Abstract
Objective:
To estimate markers of prothrombotic state and endothelial dysfunction in youths with type 1 diabetes mellitus (T1DM) and investigate possible associations with anthropometric/demographic data, glycaemic control and lipid profile.
Methods:
In a cross-sectional design, we recruited 155 youths with T1DM and determined levels of plasminogen activator inhibitor-1-antigen (PAI-1-Ag), von Willebrand factor-antigen (vWF-Ag), fibrinogen (FB), lipids and glycosylated haemoglobin (HbA1c).
Results:
Of all the participants, 76 (49%) had increased levels of at least one of prothrombotic factors. Suboptimal glycaemic control was associated with a worse lipid profile and an eightfold increased risk of elevated vWF-Ag levels. Higher vWF-Ag concentrations were also correlated with impaired lipid profile and increased HbA1c values, whereas PAI-1-Ag was positively correlated only with triglyceride levels. After adjustment for potential confounders, only HbA1c contributed independently to the variation in vWF-Ag levels.
Conclusion:
Impaired prothrombotic state and consequently endothelial dysfunction are present in youths with T1DM, representing a cumulative risk factor for future cardiovascular disease (CVD). Achievement and maintenance of euglycaemia and normolipidaemia are crucial to decelerate progress of this process.
Keywords
Introduction
Type 1 diabetes mellitus (T1DM) is characterized by increased frequency of cardiovascular disease (CVD). 1 Atherothrombosis, which is a precursor of CVD, is identified even in children with T1DM. 2 Furthermore, hyperglycaemia is associated with endothelial dysfunction that is an early feature in the development of vascular complications in patients with T1DM. 3 A number of processes regulating the thrombophilic/thrombolytic balance are impaired in these patients as a result of dysfunction of endothelial cells leading to a hypercoagulative state. 4 Endothelial function can be indirectly evaluated by blood circulating markers. In vivo, the initial reversible endothelial dysfunction is identified by increased von Willebrand factor-antigen (vWF-Ag) and tissue plasminogen activator (tPA) levels. 5 Plasminogen activator inhibitor-1 antigen (PAI-1-Ag), the major physiological inhibitor of tPA, is mainly produced by endothelium. Elevated PAI-1-Ag levels increase the risk of atherothrombosis and future CVD. 6 Moreover, fibrinogen (FB), a clotting factor and a marker of systemic inflammation, has been suggested to affect microcirculation by increasing blood viscosity, red blood cell aggregation and platelet thrombogenesis. 7
Although impaired haemostasis, fibrinolysis and endothelial dysfunction have been extensively studied in individuals with insulin resistance 8 and type 2 diabetes mellitus (T2DM), 9 published data concerning T1DM are controversial and sparse. Therefore, this study aimed to (1) estimate PAI-1-Ag, vWF-Ag and FB levels in children and adolescents with T1DM and (2) investigate possible associations with demographic/anthropometric parameters, glycaemic control and lipid profile.
Methods
Patients
In total, 155 children and adolescents with T1DM were enrolled in the study. Patients are followed up at the Pediatric Diabetes Outpatient Unit, 4th Department of Pediatrics, Faculty of Medicine, Aristotle University of Thessaloniki, Papageorgiou General Hospital. Demogra-phic data and anthropometric parameters included in the statistical analysis were gender, age at entry to study, age at diagnosis, duration of T1DM, Tanner stage, body mass index (BMI), waist circumference (WC), systolic blood pressure (SBP) and diastolic blood pressure (DBP). Furthermore, daily insulin requirements were recorded. Before entering the study, informed consent was obtained from participants older than 18 years as well as parents or guardians of individuals younger than 18 years. The research protocol was approved by the Ethics Committee of Faculty of Medicine of Aristotle University of Thessaloniki. The study was conducted according to the criteria of the Declaration of Helsinki.
Anthropometric measurements
Weight and height were measured (SECA 711; Seca GmbH & Co. KG, Hamburg, Germany; Harpenden Stadiometer; Veeder-Root, Elizabethtown, NC, USA), and BMI was calculated as weight (in kilograms)/squared height (in squared metres). Patients were classified into two groups: one comprising patients with excessive body weight (BMI > 85th percentile for gender and age) and another including patients with normal BMI for gender and age (BMI < 85th percentile). WC was measured midway between the lowest rib and the superior border of the iliac crest at the umbilical level, using a flexible tape.
A fluid column manometer was used to measure SBP and DBP in a seated relaxed position. Based on Marshall and Tanner scale, patients were classified as prepubertal (stage 1) or pubertal (stages 2–5).
Laboratory assays
Fasting blood samples were obtained from all patients for the determination of prothrombotic factors, lipid profile and glycaemic control. Levels of PAI-1-Ag and vWF-Ag were determined quantitatively in plasma with an enzyme-linked immunosorbent assay (ELISA) (ASSERACHROM® PAI-1-Ag; Diagnostica Stago, Asnieres, France) and an immunoturbidimetric assay (STA-Liatest® vWF-Ag; Diagnostica Stago), respectively. FB was determined quantitatively in plasma with the Clauss method (STA Fibrinogen®; Diagnostica Stago). Previously published age-related norms, obtained from studies conducted in paediatric populations, were used to determine the mean value as well as to define the cut-off points (the upper value of the range) for distinguishing normal from pathological levels of prothrombotic factors [PAI-1-Ag: 0.8–31.2 ng/mL (mean value: 21.0 ng/mL) for ages 1–5 years, 2.6–48.0 ng/mL (mean value: 25.3 ng/mL) for ages 6–10 years and 0.1–39.9 ng/mL (mean value: 20.0 ng/mL) for ages 11–18 years; vWF-Ag: 41.4–146.4% (mean value: 93.9%) for ages 1–5 years, 53.2–170.3% (mean value: 111.7%) for ages 6–10 years and 40.0–164.5% (mean value: 102.2%) for ages 11–18 years; and FB: 170–405 mg/dL (mean value: 276 mg/dL) for ages 1–5 years, 157–400 mg/dL (mean value: 279 mg/dL) for ages 6–10 years and 154–448 mg/dL (mean value: 300 mg/dL) for ages 11–18 years].10,11 Serum total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-c), low-density lipoprotein cholesterol (LDL-c), apolipoprotein A1 (Apo-A1), apolipoprotein B (Apo-B) and lipoprotein (a) [Lp(a)] levels were determined with standard methods using an autoanalyzer (Architect 8000c; Abbott Laboratories, Abbott Park, IL, USA), and the Apo-B/Apo-A1 ratio was calculated. Glycosylated haemoglobin (HbA1c) was measured in capillary blood sample with a specialized method (DCA 2000R+ Haemoglobin A1c; Siemens, Muenchen, Germany). Levels of HbA1c ≤ 7.5 or > 7.5% were considered as optimal or suboptimal glycaemic control, respectively. 12
Statistical analysis
Data were analysed using Statistical Package for Social Science software for Windows, version 16.0 (SPSS Inc, Chicago, IL, USA). Categorical variables were described by frequency and percentage, while continuous variables by mean ± standard deviation (SD). Kolmogorov–Smirnov test was used to investigate the normality of distributions of continuous variables. Comparisons of continuous variables between groups were performed with independent-samples t-test for normally distributed data or Mann–Whitney U test for nonparametric data. Additionally, a one-sample t-test was used to compare our individual results with the mean value of previously published age-appropriate controls, separately for each age group and each prothrombotic factor. Categorical variables were compared using chi-squared test. Correlations were calculated with Pearson’s or Spearman’s correlation coefficients, as appropriate. Standard multiple linear regression analysis was used to test the independence of variables and the magnitude of significant contribution to the variance of the dependent variables. Statistical significance was defined at p < 0.05.
Results
Characteristics of the study population
Demographic, anthropometric, metabolic and prothrombotic parameters of the patients are shown in Table 1. All participants were treated with insulin given by either multiple daily injections (n = 136, 87.7%) or continuous subcutaneous infusion (n = 19, 12.3%). None of the children or adolescents had hypertension, diabetic retinopathy, neuropathy or microalbuminuria/nephropathy. The mean age of the participants was 11.6 years, and the mean duration of diabetes was 4.2 years. The mean values of PAI-1-Ag and vWF-Ag were 40.4 ng/mL and 119.0%, respectively, and did not differ between males and females. Levels of FB were found to be greater in girls than boys (306.7 vs 277.3 mg/dL, p = 0.012). No differences were detected in the levels of all three prothrombotic factors according to pubertal stage (Table 1). We also found that 76 children and adolescents (49%) had increased levels of at least one of markers of prothrombotic state (PAI-1-Ag, vWF-Ag or FB). Abnormally high levels of PAI-1-Ag, vWF-Ag and FB were found in 39.4%, 14.2% and 5.2% of all patients, respectively.
Characteristics of the study population.
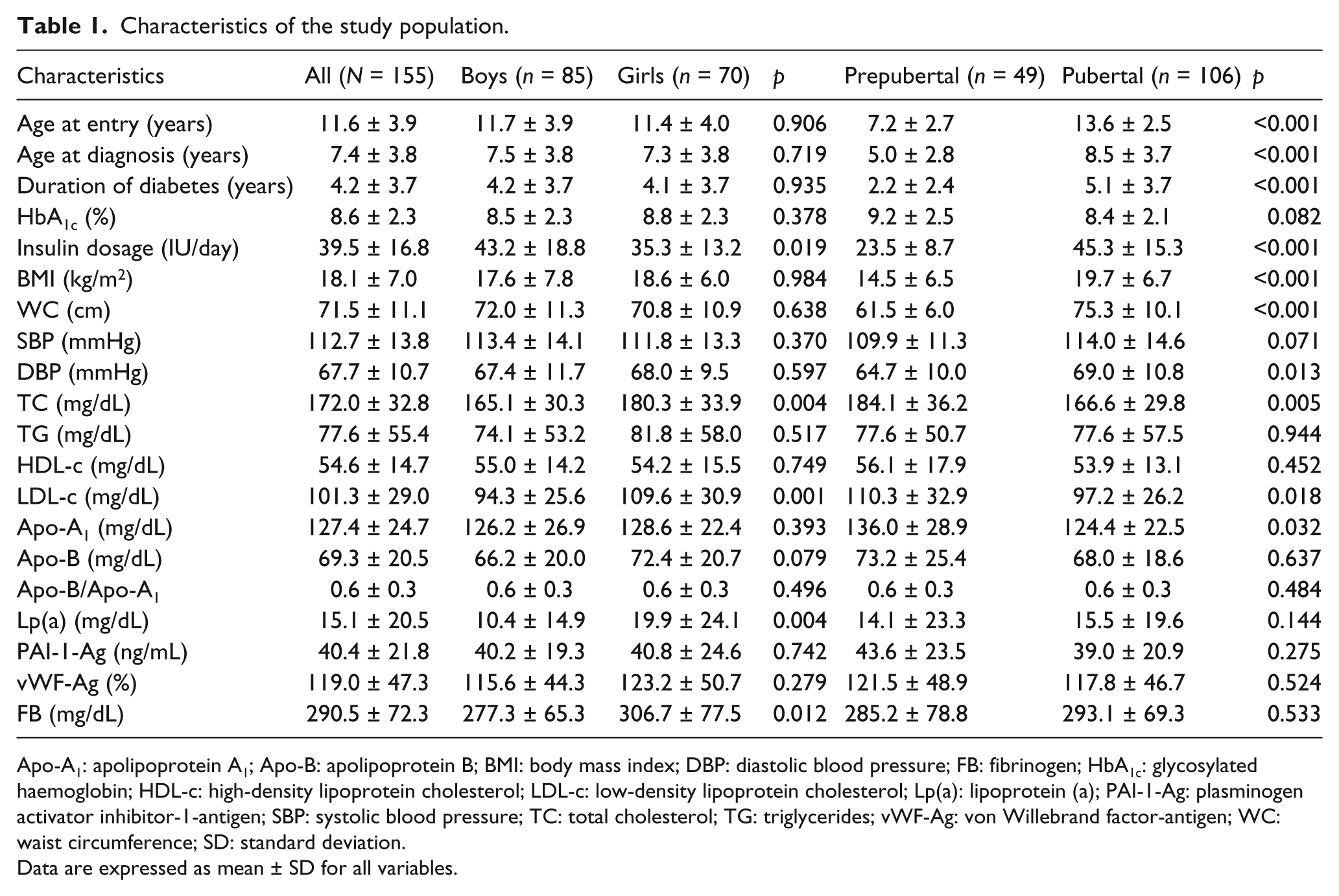
Apo-A1: apolipoprotein A1; Apo-B: apolipoprotein B; BMI: body mass index; DBP: diastolic blood pressure; FB: fibrinogen; HbA1c: glycosylated haemoglobin; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; Lp(a): lipoprotein (a); PAI-1-Ag: plasminogen activator inhibitor-1-antigen; SBP: systolic blood pressure; TC: total cholesterol; TG: triglycerides; vWF-Ag: von Willebrand factor-antigen; WC: waist circumference; SD: standard deviation.
Data are expressed as mean ± SD for all variables.
Comparisons between our individual results and the mean value of published age-appropriate controls, separately for each age group and each prothrombotic factor
The mean value of PAI-1-Ag was found to be significantly higher in diabetic subjects as compared to controls in all age groups (1–5 years: 46.5 vs 21.0 ng/mL, 6–10 years: 42.3 vs 25.3 ng/mL, 11–18 years: 38.6 vs 20.0 ng/mL, in all cases p < 0.001). On the contrary, the vWF-Ag mean value differed in the age groups of 1–5 and 11–18 years (1–5 years: 142.1 vs 93.9%, p = 0.002; 6–10 years: 108.0 vs 111.7%, p = 0.532; 11–18 years: 120.0 vs 102.2%, p = 0.001), whereas FB levels were significantly higher, between diabetics and controls, only in the 6–10 year age group (1–5 years: 285.6 vs 276.0 mg/dL, p = 0.744; 6–10 years: 289.0 vs 279.0 mg/dL, p < 0.001; 11–18 years: 292.1 vs 300.0 mg/dL, p = 0.272).
Comparisons between those with suboptimal and optimal glycaemic control
In the whole group, 40% (62 out of 155 diabetic patients) were noted to have optimal glycaemic control (a value of HbA1c ≤ 7.5%). Patients with suboptimal glycaemic control had worse lipid profile than euglycaemics. Furthermore, among prothrombotic factors, only vWF-Ag was found to be associated with suboptimal glycaemic control (p = 0.032) (Table 2). We also quantified the risk of having abnormally high levels of PAI-1-Ag, vWF-Ag and FB for patients with poor glycaemic control as compared to those with good glycaemic control. This risk was estimated to be 7.8 times greater for vWF-Ag (p = 0.004). The risk of detecting high levels of FB was 4.8 times greater, although not statistically significant, in patients with poor glycaemic control. As far as PAI-1-Ag is concerned, the estimated odds ratio did not reach the level of statistical significance (Table 3).
Comparisons between those with suboptimal and optimal glycaemic control.
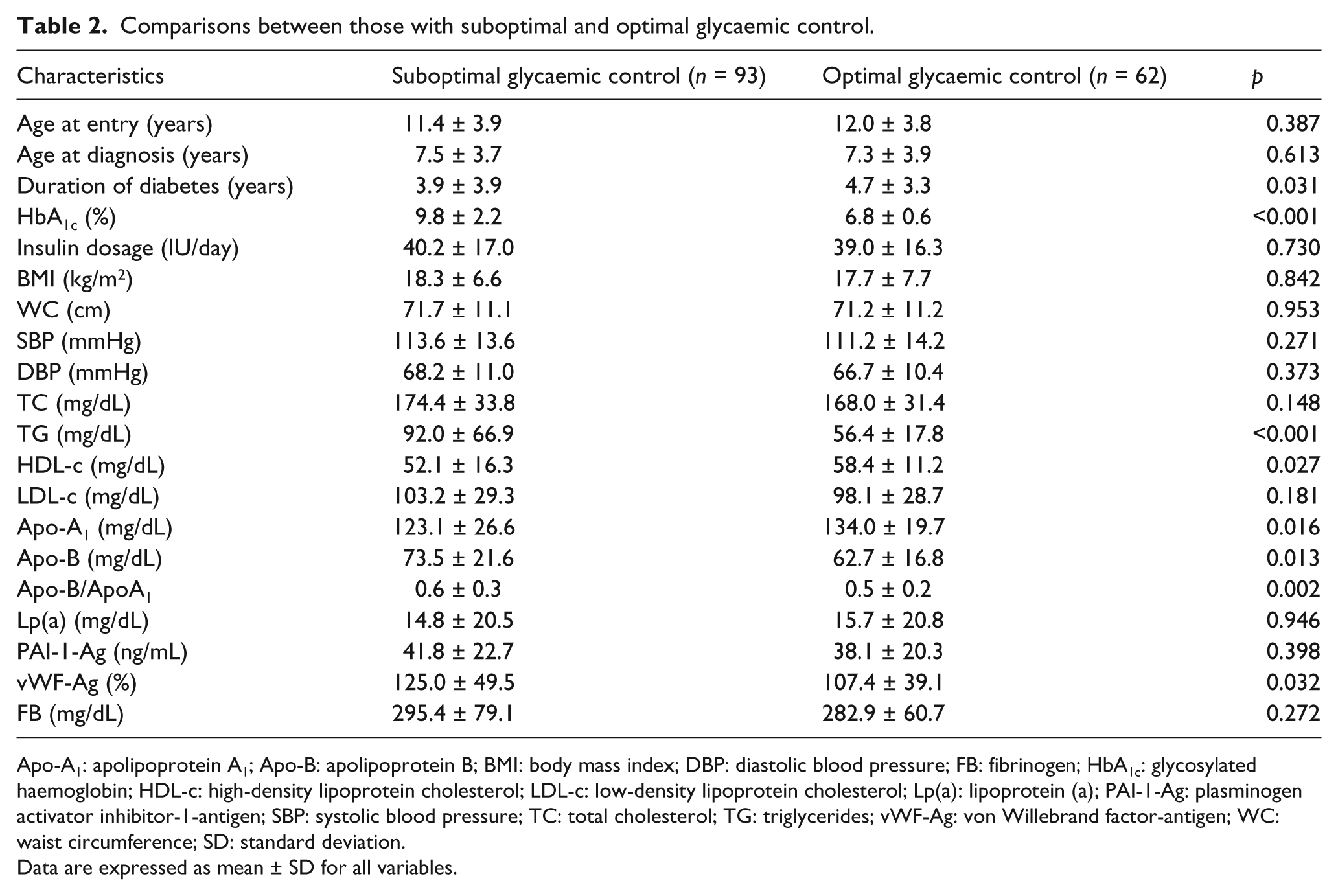
Apo-A1: apolipoprotein A1; Apo-B: apolipoprotein B; BMI: body mass index; DBP: diastolic blood pressure; FB: fibrinogen; HbA1c: glycosylated haemoglobin; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; Lp(a): lipoprotein (a); PAI-1-Ag: plasminogen activator inhibitor-1-antigen; SBP: systolic blood pressure; TC: total cholesterol; TG: triglycerides; vWF-Ag: von Willebrand factor-antigen; WC: waist circumference; SD: standard deviation.
Data are expressed as mean ± SD for all variables.
OR and CI for detection of elevated prothrombotic factors between those with suboptimal and those with optimal glycaemic control.

FB: fibrinogen; PAI-1-Ag: plasminogen activator inhibitor-1-antigen; vWF-Ag: von Willebrand factor-antigen; OR: odds ratio; CI: confidence interval.
Data are expressed as frequency and percentage (%) within those with suboptimal or optimal glycaemic control, respectively.
Comparisons between those with excessive body weight and normal-weighted
The majority of patients (n = 117) had excessive body weight (75.5%). These patients had higher daily insulin requirements (46.2 ± 20.4 vs 36.9 ± 14.6 IU/day, p = 0.042), BMI (21.9 ± 7.5 vs 16.8 ± 6.4 kg/m2, p < 0.001) and WC (77.1 ± 13.6 vs 69.1 ± 8.9 cm, p = 0.014) and tended to present higher levels of TG (80.7 ± 40.3 vs 76.6 ± 59.7 mg/dL, p = 0.084) than those with a BMI value below 85th percentile. No difference regarding HbA1c (8.7 ± 2.2 vs 8.6 ± 2.3%, p = 0.767) or SBP, DBP, TC, HDL-c, LDL-c, Apo-A1, Apo-B, Apo-B/Apo-A1 or Lp(a) levels between these two subgroups was detected (data not shown). In this subgroup, higher PAI-1 levels were found as compared to normal-weighted patients (46.3 ± 20.8 vs 38.6 ± 21.9 ng/mL, p = 0.032), whereas no differences were noted in vWF-Ag (115.8 ± 37.3 vs 120.0 ± 50.2%, p = 0.983) and FB (303.0 ± 59.7 vs 286.4 ± 75.8 mg/dL, p = 0.224) levels.
Correlations between levels of prothrombotic factors and other characteristics
The results of univariate correlation analysis for all patients are presented in Table 4. Among prothrombotic factors, vWF-Ag and FB levels were inter-correlated (r = 0.194, p = 0.017). Levels of PAI-1-Ag were positively correlated only with TG (r = 0.225, p = 0.006). Levels of vWF-Ag were also positively correlated with HbA1c, TC, TG, LDL-c and Apo-B/Apo-A1 and negatively with age at entry to study, duration of diabetes and HDL-c (Table 4). Finally, no correlation between FB and parameters of the lipid profile was detected, except a tendency for higher levels of Lp(a) with higher vWF-Ag levels (r = 0.164, p = 0.089).
Correlations between prothrombotic factors and other characteristics in the whole sample.
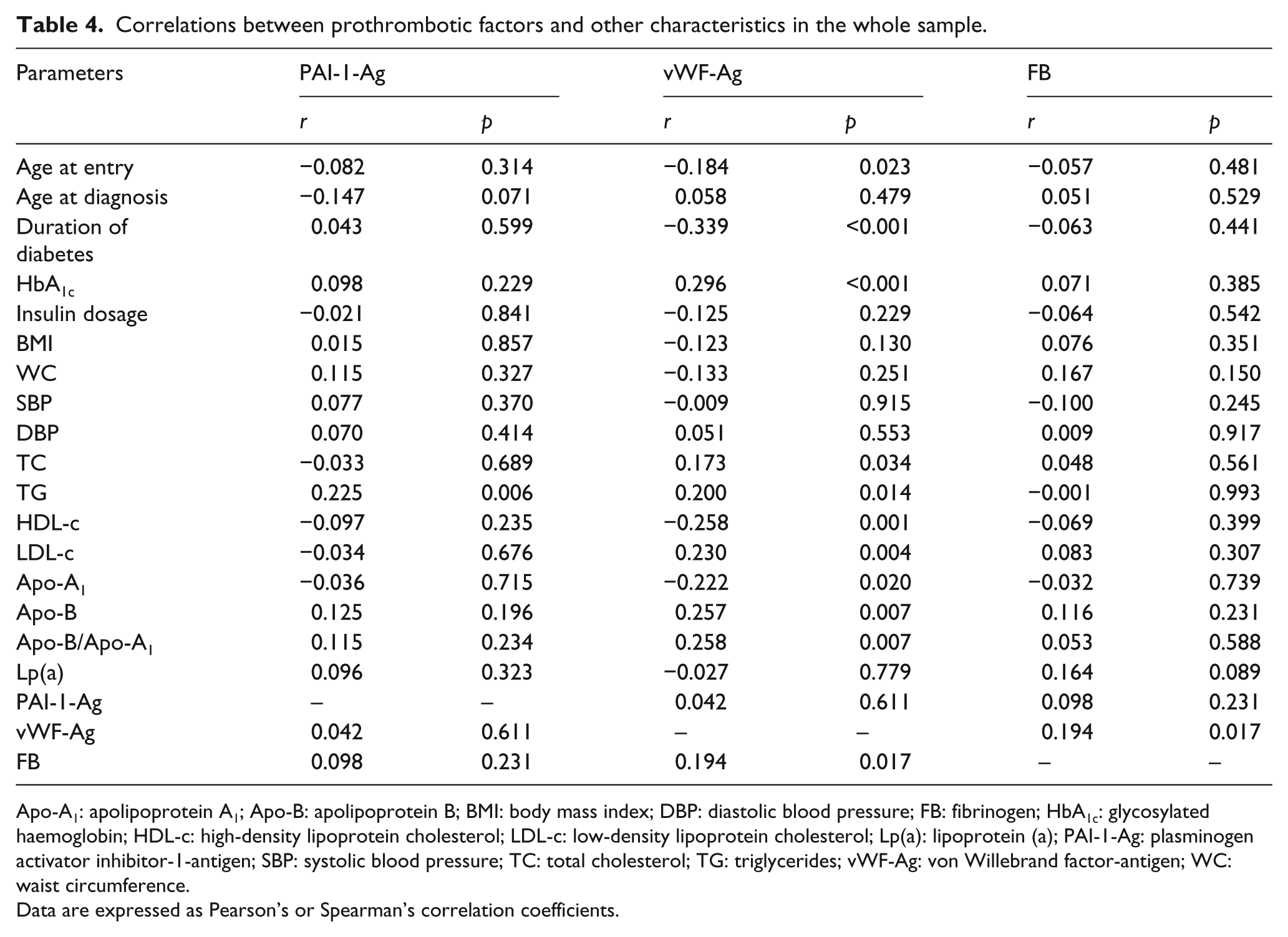
Apo-A1: apolipoprotein A1; Apo-B: apolipoprotein B; BMI: body mass index; DBP: diastolic blood pressure; FB: fibrinogen; HbA1c: glycosylated haemoglobin; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; Lp(a): lipoprotein (a); PAI-1-Ag: plasminogen activator inhibitor-1-antigen; SBP: systolic blood pressure; TC: total cholesterol; TG: triglycerides; vWF-Ag: von Willebrand factor-antigen; WC: waist circumference.
Data are expressed as Pearson’s or Spearman’s correlation coefficients.
Standard multiple linear regression analysis with PAI-1, vWF-Ag and FB levels as dependent variables
As shown in Table 5, standard multiple linear regression models were also performed on the basis of the univariate correlations (Table 4). Gender, age, HbA1c and BMI were held constant in all regression models as independent variables. When age, gender, HbA1c, BMI, duration and LDL-c were included in the regression model, only HbA1c contributed independently to the variation in vWF-Ag levels (adjusted r2 = 0.129, p < 0.001). The association between PAI-1-Ag and TG as well as other potential determinants did not remain significant in multivariable analysis (adjusted r2 = 0.005, p = 0.334). Finally, the only significant determinant for FB levels, after adjustment for all other confounding variables, was gender (adjusted r2 = 0.032, p = 0.065).
Standard multiple linear regression analysis with PAI-1, vWF-Ag and FB levels as dependent variables.
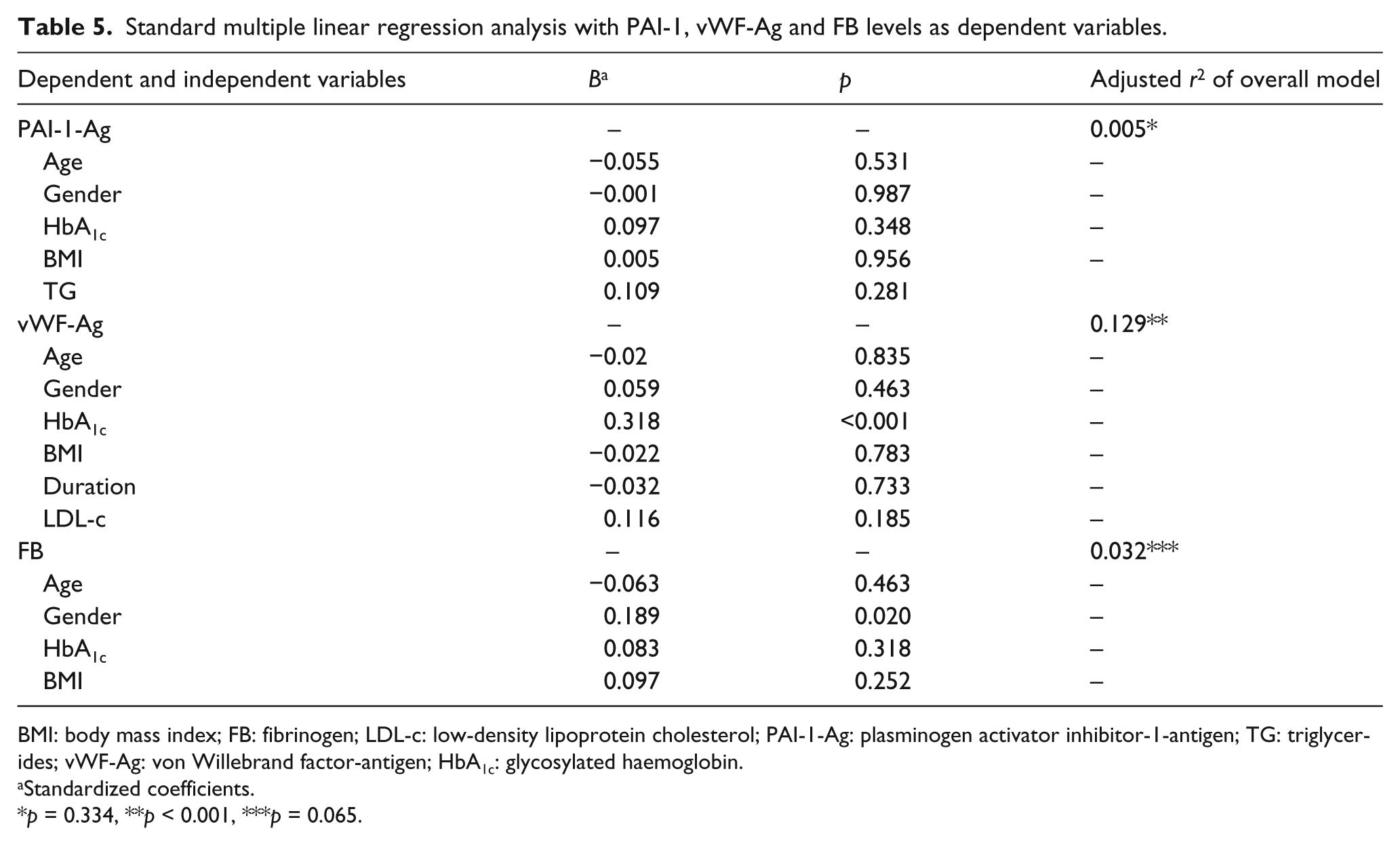
BMI: body mass index; FB: fibrinogen; LDL-c: low-density lipoprotein cholesterol; PAI-1-Ag: plasminogen activator inhibitor-1-antigen; TG: triglycerides; vWF-Ag: von Willebrand factor-antigen; HbA1c: glycosylated haemoglobin.
Standardized coefficients.
p = 0.334, **p < 0.001, ***p = 0.065.
Discussion
In this study, levels of PAI-1-Ag, vWF-Ag and FB were measured as markers of impaired prothrombotic state as well as of endothelial dysfunction in children and adolescents with T1DM. Approximately one half of the participants (49%) were characterized by increased levels of at least one of these markers, indicating impaired prothrombotic state. Significant relations were observed between elevated prothrombotic factors and suboptimal glycaemic control as well as impaired lipid profile. Among them, levels of vWF-Ag were found to be more strongly affected by glycaemic control. Indeed, this study demonstrated for the first time that the paediatric population with uncontrolled diabetes had an eightfold increased risk of detecting elevated vWF-Ag levels as compared to diabetics with good glycaemic control. All these findings validate that impaired endothelial function is already present in youths with T1DM and suggest supplementary pathways for the premature development of atherothrombosis and CVD.
Results from studies on vWF-Ag levels in children and adolescents with T1DM are conflicting, suggesting either higher13–16 or similar values17–19 compared to healthy controls. In our study, high circulating levels of vWF-Ag were detected in 14.2% of participants. Furthermore, a negative correlation between vWF-Ag levels and duration of T1DM was also observed, in concordance with the only published study in the same age group. 13 It has been speculated that during the first years of diabetes, there is a subclinical prothrombotic disorder, concerning mainly endothelium, which has been described under the term of ‘endothelial perturbation’. This phenomenon may represent either a reversible event of the initial inflammatory autoimmune process of diabetes or it can be considered an indicator of the risk for developing microvascular complications. This hypothesis remains to be clarified with further long-term studies. Moreover, we found that suboptimal glycaemic control increased the risk of detecting abnormally high levels of vWF-Ag by 7.8 times and their association remained independently significant even after adjustment for potential confounders. This finding is in accordance with previous studies in children 17 and adults,20,21 suggesting that poor metabolic control leads to endothelial activation. It is noteworthy that differences in vWF levels have also been reported between T1DM patients with and without microvascular complications.18,22 Indeed, vWF-Ag has been proposed as a non-invasive marker for endothelial dysfunction.13,23 Besides, vWF-Ag plays an essential role in the coagulation process, due to its property to contribute to normal platelet adhesion and aggregation. 24 In addition, since vWF-Ag is a carrier for factor VIII, it regulates formation of the fibrin- and platelet-rich thrombus at the position of endothelial injury. 25 In regard to the relation between vWF-Ag levels and lipid profile, a positive relationship between vWF-Ag and TC as well as TG levels has been found by Correa and Alfieri. 22 Accordingly, in this study, vWF-Ag levels were correlated positively with TC, TG, LDL-c, Apo-B levels and Apo-B/Apo-A1 ratio and negatively with HDL-c and Apo-A1 levels. Our findings confirm the fact that impaired lipid profile may additionally increase the risk of endothelial dysfunction and consequently of future CVD in youths with T1DM. 26
There is a lack of agreement on data regarding PAI-1 levels in T1DM; some studies have reported high20,27–29 and others either lower 30 or similar31 –33 PAI-1-Ag levels as compared to controls. In our study, PAI-1-Ag levels were found to be elevated in 39.4% of the whole sample. No significant association was found between PAI-1-Ag levels and glycaemic control, a finding that is in symphony with data reported previously only in adults, 30 whereas earlier studies demonstrated contradictory results.27,29,34 In this study, PAI-1-Ag levels were correlated with TG levels, a finding that is in accordance with similar data from literature,27,32,35 thus supporting the possible involvement of TG in PAI-1-Ag promoter activation. 36 However, this relationship disappeared when other confounders were taken into consideration. In participants with excessive body weight, PAI-1-Ag levels were significantly – but not independently – higher than those of normal-weighted. This finding seemed reasonable since obesity is characterized by high PAI-1 levels as well.8,37 Though the significance of PAI-1-Ag levels, as a predictor of future CVD, has already been confirmed in the metabolic syndrome and T2DM, 38 the role of PAI-1-Ag in T1DM still remains unclear and needs further elucidation.
As it is known, FB is an acute-phase protein, and its increase represents a marker of inflammation 22 and characterizes individuals at high cardiovascular risk. 7 In this study, 5.2% of the participants presented elevated FB levels. What is of great interest is that FB and gender were significantly and independently associated even after adjustment for potential confounders. In addition, no correlation with HbA1c or duration of diabetes was detected. These data are in agreement with similar studies concerning children and adolescents with T1DM18,39 and in disagreement with those reported from SEARCH study; 40 the latter showed elevated FB levels in T1DM youngsters as well as a correlation between FB concentrations and lipid profile.
Several mechanisms have been proposed to explain the role of hyperglycaemia, observed in diabetes, in alteration of prothrombotic state and endothelial function. Oxidative stress, polyol pathway/protein kinase C system activation and the presence of advanced glycation end-products (AGEs) are all potentially involved. 41 The concept of metabolic memory was recently proposed, 42 pointing out that effects of long-term mechanisms such as non-enzymatic glycation process and the excess of cellular reactive oxygen and nitrogen species, originating at the level of glycated mitochondrial proteins, appear to induce endothelial dysfunction. Consequently, heterogeneity of the reported data concerning prothrombotic factors in T1DM may be explained in some extent with the concept of metabolic memory.
The cross-sectional design can be considered as one limitation of this study. Hence, we cannot conclude to a cause–effect relationship among the parameters that were tested. The absence of a control group might also limit the value of this study; however, our aim was to test the hypothesis that glycaemic control influences the levels of prothrombotic factors; therefore, a control group was not necessary. Finally, this is a study from one research centre, and thus, further multi-centre studies that will include greater numbers of patients and from different populations are needed to confirm our findings.
In conclusion, this study suggests that impaired prothrombotic state and consequently impaired endothelial function are already present and worsen with suboptimal metabolic control in youths with T1DM, representing a cumulative risk factor for development of premature atherothrombosis and future CVD. Achievement and maintenance of euglycaemia and normolipidaemia are necessary to decelerate progress of this aggravating but modifiable process in this population.
Key messages
Abnormally high levels of PAI-1-Ag, vWF-Ag and FB are found in 39.4%, 14.2% and 5.2% of type 1 diabetic patients, respectively.
Suboptimal glycaemic control is associated with impaired lipid profile and an eightfold increased risk of elevated vWF-Ag levels.
The only significant determinant for FB levels, even after adjustment for other confounding factors, was gender.
Endothelial dysfunction and impaired prothrombotic state seem to be already present and worsen with poor metabolic control in youths with T1DM, representing a cumulative risk factor for development of premature atherothrombosis and future cardiovascular disease (CVD).
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
