Abstract
Objective:
Reduced renal function and albuminuria predict cardiovascular (CV) events and mortality in type 2 diabetes (T2D). In addition, we evaluated the role of co-existing congestive heart failure (CHF) and other CV risk factors on CV events in a large observational population-based cohort of T2D patients.
Research design and methods:
We included 66,065 patients with T2D who were reported to the National Diabetes Register (NDR) in Sweden between 2003–2006 with a follow-up of 5.7 years. Data on outcomes were collected from the cause of death and hospital discharge registers.
Results:
A total of 10% of patients experienced a CV event and 3.7% of these were fatal. Increasing levels of albuminuria and renal impairment were independently associated with increasing risk of CV events and all-cause mortality also when adjusting for CHF. In normoalbuminuric patients, a reduction in renal function is an important predictor of CV events and all-cause mortality. Glycaemic control (high HbA1c), smoking and hyperlipidaemia had important effects on risk for CV events in patients with albuminuria, while high blood pressure, but not glycaemic control, had an effect in patients with normoalbuminuric renal impairment.
Conclusion:
Albuminuria and renal impairment are independent risk factors for CV outcomes and mortality in T2D, albuminuria being the strongest risk factor and relevant at all levels of renal function. In normoalbuminuric patients, a reduction in renal function is an important predictor of CV events and all-cause mortality.
Keywords
Introduction
Diabetes increases the risk of developing cardiovascular (CV) events 4–5 times 1 and end-stage renal disease (ESRD) 10–12 times. 2 Previous studies have shown that both chronic kidney disease (CKD) with reduced renal function, based on estimated glomerular filtration rate (eGFR), and albuminuria predict CV events in the general population and in patients with type 2 diabetes (T2D).3–5 Recently, it has been suggested that an increase in the risk of developing CV events and mortality may be detected already in patients with mild renal impairment (eGFR < 90 mL/min/1.73 m2). 6 CV risk increases with decline in renal function and is strongly accentuated when renal function is lower than eGFR < 45 mL/min/1.73 m2. 4 In patients with reduced renal function, CV mortality is 10- to 20-fold compared to age-matched subjects in the general population. 7 Similarly, an increased level of albuminuria is associated with increased risk of CV events, CV mortality and all-cause mortality in patients with T2D. 3
Reduced renal function in patients with T2D is more common than previously reported, 8 and non-albuminuric renal impairment is also frequently found.9,10 The independent effects of albuminuria and reduced renal function as predictors of CV events and mortality have been analysed in epidemiological studies with different populations and a variety of outcomes as well as in two large clinical trials, Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) and Fenofibrate Intervention and Event Lowering in Diabetes (FIELD).11,12 These findings have also been verified in a meta-analysis of high-risk population-based cohorts. 13 However, to our knowledge, there have been no studies in nationwide population-based cohorts with T2D patients given routine treatments in clinical practice.
The main objective of this study was to examine the relationship between baseline renal function (eGFR), the presence or absence and degree of albuminuria, combinations of the two and CV outcomes and all-cause mortality during 5 years of follow-up in a large observational population-based cohort study of patients with T2D. In addition, we evaluated the effect of co-existing CV disease and congestive heart failure (CHF) at baseline on these relationships, and assessed the effects of different CV risk factors for CV outcomes in two different groups of patients, that is, albuminuric patients with normal renal function as compared with those with non-albuminuric renal impairment. This was done using linkage of data from the National Diabetes Register (NDR), the cause of death, and the hospital discharge registers in Sweden.
Research design and methods
Subjects
NDR was initiated in 1996. 14 Reporting to NDR is based on information collected, at least once yearly, during patient visits. The register is population-based, nationwide, and approximately 95% of hospital-based outpatient clinics and 75% of primary health care centres participate. Currently, 70% of all patients with diabetes in Sweden are estimated to be reported into the register. All patients have agreed by informed consent to be registered before inclusion, and all information is stored in a central database. The Regional Ethics Review Board at the University of Gothenburg approved the study, which was performed in accordance with the Declaration of Helsinki.
We selected patients with T2D, aged 30–79 years with reported data available in the NDR for all analysed clinical variables between 2003–2006. Patients with body mass index (BMI) < 18.5 kg/m2 (underweight) and BMI > 45 kg/m2 (extreme obesity) were excluded. Patients with an eGFR < 30 mL/min/1.73 m2 were also excluded (n = 762), since reporting of patients with diabetes and severe renal impairment into the register is limited. Altogether 66,065 patients were included and followed prospectively during 266,958 person-years for a maximum of 6 years until 2009 estimating all endpoints during the follow-up period. Mean follow-up time was 5.7 years.
Methods and definitions
Diabetes was diagnosed using Swedish and American Diabetes Association (ADA) criteria [fasting plasma glucose of 7.0 mmol/L (126 mg/dL or higher)], or current treatment with glucose-lowering agents. T2D was defined using the epidemiological definition, that is, treatment with diet or hypoglycaemic agents only, or age of 40 years or older at onset of diabetes and treatment with insulin alone or combination of oral agents.
Clinical and biochemical characteristics reported to the NDR at baseline were age, sex, diabetes duration, weight, height, blood pressure, HbA1c, total-cholesterol, low-density lipoprotein (LDL)-cholesterol, high-density lipoprotein (HDL)-cholesterol, triglycerides, albuminuria, plasma creatinine, smoking and use of glucose-lowering, antihypertensive and lipid-lowering medication. Blood pressure recording was the mean value of two supine readings (Korotkoff 1–5) with a cuff of appropriate size, after at least 5 min of rest. Smoking was defined as smoking one or more cigarettes per day, or using a pipe, or a subject who had stopped smoking within past 3 months.
Laboratory analyses of HbA1c, blood lipids levels and plasma creatinine were carried out at local laboratories. HbA1c analyses are quality assured nationwide by regular calibration (EQUALIS). In this study, all HbA1c values were reported by MonoS, a high-performance liquid chromatography (HPLC) method, and then converted to mmol/mol [International Federation of Clinical Chemistry (IFCC)] and % [National Glycohemoglobin Standardization Program (NGSP)/Diabetes Control and Complications Trial (DCCT)]. 15 LDL cholesterol values were calculated using Friedewald’s formula: LDL cholesterol = total-cholesterol − HDL-cholesterol − (0.45 × triglycerides), if triglycerides < 4.0 mmol/L. Albuminuria was defined as micro- or macroalbuminuria [urine albumin excretion (UAE) 20–200 µg/min or >200 µg/min in two out of three consecutive samples within 6 months]. Estimated eGFR (mL/min/1.73 m2) was defined according to 175 modification of diet in renal disease (MDRD). 16 Renal impairment was defined as eGFR < 60 mL/min/1.73 m2 and CKD stage 3. 17 A supplementary analysis also comprised estimation of eGFR (mL/min/1.73 m2) defined according to Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI). 18
Follow-up and definition of endpoints
All patients were followed from the baseline examination until an event or death or otherwise until 31 December 2007. The following endpoints were used: fatal or non-fatal coronary heart disease (CHD), fatal or non-fatal cardiovascular disease (CVD) and all-cause mortality. Fatal CHD was defined as International Classification of Diseases (ICD)-10 codes I20-I25, and non-fatal CHD event was defined as non-fatal myocardial infarction (ICD-10 code I21), unstable angina (ICD-10 code I20.0), percutaneous coronary intervention (PCI) and/or coronary artery bypass graft (CABG). Fatal/non-fatal stroke was defined as fatal or non-fatal intracerebral haemorrhage, cerebral infarction or unspecified stroke (ICD-10 codes I61, I63, I64 and I67.9). Fatal/non-fatal CVD was the composite of fatal/non-fatal CHD or fatal/non-fatal stroke, whichever came first. A history of CHF was coded as ICD-10 code I50, and atrial fibrillation I48.
All endpoint events were retrieved by data linkage with the cause of death and hospital discharge registers (National Board of Health and Welfare, Sweden), a reliable validated alternative to revised hospital discharge records and death certificates. 19
Statistical methods
Mean values ± 1 standard deviation (SD) and frequencies (%) of baseline clinical and biochemical characteristics are given in Table 1. Cox proportional hazards regression was used to estimate hazard ratios (HRs) with 95% confidence intervals (95% CIs) for CV diseases and mortality with albuminuria or renal impairment, adjusted for covariates as given (Tables 2 to 4). The proportional hazards assumption was analysed with Schoenfeld residuals and with the test of all time-dependent covariates simultaneously. The assumption was fulfilled for all covariates except age, which was included as a strata variable by stratification in quartiles (Tables 2 to 4). Albuminuria was analysed with micro- and macroalbuminuria as dummy variables, and normoalbuminuria as a reference, as shown in Table 2. Various combinations of albuminuria and renal impairment were used in Table 3 and Figure 1. Interaction between albuminuria and renal impairment was analysed with maximum likelihood estimation. HRs for clinical characteristics and other CV risk factors, used as covariates, as shown in Tables 2 and 3, were estimated in two subgroups consisting of combinations of albuminuria and no renal impairment, or renal impairment and no albuminuria (Table 4). The c statistic was estimated for discrimination, using a method allowing for censoring at Cox regression. 20
Clinical and biochemical characteristics at baseline in all patients, patients with or without albuminuria and patients with or without renal impairment.
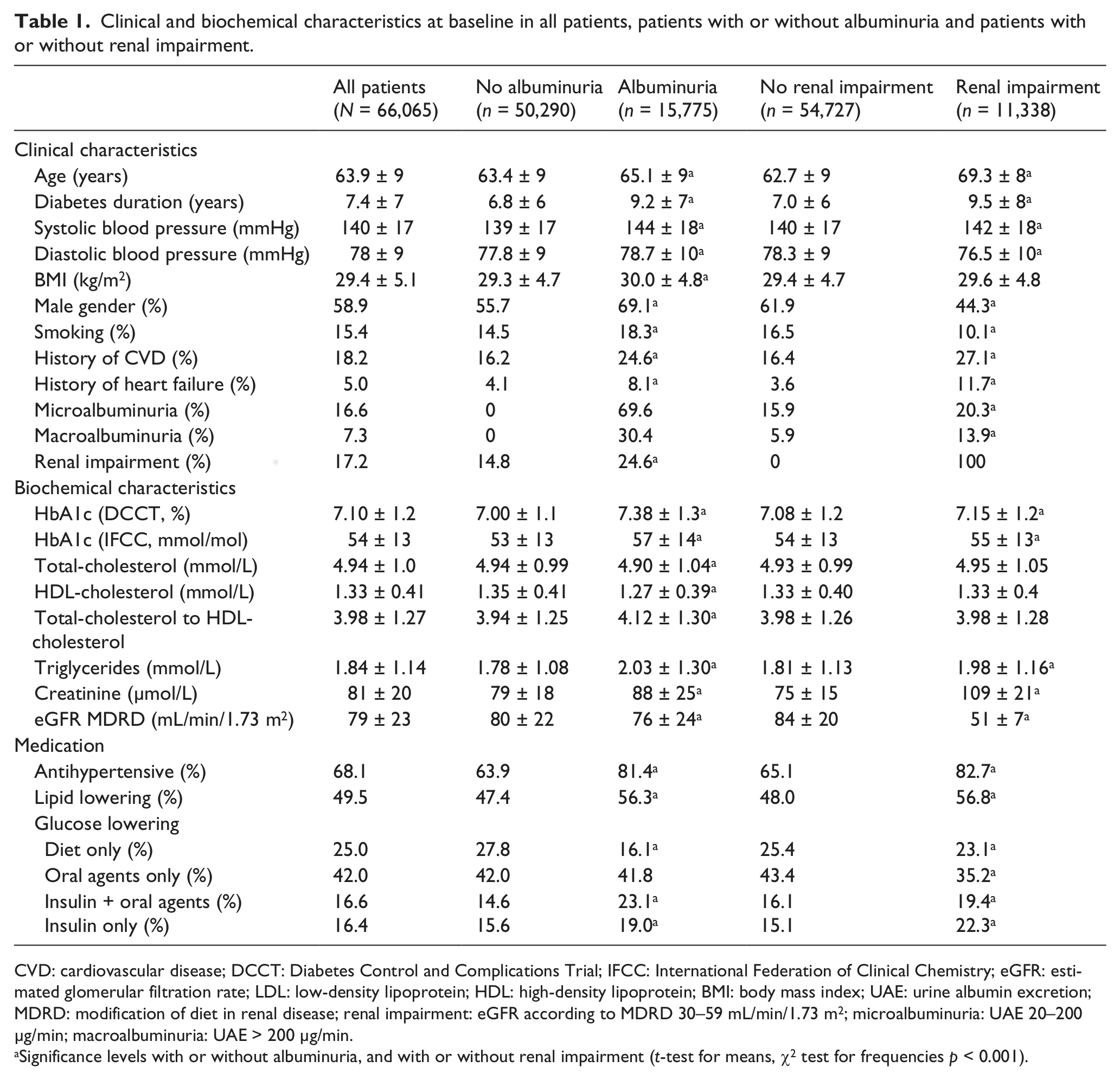
CVD: cardiovascular disease; DCCT: Diabetes Control and Complications Trial; IFCC: International Federation of Clinical Chemistry; eGFR: estimated glomerular filtration rate; LDL: low-density lipoprotein; HDL: high-density lipoprotein; BMI: body mass index; UAE: urine albumin excretion; MDRD: modification of diet in renal disease; renal impairment: eGFR according to MDRD 30–59 mL/min/1.73 m2; microalbuminuria: UAE 20–200 µg/min; macroalbuminuria: UAE > 200 µg/min.
Significance levels with or without albuminuria, and with or without renal impairment (t-test for means, χ2 test for frequencies p < 0.001).
Hazard ratios (95% CI) for cardiovascular outcomes and all-cause mortality by different degrees of albuminuria and renal impairment in all 66,065 patients with type 2 diabetes followed for 5.7 years.
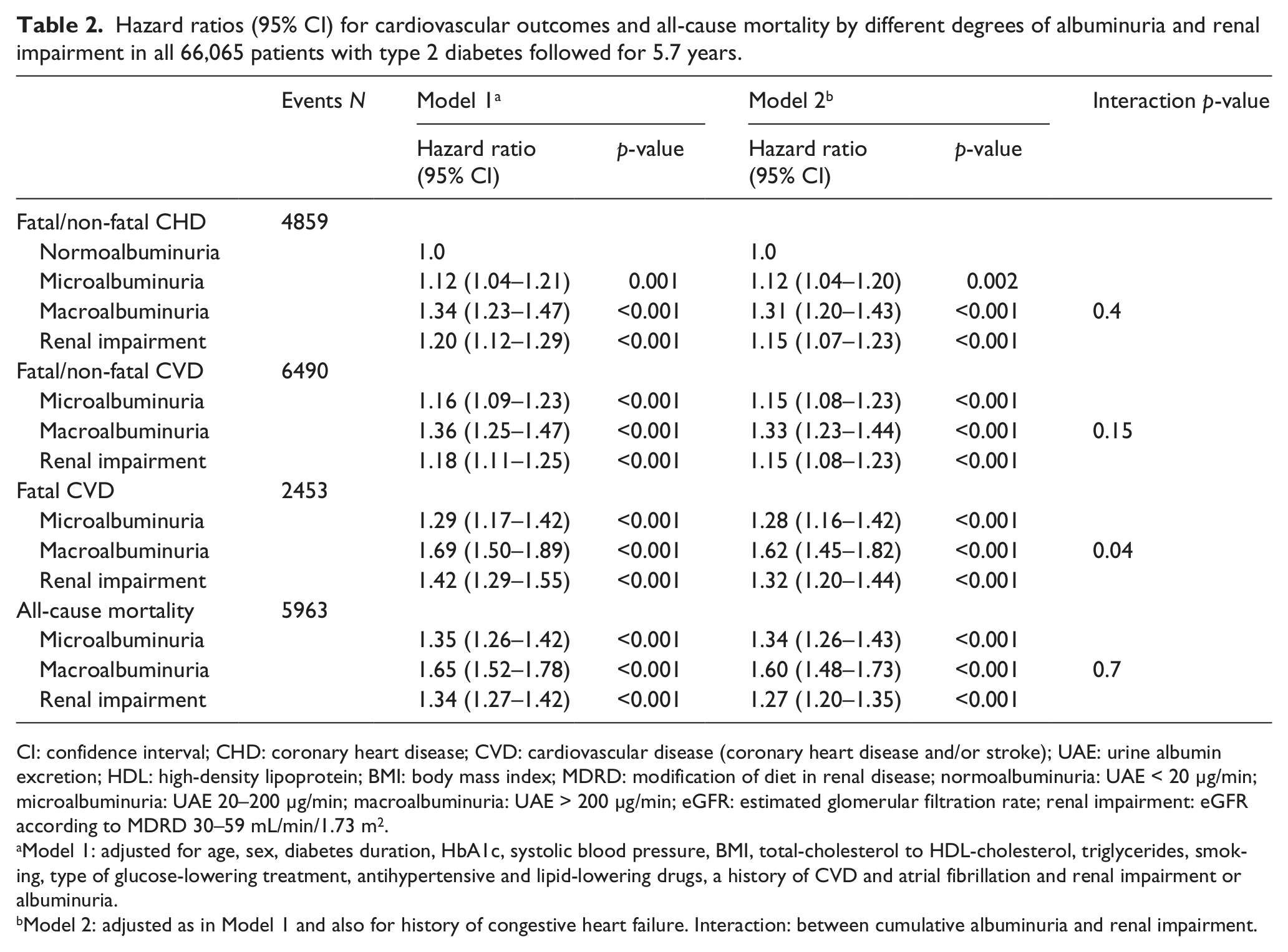
CI: confidence interval; CHD: coronary heart disease; CVD: cardiovascular disease (coronary heart disease and/or stroke); UAE: urine albumin excretion; HDL: high-density lipoprotein; BMI: body mass index; MDRD: modification of diet in renal disease; normoalbuminuria: UAE < 20 µg/min; microalbuminuria: UAE 20–200 µg/min; macroalbuminuria: UAE > 200 µg/min; eGFR: estimated glomerular filtration rate; renal impairment: eGFR according to MDRD 30–59 mL/min/1.73 m2.
Model 1: adjusted for age, sex, diabetes duration, HbA1c, systolic blood pressure, BMI, total-cholesterol to HDL-cholesterol, triglycerides, smoking, type of glucose-lowering treatment, antihypertensive and lipid-lowering drugs, a history of CVD and atrial fibrillation and renal impairment or albuminuria.
Model 2: adjusted as in Model 1 and also for history of congestive heart failure. Interaction: between cumulative albuminuria and renal impairment.
Hazard ratios for cardiovascular outcomes and combinations of albuminuria (normoalbuminuria vs albuminuria) and renal impairment (eGFR ≥60 vs 30–59 mL/min/1.73 m2) and different outcomes in all patients (N = 66,065).
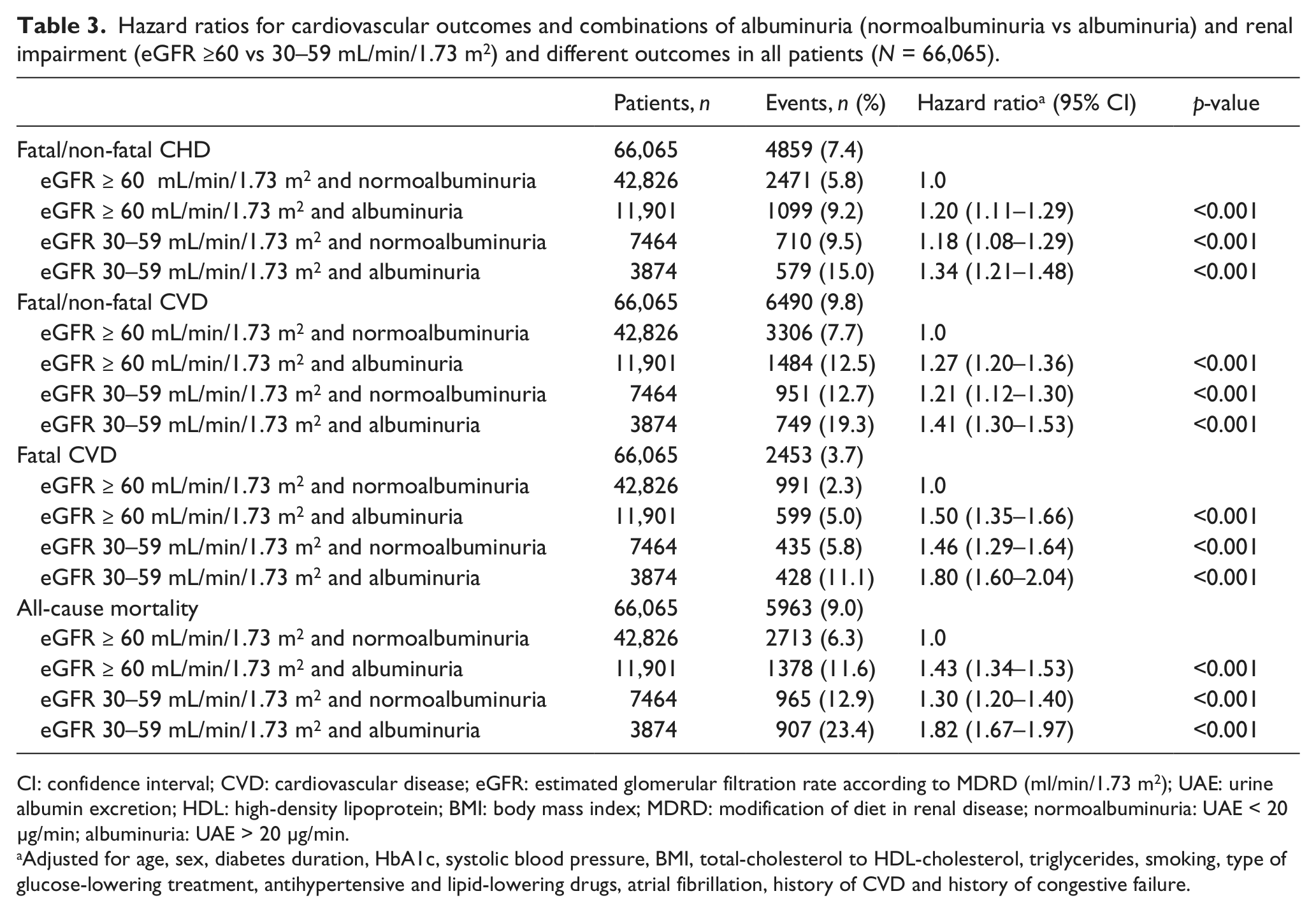
CI: confidence interval; CVD: cardiovascular disease; eGFR: estimated glomerular filtration rate according to MDRD (ml/min/1.73 m2); UAE: urine albumin excretion; HDL: high-density lipoprotein; BMI: body mass index; MDRD: modification of diet in renal disease; normoalbuminuria: UAE < 20 µg/min; albuminuria: UAE > 20 µg/min.
Adjusted for age, sex, diabetes duration, HbA1c, systolic blood pressure, BMI, total-cholesterol to HDL-cholesterol, triglycerides, smoking, type of glucose-lowering treatment, antihypertensive and lipid-lowering drugs, atrial fibrillation, history of CVD and history of congestive failure.
Hazard ratios for fatal/non-fatal CVD with various risk factors in patients with albuminuria (micro- or macroalbuminuria) only, or in patients with renal impairment (eGFR ≥ 60 vs 30–59 mL/min/1.73 m2) only.
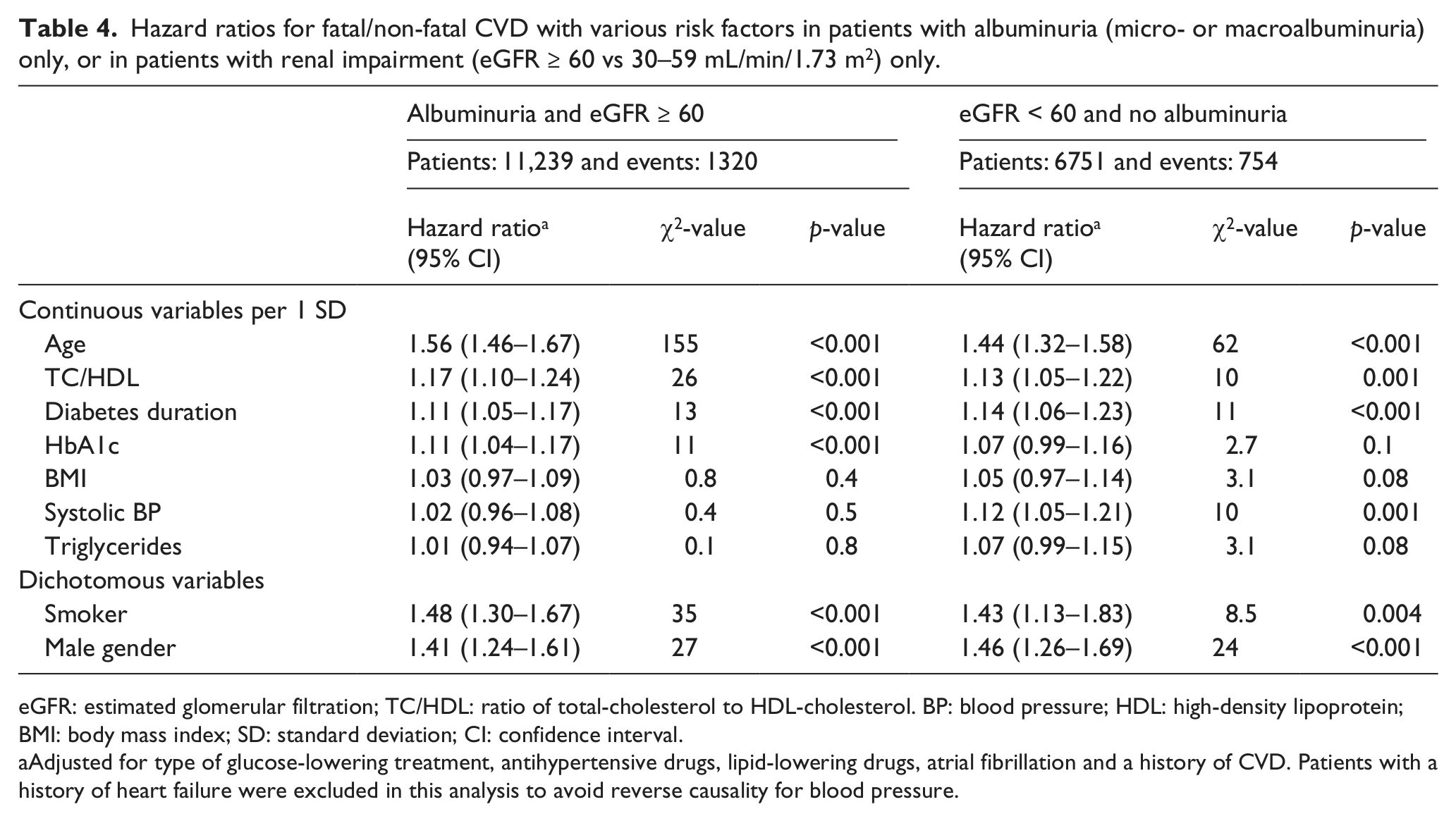
eGFR: estimated glomerular filtration; TC/HDL: ratio of total-cholesterol to HDL-cholesterol. BP: blood pressure; HDL: high-density lipoprotein; BMI: body mass index; SD: standard deviation; CI: confidence interval.
Adjusted for type of glucose-lowering treatment, antihypertensive drugs, lipid-lowering drugs, atrial fibrillation and a history of CVD. Patients with a history of heart failure were excluded in this analysis to avoid reverse causality for blood pressure.
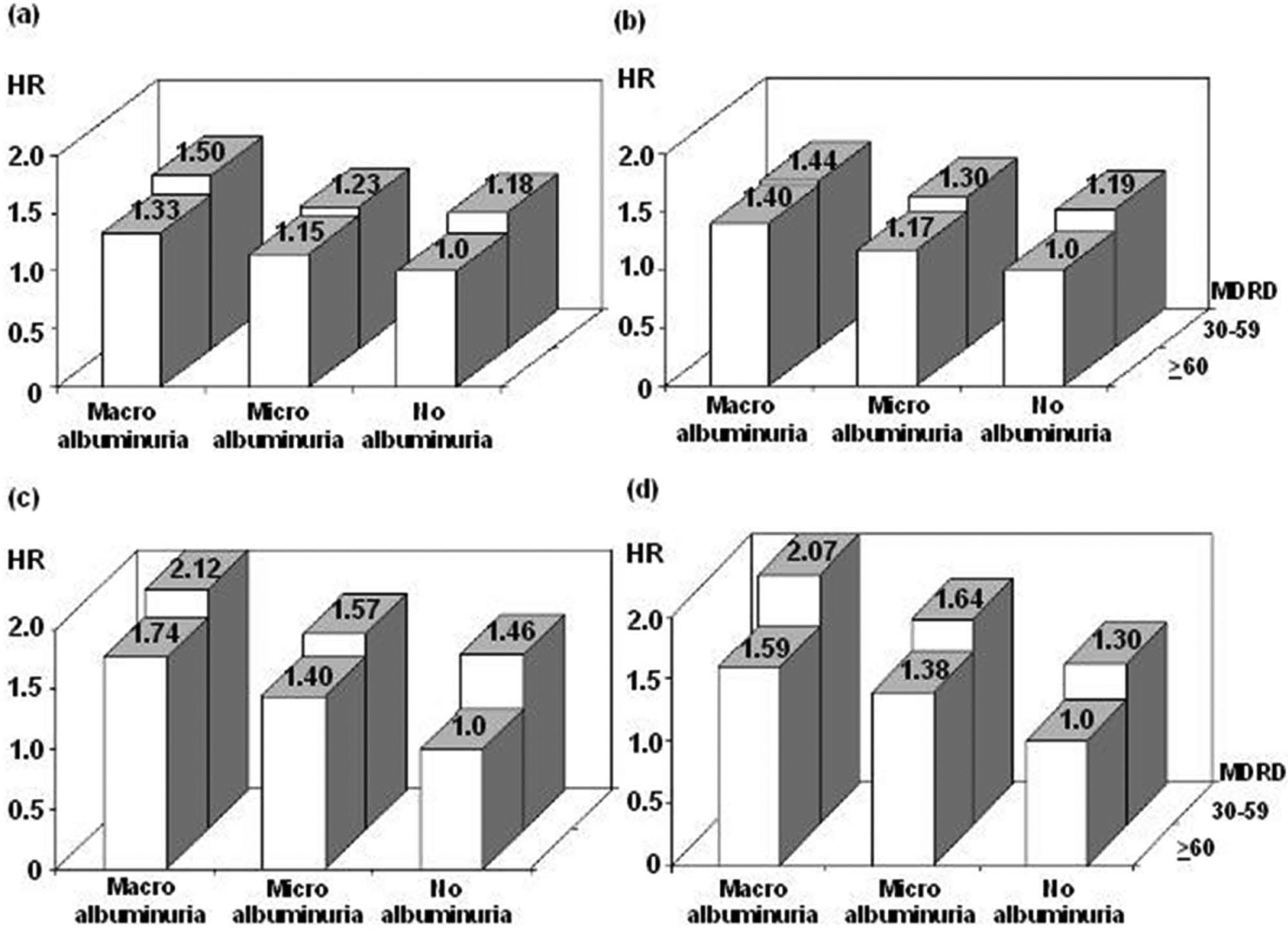
(a–d) HR for cardiovascular outcomes, all-cause mortality and combinations of albuminuria and renal impairment, adjusted for age, sex, diabetes duration, HbA1c, systolic blood pressure, BMI, ratio of total-cholesterol to HDL-cholesterol, triglycerides, smoking, type of glucose-lowering treatment, antihypertensive and lipid-lowering drugs, atrial fibrillation, a history of CVD and a history of congestive failure: (a) Fatal/non-fatal CHD; (b) Fatal/non-fatal CVD; (c) Fatal CVD and (d) all-cause mortality.
All statistical analyses were performed with SAS version 9.3 (SAS Institute, USA). A two-sided p-value < 0.05 was considered statistically significant.
Results
Clinical and biochemical characteristics at baseline
Clinical and biochemical characteristics at baseline are displayed in Table 1. The mean age of all patients (N = 66,065, 59% men) was 64 years with mean diabetes duration of 7 years. The mean eGFR was 79 mL/min/1.73 m2, and 17% had renal impairment (eGFR < 60 mL/min/1.73 m2) and 24% any degree of albuminuria, that is, micro- or macroalbuminuria. The mean HbA1c (IFCC) was 54 mmol/mol and the mean blood pressure 140/78 mmHg.
Patients with albuminuria at baseline (n = 15,775) were significantly older, had longer diabetes duration, lower renal function, worse glycaemic control, higher systolic blood pressure despite a larger proportion of antihypertensive treatment, lower total- and HDL-cholesterol but a higher total- to HDL-cholesterol ratio, higher triglycerides (influenced by a larger proportion of lipid-lowering treatment) and they were more often men, smokers and had a history of CVD and CHF compared to those without albuminuria at baseline. Three quarters (75%) of the patients with albuminuria had normal renal function.
Patients with renal impairment (n = 11,338) at baseline were significantly older compared with patients with normal renal function, and had a longer diabetes duration; worse glycaemic control; higher systolic but lower diastolic blood pressure, that is, a higher pulse pressure, in spite of more antihypertensive treatment; similar cholesterol levels; higher triglycerides, but were more often on lipid-lowering treatment; and they were more often women, non-smokers and had a history of CVD and CHF at baseline. A total of 67% of patients with renal impairment were normoalbuminuric, that is, had no micro- or macroalbuminuria.
Independent effects of albuminuria or renal impairment on risk for CV outcomes
A total of 6490 (9.8%) patients experienced a CV event (CHD or stroke), of which 2453 (3.7%) were fatal (Table 2). In addition, 3510 (5.3%) patients died from other non-CV events. The all-cause mortality rate was 1.6%/year.
Increasing graded levels of albuminuria (normo- vs micro- and macroalbuminuria, respectively) at baseline were associated with significantly increasing risks (increasing HRs) of CV events and all-cause mortality after adjustments for renal impairment and covariates, as shown in Table 2, model 1. A change from one clinical stage of albuminuria to the next (i.e. from normo- to micro- or from micro- to macroalbuminuria) was associated with a higher risk for all CV outcomes (fatal or non-fatal CHD, fatal or non-fatal CVD and fatal CVD) and all-cause mortality. Similarly, the risk for each CV outcome and all-cause mortality increased with the presence of renal impairment, but to a lesser extent, when adjusted for albuminuria and the covariates as illustrated in Figure 1.
When adjusting for co-existing CHF at baseline (Table 2, model 2), increasing levels of albuminuria at baseline were still associated with highly significantly (p < 0.001) increased risks of CV outcomes and all-cause mortality, although the risk when having macroalbuminuria exceeded the risks for normo- and microalbuminuria. This increase in risk was somewhat smaller than when adjusting with model 1. The presence of renal impairment at baseline was also associated with a significantly increased risk for fatal/non-fatal CHD or CVD, fatal CVD and all-cause mortality in model 2. Taken together, macroalbuminuria renders the largest increase in risk for CV outcomes and all-cause mortality. The effects of albuminuria and renal impairment were found to be additive for the outcomes when interaction was analysed with maximum likelihood estimation (not shown).
Effects of combinations of albuminuria and renal impairment on risk for CV outcomes
Table 3 shows combinations of normoalbuminuria, albuminuria and renal impairment as risk factors for CV outcomes. The presence of any degree of albuminuria inflicts increased risks for all CV outcomes and all-cause mortality of similar degree irrespective of the presence or absence of renal impairment, as displayed in Table 3. Albuminuria and renal impairment are both independent risk factors for development of CV events, albuminuria being the strongest one and relevant at all levels of renal impairment. In patients with normoalbuminuria, a reduction of renal function dramatically increases the risk of fatal CVD outcomes and all-cause mortality.
Effects of individual CV risk factors on risk for CV outcomes in two different subgroups of patients
Table 4 displays the effect of individual CV risk factors on the risk for CV outcomes in patients with normal renal function and albuminuria and patients with normoalbuminuric renal impairment. In both patient groups, age and male gender have a strong influence upon the risk for CV outcomes. Glycaemic control (high HbA1c), hyperlipidaemia and smoking, but not systolic blood pressure, have important effects on risk for CV events in patients with albuminuria and normal renal function. Slightly in contrast, hyperlipidaemia, smoking and increased systolic blood pressure, but not glycaemic control (high HbA1c), have important effects on risk for CV events in patients with normoalbuminuric renal impairment.
Complementary analysis of eGFR according to the CKD-EPI equation
Supplementary Tables 1 and 2 demonstrate that the effects of eGFR 30–59 mL/min/1.73 m2 on risks for CV outcomes or all-cause mortality were similar to corresponding eGFR effects estimated using the MDRD equation. The HRs were generally the same or differed only slightly by the second decimal. The c statistic (discrimination measure) for CVD was 0.77 (95% CI: 0.75–0.79) with eGFR according to MDRD and 0.73 (95% CI: 0.71–0.75) with eGFR according to CKD-EPI, and for all-cause mortality 0.72 (95% CI: 0.70–0.74) MDRD and 0.66 (95% 0.63–0.68) CKD-EPI, respectively.
Discussion
This nationwide, population-based study in clinical practice confirms that albuminuria and renal impairment are independent and additive risk factors for development of CV outcomes and all-cause mortality in patients with T2D, as previously found in smaller community-based studies in various populations of different ethnicities,21–25 and clinical trials.11,12 We show that in a general T2D population with mild to moderate renal impairment, presence of albuminuria carries the majority of increase in risk for CV and all-cause mortality, when adjusting for traditional CV risk factors, and also for a history of CHF.
Systemic endothelial dysfunction and accelerated atherosclerosis are plausible mechanisms for the relationship between albuminuria and reduced renal function, and CV outcomes and all-cause mortality.26,27 Albuminuria has been suggested as a marker for microvascular changes of CHD in diabetes, that is, hyperglycaemia-induced coronary vessel disease, while macroangiopathic ischaemic heart disease is generally related to traditional CV risk factors, such as hypertension and dyslipidaemia.28–30 This is supported by our study as we demonstrate that albuminuria has the strongest effect on CVD risk, independently of traditional risk factors and the presence of mild to moderate renal impairment.
The independent effect of reduced renal function and the interaction between CKD and CVD has, in addition to the presence of traditional risk factors, been suggested to be related to non-traditional risk factors. 31 This is supported by the fact that the impact of CKD increases with declining renal function and is extremely high in patients with ESRD and dialysis treatment. 4 In our study, both albuminuria and moderately reduced renal function are independent risk factors for CV outcomes and all-cause mortality, albuminuria being the strongest risk factor.
In this study, we also show that individual CV risk factors may have different effects on risk for CV outcomes depending on renal status. Hyperlipidaemia, smoking and glycaemic control have important effects on risk for CV events in patients with albuminuria and normal renal function, but increased HbA1c had no effect on this risk in patients with normoalbuminuric renal impairment. Instead, we show that systolic blood pressure has a strong effect on risk for CV events in patients with normoalbuminuric renal impairment but not in patients with normal renal function and albuminuria only. This finding may have implications on treatment of CV risk factors in these two subgroups of patients.
The mean eGFR in patients with renal impairment was 51 mL/min/1.73 m2, a moderate renal impairment, and the range of reduced renal function was narrow (30–59 mL/min/1.73 m2) since patients with eGFR < 30 mL/min/1.73 m2 were excluded from the study. The majority of patients with renal impairment were normoalbuminuric, thus representing a low-risk population for CV outcomes. 32
The major strengths of this cohort are the nationwide scale and large number of patients, many person-years of observation and numbers of events not constrained to specialized clinics. We included patients who received routine treatment according to national guidelines in both primary and secondary care, supporting high external validity and generalizability to the T2D population of our findings. A large number of relevant clinical characteristics, traditional CV risk factors, previous CV diseases and treatments for diabetes, hypertension and hyperlipidaemia were included as covariates in the Cox regression analyses, although unknown covariates of importance cannot be completely excluded. CV outcomes and all-cause mortality in this study were retrieved by linkage with the national cause of death and hospital discharge registers in Sweden. This has been found to be a reliable validated alternative to revised hospital discharge records and death certificates. 19
We excluded patients with severe renal impairment (eGFR < 30 mL/min/1.73 m2) since only few departments of nephrology report to the NDR. This is a limitation of the study since we are not able to analyse the predictive relationships in this high-risk population where severely reduced renal function is a major and independent risk factor. The narrow range of reduced renal function (30–59 mL/min/1.73 m2) also gives us little contrast between normal and impaired renal function in the analyses. In addition, a significantly larger proportion of women (58%) were found in patients with renal impairment as compared to those with albuminuria, indicating a potential effect of sex when estimating renal function GFR using MDRD. This gender distribution may be of importance for the risk of new CV events in patients with renal impairment since women in general have a lower CV risk. 1
Other possible limitations and weaknesses in this study are the use of reported laboratory values from local laboratories and that eGFR was calculated at baseline using the one reported plasma creatinine value available; however, the observations were highly statistically significant. Laboratory methods to analyse creatinine and absence or presence of albuminuria may vary between centres and laboratories over time, and reported data from participating centres may vary slightly in accuracy and precision. The effects of within-individual variation for plasma creatinine and the knowledge that eGFR may be imprecise on an individual level. However, the possible effects of these limitations would generate an underestimation rather than overestimation of the true risk. With reported clinical staging of albuminuria at baseline as the only available variable for urinary albumin excretion and without quantitative measurements available, we were not able to assess trends over time or within categories.
In addition, no detailed data on medication with different subgroups of antihypertensive agents were available in the NDR at baseline, but as expected, patients with albuminuria or renal impairment at baseline had a larger proportion of antihypertensive treatment as compared to patients without. The role of renin–angiotensin system (RAS) blockade could not be evaluated in this study. These agents have well-known effects on both eGFR and degree of albuminuria, but we have recently described that 25% of normoalbuminuric patients with T2D and renal impairment have no ongoing treatment with RAS-blocking agents. 33 RAS-blocking agents also have a specific effect on outcomes, that is, CV event and mortality. Angiotensin converting enzyme (ACE)-inhibitors clearly improve morbidity and survival in non-uraemic patients with heart failure 34 and in patients with CKD. 3 Unfortunately, this could not be evaluated in this study.
Matsushita et al. recently published a large meta-analysis, where they compared GFR estimations according to the MDRD and CKD-EPI equations as predictors of risk of CV events and all-cause mortality. They found that the adjusted HRs for CV events and all-cause mortality using either the MDRD or the CKD-EPI equation for estimation of eGFR were equivalent but concluded that the CKD-EPI equation more accurately categorized the risk for mortality than the MDRD equation. 35 In our study (see Supplementary Tables 1 and 2), we found similar adjusted HRs for CV events and all-cause mortality using the MDRD or the CKD-EPI equation, but we emphasize that the direct estimation of the magnitude of HRs for these outcomes should be the relevant method to evaluate and compare the two eGFR-equations. In addition, discrimination based on the c statistic was actually slightly better with MDRD than CKD-EPI.
In conclusion, we confirm that albuminuria and renal impairment are independent risk factors for development of CV events in patients with T2D in clinical practice. Albuminuria is the strongest risk factor at all levels of renal impairment. Importantly, in patients with normoalbuminuria, a reduction in renal function is a risk factor for CV outcomes and all-cause mortality. Interestingly, hyperlipidaemia, smoking and glycaemic control (high HbA1c) had important effects on risk for CV events in patients with albuminuria and normal renal function, while no effect of increased HbA1c was found in patients with normoalbuminuric renal impairment. In contrast, systolic blood pressure has a strong effect in patients with normoalbuminuric renal impairment but not in patients with albuminuria only. These findings may have implications on treatment of CV risk factors in these two subgroups of patients.
Footnotes
Acknowledgements
We would like to thank the regional NDR coordinators, all participating nurses, physicians and other staff who have contributed to the NDR. Most of all, we would like to thank the patients who support the NDR, both individually and collectively through their patient organization, the Swedish Diabetes Federation. M.S. and J.C. researched the data. J.C. performed the statistical analyses. M.S. wrote the manuscript. All authors contributed to the discussion and reviewed the manuscript.
Declaration of conflicting interests
Results and views of the present study represent the authors and not necessarily any official views of the Medical Products Agency where two of the authors are employed (M.S. and B.Z.). The results presented in this article have not been published previously in whole or part. None of the authors have any conflict of interest to declare.
Funding
The Swedish Association of Local Authorities and Regions funds the NDR: Swedish Heart and Lung Foundation project 20100648.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
