Abstract
Cardiovascular (CV) disease is the leading cause of mortality and morbidity in patients with type 2 diabetes mellitus (T2DM). However, improving glycaemic control alone has not decreased CV events. Therapies that improve glycaemic control, CV disease risk factors and CV function are more likely to be successful. Dipeptidyl peptidase-4 (DPP-4) inhibitors prevent breakdown of incretin hormones glucagon-like peptide-1(GLP-1) and glucose-dependent insulinotropic peptide and improve glycaemic control in patients with T2DM. DPP-4 acts on other substrates, many associated with cardioprotection. Thus, inhibition of DPP-4 may lead to elevations in these potentially beneficial substrates. Data from animal studies and small observational studies in humans suggest that DPP-4 inhibitors may potentially reduce CV risk. However, recently completed CV outcome trials in patients with T2DM and CV disease or at high risk of adverse CV events have shown that the DPP-4 inhibitors saxagliptin and alogliptin neither increased nor decreased major adverse CV events.
Introduction
Cardiovascular (CV) disease remains the leading cause of mortality and morbidity in patients with type 2 diabetes mellitus (T2DM). 1 The risks of microvascular complications, large vessel disease including myocardial infarction (MI) and diabetes-related death are closely related to hyperglycaemia. 2 However, reducing glycated haemoglobin (HbA1c) levels to <7% in patients with established T2DM and CV disease or at high CV risk has not been effective in decreasing CV outcomes. 3 Therefore, the reduction of CV risk in T2DM may require more than controlling glycaemia. Given the many non-glucose-related metabolic abnormalities associated with T2DM, approaches that improve many of these other CV disease risk factors and/or directly improve vascular function are more likely to be successful.
The incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP) are released by the intestine in response to meals. GLP-1 and GIP enhance glucose-dependent insulin secretion. GLP-1 also suppresses glucagon secretion, decreases the rate of gastric emptying and decreases food intake. All of these effects contribute to a timely and coordinated regulation of glycaemic control. 4
The enzyme dipeptidyl peptidase-4 (DPP-4) rapidly inactivates GLP-1 and GIP in vivo, thereby reducing their concentrations and limiting their effects. 4 There is some evidence that DPP-4 activity is increased in T2DM. 5 Recent data also show increased DPP-4 expression in visceral adipose tissue and augmented release of DPP-4 into circulation in obese non-diabetic individuals, 6 raising the possibility that increased DPP-4 activity may play a pathophysiologic role in the early stages of diabetes development in obesity.
In addition to GLP-1 and GIP, DPP-4 acts on other substrates, many of which are associated with cardioprotection in experimental models. Most notable of these is stromal cell–derived factor-1α (SDF-1α), which has been shown to facilitate endothelial progenitor cell (EPC) mobilization and vascular repair following ischaemic injury 7 (Figure 1). B-type natriuretic peptide (BNP) is also cleaved by DPP-4, and although it is positively associated with fluid overload states and congestive heart failure (CHF), there is also some evidence for favourable effects of BNP on cardiorenal function upon DPP-4 inhibition 8 (Figure 1). DPP-4 also appears to be involved in the degradation of the potent vasodilation molecules substance P and bradykinin.9,10 In addition to their vasorelaxation effects, both bradykinin and substance P enhance fibrinolysis by stimulation of tissue plasminogen activator (tPA).11,12 Thus, inhibition of DPP-4 may also lead to elevations in several substrates with potentially favourable effects on vascular function and anti-coagulation. On the other hand, substance P has been shown to stimulate sympathetic nervous system activity, which may result in increased heart rate and vasoconstriction. 13 Similarly, as pointed out above, BNP is a relatively strong predictor of CHF and mortality, so its overall role in disease prevention remains unclear.
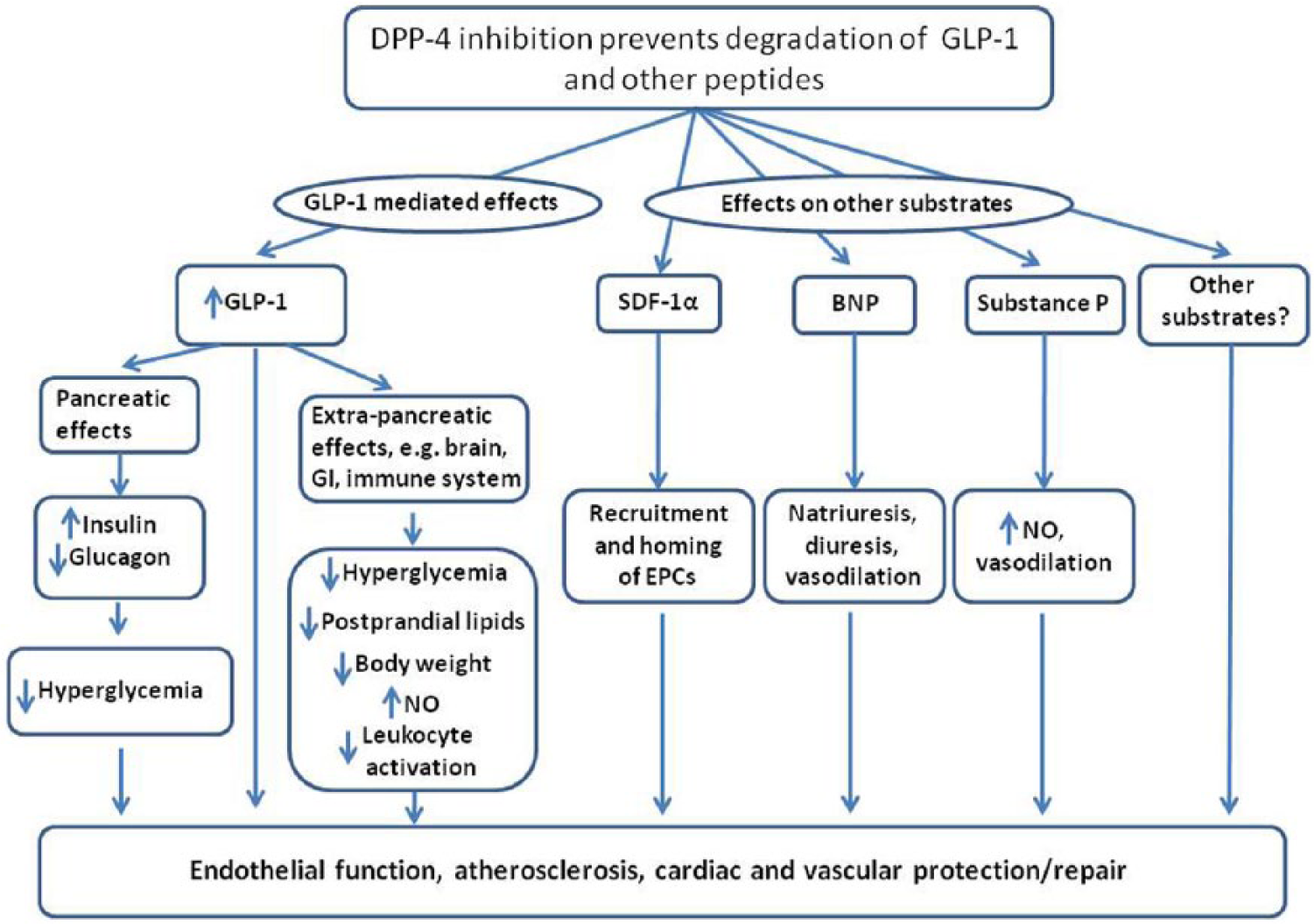
DPP-4 substrates may affect cardiovascular function. In addition to GLP-1, DPP-4 acts on other substrates that may play a role in the regulation of the cardiovascular system.
Evidence from preclinical studies and small observational studies in humans suggests that incretin-based therapies (e.g. DPP-4 inhibitors and GLP-1 receptor agonists) may have favourable effects on several CV risk factors and mechanisms contributing to CV pathology. The effects of DPP-4 inhibitors may be dependent on their ability to increase GLP-1 levels or could be due to elevation in other DPP-4 substrates. Moreover, the GLP-1-dependent CV effects of DPP-4 inhibitors may be indirect (e.g. through reduction of plasma glucose and lipids) or direct (e.g. via direct action on the vascular wall and on cardiomyocytes).
Currently approved DPP-4 inhibitors are sitagliptin, saxagliptin, linagliptin, alogliptin and vildagliptin (Europe only). This review discusses the potential effects of DPP-4 inhibitors on CV risk factors and CV disease.
Dyslipidemia, inflammation, endothelial dysfunction and atherosclerosis
Accelerated atherosclerosis is believed largely responsible for the increased risk of CV disease in T2DM. 14 The initiation and propagation of atherosclerotic plaques are complex processes, and current conceptual understanding of these events has been excellently discussed in prior reviews.15,16 Three key aspects of atherosclerotic lesion formation include lipoprotein entry and retention in the arterial wall, induction of systemic and local inflammation and endothelial dysfunction. Each of these processes is present at an early stage in the evolution of T2DM and undoubtedly contributes to accelerated formation of atherosclerosis in T2DM.16,17
Fasting dyslipidemia, comprising elevated triglyceride (TG)-rich lipoproteins and small, dense low-density lipoprotein (LDL) and reduced high-density lipoprotein (HDL), is quite common in T2DM. 18 Excess TG-rich lipoprotein production from both the liver (very low-density lipoprotein (VLDL)) and the gut (chylomicrons) has been described in insulin-resistant individuals and in patients with diabetes. 19 The abnormal lipid metabolism in T2DM leads to increased lipoprotein entry and, through subsequent chemical and enzyme modification, to their retention in susceptible regions in the arterial wall. 16 This begins a complex series of events that leads to inflammatory cell influx, their uptake of lipoproteins and local inflammation, all key events in the initiation of atherosclerosis. 16
Obesity is a major risk factor for insulin resistance and T2DM and is associated with increased adipose tissue and systemic inflammation. 20 This becomes further exacerbated with the development of impaired glucose tolerance, T2DM and hyperglycaemia, which are associated with further increases in levels of systemic inflammation markers, such as interleukin (IL)-6, IL-18 and tumour necrosis factor-α (TNF-α). 21 This resulting local and systemic inflammation creates a favourable milieu for atherosclerosis by contributing to, and/or working in concert with, dyslipidemia, hyperglycaemia, dysfunctional HDL and influx of more inflammatory cells.
Endothelial dysfunction is characterized by increased vascular tone and increased generation of procoagulation and proinflammatory factors, a combination of properties that would presumably greatly facilitate the formation of atherosclerotic plaques and increase the risk of thrombosis. 22 It is not surprising, therefore, that endothelial dysfunction is associated with progression of atherosclerosis 23 and is a powerful predictor of future adverse CV events. 24 A key step underlying each of these consequences of endothelial dysfunction is the reduced bioavailability of endothelial cell nitric oxide (NO) via its decreased generation and/or increased degradation. 22 Importantly, multiple CV risk factors and metabolic abnormalities, such as hyperglycaemia, inhibit endothelial NO production 25 and/or enhance its degradation, thereby explaining the reported association 26 of endothelial dysfunction with conditions such as obesity, dyslipidemia, hypertension and hyperglycaemia as well as smoking and physical inactivity.27,28 Consistent with this, endothelial dysfunction is associated with and occurs early in the development of T2DM 17 and may account in part for the increased CV risk in T2DM.
Effects of DPP-4 inhibition on CV disease risk factors, inflammation and vascular function
Glycaemic control
Epidemiology studies and meta-analyses in patients with and without diagnosed diabetes have shown that hyperglycaemia is associated with an increased CV risk.2,29,30 In clinical trials, DPP-4 inhibitors when used as monotherapy or as add-on therapy to other anti-hyperglycaemic agents reduced HbA1c by 0.5%–0.9%. 31 DPP-4 inhibitors also reduce fasting and postprandial plasma glucose concentrations 31 and have a low propensity to cause hypoglycaemia. 32
Dyslipidemia
GLP-1 receptor agonists have a multitude of favourable effects on dyslipidemia. These include reducing lipoprotein production from the gut and possibly the liver 33 and reducing postprandial elevations in TGs. 34 Because DPP-4 inhibitors cause GLP-1 elevation, there has been interest in determining whether this class of agents has similar or different effects on lipid metabolism.
A small clinical study found that compared with placebo administration, treatment with vildagliptin for 4 weeks decreased postprandial plasma TGs and intestinally derived apolipoprotein B-48–containing TG-rich lipoprotein particles after a fat-rich meal in patients with T2DM. 35 Other studies in patients with T2DM observed that sitagliptin reduced postprandial TG levels in a 2-week cross-over study, 36 and alogliptin alone or co-administered with pioglitazone reduced postprandial TGs and TG-rich lipoproteins compared with placebo. 37 Finally, a meta-analysis of 17 clinical trials concluded that DPP-4 inhibitors decreased total cholesterol and TGs when compared with placebo. 38
Blood pressure and body weight
Although results have been variable, DPP-4 inhibitors have generally had neutral effects on or have shown modest reductions in systolic and diastolic blood pressures and body weight in phase 3 clinical trials.39,40
Endothelial function and atherosclerosis
In animal models of endothelial dysfunction, DPP-4 inhibitors increased the bioavailability of NO, improved endothelial function and reduced inflammation.41,42 In patients with T2DM treated with metformin with or without a sulphonylurea, vildagliptin improved endothelium-dependent (acetylcholine) forearm blood flow but not endothelium-independent (sodium nitroprusside) blood flow. 43 In patients with uncontrolled T2DM and coronary artery disease, the addition of sitagliptin (50 mg/day for 6 months) increased a peripheral arterial tonometry index of endothelial function and decreased levels of C-reactive protein. 44 In contrast, a recent study found that 6 weeks of treatment with sitagliptin (50 mg/day) or alogliptin (25 mg/day) significantly reduced flow-mediated vasodilation in patients with T2DM. 45 Although it is difficult to understand how these DPP-4 inhibitors would worsen endothelial function, reasons for these conflicting results may relate to the different methodologies used for measures of vascular function or possibly to characteristics of patients, study design or different DPP-4 inhibitors. 45
In animal models of diabetes and atherosclerosis, DPP-4 inhibitors reduced atherosclerotic lesions and the expression of proinflammatory cytokines in these lesions 46 and also reduced monocyte activation and chemotaxis. 47 Similarly, DPP-4 inhibitors reduced levels of inflammatory chemokines 48 and markers of inflammation and oxidative stress in patients with T2DM. 49 Moreover, in patients inadequately controlled with metformin, treatment with sitagliptin or vildagliptin for 12 weeks decreased intima-media thickness of the carotid artery, a validated imaging measure of carotid atherosclerosis. 49
Cardiac function and vascular repair
Many, but not all, studies in experimental animals have found that DPP-4 inhibition improves cardiac function and survival following cardiac damage. For example, in a porcine model of pacing-induced heart failure, treatment with sitagliptin preserved glomerular filtration rate, reduced heart rate, increased stroke volume and potentiated the positive inotropic effects of infused BNP.
8
In a mouse model of diabetes, pretreatment with sitagliptin improved survival after experimentally induced MI and also increased cardiac expression of atrial natriuretic peptide (ANP) and other proteins associated with cardiomyocyte survival.
50
Moreover, in this same study, normoglycaemic
In contrast to the preceding studies, 12 weeks’ treatment with vildagliptin did not reverse elevations in myocardial BNP and ANP gene expression and plasma concentration of BNP or cardiac remodelling in a rat model of post-MI heart failure. 52 The reasons for these discordant results are not known but may reflect study differences in treatment duration and types of animal models used. Furthermore, it is difficult to distinguish whether changes in BNP levels after treatment with DPP-4 inhibitors reflect compensatory responses to altered myocardial function or reduced BNP cleavage.
The effect of DPP-4 inhibition on cardiac function in humans has been explored only in a few studies and in a limited number of patients. Sitagliptin, given before an oral glucose load, improved left ventricular performance in response to dobutamine stress echocardiography and decreased post-ischemic stunning in patients with coronary artery disease (n = 14). 53 In another small study (n = 12) in patients without diabetes and with non-ischemic cardiomyopathy, therapy with sitagliptin for 4 weeks resulted in increased myocardial glucose uptake. 54 In contrast, a recent large clinical trial on CV safety of saxagliptin [Saxagliptin Assessment of Vascular Outcomes Recorded in patients with diabetes mellitus (SAVOR) trial] showed an increased rate of hospitalization for heart failure in patients with T2DM at high CV risk. 55 Note, however, that this was a component of a secondary composite end point, and the analysis was not controlled for multiple testing. Therefore, this finding needs to be evaluated in other ongoing CV outcome studies with DPP-4 inhibitors.56–58
There is some evidence that DPP-4 inhibition may facilitate vascular repair, mainly by protecting SDF-1α from degradation. SDF-1α is a DPP-4 substrate that participates in recruiting bone marrow–derived EPCs to sites of vascular damage. 48 EPCs play a role in endothelium integrity and repair and in neoangiogenesis. 48 SDF-1α may also protect other types of progenitor cells. For example, in mice, administration of a DPP-4 inhibitor together with granulocyte colony-stimulating factor stabilized SDF-1α in the heart, enhanced myocardial progenitor cell homing and function, reduced cardiac remodelling and improved heart function and survival after MI. 59
There is also some evidence that DPP-4-mediated effects on SDF-1α, and EPCs may be relevant in humans. In patients with T2DM, EPC levels are decreased, 60 and EPC proliferation, adhesion and ability to repair damaged vasculature are impaired. 61 Treatment for 4 weeks with sitagliptin plus metformin with or without insulin secretagogues in patients with poorly controlled T2DM increased EPCs twofold and SDF-1α by 50% and decreased the proinflammatory chemokine monocyte chemotactic protein-1 by 25%. 48
GLP-1-dependent and -independent actions of DPP-4 inhibitors
GLP-1 receptors are present in blood vessels and the heart, 33 suggesting that some of the aforementioned beneficial effects of DPP-4 inhibition may be due to elevated levels of GLP-1. For example, infusion of GLP-1 increased flow-mediated vasodilation in patients with T2DM and stable coronary artery disease 62 and acetylcholine-induced blood flow response in healthy individuals. 63 This is supported by studies showing GLP-1 receptor agonists may improve cardio vascular function. This includes lowering of both systolic and diastolic pressure, a rather consistent finding with treatment with GLP-1 receptor agonists. 64 There is also evidence for an indirect sitagliptin-mediated augmentation of cardiac function (during dobutamine stress testing) as this improvement was accompanied by an increase in GLP-1. 53 Thus, the observed benefits of DPP-4 inhibition on cardiac and endothelial function appear to be, in part, due to elevation of GLP-1 levels or its receptor activation.
However, other CV effects of DPP-4 inhibition may be independent of GLP-1. The inhibition of SDF-1α degradation by DPP-4 and the consequences on EPCs mobilization is one example. 48 Another example is the direct in vitro inhibition of proinflammatory cytokine expression in mononuclear cells treated with DPP-4 inhibitors. 46 Similar in vitro data exist for T-cell proliferation and activation, suggesting this treatment could reduce inflammation in tissues and atherosclerotic plaques by more direct mechanisms. 46 On the other hand, not all indirect effects of DPP-4 inhibitors may be beneficial. A recent study in humans showed that sitagliptin did not augment vasodilation effects, despite raising substance P levels but instead increased heart rate when co-administered with a high dose of angiotensin-converting enzyme inhibitors. 65 Therefore, the effects of DPP-4 inhibition on CV disease risk factors and on cardiac function and vascular repair likely represent contributions from and a balance of both GLP-1-dependent actions of DPP-4 inhibitors as well as mechanisms independent of GLP-1 (Figure 1).
Initial studies of DPP-4 inhibition on CV outcomes
Given the many beneficial effects of DPP-4 inhibitors on CV risk factors and vascular function, there was great interest in their potential to reduce adverse CV events. This enthusiasm was supported by several compilations of earlier shorter-duration studies.
Meta-analyses of clinical trial data of individual DPP-4 inhibitors sitagliptin, 66 saxagliptin, 67 linagliptin, 68 vildagliptin 69 and alogliptin 70 found that these DPP-4 inhibitors were not associated with an increased risk of adverse CV events, and some analyses suggested a potential reduction of adverse CV events.67,68 An additional meta-analysis of 70 clinical trials (each ⩾24 weeks duration) found that DPP-4 inhibitors reduced major adverse CV events and all-cause mortality in patients with T2DM. 40 However, there were several limitations to these studies and the subsequent meta-analyses. The original studies were not intended as CV outcome studies, and therefore, they tended to be short in duration; contained relatively small sample sizes; and CV outcome definitions, and determination and adjudication of events were not consistent across studies. Thus, although these initial reports were positive, more definitive studies were clearly needed.
Since 2008, regulatory authorities have required that all new anti-diabetic agents undergo a rigorous long-term CV risk assessment. 71 The US Food and Drug Administration (FDA) guidance states that trials should include patients with advanced disease, elderly patients and patients with some degree of renal impairment. An upper bound of the 95% confidence interval (CI) of 1.3 for the risk ratio of important CV events (e.g. CV mortality, MI and stroke) was also instituted to exclude unacceptable CV risk for new treatments of T2DM. This FDA guidance recommended more systemic and prolonged evaluation of CV outcomes for DPP-4 inhibitors. In fact, several large trials have been recently completed or are ongoing to evaluate CV safety of new DPP-4 inhibitors in patients with T2DM and high risk of CV disease (Table 1). The results from two of these trials have recently been published.
DPP-4 inhibitor cardiovascular outcome trials.
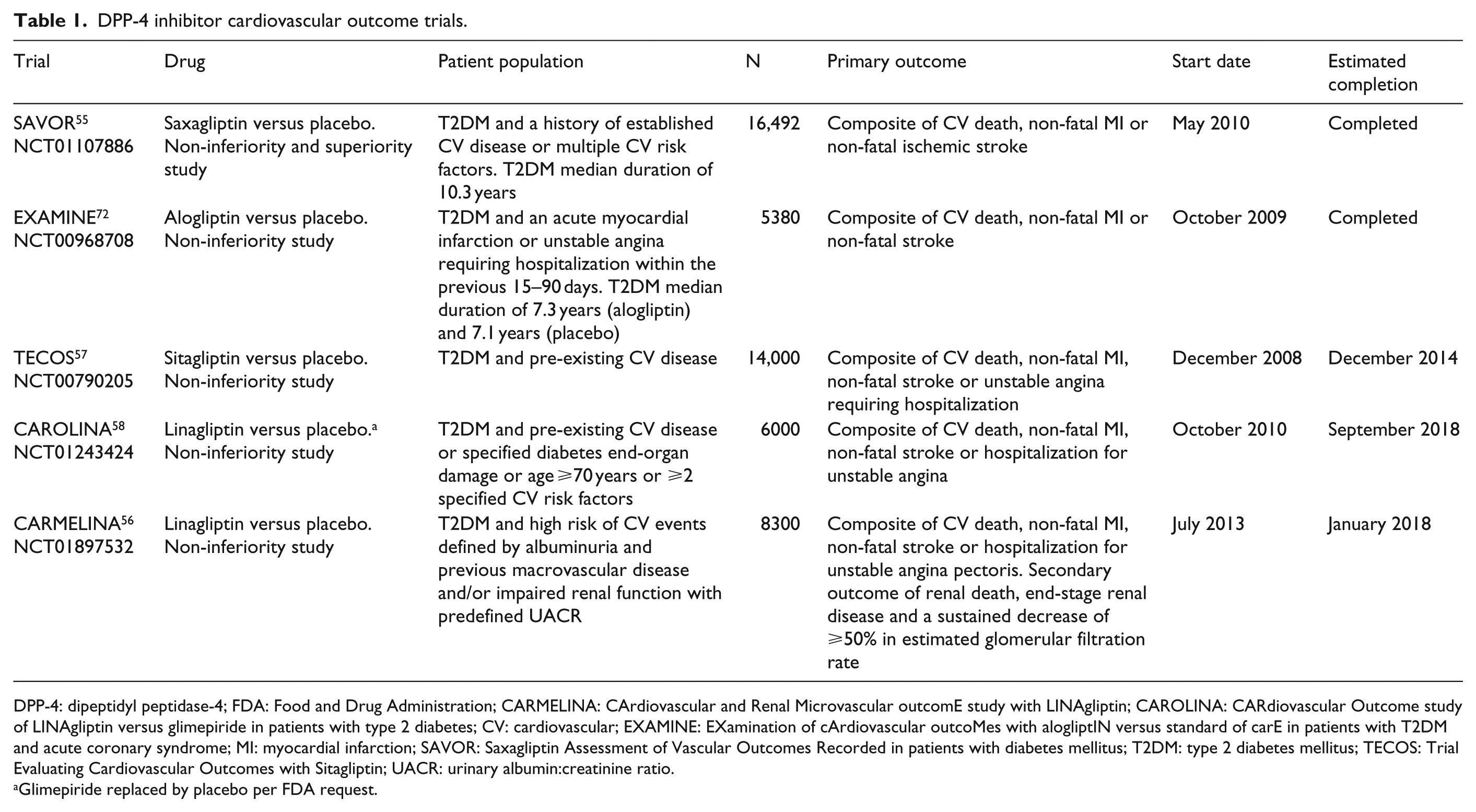
DPP-4: dipeptidyl peptidase-4; FDA: Food and Drug Administration; CARMELINA: CArdiovascular and Renal Microvascular outcomE study with LINAgliptin; CAROLINA: CARdiovascular Outcome study of LINAgliptin versus glimepiride in patients with type 2 diabetes; CV: cardiovascular; EXAMINE: EXamination of cArdiovascular outcoMes with alogliptIN versus standard of carE in patients with T2DM and acute coronary syndrome; MI: myocardial infarction; SAVOR: Saxagliptin Assessment of Vascular Outcomes Recorded in patients with diabetes mellitus; T2DM: type 2 diabetes mellitus; TECOS: Trial Evaluating Cardiovascular Outcomes with Sitagliptin; UACR: urinary albumin:creatinine ratio.
Glimepiride replaced by placebo per FDA request.
The SAVOR trial
55
assessed the CV safety and efficacy of saxagliptin [5 or 2.5 mg/day in those with an estimated glomerular filtration rate (eGFR) of ⩽50 mL/min] compared with placebo in patients with T2DM and a history of established CV disease or multiple CV risk factors, such as dyslipidemia, hypertension or active smoking (N = 16,492). Patients continued to receive standard-of-care anti-hyperglycaemic (except for other DPP-4 inhibitors or GLP-1 receptor agonists) and CV disease therapy. Median follow-up was 2.1 years. Saxagliptin did not increase or decrease the risk of the primary composite end point of CV death, non-fatal MI or non-fatal ischemic stroke, compared with placebo [hazard ratio, 1.00 (95% CI, 0.89–1.12);
In the EXamination of cArdiovascular outcoMes with alogliptIN versus standard of carE in patients with T2DM and acute coronary syndrome (EXAMINE) trial,
72
patients (N = 5380) with T2DM and an acute coronary syndrome within 15–90 days before randomization received alogliptin (25 mg/day for those with eGFR ⩾ 60 mL/min/1.73 m2; 12.5 mg/day for eGFR = 30–<60; 6.25 mg/day for eGFR < 30) or placebo for a median follow-up of 18 months. Patients continued to receive standard-of-care medications for T2DM and CV risk factors. The primary end point of CV death, non-fatal MI or non-fatal stroke occurred in 11.3% of patients receiving alogliptin and 11.8% of patients receiving placebo (hazard ratio, 0.96; upper boundary of the one-sided repeated CI, 1.16;
So, despite many promising effects of DPP-4 inhibitors on CV risk factors, vascular function and animal models of atherosclerosis, two of the five ongoing studies of DPP-4 inhibitors in humans have shown no benefit on CV outcomes during their relatively short follow-up periods. We will, of course, need to wait for the results of all the studies to fully appreciate the consequences of DPP-4 inhibition therapy on CV disease. It is possible that these neutral effects on CV disease may result from several limiting features of safety outcome studies. 76 First, as a higher event rate permits enrolling lower numbers of participants (thereby reducing study duration and costs), both studies presented to date included participants with significant CV disease or at high risk for CV disease. It is, therefore, possible that CV effects of DPP-4 inhibition may be less apparent when initiated later in the course of disease, when more advanced atherosclerosis is present. This appears true for glucose lowering itself where there is evidence that it is more effective in those with less extensive vascular injury [as suggested in the Veterans Affairs Diabetes Trial (VADT) subset analysis 77 and the Action to Control Cardiovascular Risk in Diabetes (ACCORD) subgroup analysis 78 ]. Therefore, future studies may be needed to evaluate the effectiveness of DPP-4 inhibitors in those with lower CV risk or on progression of sub-clinical atherosclerosis as measured by gold standard imaging techniques, such as computed tomography angiogram scans of coronary arteries or carotid magnetic resonance imaging or ultrasound. Second, consistent with the FDA guidance, the primary end point of most studies is typically CV safety. CV safety studies are primarily designed as non-inferiority trials, including SAVOR and EXAMINE (Table 1), and thus, they may not be adequately powered to evaluate CV benefits of DPP-4 inhibitors. For a trial testing CV efficacy of diabetes therapies, that is, a superiority trial, the required number of events may be considerably larger than for a non-inferiority trial and would depend on the size of the drug’s postulated effect on CV events versus the chosen comparator(s). 76
Possible evidence for increased risk for CHF
As noted above, the SAVOR study reported an increased risk for hospitalization for heart failure. Although not expected based on earlier study findings, and possibly a result of chance, this has increased the awareness and monitoring of this adverse event. This finding was not corroborated in the EXAMINE study, although a trend for increased CHF was noted. Subsequently, two recent meta-analyses showed a significant increase in hospitalization for heart failure in patients receiving DPP-4 inhibitors, raising further concern for this adverse event.79,80 However, it is important to note that the meta-analyses were largely influenced by results of the large SAVOR and EXAMINE trials. In further analyses of the many DPP-4 studies, it appears that the risk for CHF is most apparent in trials enrolling individuals with the greatest underlying CV risk.79,80 This suggests that health-care providers may wish to be more cautious in using DPP-4 inhibitors in patients prone to heart failure.79,80 The mechanisms underlying a potential increased risk of heart failure with use of DPP-4 inhibitors are not known. Increased levels of substance P, which stimulates sympathetic activity, are one possibility. 65 Moreover, Devin et al. 65 recently speculated that this effect may be particularly relevant in patients receiving angiotensin-converting enzyme inhibitors, as this combination may further increase substance P levels. Although supported by experimental data, this hypothesis requires further examination.
Summary and conclusion
Patients with T2DM are at increased risk of CV disease. In addition to hyperglycaemia that contributes to increased CV risk, patients with T2DM often have other conditions, such as hypertension and dyslipidemia that contribute to the development of CV complications. Therapies that improve glycaemic control, CV disease risk factors and vascular function are more likely to be successful. DPP-4 inhibitors improve glycaemic control and, based on animal studies and small trials in humans, have the potential to provide beneficial CV effects. However, recently completed CV safety trials have shown that DPP-4 inhibitors do not increase or decrease major adverse CV events over several years of use in individuals with T2DM and CV disease or at high risk for CV disease. These results, together with future results from ongoing CV disease trials of other DPP-4 inhibitors, will provide the body of evidence that health-care providers will need to better prioritize the use of these agents when selecting optimal glucose-lowering therapy for patients with T2DM.
Footnotes
Declaration of conflicting interests
J.K., M.S. and C.B. declare no conflicts.
Funding
Editorial support was provided by Richard M. Edwards, PhD, and Janet E. Matsuura, PhD, from Complete Healthcare Communications, Inc. (Chadds Ford, PA), and was funded by Bristol-Myers Squibb and AstraZeneca LP. P.R. has received research grants from Bristol-Myers Squibb.
