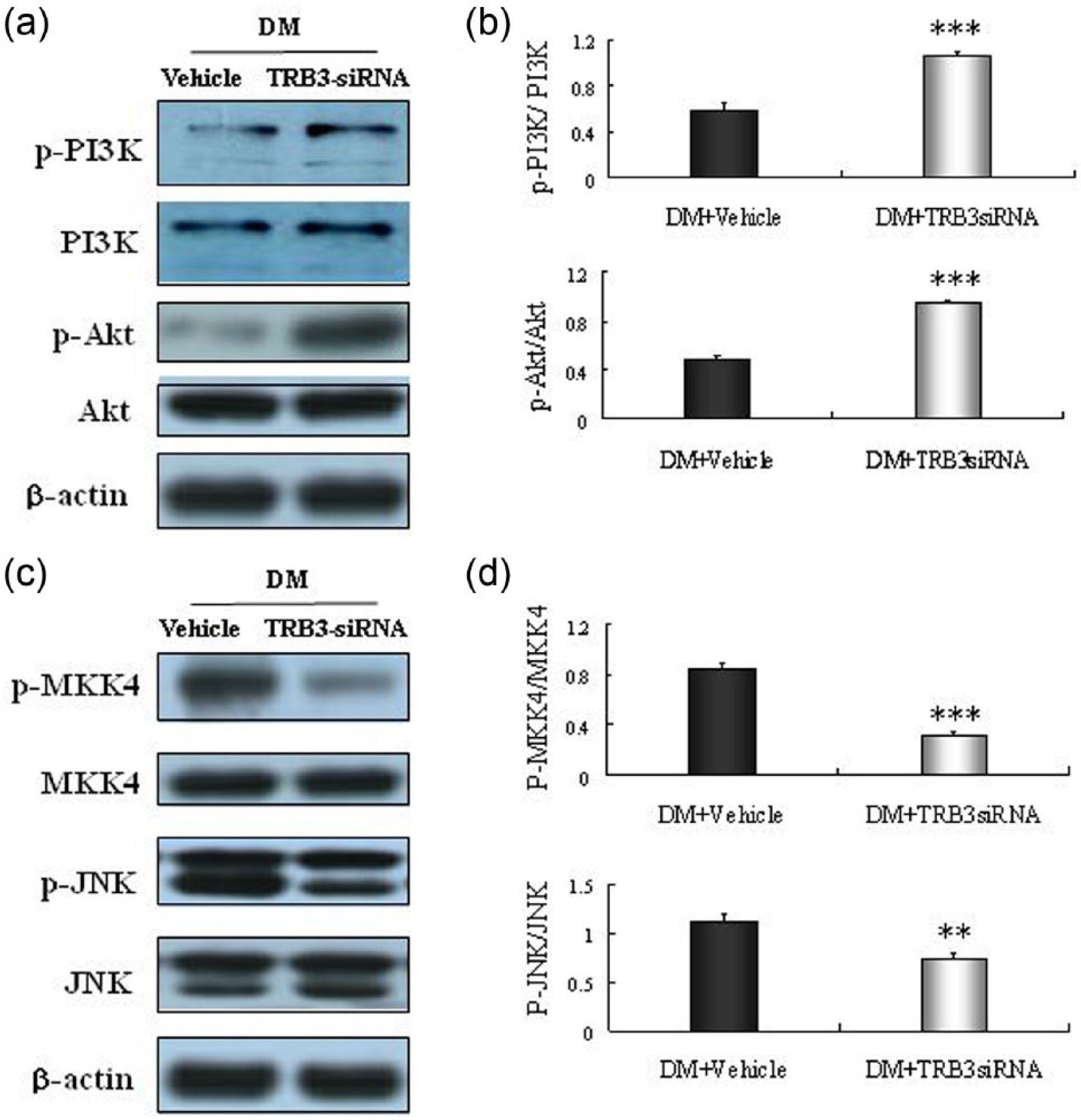
TRB3 silencing exerted differential regulations of PI3K/Akt and JNK pathways. (a and b) Representative western blots of PI3K, Akt. The blunted PI3K/Akt pathway was recovered after TRB3 silencing. (c and d) Representative western blots of MKK4 and JNK. Significant activations of MKK4/JNK were abolished with silence of TRB3. Bar graph showed mean ± SEM of western blot analysis. **p < 0.01, ***p < 0.001 versus DM + vehicle.
Discussion
The salient finding of this study is that TRB3 plays a pivotal role in diabetic aortic remodelling. More importantly, silence of TRB3 reverses pathological aortic remodelling possibly through MKK4/JNK pathway. Thus, TRB3 could be an effective therapeutic candidate against aortic remodelling in type 2 diabetes.
In this study, the type 2 diabetic rats exhibited decreased phosphorylation of Akt with up-regulated TRB3 in aortic tissue. This finding was consistent with previous studies,8,9 that is TRB3 inhibiting insulin signalling by binding and preventing activation of Akt in diabetes. Additionally, the activity of PI3K was also reduced in parallel to TRB3 elevation. As we know, the PI3K/Akt pathway plays an important role in the metabolic effects of insulin. 10 In this study, elevated blood glucose and impaired insulin sensitivity were observed in diabetic model. Thus, we deduced that the metabolic disturbance observed in diabetes was attributed to blunted PI3K/Akt activity induced by overexpressed TRB3.
To our knowledge, insulin resistance is known as ‘pathway selective’. 10 That is, PI3K/Akt pathway appears to be selectively inhibited, compared with fully activated MAPK pathway. Intriguingly, Kiss-Toth et al. 11 reported that TRB3 serves as a molecular switch and regulates the relative activation of the three classes of MAPK. In this study, we noticed that MKK4/JNK was profoundly activated in accompanied by TRB3 elevation. Meanwhile, there were obvious collagen deposition and elastin disruption in aortic tissues, in keeping with decreased vascular compliance in diabetic rats. However, there were no differences in blood pressures. These observations suggested that evident aortic remodelling existed in type 2 diabetic rats, without blood pressure modification. It was reported that activated JNK was contributable to vascular fibrosis 22 and remodelling. 23 Thus, we hypothesized TRB3 was implicated in diabetic aortic remodelling possibly mediated by JNK pathway.
Thus, TRB3 is a key regulator for ‘selective’ insulin resistance through modulating the PI3K/Akt and JNK pathways. Meanwhile, insulin resistance was the main trigger to vascular remodelling in diabetes. 4 These data raise the possibility that TRB3 elevation contributed to the development of aortic remodelling.
Given the involvement of overexpressed TRB3 in diabetic aortic remodelling, we aimed to explore the potential therapeutic effect of TRB3 as a novel treatment of aortic remodelling. Accordingly, TRB3-siRNA was used in vivo. After the systemic silencing, there was no notable adverse effect and no death in rats receiving TRB3-siRNA or vehicle. Analysis of TRB3 protein expression demonstrated that TRB3 was significantly decreased by 68%. On the basis of this finding, the systemic gene therapy was secure and feasible.
We noticed that the blood glucose became lower and insulin sensitivity was increased in TRB3-siRNA group. Accompanied with these alterations, activities of PI3K/Akt were recovered. Most likely, this improved metabolic disturbance was associated with normalized activation of PI3K/Akt. 24
More importantly, aortic thickness, perivascular fibrosis, collagen content and the medial collagen/elastin ratio were significantly decreased in TRB3-siRNA group. In addition, according to the echocardiography results, the vascular stiffness was markedly reduced and the compliance was improved. These observations revealed a role of TRB3 knockdown in reversing pathological vascular remodelling in diabetes. Intriguingly, the activations of MKK4/JNK were abolished in keeping with silence of TRB3. With the aforementioned, it was demonstrated that silence of TRB3 decreased MKK4/JNK phosphorylations, thereby diminishing their contribution to aortic remodelling. On the other hand, PI3K and Akt are important positive regulators of endothelial nitric oxide synthase (eNOS), which generates nitric oxide (NO) and ultimately leads to vasorelaxation.25,26 Previous study 27 has shown that TRB3 R84 variant impairs NO production in human endothelial cells. Accordingly, the restored PI3K/Akt pathway might also contribute to these alterations, in relation to its role in NO production.
Collectively, our observations highlight the importance of PI3K/Akt and JNK pathways in TRB3-related aortic remodelling. Silence of TRB3 effectively alleviated ‘selective’ insulin resistance, thereby the pathological remodelling was remarkably improved. Our findings strongly suggest a therapeutic potential for TRB3 silencing in the aortic remodelling under diabetes. There have since been studies on the phenotype of TRB3 knockout mice. TRB3 knockout mice are protected from HF diet-induced insulin resistance in skeletal muscle. 28 According to these studies, TRB3 is likely to be a safe target in humans.
Conclusion
TRB3, being a key regulator involved in insulin resistance, played an important role in diabetic aortic remodelling. Furthermore, our study has convincingly shown that TRB3 is a potent target mediating regression of aortic remodelling in type 2 diabetic rats.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the research grants from the National Basic Research Program of China (973 Program, Grant No. 2012CB722406), the National Natural Science Foundation of China (81070192, 81070141, 81100605, 81270352, 81270287, 81300168, 81471036 and 81470560), the Natural Science Foundation of Shandong Province (BS2013YY017, ZR2014HQ037), the Independent Innovation Foundation of Shandong University (2012JC034), cardiovascular exploration research foundation of Chinese Medical Doctor Association (DFCMDA201320) and the Specialized Research Fund for the Doctoral Program of Higher Education (SRFDP 20130131120065).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.