Abstract
Human islet transplant success is partially impaired by slow revascularisation. Our study investigated the potential for rotational cell culture (RC) of human islets combined with thiazolidinedione (TZD) stimulation of peroxisome proliferator-activated receptor gamma (PPARγ) to upregulate vascular endothelial growth factor (VEGF) expression in the islets. Four groups of human islets were studied: static culture (SC) with and without 25 mmol/L TZD and RC with and without 25 mmol/L TZD. These were assessed for insulin secretion and soluble VEGF-A release. Both proteins were quantified by enzyme-linked immunosorbent assay (ELISA), supported with qualitative immunofluorescence staining. RC + TZD increased insulin secretion by >20% (p < 0.05–0.001) in response to 16.7 mmol/L glucose and 16.7 mmol/L glucose + 10 mmol/L theophylline (G + T). This effect was seen at all time intervals compared with SC and without addition of TZD. Soluble VEGF-A release was significantly augmented by RC and TZD exposure with an increased effect of >30% (p < 0.001) at 72 h under both SC and RC conditions. RC supplemented with a TZD enhances and prolongs the release of insulin and soluble VEGF-A by isolated human islets.
Keywords
Introduction
Since the successful clinical islet transplantations performed by Shapiro et al. 1 in Edmonton in 2000, several trials have validated the procedure of intrahepatic islet transplantation to treat Type 1 diabetes via portal vein infusion with the results collated by the Collaborative Islet Transplant Registry. 2 By 2002, 35 patients had been transplanted by the Edmonton group. Of these, 87% remained insulin independent at 1 year; 3 however, although 80% of grafts were still producing C-peptide, 5 years after transplantation only 10% of islet graft recipients remained insulin independent.4,5 The gradual loss of graft function invokes consideration of the following issues: improvements to increase the islet yield from donor pancreata,6,7 reduction in β-cell toxicity of immunosuppressants, 8 development of immunomodulation of islets prior to implantation,9,10 enhancement of islet viability before and after implantation 11 and improvement in the rate of islet engraftment post transplant.12,13
Without specific intervention to aid the post-transplant engraftment, portal vein–infused islets require up to 14 days to revascularise and by 1 month achieve only 5% of the blood perfusion of native islets. 14 Current islet isolation protocols employ harsh enzymatic digestion to free islets from pancreatic exocrine tissue, resulting in damage to the islet microvasculature. 15 Thus, strategies to improve post-transplant islet revascularisation may restore adequate oxygenation and, as such, support graft function.
The vascular endothelial growth factor (VEGF) group of genes plays a vital role in the regulation of growth and differentiation of vascular endothelial cells. 16 VEGF-A regulates vasculogenesis (new blood vessel development in the absence of pre-existing vasculature) and angiogenesis (new blood vessel development from pre-existing vasculature) as well as microvessel permeability.17,18 VEGF-B directs myocardial angiogenesis 19 and VEGF-C and D specifically regulate lymphangiogenesis. 20 Human VEGF-A consists of five major isoforms having 121, 145, 165, 189 and 206 amino acid residues in the mature monomer. VEGF165 is a heavily glycated dimer of about 45 kDa and is the most abundant form, while VEGF206 is very rare. 21 A significant fraction of VEGF-A165 is not secreted but remains bound to the cell surface and the extracellular matrix (ECM), providing a target for immunocytochemistry. 22
Agonists of the peroxisome proliferator-activated receptor gamma (PPARγ), such as the thiazolidinedione (TZD) class of agents (e.g. troglitazone, pioglitazone and rosiglitazone), have been used experimentally to increase VEGF-A expression and action in endothelial cells. 23 Kidszun et al. 24 have shown that adding VEGF-A to islet cultures increased sprouting of capillaries from mouse islets, while Hsu et al. 25 employed systemic administration of rosiglitazone prior to and after transplant to improve the reversal of streptozotocin-induced diabetes and islet graft survival in mice. Additionally, ex vivo transfection for human VEGF gene expression prior to islet transplantation also improved graft revascularisation. 26
Islet β-cells stain positively for VEGF-A, and islets have been shown to produce, sequester and secrete this growth factor, suggesting a role for VEGF-A in islet angiogenesis and to regulate microvessel permeability to facilitate rapid transfer of released insulin into the blood.18,27,28 Hypoxia and prolonged maintenance in culture increase islet VEGF-A messenger RNA (mRNA) levels, indicating that conditions in which vascular flow is reduced or abolished may enhance signals for islet angiogenesis. 27 While hypoxia cannot be avoided, it should be minimised as it will clearly detract from the retention of optimal islet viability prior to transplantation.
Since PPARγ is expressed by β-cells 29 as well as by endothelium 30 and it has been shown that donor-derived endothelium contributes to revascularisation of transplanted islets,31,32 upregulation of PPARγ by a TZD prior to transplantation could potentially increase VEGF-mediated vascularisation of islets. Additional potential advantages to this approach are that TZDs have been reported to increase islet β-cell granulation, support β-cell insulin secretion 33 and exert genomic effects such as downregulation of caspase-3 and P-JNK and upregulation of insulin that improve β-cell survival34,35 and preserve β-cell function in patients with adult-onset latent autoimmune diabetes. 36
The purpose of this study was to test the hypothesis that in vitro islet pre-treatment by stimulation of PPARγ can improve endocrine islet function and upregulate islet VEGF-A with a view to support post-transplant insulin secretory capacity and enhance revascularisation. Previous studies published by our laboratory have shown that rotational cell culture (RC) supports maintenance of the three-dimensional structure and insulin secretory function of human islets (for up to 10 days)37,38 and clonal β-cell-derived ‘pseudoislets’; 39 hence, we employed this culture technique for the study reported here. The Synthecon rotational cell culture system used in our laboratory has been shown to improve oxygenation and distribution of nutrients and dilution of metabolic waste in the culture medium compared with conventional static culture (SC). 40 We emphasise that the techniques described here are designed as pre-implantation treatment with cessation of islet exposure to the TZD immediately prior to transplantation.
Methods
Human islet isolation and culture
Local research ethics committee approval was gained for this study, and human pancreata were obtained through the UK Human Tissue Bank (De Montfort University, Leicester, UK – now Abcellute, Cardiff, UK). Pancreata from four donors (3 male, 1 female, aged 20, 47, 54 and 42 years, respectively) were utilised during the course of this investigation and were enzymatically digested using ductal distention with Liberase (Roche Diagnostics, Sussex, UK), and islets were isolated by a manual method established in our laboratory. 41 In brief, Liberase was infused into the gland following cannulation of the pancreatic duct. Digestion of the pancreas took place at 37°C and was frequently monitored by staining samples of the digest with dithizone, which stains islets red by binding to the zinc that is packaged with insulin in the secretory vesicles. Once islets were observed that were cleaved from the exocrine tissue, digestion was stopped by the addition of cold Hanks’ balanced salt solution (HBSS; Sigma–Aldrich, Dorset, UK – henceforth named as Sigma) supplemented with 2.5% (w/v) bovine serum albumin (BSA; Sigma). The digestate was centrifuged at 300×g for 3 min and the supernatant, containing Liberase, was discarded. This washing procedure was repeated twice more, after which the digestate was suspended in cold University of Wisconsin (UW) solution (Bristol-Myers Squibb, Uxbridge, UK) for 30 min. Following centrifugation at 300×g for 3 min, islets were separated from the exocrine tissue with a discontinuous density gradient consisting of HISTOPAQUE-1077 containing the pancreatic digest, with a covering layer of HBSS supplemented with 0.5% BSA (all purchased from Sigma). Centrifugation at 800×g for 5 min resulted in the islets accumulating at the interface between the two layers. Isolated islets were cultured overnight in a humidified incubator at 30°C with 5% CO2 using 90 mm Petri dishes (Appleton Woods, Birmingham, UK) and medium 199 (m199; Sigma) supplemented with 10% fetal calf serum (FCS; First Link (UK) Ltd, Birmingham, UK), 100 U/mL penicillin, 100 µg/mL streptomycin and 10 µg/mL amphotericin B (all purchased from Sigma).
After 24 h, the islets were distributed into four experimental groups at a density of 750 islet equivalents (IEQ) per millilitre with supplemented m199 as the base medium. As most islet preparations contain islets and islet fragments of varying sizes, a formula has been developed to standardise all islets to a diameter of 150 µm. A quantity of 3 × 100 µL samples of the islet preparation are examined and allocated into specific size groups. Each size group is then multiplied by its conversion factor to adjust the number of islets counted within that size group to IEQs. 42 A concentration titration experiment indicated that insulin secretion and VEGF expression were best supported by 25 mmol/L of the TZD rosiglitazone maleate. The four groups were SC, SC plus 25 mmol/L TZD (SC TZD), RC and RC plus 25 mmol/L TZD (RC TZD). A Synthecon RC system was employed to maintain RC islets in a microgravity suspension culture in 10 mL high-aspect rotational vessels (HARV; CELLON, Luxembourg).
Insulin secretion
Insulin secretion was assessed at 24, 48 and 72 h of treatment by static incubation. Low glucose (1.67 mmol/L glucose), high glucose (16.7 mmol/L glucose) and high glucose with theophylline (16.7 mmol/L glucose + 10 mmol/L theophylline) were prepared in HEPES-buffered Hank’s solution (10 mmol/L HEPES, 113.28 mmol/L NaCl, 4.17 mmol/L NaHCO3, 0.336 mmol/L Na2HPO4, 5.37 mmol/L KCl, 0.952 mmol/L CaCl2, 0.811 mmol/L MgSO4.7H2O, 0.441 mmol/L NH2PO4) of pH 7.4, with 0.5% BSA as insulin secretagogues (all purchased from Sigma). Islet aliquots from all four experimental groups were incubated in low glucose for 1 h at 37°C, after which 18 LP2 tubes containing 20 IEQ each were prepared per experimental group, 6 tubes for the low glucose control and 6 tubes for each of the other two secretagogues. A volume of 2 mL of each secretagogue was added to the allocated tubes per experimental group and incubated at 37°C for 1 h prior to centrifugation and collection of the supernatants for determination of insulin content by enzyme-linked immunosorbent assay (ELISA) (Mercodia human insulin kit from Diagenics Ltd, Milton Keynes, UK). Insulin secretion responses to high glucose ± theophylline were expressed as percentage increase compared with basal secretion achieved by 1.67 mmol/L glucose control (expressed as 100%).
Human islet VEGF production and release
Human islets maintained in m199 in SC and RC ± TZD for 24 h (48 h post isolation) were plated into 24-well plates (Corning Costar plates; Sigma) at a density of 20 IEQ/well to ensure consistency of islet numbers. The islets received m199 ± TZD as per their experimental group, and tissue culture medium was collected at 24 h (48 h exposure, 72 h post isolation) and 48 h (72 h exposure, 4 days post isolation). Soluble VEGF concentration was determined by ELISA (RayBiotech human VEGF kit; Insight Biotechnology Ltd, Wembley, UK). VEGF secretion responses to TZD were expressed as percentage increase compared with no TZD (expressed as 100%).
Immunostaining
Islets from the four experimental groups were plated at a density of 20 IEQ/well onto glass cover slips (VWR International Ltd, Leicestershire, UK) placed into sterile 24-well plates. A 24-h period of culture in a humidified incubator with 5% CO2 at 30°C allowed the islets to adhere and was followed by fixing of the tissue with 4% paraformaldehyde solubilised in phosphate buffered saline (PBS; Sigma).
Membrane permeabilisation was performed with 0.05% Triton X-100 (Sigma) at room temperature for 30 min, followed by blocking non-specific binding sites using a solution consisting of 2% Roche blocking buffer (Roche Diagnostics) and 5% normal goat serum (Sigma) in PBS at room temperature for 1 h.
The primary antibodies used were rabbit anti-pig polyclonal insulin (whole antiserum, isotype IgG; Abcam, Cambridge, UK) and mouse anti-human monoclonal VEGF (clone 56-1; R&D Systems, Minneapolis, MN, USA) and were used at 1:100 dilution with incubation at room temperature on a plate shaker for 1 h and then at 4°C overnight.
Goat anti-rabbit tetramethylrhodamine isothiocyanate (TRITC) and goat anti-mouse fluorescein isothiocyanate (FITC; both were purchased from Southern Biotech, Alabama, USA) secondary antibodies were diluted (1:100) in PBS with 0.05% TWEEN-20 (Sigma) and incubated for 3 h at room temperature in the dark. TRITC was selected to give a red stain for insulin and FITC to produce green staining for VEGF. Negative control staining was performed using rabbit and mouse IgG and indicated no non-specific binding of the secondary antibodies. Representative images were captured using a ZEISS Axioskop 40 microscope fitted with an AxioCam camera linked to a computer with AxioVision software (microscope, camera and software were purchased from Carl Zeiss Ltd, Welwyn Garden City, UK).
Statistical analysis
A two-way analysis of variance (ANOVA; SigmaStat) was used to test for significant differences between the insulin secretory responses to 1.67 mmol/L, 16.7 mmol/L and G + T in the four treatment groups. A Bonferroni post hoc test for multiple comparisons was performed to identify which responses were statistically significant (p < 0.05). VEGF data were analysed using Student’s t-test to determine whether there were significant differences (p < 0.05) in soluble VEGF release between TZD-treated and untreated groups in both SC and RC at 48 and 72 h of exposure.
Results
Human islet isolation and culture
At 48 h of treatment, a sample from each culture group stained with dithizone showed normal morphology (Figure 1(a) to (d)). After 5 days in SC, islets showed some loss of structural integrity, while RC supported the three-dimensional structure of the islets (Figure 2(a) and (b)).
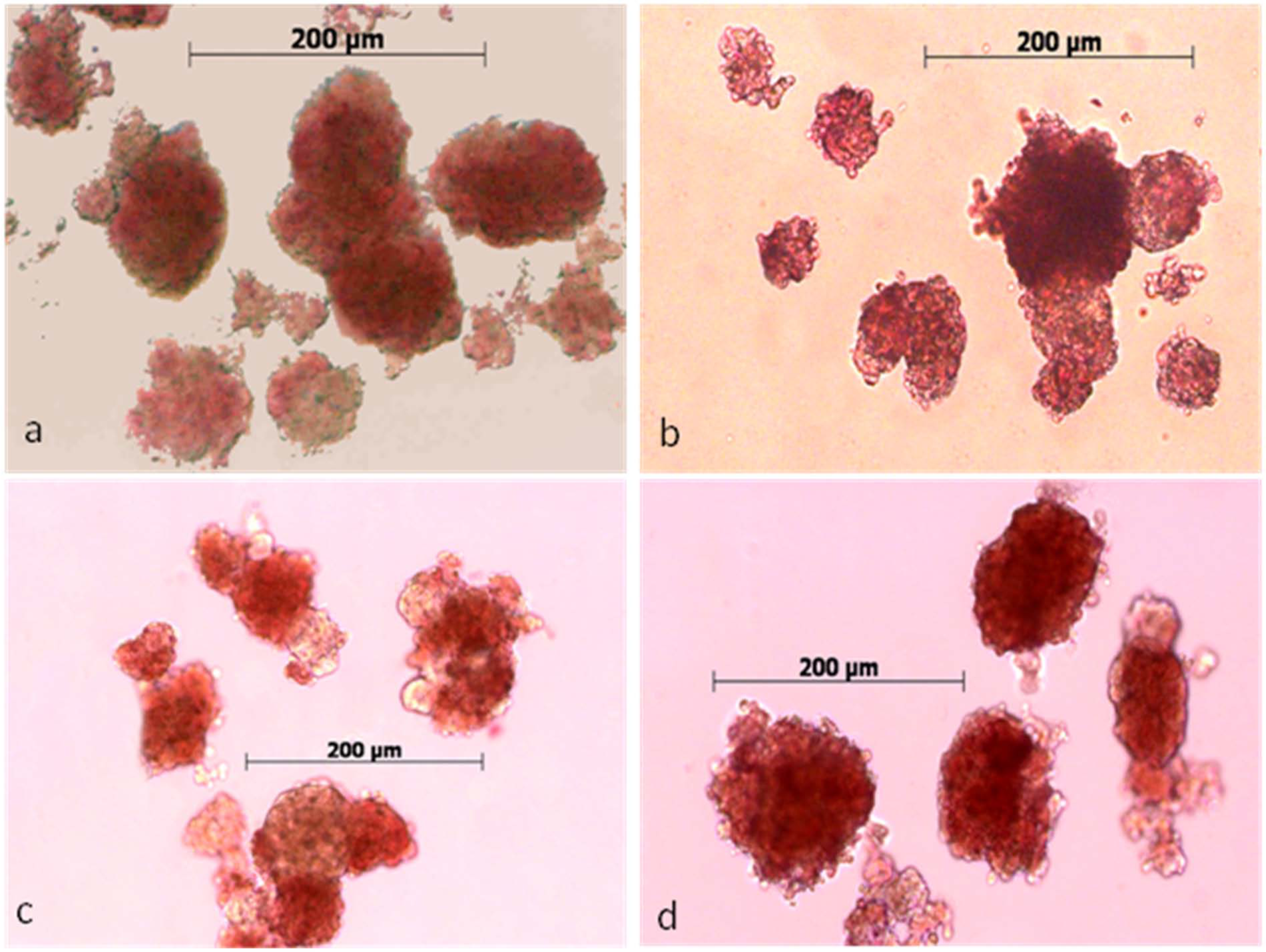
Human islets from the four experimental groups at 24 h of treatment (48 h post isolation): (a) SC, (b) SC TZD, (c) RC and (d) RC TZD.
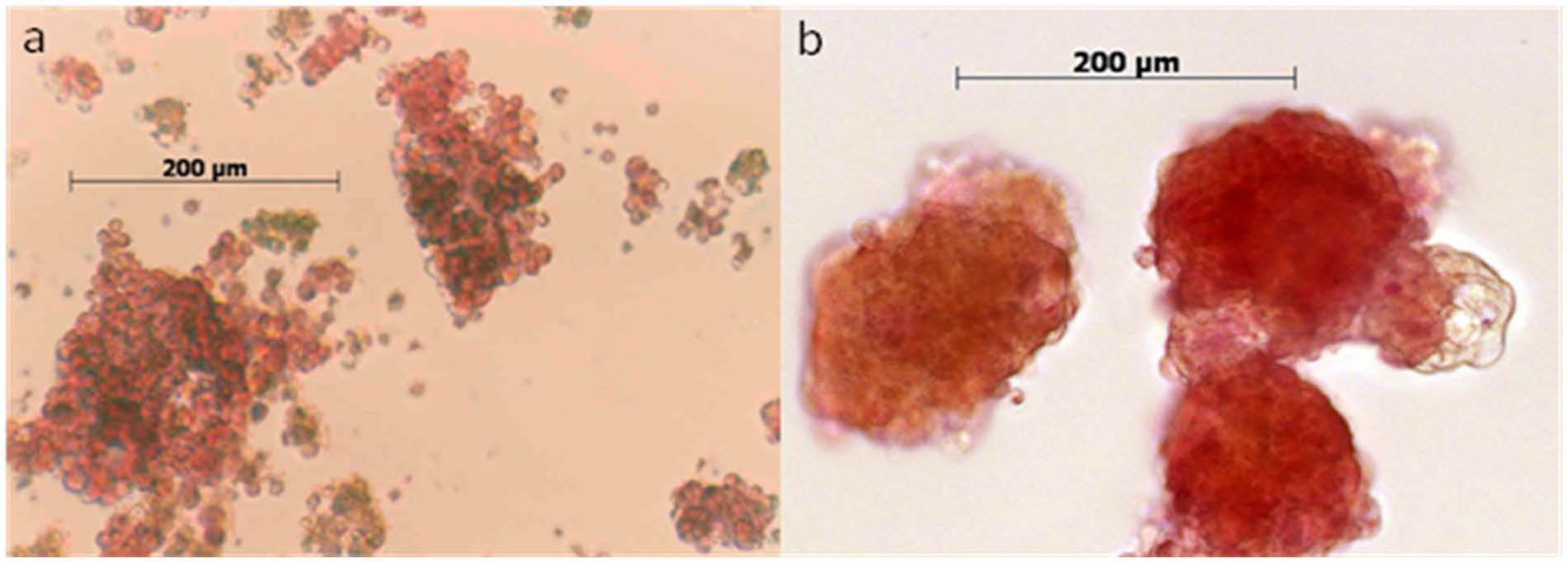
Human islets cultured in m199 for 5 days: (a) SC and (b) RC.
Insulin secretion
Figure 3 shows the combined 24-h post-isolation insulin secretion data for the four donor pancreata used: there was more than twofold increased response to high glucose that was exaggerated in the presence of theophylline. Figure 4(a) to (c) shows the insulin secretion data for the four islet groups over 72 h. For convenience, the levels of statistical significance between treatment groups are shown in Table 1. At each time point, islets responded to high glucose, and this response was enhanced with theophylline. While not always statistically significant, the mean values for these responses were consistently greater for RC than SC, and the presence of TZD further increased these responses for both SC and RC, consistently achieving mean increases >20% (p < 0.05) versus SC alone.
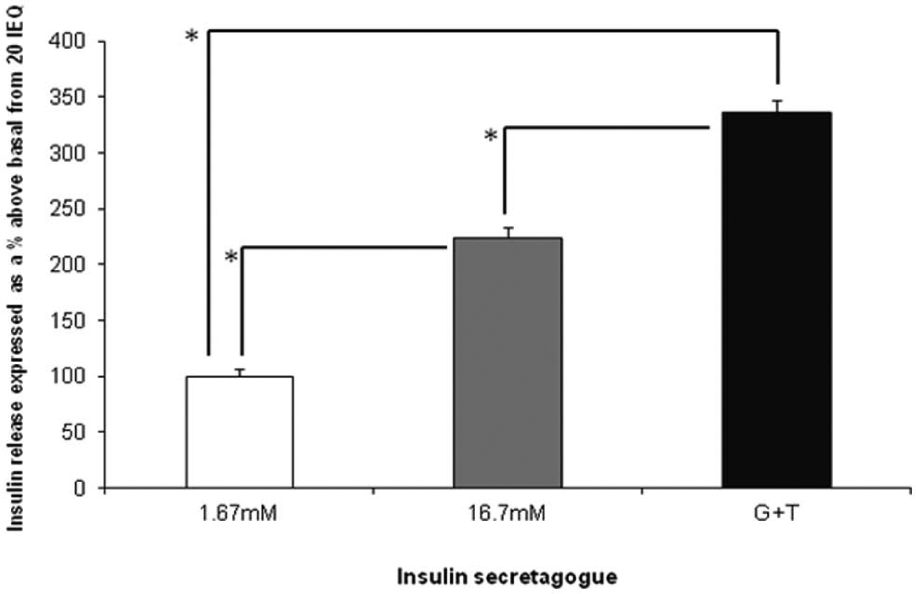
Insulin secretion data from human islets of four donors 24 h post isolation, with islets maintained in SC.
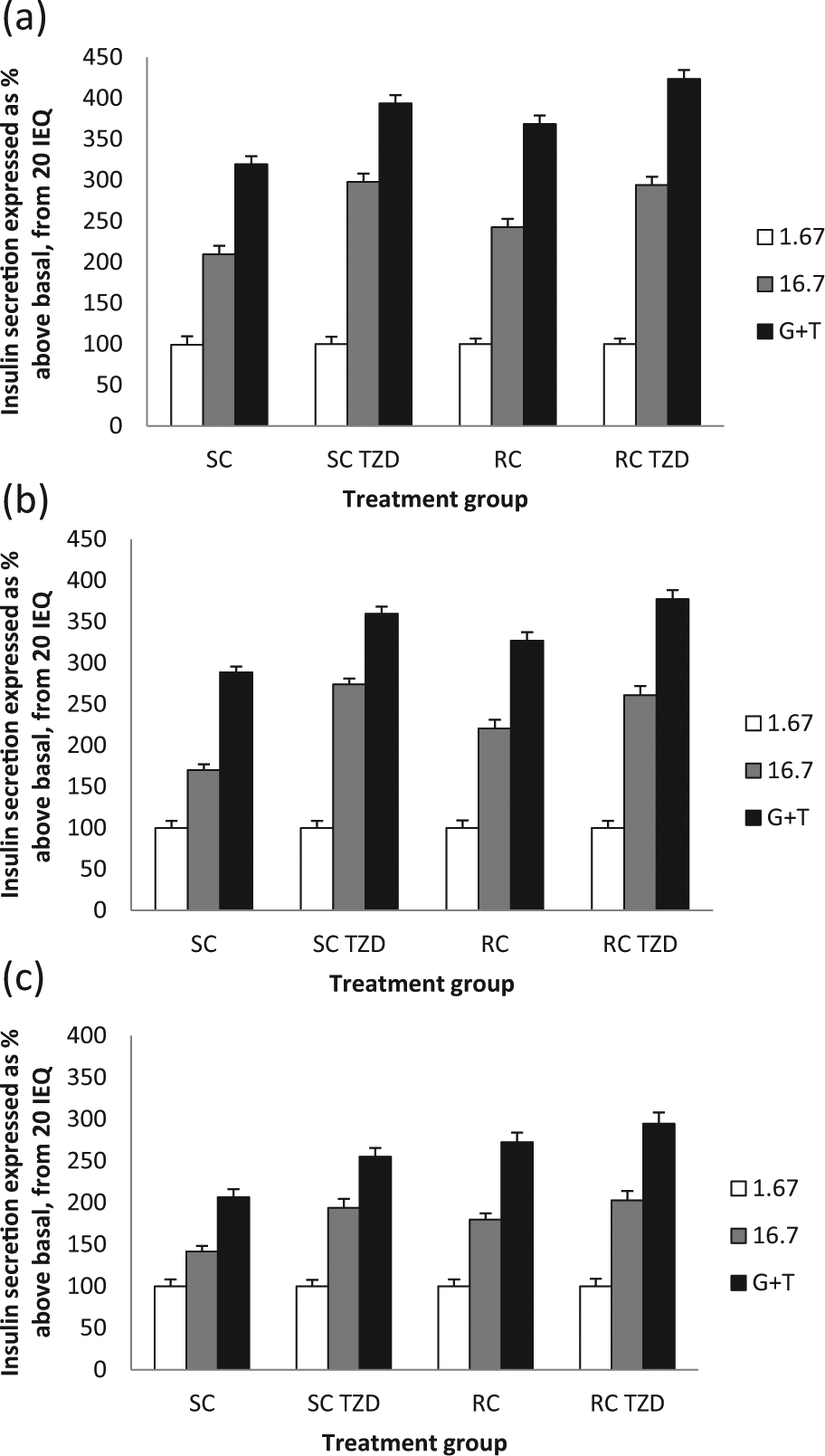
Insulin secretion results for human islets from the four experimental groups at (a) 24 h of treatment (48 h post isolation), (b) 48 h of treatment (72 h post isolation) and (c) 72 h of treatment (4 days post isolation).
Significant differences (p < 0.05) in insulin secretion found in and between experimental groups of islets with or without 25 mmol/L TZD at 24, 48 and 72 h of treatment in SC and RC (the results in this table relate to Figure 4(a) to (c)).

ANOVA: analysis of variance; TZD: thiazolidinedione; SC: static culture; RC: rotational cell culture; G + T: 16.7 mmol/L glucose + 10 mmol/L theophylline.
Human islet immunostaining
Figure 5(a) to (d) shows the images obtained at 72 h after dual staining of islets for insulin and VEGF. The expression of VEGF within the islet often appeared to be co-localised with insulin expression in SC and RC, resulting in an orange fluorescence as red and green overlapped. Increased expression of insulin and VEGF following TZD treatment allowed these two proteins to be more distinctly visualised in the SC TZD and RC TZD groups with red and green fluorescence clearly apparent, while some degree of overlap (orange) persisted. These qualitative observations are consistent with our quantitative insulin and VEGF data.
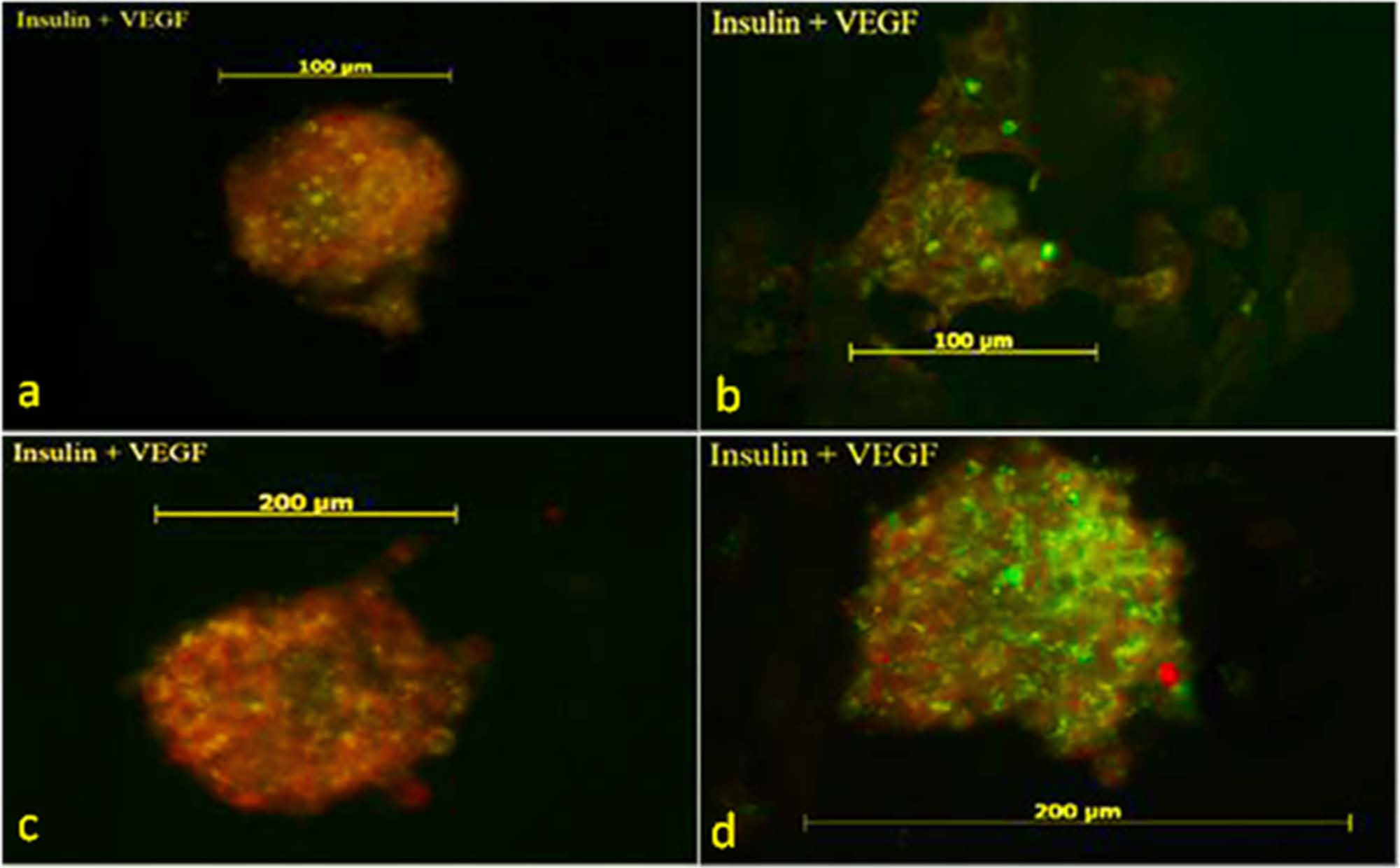
Fluorescence immunostaining for insulin (red – TRITC) and VEGF (green – FITC) in human islets maintained in (a) SC, (b) SC TZD, (c) RC and (d) RC TZD.
VEGF release
Soluble VEGF-A release from all four experimental groups after exposure to 25 mmol/L TZD for 48 and 72 h is shown in Figure 6(a) and (b), respectively. By 72 h, the TZD induced a doubling in SC VEGF release and increased RC VEGF release more than threefold.
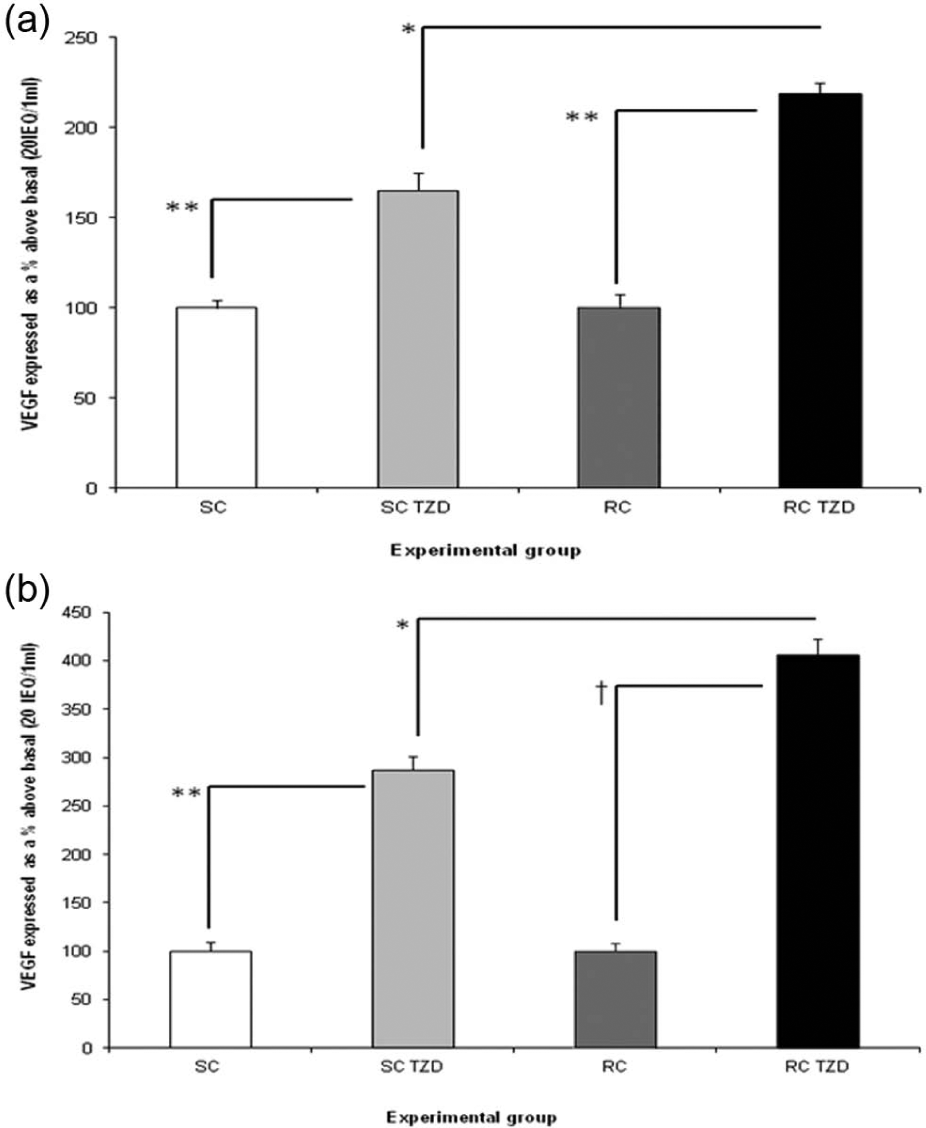
VEGF release from human islets in SC (white bars), SC TZD (light grey bars), RC (dark grey bars) and RC TZD (black bars) at (a) 48 and (b) 72 h of exposure to TZD with n = 8 per experimental group at each time point. (a) 100% SC control = 1069.77 pg/mL, 100% RC control = 1020.31 pg/ml and (b) 100% SC control = 1187.55 pg/mL, 100% RC control = 1159.42 pg/mL.
Discussion
The morphological and functional integrity of human islets deteriorates progressively after isolation and maintenance in conventional SC.37,38 After 5 days of SC, the islets suffer extensive fragmentation, while islets in RC retain a more intact three-dimensional structure. These observations support evidence previously published by our laboratory regarding the beneficial effect of RC on the maintenance of islet structural integrity.37–39
Rotational microgravity culture of islets was chosen for this study since previous studies from this laboratory have established that RC confers higher islet and β-cell viability compared to SC.37–39 Experiments in the present series confirmed and extended our previous studies,38,39 demonstrating that RC preserves islet/pseudoislet integrity and insulin secretory function better than SC through 4 days (Figures 2 and 4). While islets from all four experimental groups showed a gradual reduction of insulin secretory activity between 24 and 72 h, the SC group suffered the largest reduction in the insulin response to high glucose and G + T at 24–72 h post isolation. As islets in the other three groups maintained greater responsiveness to high glucose and G + T, and treatment of these groups involved TZD exposure and/or RC, it is reasoned that both RC and TZD treatment support islet viability. This is confirmed by our observation that the TZD enabled increased glucose-induced insulin secretion in SC and RC when compared with the SC control group. Also, the increased insulin secretory responsiveness of SC and RC islets to TZD, which became relatively greater at 48–72 h, coincides with the transcriptional time course of action of TZDs in other culture systems. 43
The molecular mechanism pertaining to increased insulin secretion following exposure of islets to rosiglitazone has been studied by a number of groups. Richardson et al. 35 have shown that rosiglitazone induced the nuclear accumulation of insulin promoter factor 1 (IPF1) and forkhead homeobox A2 (FOXA2), independent of glucose concentration, and also resulted in a twofold increase in the activity of the IPF1 gene promoter. Yang et al. exposed islets to high levels of free fatty acids that caused a significant reduction in insulin receptor substrate-2 (IRS-2) protein and the association of IRS-2 with the p85 subunit of phosphatidylinositol 3-kinase (PI 3-kinase), resulting in increased basal insulin secretion and reduced glucose-stimulated insulin secretion. Subsequent culture with rosiglitazone restored the insulin secretory profile to near normal and increased both IRS-2 protein levels and its association with the p85 subunit of PI 3-kinase. 44 Thus, the effects of rosiglitazone on insulin secretion could be mediated through an IRS-2-associated PI 3-kinase signalling pathway as well as by upregulation of IPF1.
Following transplantation, islet viability is reduced by slow revascularisation and immunological assault resulting in the need for islets from two or more donors per recipient to secure successful graft function.1,4,5 Since vascularisation can be enhanced by VEGF,27–30,45 which is produced by human islets,27,45 this study examined whether stimulation of endogenous islet VEGF secretion using a TZD could increase the vascularisation potential of newly isolated human islets in culture.
PPARγ, a molecular target for TZDs, is a nuclear-based ligand-activated transcription factor affecting both glucose metabolism and expression of genes encoding the production of VEGF and its receptors in several cell types.46–48 Furthermore, VEGF-induced neovascularisation via PPARγ-mediated pathways has been established in vivo in a murine corneal angiogenic model. 23 In this study, the TZD rosiglitazone was employed as a potent PPARγ agonist, to assess the potential to prime revascularisation of isolated human islets in vitro. The increased VEGF expression observed with a TZD in SC and RC islets by 48 h was enhanced in both groups by 72 h, with RC outperforming SC. This observation, coupled with improved insulin secretory function, suggests that a combination of RC and TZD exposure provides a favourable pre-transplant environment for maintaining functional and structural viability of human islets in culture for at least 4 days.
While the clinical use of rosiglitazone has been discontinued, our study has employed its ability to upregulate VEGF expression in islet endothelium in vitro as a pre-transplant intervention with cessation of exposure prior to islet implantation. Additionally, the proof of concept gained from this investigation can be applied using other PPARγ agonists. A number of agents as diverse as the flavonoid psi-baptigenin, 49 pioglitazone 50 and 15-deoxy-prostaglandin J2 46 have been shown to activate PPARγ, and the latter two agents also upregulate VEGF46,51 and, as such, could be utilised like rosiglitazone to improve pre-transplant islet viability and angiogenic potential.
In this study, islets stained strongly for insulin and VEGF, especially after RC and exposure to TZD. This finding suggests an increased production of VEGF and reflects previous evidence that β-cells and islet vascular endothelial cells are endowed with insulin receptors52,53 and β-cells have been shown to sequester VEGF. 28 Indeed, both the insulin and VEGF signals appeared to be stronger, and these qualitative observations are supported by the increase in both insulin and VEGF production, which we quantified by employing ELISA assays.
The differences in glucose-induced insulin secretion between static and RC are more evident without a TZD, as both RC and TZD exert positive effects on β-cell function.37–39 RC was originally optimised to favour three-dimensional integrity and insulin production/secretion by islet β-cells. The improved insulin secretory profile after RC might be explained by the ‘β-cell recruitment’ phenomenon in which the detection of increased extracellular glucose by a few individual β-cells initiates the insulin secretory cascade and recruitment of adjacent β-cells to participate in insulin secretion. 54 Additionally, TZDs have been shown to support β-cell function and viability,34–36 and our data corroborate these findings under both SC and RC conditions. While β-cells did not show an additive increase in insulin secretion in response to the combined effects of RC and exposure to a TZD, there was an additional increase in measured soluble VEGF-A release from the combined RC + TZD treatment group. There may be a lower threshold for upregulation of insulin secretion from β-cells than for the release of VEGF from islet endothelial cells in the presence of ischaemia.
Our further investigations will include more extensive characterisation of the effect of PPARγ agonism on islet morphology, followed by in vivo assessment of islet function and revascularisation. In conclusion, in vitro pre-treatment of human islets with a TZD in RC may improve islet revascularisation and support β-cell function in preparation for transplantation.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest associated with this manuscript.
Funding
This study received financial support from the Rowlands Trust, The Eveson Charitable Trust, Worcestershire Acute Hospitals NHS Trust and the South Warwickshire and Wyre Forest Diabetes UK Voluntary Support Groups.
