Abstract
Borderline ankle–brachial index is increasingly recognised as a marker of cardiovascular risk. We evaluated the impact of borderline ankle–brachial index in 12,772 Chinese type 2 diabetes patients from the Joint Asia Diabetes Evaluation Program between 2007 and 2012. Cardiovascular risk factors, complications and health-related quality of life were compared between patients with normal ankle–brachial index (1.0–1.4), borderline ankle–brachial index (0.90–0.99) and peripheral arterial disease (ankle–brachial index < 0.9). The prevalence of peripheral arterial disease and borderline ankle–brachial index was 4.6% and 9.6%, respectively. Borderline ankle–brachial index patients were older, more likely to be smokers and hypertensive, had longer duration of diabetes, poorer kidney function and poorer health-related quality of life than patients with normal ankle–brachial index. After adjustment for traditional cardiovascular risk factors, borderline ankle–brachial index was an independent predictor of diabetes-related micro- and macrovascular complications including retinopathy (odd ratios: 1.19 (95% confidence interval: 1.04–1.37)), macroalbuminuria (1.31 (1.10–1.56)), chronic kidney disease (1.22 (1.00–1.50)) and stroke (1.31 (1.05–1.64)). These findings suggest that patients with diabetes and borderline ankle–brachial index are at increased cardiovascular risk and may benefit from more intensive management.
Keywords
Introduction
Asia is currently facing a burgeoning epidemic of type 2 diabetes.1,2 Cardiovascular (CV) diseases remain the leading causes of morbidity and pre-mature death in the diabetic population, but the assessment of CV risk is often inadequate.1,3 The measurement of ankle–brachial index (ABI) is a simple and useful method for estimating CV risk and is recommended by the American Diabetes Association for all diabetic patients older than 50 years. 4 An ABI value ⩽0.9 is conventionally used as the threshold to define peripheral arterial disease (PAD) and high CV risk.5,6 Recent studies have reported that ABI value of 0.91–0.99 is also associated with increased risk of CV diseases (CVDs) in non-diabetic patients.7,8 The 2011 American College of Cardiology Foundation (ACCF) and American Heart Association (AHA) guidelines for the management of PAD have recommended ABI values of 0.90–0.99 as ‘borderline’ rather than normal. 9 However, there is a paucity of data on the impact of borderline ABI in diabetic patients. In this study, we aim to evaluate the prevalence of borderline ABI and its associations with diabetes-related CV complications in a large cohort of Chinese patients with type 2 diabetes.
Methods
Study subjects and setting
Between November 2007 and July 2012, Chinese patients with type 2 diabetes aged 18 years or above enrolled into the Joint Asia Diabetes Evaluation (JADE) Program from three public hospitals and three community-based out-patient clinics in Hong Kong were studied. Exclusion criteria included patients with type 1 diabetes as defined by presentation with ketoacidosis, unprovoked ketosis or requirement of insulin within 1 year of diagnosis; ABI > 1.4 indicating non-compressible arteries; and missing data on ABI. The design, implementation and adoption of the JADE Program have been published.10–12 In brief, the JADE Program is a web-based programme that uses information technology to facilitate healthcare professionals to create a diabetes registry and to deliver evidence-based care tailored to patients’ risk profiles. All enrolled patients gave written informed consent to undergo comprehensive assessment, which includes clinical and demographic information and laboratory investigations at enrolment and at regular intervals thereafter. At each review visit, key parameters as recommended by international guidelines are captured to document clinical progress, risk factor control and complications since last visit. The JADE Program is governed by the Asia Diabetes Foundation, which is a non-profit making organisation incorporated under the Chinese University of Hong Kong Foundation.
Clinical assessments
All patients recruited in Hong Kong JADE Program undertook comprehensive assessment including medical history taking, physical examination, laboratory testing and quality of life assessment using a structured protocol. History of complications, comorbidities and medication use were cross-checked with the Hong Kong Hospital Authority (HA) Clinical Management System (CMS), which captured all admissions to public health facilities used by over 90% of Hong Kong residents. Physical examination included standardised anthropometric measurements of body weight, height and waist circumference, as well as blood pressure (BP). Fundoscopy or retinal photography was used to assess the presence of retinopathy. Peripheral sensory neuropathy was assessed using monofilament and graduated tuning fork. All patients underwent routine ABI assessment during annual assessments regardless of PAD status or symptoms suggestive of PAD. ABI was measured by a trained nurse using an 8-mHz hand-held Doppler device (Summit Doppler System, Trumbull, CT) and a sphygmomanometer. Systolic BP from both brachial arteries and dorsalis pedis arteries were measured with the patient in the supine position. ABI was calculated for each leg by taking the higher pressure of the two arteries at the ankle, divided by the higher of the two arm brachial arterial systolic pressure. The lower ABI of each patient was used for analysis. ABI was classified into one of the three categories: PAD (ABI ⩽ 0.90), borderline ABI (0.91–0.99) and normal ABI (1.00–1.40).
Fasting blood samples after at least an 8-h fast overnight were collected for measurement of fasting plasma glucose, glycated haemoglobin (HbA1c), lipid profile [total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C) and triglycerides] and renal function. The abbreviated Modification of Diet in Renal Disease study formula recalibrated for the Chinese population was used to determine the estimated glomerular filtration rate (eGFR). 13 A random spot urine sample was collected to measure the albumin-to-creatinine ratio (ACR).
Health-related quality of life (HRQoL) was assessed using the EuroQoL 5-dimension (EQ-5D) tool.14,15 Patients were asked to rank their health status of self-care, usual activities, pain/discomfort, mobility and anxiety/depression using three levels: ‘no problems’, ‘moderate problems’ and ‘extreme problems’. The UK tariff was used to calculate the index. 16 ‘Moderate problems’ and ‘extreme problems’ were classified as ‘having problems’ in analysis.
Definitions of complications
Macroalbuminuria was defined as urinary ACR > 25.0 mg/mmol. Chronic kidney disease (CKD) was defined as eGFR < 60 mL/min/1.73 m2. History of CVD [coronary heart disease (CHD), congestive heart failure or stroke] was based on medical history confirmed by the referring doctor and/or Hong Kong HA record. Peripheral sensory neuropathy was defined by meeting two of three criteria of abnormal sensation over lower limb, reduced sensation to monofilament over any part of the sole and a score of ⩽6 of 8 (aged >65 years) or ⩽4 of 8 (aged ⩾65 years) using a graduated tuning fork. Retinopathy was defined by typical retinal changes including vitrectomy, haemorrhages and exudates on fundoscopy. General obesity was defined as body mass index (BMI) ⩾27.5 kg/m2 while central obesity was defined as waist circumference ⩾90 cm in men or ⩾80 cm in women. Hypertension was defined as BP ⩾ 140/90 mmHg and/or treatment with anti-hypertensive drugs. Dyslipidaemia was defined as a plasma LDL-C ⩾ 2.6 mmol/L, HDL-C < 1 mmol/L, triglycerides ⩾ 2.3 mmol/L 17 or treatment with any lipid-regulating drugs. The ‘ABC’ treatment goals were defined as (A) HbA1c < 7.0%, (B) BP < 130/80 mmHg and (C) LDL-C < 2.6 mmol/L. 18 We also calculated the 5-year risk of CHD and stroke using the equations derived from the Hong Kong Diabetes Registry.19,20 We then used 5-year CHD risk ⩾20% and 5-year stroke risk ⩾10% to identify high-risk subjects for future CV events in those without prior history of CHD or stroke.
Statistical analysis
All data were reported as number (percentages), mean ± standard deviation (SD) or median [interquartile range (IQR)] as appropriate. χ2-test, Kruskal–Wallis H test and one-way analysis of variance (ANOVA) were used for comparisons as appropriate. Multiple logistic regression models were used to examine the associations between ABI and microvascular (retinopathy, CKD, sensory neuropathy) and macrovascular complications (stroke, CHD) using normal ABI as the reference and expressed as odds ratios (OR) with 95% confidence interval (CI). The covariates entered into the model were based on clinical a priori knowledge, including age, sex, duration of diabetes, high school education, smoking, waist circumference, hypertension, dyslipidaemia and HbA1c. Statistical analysis was performed using the SPSS (version 20.0, IBM). A p-value < 0.05 was considered to be statistically significant. Bonferroni correction was used for post hoc comparisons between normal, borderline ABI and PAD.
Results
Of the 13,228 patients with diabetes enrolled into the JADE Program, we excluded 280 patients with type 1 diabetes, 112 patients with missing data for ABI and 64 (0.5%) patients with an ABI > 1.4. Of the remaining 12,772 patients (mean age: 58.8 ± 11.9 years), 54.6% (n = 6972) were men and median duration of diabetes was 7 (IQR: 2–14) years. Anthropomorphically, 63.6% of patients were centrally obese, 63.6% had hypertension and 87.4% had dyslipidaemia. In these subjects, 9.2% had CKD, 6.4% had history of stroke and 10.3% had known CHD.
Overall, 9.6% had borderline ABI and 4.6% had PAD with higher rates in women (borderline ABI 11.6% and PAD 5.4%) than men (borderline ABI 8.0%, PAD 4.0% and both p < 0.001). Compared to patients with normal ABI, those with borderline ABI had lower education and were more likely to be active smokers (Table 1). They also had longer duration of diabetes, higher systolic BP and worse renal function but had comparable BMI, waist circumference, HbA1c, LDL-C and triglyceride. Fewer patients in the borderline ABI group attained all three ‘ABC’ targets than normal ABI group (7.5% vs 10.0%, p = 0.002, Table 2). Among the three groups, patients with PAD had the worst metabolic profile with the highest HbA1c, systolic BP and waist circumference, although waist circumference was only significantly higher in women with PAD. Overall, 21.8% of patients were treated with insulin with a progressive increase in insulin usage across normal ABI, borderline ABI and PAD groups. While patients with borderline ABI were more likely than the normal ABI group to receive anti-hypertensive drugs, they were less likely to be treated with statins and renin–angiotensin system (RAS) inhibitors than those with PAD (both p < 0.001) (Table 2).
Demographic and clinical characteristics of Chinese patients with type 2 diabetes categorised according to ankle–brachial index (ABI).
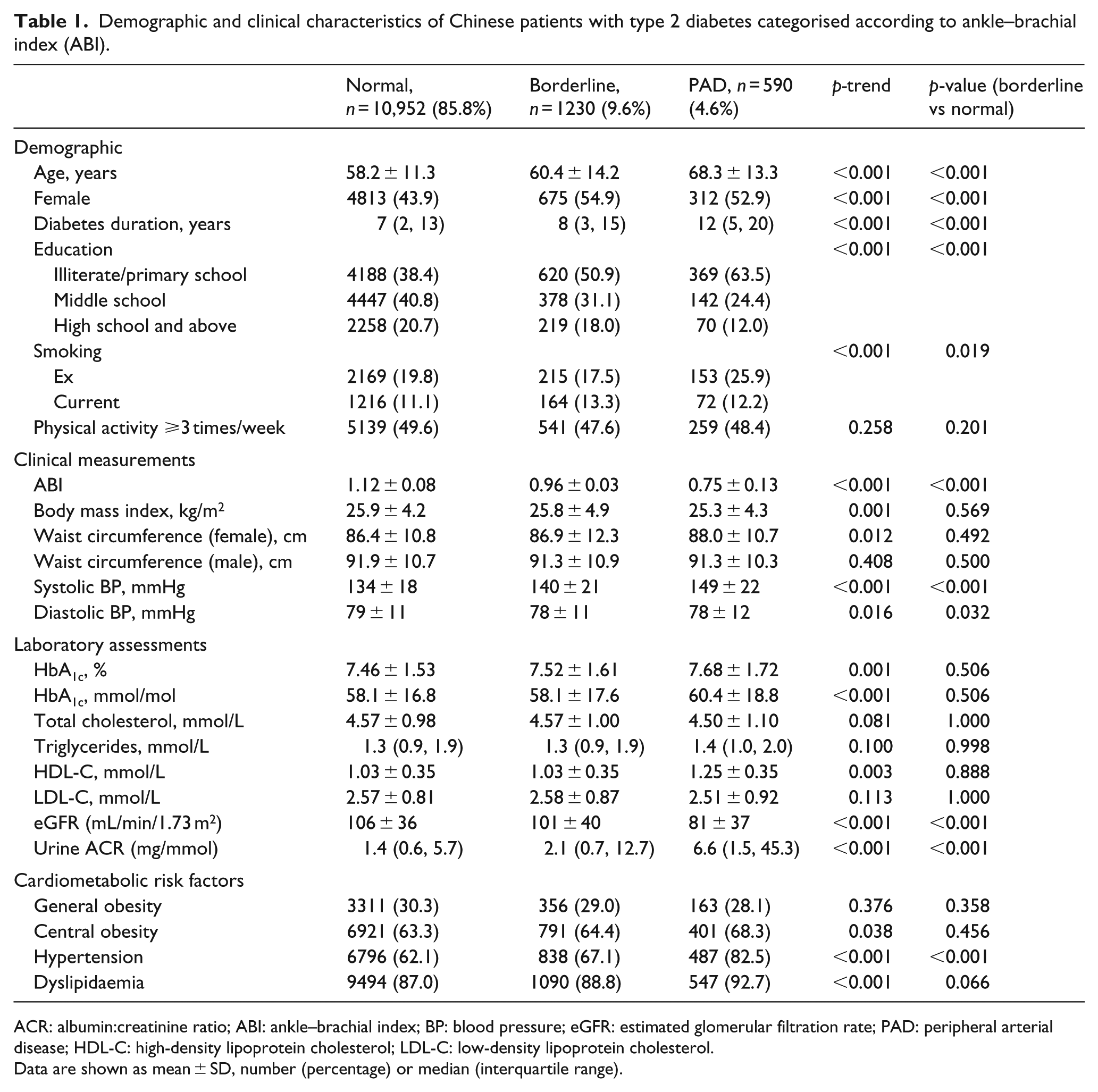
ACR: albumin:creatinine ratio; ABI: ankle–brachial index; BP: blood pressure; eGFR: estimated glomerular filtration rate; PAD: peripheral arterial disease; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol.
Data are shown as mean ± SD, number (percentage) or median (interquartile range).
Diabetes-related complications, treatment and achievement of therapeutic targets in type 2 diabetic patients according to ABI category.
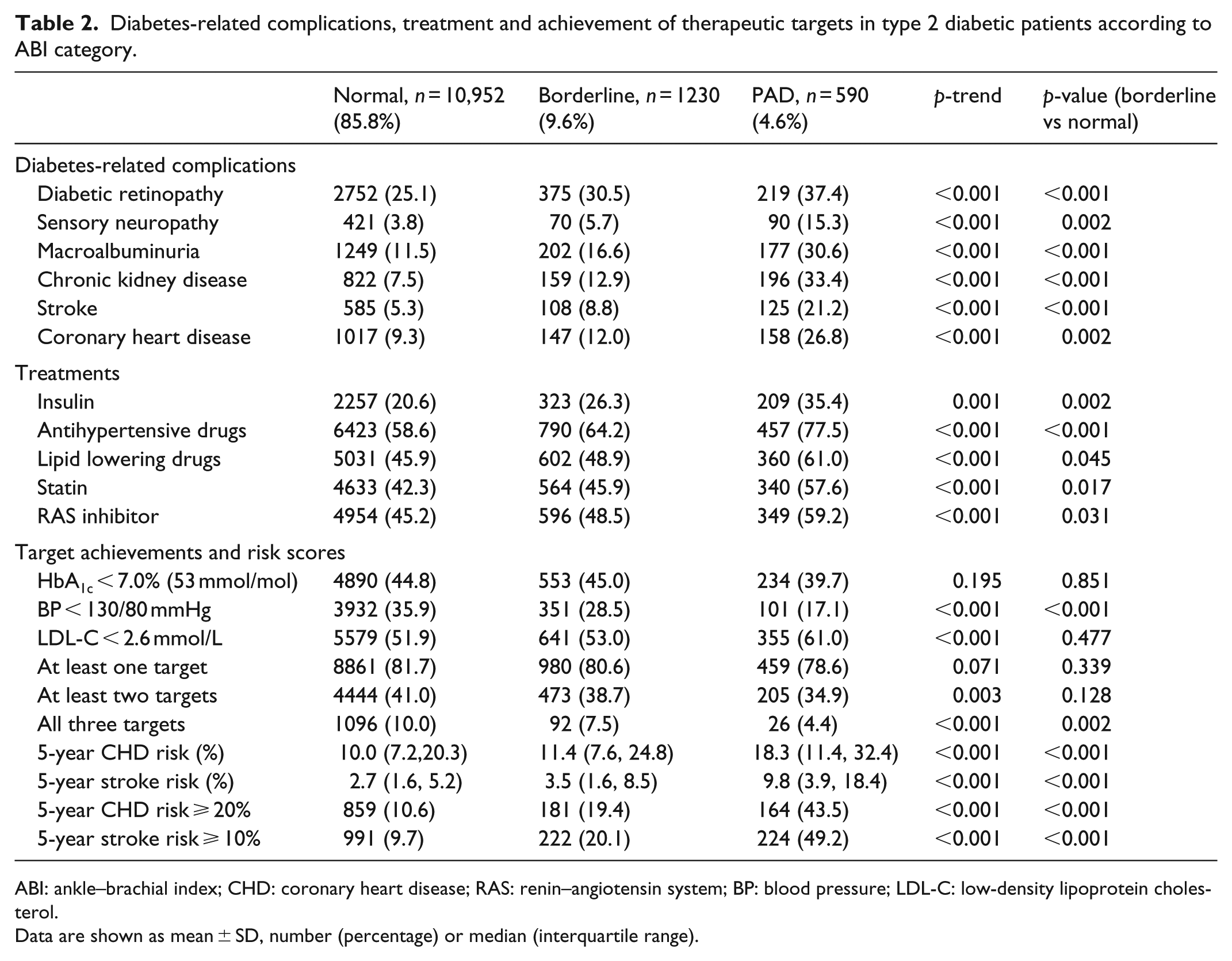
ABI: ankle–brachial index; CHD: coronary heart disease; RAS: renin–angiotensin system; BP: blood pressure; LDL-C: low-density lipoprotein cholesterol.
Data are shown as mean ± SD, number (percentage) or median (interquartile range).
Based on the risk equations derived from the Hong Kong Diabetes Registry,19,20 the 5-year risk of stroke and CHD increased with declining ABI measurements (p-trend < 0.001) (Table 2). Additionally, more patients with borderline ABI had higher frequencies of CHD, stroke, CVD, macroalbuminuria and CKD than those with normal ABI. They also had higher rates of retinopathy and sensory neuropathy (Table 2). After adjusting for traditional CV risk factors, including age, sex, disease duration, higher school education, smoking, waist circumference, hypertension, dyslipidaemia and HbA1c, borderline ABI remained independently associated with 1.2–1.3 increased odds of retinopathy, macroalbuminuria, CKD and stroke (Figure 1).
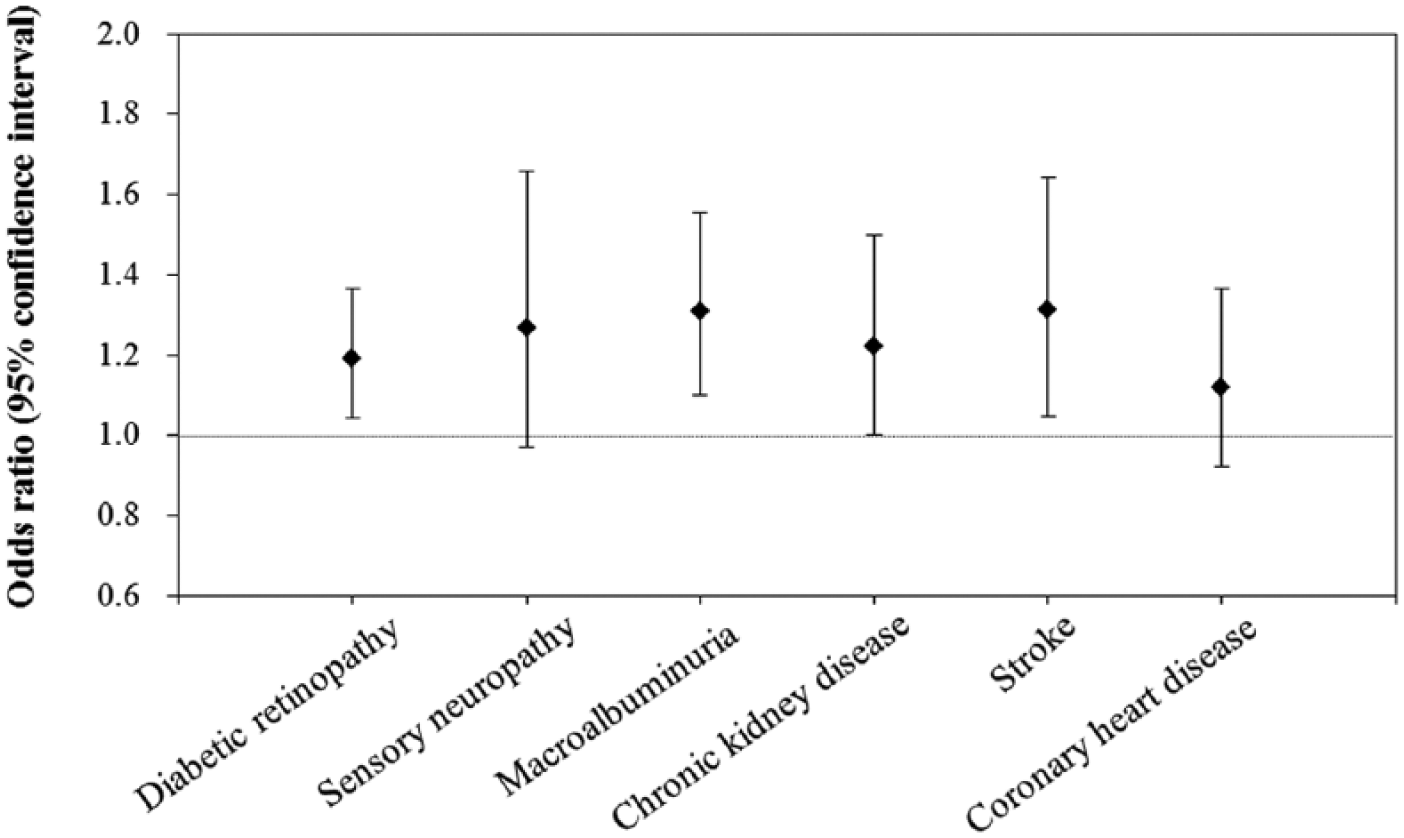
Adjusted odds ratios for micro- and macrovascular complications in patients with borderline ankle–brachial index (ABI) compared to those with normal ABI.
Patients with PAD and borderline ABI reported lower HRQoL than patients with normal ABI as evidenced by lower EQ-5D health utility index. Across the five dimensions of EQ-5D evaluated, patients with borderline ABI were more likely to experience problems in mobility, self-care and usual activity, but not anxiety/depression or pain/discomfort than those with normal ABI (Figure 2).
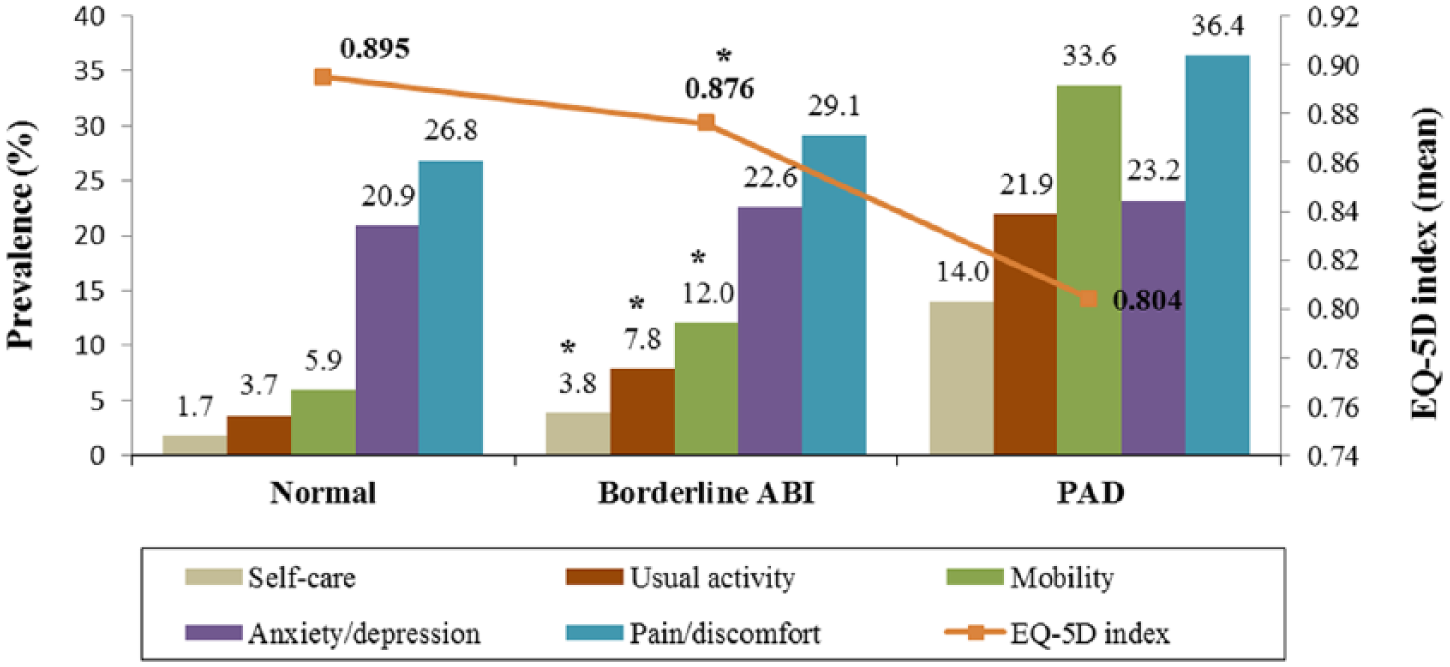
Prevalence of reported problems in EQ-5D by ABI groups.
Discussion
To our best knowledge, this is the first report on the prevalence of borderline ABI and its associations with micro- and macrovascular complications in a large cohort of Chinese patients with type 2 diabetes. We demonstrated that twice as many diabetic patients had borderline ABI (9.6%) than PAD (4.6%). Apart from having poorer HRQoL, patients with borderline ABI had 20%–30% increased odds of having micro- and macrovascular complications compared to those with normal ABI. However, these high-risk subjects were less likely to be prescribed disease-modifying drugs such as RAS inhibitors and statins than those with PAD. These findings suggest that borderline ABI identifies a significant population of high-risk type 2 diabetic patients for whom intensive management may reduce future risk of CV–renal complications. PAD has been reported in 6%–30% of Chinese type 2 diabetes patients,21–23 but the prevalence of borderline ABI remained less studied. In our cohort, the prevalence of borderline ABI (9.6%) was comparable to reported rates of 8.9% and 8.7% in a recent Japanese study of 3981 diabetic patients and in US National Health and Nutrition Examination Survey (NHANES, 1999–2002) involving 4895 subjects aged ⩾40 years in the community, respectively.8,24 Women in our cohort were more likely to have borderline ABI (11.6%) than men (8.0%). Similarly, in the NHANES (1999–2002) and the Multi-Ethnic Study of Atherosclerosis (MESA), the prevalence of borderline ABI nearly doubled in women (11.7% and 10.6%) than men (6.0% and 4.3%).7,8 The increased prevalence of borderline ABI in women may be due to older age compared to men.
Borderline ABI is increasingly recognised as a marker of CV risk. In the MESA study, patients with borderline ABI were more likely to have increased carotid artery intima-media thickness and a coronary artery calcium score >20 than those with normal ABI. 7 In a meta-analysis, patients with borderline ABI had a hazard ratio (HR) of 1.43 in men and 1.53 in women for major adverse coronary events. 25 In a recent study of Japanese diabetic patients, the risks for all-cause death in patients with PAD (HR = 2.16; 95% CI: 1.46–3.14) and borderline ABI (HR: 1.78; 95% CI: 1.14–2.70) were significantly higher than in those with normal ABI. 24 In our study, 5-year CHD and stroke risk were increased by 14%–30% in patients with borderline ABI. The latter was also independently associated with retinopathy and macroalbuminuria, which predicted CV and all-cause death in both general 26 and diabetic populations. 27
The association of impaired physical function and HRQoL with PAD, with or without symptoms, has been reported.28,29 In our study, patients with borderline ABI had worse HRQoL and lower scores in all EQ-5D (mobility, self-care and usual activity) except for anxiety/depression and pain/discomfort compared to those with normal ABI. Other researchers have also reported poorer HRQoL in individuals with borderline ABI than in those with normal ABI. 30 In a 5-year prospective study, borderline ABI was associated with a HR of 3.07 (95% CI: 1.21–7.84) for loss of mobility compared to those with normal ABI. 31 These findings highlighted that in addition to being at risk for clinical events, patients with borderline ABI also had functional impairment and suboptimal quality of life.
Akin to the prognostic significance of ‘pre-hypertension’ (systolic BP: 120–139 mmHg and/or diastolic BP: 80–89 mmHg), 32 our findings strongly support the use of borderline ABI to identify high-risk diabetic patients for early intervention. In the NHANES cohort, subjects with borderline ABI were more likely to have worse modifiable cardio-metabolic risk factors such as hypertension and abdominal obesity. 8 Clairotte et al. 33 found that an ABI threshold of 0.90 had a high specificity (96.8%) but a medium sensitivity (54.4%) for two-dimensional ultrasound-identified arterial stenosis in diabetic patients compared to a sensitivity of 98.3% and specificity of 72.6% in non-diabetic subjects. However, using a cut-off value of 1.0–1.1 had better performance and identified >80% of diabetic patients with PAD. 33 In support of these findings, our results also suggested the use of a higher cut-off value for PAD screening in diabetic patients.
In both randomised clinical trials and observational cohorts, optimal control of multiple risk factors reduced micro- and macrovascular complications and mortality in type 2 diabetes.2,18 However, less than one-third of our patients with borderline ABI attained BP target (<130/80 mmHg), and only half were at LDL-C goal (<2.6 mmol/L). Compared to patients with PAD, they were also less likely to be treated with statins or RAS inhibitors. Given the increased risk of complications in those with borderline ABI, using a higher cut-off value of ABI may identify high-risk subjects for intensive management.
Several limitations of this study need to be acknowledged. First, this study only involved Chinese patients and therefore might not apply to other ethnic populations. Second, we did not repeat ABI measurements since all nurses had undergone training. Besides, measurement bias in this large population may be assumed to be random and are not likely to affect the conclusions. Third, despite the relatively large sample size and careful clinical assessment, the cross-sectional nature of our study does not allow causal inference of the associations between borderline PAD and diabetic complications. Further research is required to validate the predictive value of borderline ABI for risk stratification and clinical outcomes in type 2 diabetes.
In Chinese patients with type 2 diabetes, twice as many subjects had borderline ABI as those with PAD which was associated with increased prevalence of micro- and macrovascular complications and decreased HRQoL. Diabetic patients with not only abnormal ABI < 0.9 but also borderline ABI (0.91–0.99) could be at high risk of CV complications. We proposed using a higher cut-off value of ABI (<1.0) to screen for CV risk, potentially identifying an additional 10% of high-risk diabetic patients for intensive treatment.
Footnotes
Acknowledgements
We thank all participating doctors, nurses and patients for contributing to the JADE Program. Special thanks are extended to Ms Harriet Chung, Ms Rebecca Wong, Ms Grace Lin, Ms Maggie Lau and Ms Marina Cheung, for coordinating the diabetes comprehensive assessment programmes. B.P.Y., Y.Z., A.P.S.K. and J.C.N.C. participated in the study design and conception. B.P.Y., Y.Z., A.P.S.K., A.O.Y.L., R.O., R.Y., P.C.Y.T., C.T., K.L., T.W., G.L., W.S., R.C.W.M., F.C.C.C. and J.C.N.C. performed the statistical analysis. B.P.Y., Y.Z., A.O.Y.L., R.O., R.Y., P.C.Y.T., C.T., K.L., W.S., R.C.W.M., F.C.C.C. and J.C.N.C. performed the data interpretation. B.P.Y. and Y.Z. drafted the manuscript. B.P.Y., Y.Z., A.P.S.K., A.O.Y.L., R.O., R.Y., P.C.Y.T., C.T., K.L., T.W., G.L., W.S., R.C.W.M., F.C.C.C. and J.C.N.C. revised the manuscript. A.P.S.K. and J.C.N.C. participated in the data acquisition. All authors read and approved the final manuscript. B.P.Y. and Y.Z. have equal contributions.
Declaration of conflicting interests
Dr Juliana CN Chan has received honorarium for consultancy or giving lectures from Bayer, Boehringer Ingelheim, Daiichi-Sankyo, Eli-Lilly, GlaxoSmithKline, Merck Sharp & Dohme, Merck Serono, Pfizer, Astra Zeneca, Sanofi, Novo-nordisk and/or Bristol-Myers Squibb. Troels Wolthers and Greg Lyubomirsky are also employees of Merck Sharp & Dohme. The rest of authors declare that they have no conflicts of interest.
Funding
This work was partially supported by the Asia Diabetes Foundation, a charitable organisation under the auspices of the Chinese University of Hong Kong Foundation, and an unrestricted grant from Merck Sharpe & Dohme and Liao Wun Yuk Diabetes Memorial Fund, the latter under the Chinese University of Hong Kong.
