Abstract
Objective:
To analyse the association between serum C-peptide and coronary artery disease in the general population.
Methods:
Follow-up study of 6630 adults from the general population. They were stratified into group 1 (no insulin resistance: C-peptide < third tercile and glycaemia < 100 mg/dL), group 2 (initial insulin resistance: C-peptide ⩾ third tercile and glycaemia < 100 mg/dL) and group 3 (advanced insulin resistance: glycaemia ⩾ 100 mg/dL).
Results:
After 3.5 years of follow-up, group 2 had a higher incidence of myocardial infarction (relative risk (RR) = 4.2, 95% confidence interval (CI) = 1.7–10.6) and coronary artery disease (RR = 3.5, 95% CI = 1.9–6.6) than group 1. Group 3 also had increased incidences of both diseases. In multivariable analysis of the entire population, groups 2 and 3 showed significant risks of myocardial infarction and coronary artery disease (RR > 3 and RR > 2, respectively). However, when people with diabetes were excluded, the increased risks were corroborated only in group 2 for myocardial infarction (RR = 2.8, 95% CI = 1.1–6.9; p = 0.025) and coronary artery disease (RR = 2.4, 95% CI = 1.3–4.6; p = 0.007).
Conclusion:
Elevated C-peptide is associated with the incidence of myocardial infarction and coronary artery disease in the general population. It can be an earlier predictor of coronary events than impaired fasting glucose.
Background
C-peptide was discovered by Steiner et al. 1 in 1967 as a by-product of proinsulin. Later, it has been shown to play a physiological role in different types of cells, including endothelial cells and fibroblasts. 2 In recent years, this small 31-amino acid peptide has been revealed to play an active role in the pathogenesis of atherosclerosis 3 and possibly a risk factor for coronary artery disease (CAD).
Patients with recently diagnosed diabetes and insulin resistance (IR) have elevated blood concentrations of C-peptide, and diffuse, widespread atherosclerosis has been reported in these patients. 4 Experimental data support the hypothesis that elevated C-peptide contributes to increased cardiovascular risk in these patients. 5 Research has found that the endothelial dysfunction in patients with IR stimulates the deposition of C-peptide molecules in the vascular wall intima, where these molecules, through chemotaxis, favour the accumulation of monocytes, and that these inflammatory cells can then trigger the process of atherosclerosis. 6 Subsequently, C-peptide can perpetuate the process by promoting macrophages differentiation into foam cells, 7 and the proliferation of smooth muscle cells. 8
The few epidemiological studies to date that have evaluated the relationship between C-peptide and atherosclerosis have involved populations of patients with diabetes and have reported contradictory findings.9–11 In individuals who underwent coronary artery angiography, elevated C-peptide levels were associated with increased overall and cardiovascular mortality, and with the severity of the coronary lesions. 12 To our knowledge, no prospective cohort studies have attempted to determine whether elevated C-peptide levels modify the risk of CAD in the general population.
The aim of this study was to explore the association between C-peptide and the incidence of acute myocardial infarction (AMI) and CAD in the general population with and without hyperglycaemia.
Research design and methods
Participants
The participants in this follow-up study were members of the CDC de Canarias cohort (CDC is the acronym for cardiovascular, diabetes and cancer), which was recruited randomly from the general population of the Canary Islands between 2000 and 2005, and which constitutes a representative sample of the adult population. The sampling strategy was stratified multistage: island, zone (north and south in each island), random selection of at least one municipality, random sample of individuals from the database of those who were issued health cards. The methods used in the CDC de Canarias study were reported previously. 13 The participation rate for the analyses reported here was 70% of all recruited participants (n = 6729; aged 18–75 years).
Ethics statement
This study was approved by the Bioethics Committee of Nuestra Señora de la Candelaria University Hospital, and all participants provided their informed consent in writing.
Anthropometric and biochemical data
The participants were interviewed with a questionnaire (available at http://www.cdcdecanarias.org) to obtain information on personal and family health-related antecedents and lifestyle: Smoking (at least 1 cigarette per day), alcohol drinking (none, moderate if ⩿30 g/day and excessive when >30 g/day), sedentarism 14 and so on. Each participant underwent a physical examination to obtain data on anthropometric measures. General obesity was defined as a body mass index ⩾ 30 kg/m2, and abdominal obesity was defined as a waist/height ratio ⩾0.55. 15
A fasting venous blood sample was obtained; all samples were centrifuged in situ at room temperature for 10 min at 2000 r/min and were transported daily to the hospital laboratory. Glucose and lipid concentrations were measured on the same day the blood samples were drawn, and the values were reported as mg/dL. Serum samples were stored at −80°C, and aliquots were thawed to measure C-peptide concentration by enzyme immunoanalysis (Biosource®, ng/mL, intra-assay coefficient of variation 3.98%, inter-assay coefficient of variation 11.86%). To calculate the coefficients of variation, a pool was prepared previously with serum from 600 patients seen during 1 week at the Immunology Unit of our hospital, and C-peptide concentration was measured in 32 aliquots for the intra-assay coefficient of variation and 42 aliquots for the inter-assay coefficient.
A participant was considered to have type 2 diabetes if he or she stated having the disease and receiving antidiabetic treatment (85% of cases), or if fasting blood glucose concentration was ⩾126 mg/dL in individuals who were not aware of their status. Participants with type 1 diabetes (n = 23) were excluded from the present analysis. Hyperglycaemia was defined as a serum glucose concentration > 100 mg/dL after an 8-h fast (therefore including diabetes). Impaired fasting glucose (IFG) was considered to exist when the serum concentration was between 100 and 125 mg/dL in participants without diabetes. Hypertension was considered present if the participant stated having the disease and receiving treatment for it, or if systolic blood pressure was ⩾140 mmHg or diastolic blood pressure was ⩾90 mmHg in persons who were unaware of their status.
Cardiovascular risk was calculated with the Framingham function calibrated for the population of the Canary Islands. 16 In accordance with earlier studies, the third tercile (T3) of C-peptide concentration was used as the cut-off value for high-level exposure.9,11,12 Serum concentrations of glucose and C-peptide were combined as proposed in the homeostasis model assessment-2 (HOMA2)-IR model,17,18 and IR was considered to exist in persons in the 75th percentile (P75) or higher. Apart from the HOMA2 model, we combined values of C-peptide and glucose concentrations to build a three-level IR ordinal variable: group 1 (individuals without IR: with C-peptide < T3 and glucose < 100 mg/dL), group 2 (individuals with initial IR: C-peptide ⩾ T3 and glucose < 100 mg/dL) and group 3 (individuals with advanced IR: hyperglycaemia). AMI was considered to exist when the individual stated having a history of AMI and the diagnosis was verified in the digital medical records held by public primary care centres and hospitals (n = 35). In addition, AMI was recorded for cases (n = 10) in which a relative stated that the individual had died of AMI and the diagnosis was verified in the medical record. The diagnosis of CAD included, in addition to the 45 cases of AMI, all cases self-reported as diagnosed angina pectoris during the interview (n = 32).
Statistics
Variables measured with a normal scale are reported as summary statistics, that is, the mean ± standard deviation, and nominal variables are reported as absolute and relative frequencies (%) for each category. Chi-squared tests were used to compare proportions among groups, and analysis of variance (ANOVA) was used to compare mean values, with logarithmic transformation as needed to approximate the variable of interest to a normal distribution (Framingham function). The incidence density of AMI and CAD was obtained with the application included in OpenEpi–Epidemiologic Calculators and was expressed per 10 5 persons. Relative risk (RR) of AMI and CAD was estimated with a 95% confidence interval (95% CI) for individuals with initial IR (group 2) and advanced IR (group 3) compared to group 1, which was used as the reference group. As a worst-case analysis, we performed a sensitivity analysis including individuals lost to follow-up (n = 537), on the conservative assumption that none of them had had a cardiovascular event.
Cox regression models were used to adjust the IR effect for age and sex, with time to the diagnosis of AMI or CAD as the dependent variable. The assumption of proportionality of the risks was verified by testing the interaction between C-peptide concentration and time to the event. In addition, the models were adjusted for the factors significantly associated with MI or CAD in the univariate analysis (smoking, alcohol, sedentarism, obesity, lipids, diabetes, hypertension and previous CAD). Given the small numbers of events during the relatively brief median follow-up period used in this analysis, no more than two cardiovascular risk factors were simultaneously adjusted for, to avoid over-parameterizing the models. In addition, models were fitted for the value obtained for each participant with the Framingham function, which includes the main factors. Finally, Cox models were also adjusted for a sample paired according to the three Framingham risk score categories (low, medium, high), with 10 controls per each case of AMI or CAD; for these paired models, we use Cox regression stratified on matched pairs.
Apart from the IR group analysis, we repeated the multivariable adjustments described above to analyse the effect of elevated C-peptide (T3) compared to its normal values (terciles 1 plus 2) in the population with blood glucose < 126 mg/dL. These models were also repeated with C-peptide as a continuous variable. All calculations were done with SPSS© v.21 (Statistical Package for the Social Sciences) and program R v.2.10 (R Foundation for Statistical Computing).
Results
C-peptide concentration was measured in 6630 participants, of whom 6093 completed follow-up. Mean serum concentration was 2.16 ± 1.23 ng/mL. The cut-off value for elevated C-peptide (T3) was 2.26 ng/mL.
Cardiovascular risk was higher in IR groups 2 and 3 than group 1, except for smoking (Table 1). None of the participants in group 1, and slightly more than half of those in groups 2 and 3, had HOMA2 > P75 (Table 1). Excluding persons with diabetes reduced the prevalence of HOMA2 > P75 only in people with advanced IR (group 3 = 46.5%, 95% CI = 44.1–49.9). Figure 1 shows the association of AMI and CAD with the risk factors considered in our analysis.
Prevalence (95% CI) of cardiovascular risk factors in the three insulin resistance (IR) groups.
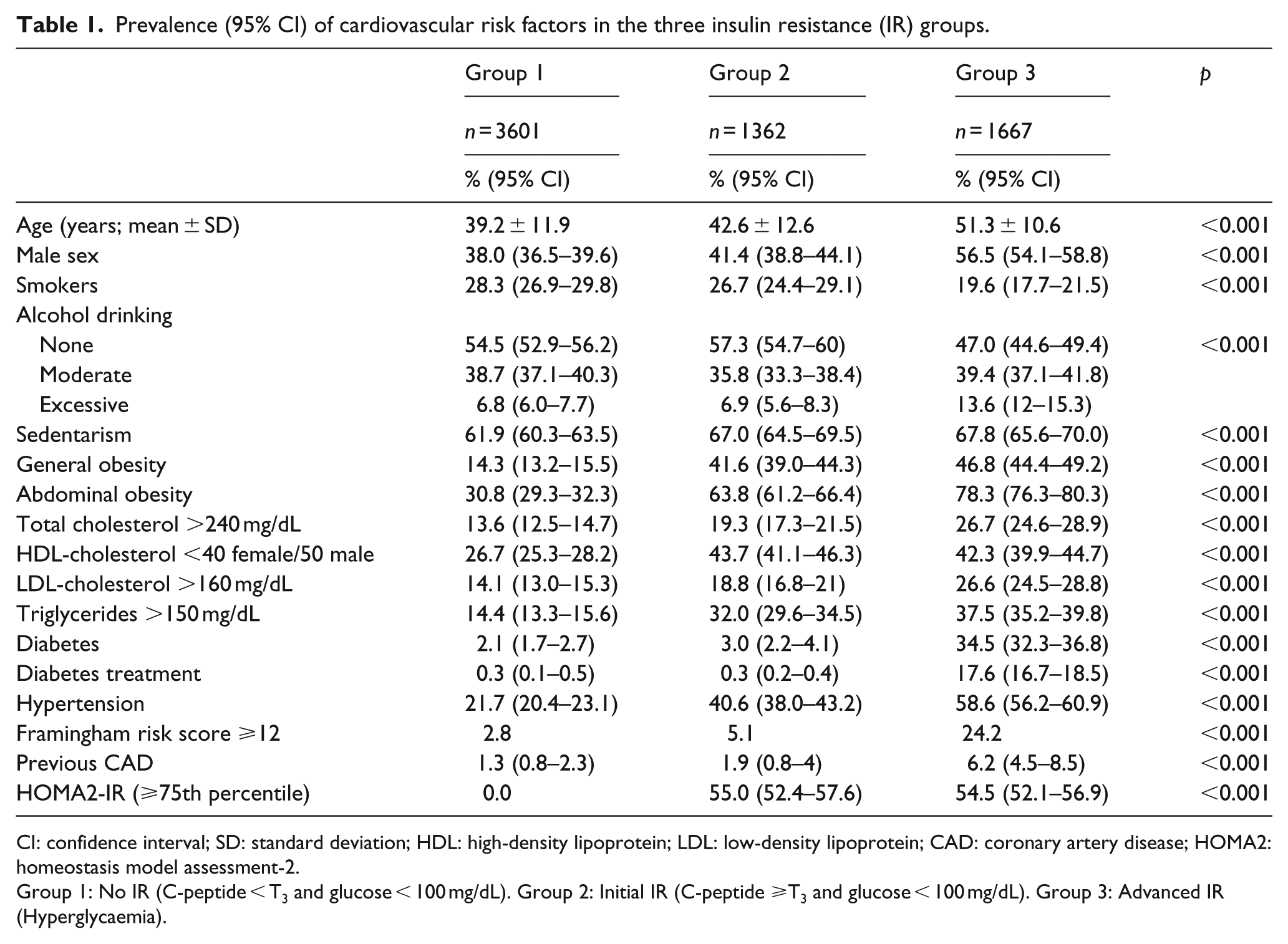
CI: confidence interval; SD: standard deviation; HDL: high-density lipoprotein; LDL: low-density lipoprotein; CAD: coronary artery disease; HOMA2: homeostasis model assessment-2.
Group 1: No IR (C-peptide < T3 and glucose < 100 mg/dL). Group 2: Initial IR (C-peptide ⩾T3 and glucose < 100 mg/dL). Group 3: Advanced IR (Hyperglycaemia).
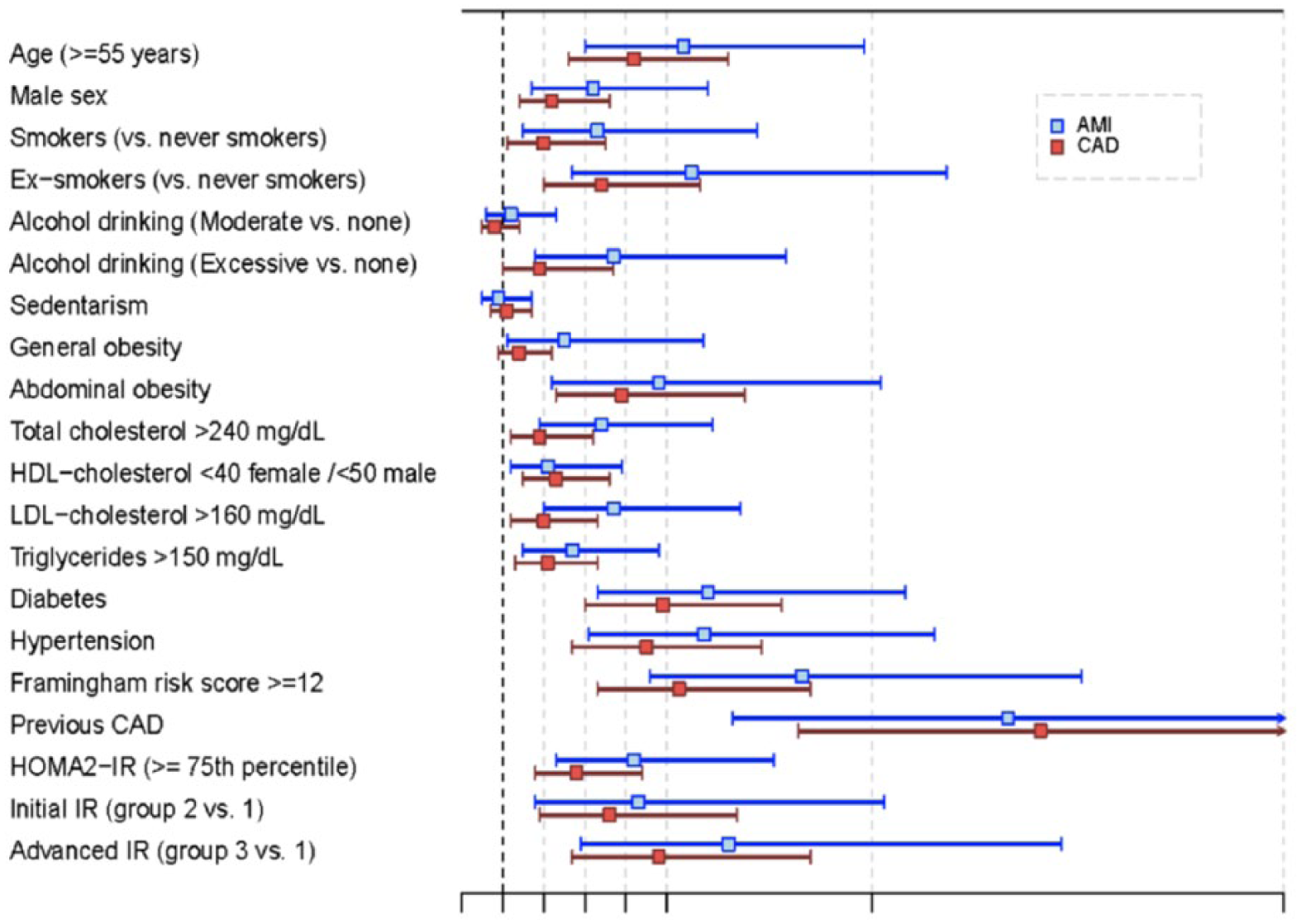
The forest plot showing the odds ratios for incident AMI and CAD (95% CI) exposed to each cardiovascular risk factor versus the absence of the factor for a sample of 6630 adults from the general population.
During the 3.52-year follow-up period, there were 45 cases of AMI and 77 cases of CAD. Table 2 shows that the incidence of AMI (RR = 4.2) and CAD (RR = 3.5) was higher in group 2 than group 1 (the reference group); in group 3, the incidence of AMI (RR = 6.8) and CAD (RR = 4.9) was even higher. When individuals with diabetes were excluded from the analysis, the RR for AMI and CAD showed little change in group 2, but decreased by about half in group 3. Sensitivity analysis was done for individuals lost to follow-up, but this analysis disclosed no changes in the RR for AMI or CAD (Table 2).
Incidence density per 105 persons/year (95% CI), number of cases/number of participants in each group and relative risk (95% CI) of AMI and CAD in groups 2 and 3 versus group 1.
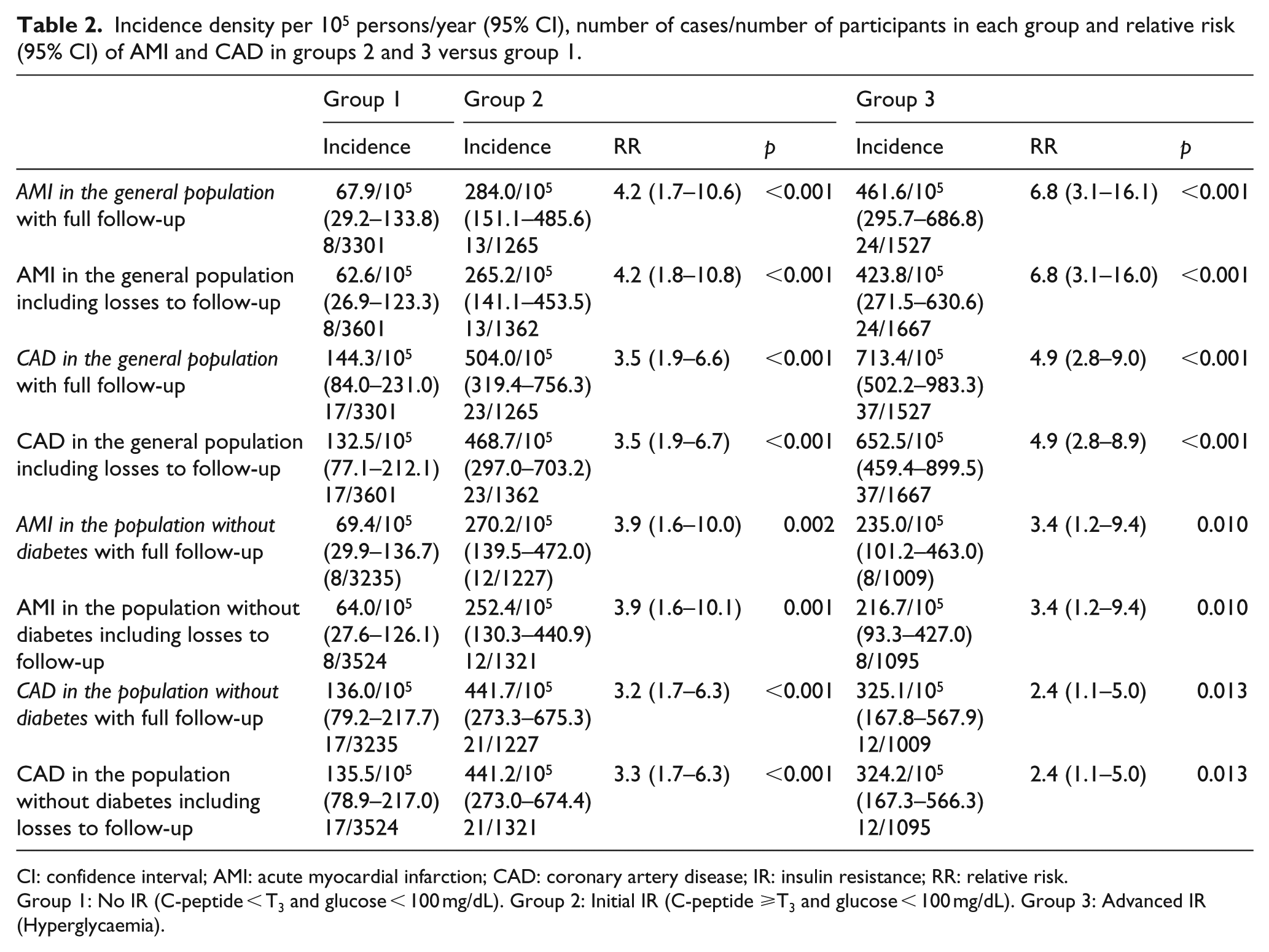
CI: confidence interval; AMI: acute myocardial infarction; CAD: coronary artery disease; IR: insulin resistance; RR: relative risk.
Group 1: No IR (C-peptide < T3 and glucose < 100 mg/dL). Group 2: Initial IR (C-peptide ⩾T3 and glucose < 100 mg/dL). Group 3: Advanced IR (Hyperglycaemia).
In the entire population, adjusting for age and sex (Table 3, models M1 and M2) corroborated the higher risk of AMI (RR = 3.1) and CAD (RR = 2.7) in group 2 compared to group 1; in fact, the risks were higher than in group 3 (2.6 and 2.2, respectively). The results were similar when the models were adjusted with the Framingham score in place of age and sex (Table 3, models M3 and M4).
Multivariable analysis with Cox models for initial IR (group 2 vs group 1) and advanced IR (group 3 vs group 1) after a median follow-up period of 3.5 years. Data are hazard ratios followed by 95% CI in parentheses and p values.
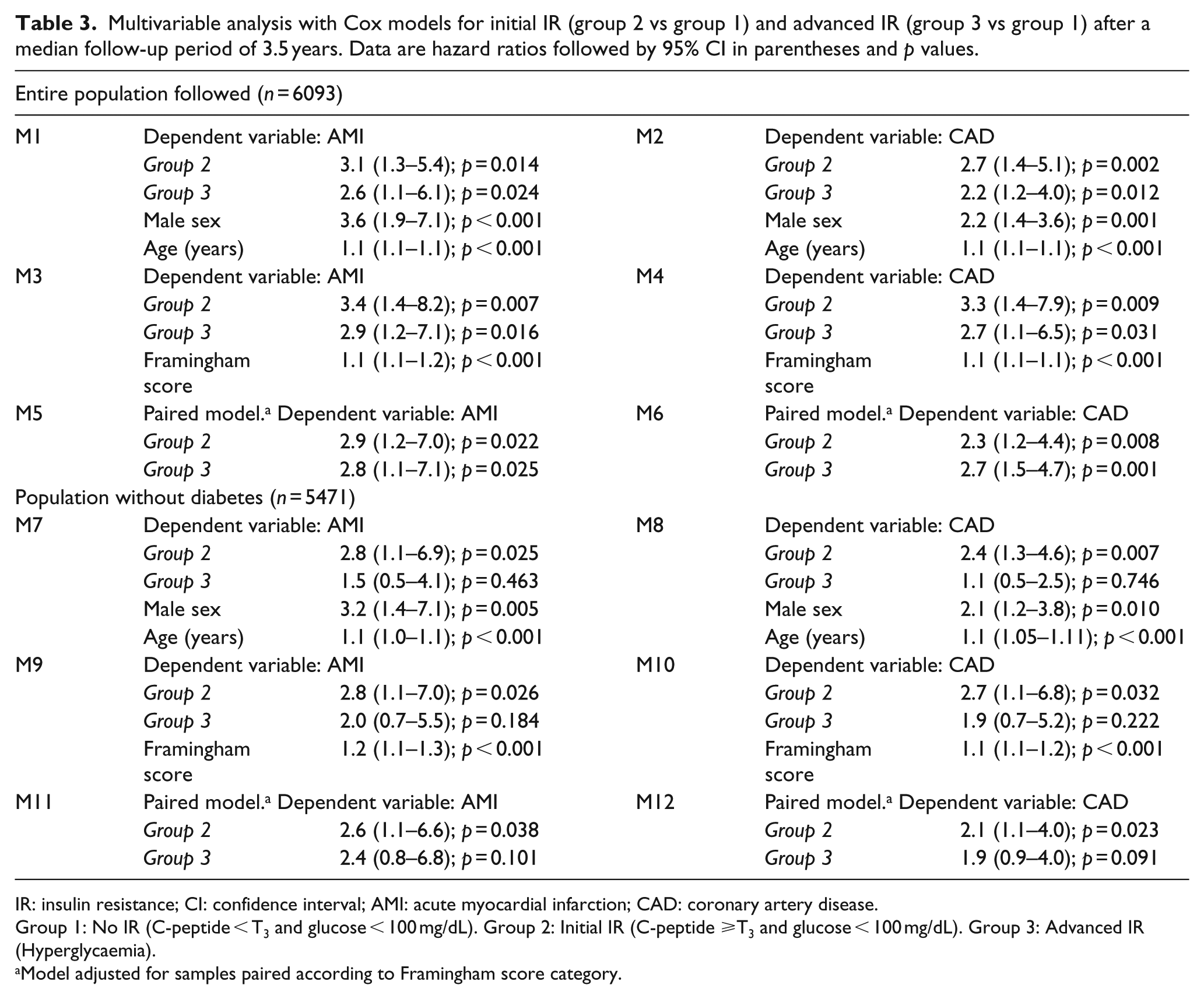
IR: insulin resistance; CI: confidence interval; AMI: acute myocardial infarction; CAD: coronary artery disease.
Group 1: No IR (C-peptide < T3 and glucose < 100 mg/dL). Group 2: Initial IR (C-peptide ⩾T3 and glucose < 100 mg/dL). Group 3: Advanced IR (Hyperglycaemia).
Model adjusted for samples paired according to Framingham score category.
When individuals with diabetes were excluded (Table 3, models M7 and M8), the risk remained significant for AMI and CAD only in initial IR (group 2); Figure 2 illustrates the survival function obtained with model M7. The results did not vary when these Cox models were adjusted with the cardiovascular risk factors of Table 1 in place of age or sex: only group 2 showed a significant risk of AMI (RR > 3) and CAD (RR > 2); risk factors that were significant included smoking (p = 0.001), hypertension (p = 0.049), total cholesterol > 240 mg/dL (p = 0.033) and previous CAD (p < 0.001); factors that were not significant included obesity, sedentarism and alcohol consumption. As before, the results were similar when the Cox models were adjusted with the Framingham score in place of these factors (Table 3, models M9 and M10): only group 2 showed a significant risk of AMI (RR = 2.8) and CAD (RR = 2.7). In the models adjusted for samples paired according to Framingham score category (Table 3: models M5 and M6 for the entire population, and models M11 and M12 for the population without diabetes), group 2 was always associated with an increased risk of AMI and CAD.
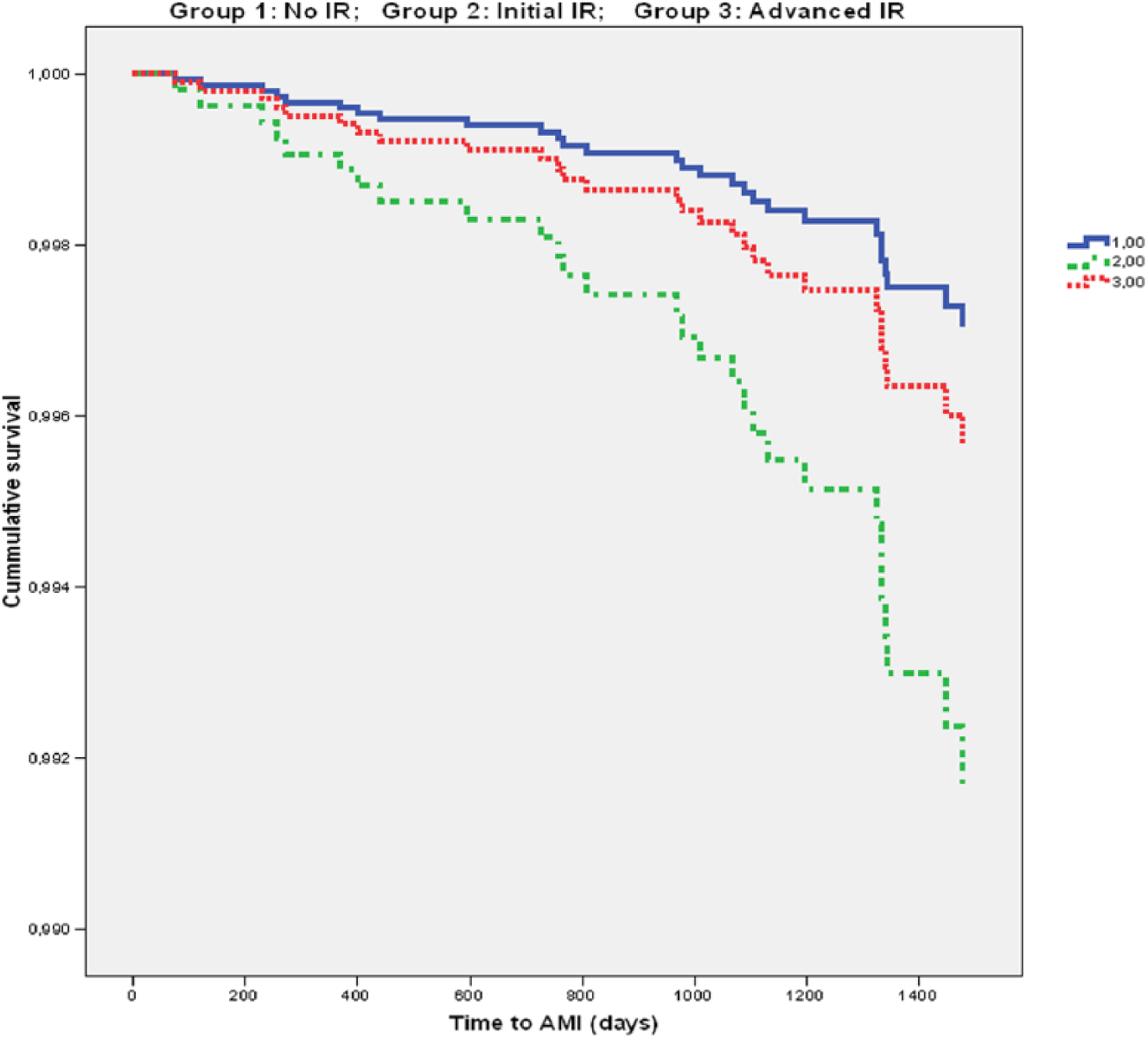
Survival function (time to acute myocardial infarction) in three groups in the general population according to model M7. Only the association in group 2 was significant (RR = 2.8; p = 0.025).
Multivariable analyses to determine the effect of elevated C-peptide (T3) compared to its normal values (terciles 1 plus 2) in the population with blood glucose < 126 mg/dL (n = 5673) consistently showed a significant effect of T3 for AMI (RR = 2.4 when adjusted by Framingham score; 95% CI = 1.1–5.0; p = 0.023) and CAD (RR = 1.9 when adjusted by Framingham score; 95% CI: = 1.1–3.2; p = 0.018). Multivariable analysis of C-peptide as a continuous variable also showed a significant effect (RR = 1.2 for AMI when adjusted by Framingham score; 95% CI = 1.0–1.4; p = 0.029).
Conclusion
We report a direct association between serum C-peptide concentration and the incidence of CAD in the general population. Furthermore, elevated C-peptide is a risk factor for CAD risk in the population with normal glucose concentration and can therefore predict coronary events sooner than IFG. In the population without hyperglycaemia, the risk is threefold as high in individuals with elevated C-peptide as in individuals with normal C-peptide concentration.
Currently, most individuals in group 2 (20% of the general population) are not identified as a coronary risk population because they fall outside the concept of prediabetes. Since hyperglycaemia per se is already a known cardiovascular risk factor, 19 it is probably more useful in clinical terms to identify risk factors in individuals with normal blood glucose concentrations. A notable finding is that after 3.5 years of follow-up, there was still no evidence of a significant raise in the risk of coronary events in persons with hyperglycaemia, whereas such evidence was seen in persons with normal glucose but increased C-peptide concentrations. This increase, a consequence of initial IR, precedes the appearance of prediabetes and diabetes; 20 it is plausible then, in pathophysiological terms, that elevated C-peptide constitutes an earlier risk factor for coronary risk than hyperglycaemia.
Also of note is our finding that the prevalence of classical cardiovascular risk factors was higher in persons with elevated C-peptide but normal glucose concentrations (group 2) than in individuals from the apparently healthy population (group 1). This finding has also been reported previously11,21,22 and may partly explain the excess cardiovascular risk. However, even after we adjusted the regression models for sex, age, Framingham function score and classical risk factors, the high risk of AMI and CAD remained evident in group 2.
Our analysis did not corroborate a relationship between C-peptide levels and CAD in individuals with hyperglycaemia (group 3). There are several potential explanations for the conflicting results in the population with diabetes or prediabetes. Confounding of the relationship between C-peptide and CAD in diabetes may include a decrease of insulin and C-peptide secretion along with an increase in cardiovascular risk due to diabetes. Persons with type 2 diabetes have been previously exposed to elevated concentrations of C-peptide, because IR constitutes the initial pathophysiological component of type 2 diabetes. In these people, a low serum concentration of C-peptide at the time of analysis may be confused with the effect of exposure to elevated C-peptide levels in the past. Another possible explanation is that a large proportion of these individuals are treated with drugs that can modify endogenous C-peptide concentrations.
We found no published cohort studies that prospectively evaluated the association between C-peptide and cardiovascular disease in the general population, although some such studies have been published for the population with diabetes. The results to date, however, have been inconsistent.9,10
The 75th percentile in the HOMA model identified only approximately half of the individuals in groups 2 and 3 as having IR; this result raises the issue of the best cut-off value for this model. Moreover, excluding persons with diabetes mitigated the effect on AMI and CAD risk in group 3 in the bivariate analysis and eliminated the effect in this group in the multivariable analysis. This finding can be interpreted as evidence that IFG in the rest of the members of this group was not a significant risk factor during the relatively brief 3.5-year follow-up period we used for this cohort. However, excluding participants with diabetes had little effect, if any, on the results in group 2, in which the prevalence of the disease was irrelevant.
Several studies have shown a direct relationship between IR and cardiovascular risk.23,24 In our cohort, however, the prevalence of IR was similar in groups 2 and 3, so the greater cardiovascular risk in group 2 is unlikely to be attributable to IR as estimated with the HOMA model. In contrast, if we accept that elevated C-peptide levels in group 2 are a consequence of initial IR, it is plausible that this molecule is related with the cardiovascular effects we detected. Nonetheless, because C-peptide is secreted in an equimolar fashion with insulin and the latter was not measured in this study, we cannot rule out that the relationship we found between C-peptide and coronary disease is the result of a direct action of insulin. 25 Measuring C-peptide to estimate IR could have some advantages compared with measurement of insulin. (The half-life of C-Peptide in the circulation is between two and five times longer than that of insulin. Therefore, C-Peptide levels are a more stable indicator of insulin secretion than the more rapidly changing levels of insulin.) In addition, in patients who underwent coronary angiography, C-peptide was found to be associated with cardiovascular mortality even after adjustment for insulin concentration. 12 In addition, C-peptide per se is known to activate a number of intracellular pathways 26 and to induce pro-inflammatory activities in the vascular wall – activities that suggest an active role in the occurrence of coronary events.3,5–8 Taken together, these findings are indications that C-peptide is in itself a cardiovascular risk factor – a possibility that enhances its potential clinical usefulness as a predictor of cardiovascular disease. Recently, two studies found that C-peptide predicts cardiovascular death in adults who do not have diabetes.27,28 Their results strongly support our findings, and these authors conclude that prospective studies are needed. However, we must be cautious as established procedures to estimate cardiovascular risk associated with diabetes or IR (oral glucose tolerance test (OGTT), HbA1c) were not performed in our study.
Additional epidemiological studies with longer follow-up periods, as well as cost-effectiveness studies, will be needed to characterize the effect of C-peptide in better detail. Nevertheless, the characteristics of our study population suggest that its potential clinical usefulness as a cardiovascular risk marker is likely to be the greatest for individuals around 40 years of age with normal blood glucose levels in whom any of the classical cardiovascular risk factors (smoking, hypertension or dyslipidaemia) has already appeared. In these people, the finding of an elevated C-peptide concentration would lead to a higher risk estimated with the Framingham function. Accordingly, future studies should investigate the effectiveness of early treatment (before the appearance of IFG) with insulin sensitizers, and the effectiveness of health education programmes aimed at increasing physical activity in this group with a view to lowering their IR.
The main limitation of our study is the short follow-up period, during which only 45 cases of AMI and 77 cases of CAD occurred. The loss to follow-up of 8% of the participants is an additional limitation; nevertheless, our sensitivity analysis was conservative and corroborated that the risk of AMI and CAD remained elevated. One more limitation is that, for economic reasons, we have no data on this cohort about serum values of proinsulin, free fatty acids, as well as C-reactive protein or glomerular filtration rate. It is also a limitation that we have no measurements of insulin, HbA1c, no OGTT and no repeated measurements. Finally, we recognize as limitation that the diagnosis of CAD included self-described cases of angina, although it is notable that the results match those of IMA.On the other hand, the main strength of our study lies in the large size of the sample from the general population, the random selection of members of the cohort and the prospective nature of the study design.
To conclude, elevated serum C-peptide is directly associated with the incidence of coronary disease in the general population. An elevated C-peptide value is a risk factor for CAD in the population with normal blood glucose levels and is thus potentially useful as an earlier predictor of coronary events than IFG.
Footnotes
Acknowledgements
The authors thank Karen Shashok for translating the original manuscript into English and Miguel Ángel García Bello for his help with the graphics.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by Canary Islands Health Service and Spanish Government (Instituto de Salud Carlos III, project 0901314 and RD12/0042/0031. FEDER).
