Abstract
The Global Partnership for Effective Diabetes Management, established to provide practical guidance to improve patient outcomes in diabetes, has developed and modified recommendations to improve glycaemic control in type 2 diabetes. The Global Partnership advocates an individualized therapeutic approach and, as part of the process to customize therapy, has previously identified specific type 2 diabetes patient subgroups that require special consideration. This article builds on earlier publications, expanding the scope of practical guidance to include newly diagnosed individuals with complications and women with diabetes in pregnancy. Good glycaemic control remains the cornerstone of managing type 2 diabetes, and plays a vital role in preventing or delaying the onset and progression of diabetic complications. Individualizing therapeutic goals and treatments to meet glycaemic targets safely and without delay remains paramount, in addition to a wider programme of care to reduce cardiovascular risk factors and improve patient outcomes.
Introduction
The Global Partnership for Effective Diabetes Management,* established to provide practical guidance to improve patient outcomes in diabetes, has previously developed 10 key recommended steps to assist health-care professionals to ensure that patients achieve glycaemic targets (Table 1). 1 Following the initial proposal of the 10 steps to improve glycaemic control, results of large intervention trials have been published. Whereas trials have consistently reported a reduction in microvascular complications with intensive glycaemic control,2–6 a reduction in macrovascular complications with this approach remains the subject of debate. 7
Ten steps to get more type 2 diabetes patients to goal. 16

A main outcome of such a discussion is the appreciation that a ‘one-size-fits-all’ strategy for managing type 2 diabetes will not provide a great deal of treatment success. Such an approach does not take into consideration the heterogeneity of the patient population (i.e. phenotype, genotype, age, disease duration, complications status, susceptibility to medication adverse effects, education etc.), requiring that individualized management should be employed along with full consideration of cardiovascular (CV) risk factors in the context of multifactorial interventions. 7 Moreover, in the past decade, new pharmacological options have been made available for the treatment of type 2 diabetes after metformin failure, and more options are advanced in development such as sodium glucose co-transporter 2 (SGLT2) inhibitors.
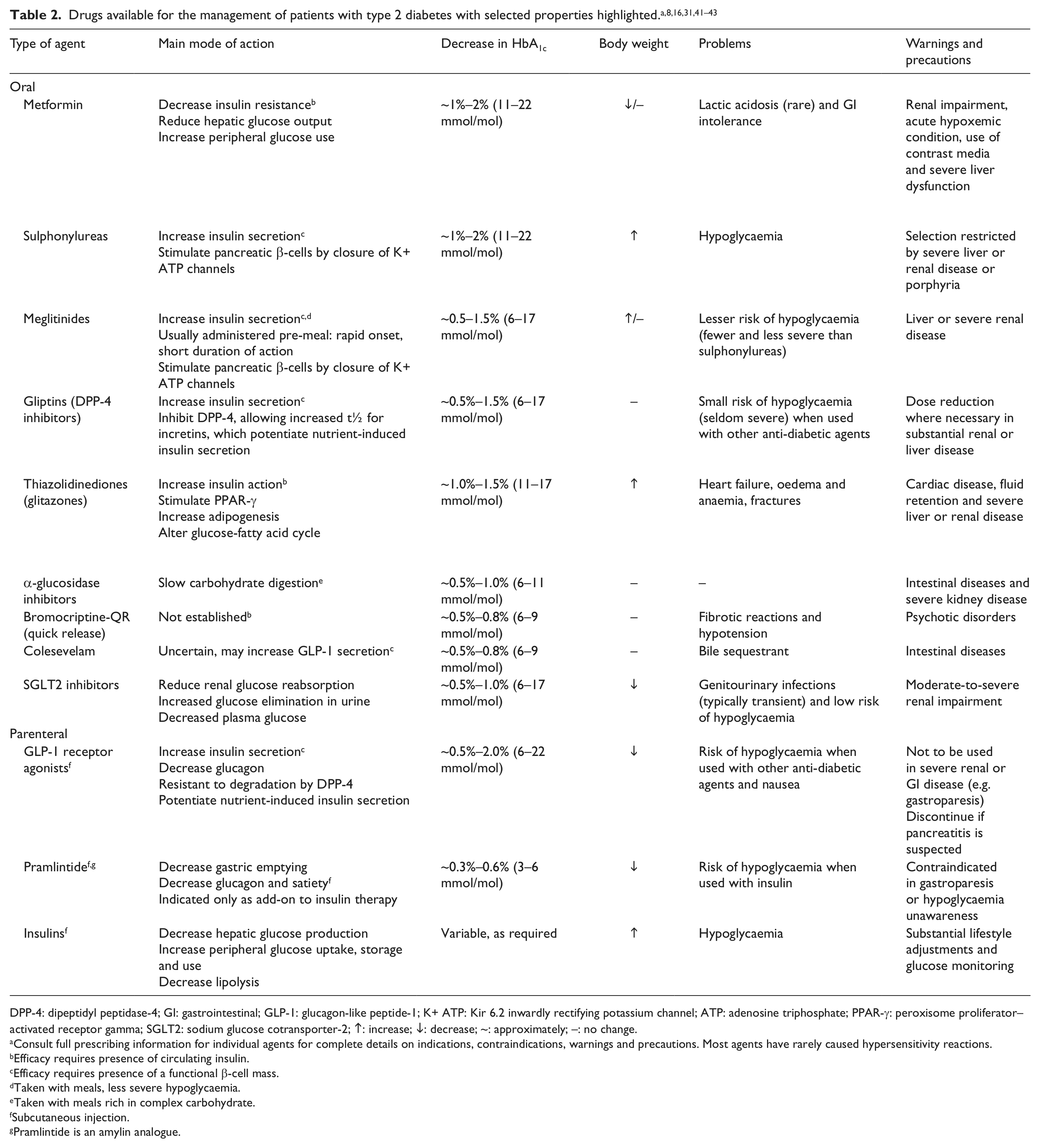
Added on top of metformin, the different pharmacological options are associated with a similar level of efficacy, 3 although they differentiate with respect to other features (effect on body weight, risk of hypoglycaemia, potential CV safety/protection etc. [Table 2]). Given these new options, the Global Partnership already advocated the need for an individualized therapeutic approach, and in an attempt to facilitate customized treatment, we identified specific type 2 diabetes patient subgroups that require special consideration and proposed practical advice for setting glycaemic targets for these groups. 7 A patient-centred approach has also been recommended by several organizations, such as the Canadian Diabetes Association and the American Association of Clinical Endocrinologists, and more recently, this approach was included in the updated joint position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). 8 Herein, we review the recent evidence to refine our recommendations on individualization of therapy in terms of glycaemic targets and choice of treatment.
DPP-4: dipeptidyl peptidase-4; GI: gastrointestinal; GLP-1: glucagon-like peptide-1; K+ ATP: Kir 6.2 inwardly rectifying potassium channel; ATP: adenosine triphosphate; PPAR-γ: peroxisome proliferator–activated receptor gamma; SGLT2: sodium glucose cotransporter-2; ↑: increase; ↓: decrease; ~: approximately; –: no change.
Consult full prescribing information for individual agents for complete details on indications, contraindications, warnings and precautions. Most agents have rarely caused hypersensitivity reactions.
Efficacy requires presence of circulating insulin.
Efficacy requires presence of a functional β-cell mass.
Taken with meals, less severe hypoglycaemia.
Taken with meals rich in complex carbohydrate.
Subcutaneous injection.
Pramlintide is an amylin analogue.
Defining glycaemic targets: the evidence
Hyperglycaemia defines diabetes, and glycaemic control is fundamental to management of the disease. 9 The UK Prospective Diabetes Study (UKPDS) established that early intensive glycaemic control in newly diagnosed type 2 diabetic patients is associated with significantly decreased rates of microvascular complications (retinopathy, neuropathy and nephropathy). 10 For macrovascular complications, a trend towards a reduced rate of myocardial infarctions (MIs) was observed in patients treated with intensive therapy, although this was not statistically significant. 10 Ten-year follow-up of patients in the UKPDS, during which glycaemic control was similar between groups, demonstrated that the benefit of intensive therapy (i.e. reduction in microvascular complications) 10 was maintained over the long term. 11 Analysis of the long-term UKPDS data also showed that those patients treated intensively to achieve glycaemic control received a benefit in terms of cardiovascular disease (CVD) and total mortality reduction. 11 These findings from the long-term follow-up led to the concept of a ‘legacy effect’ for both micro- and macrovascular complications resulting from early and intensive glycaemic control, 11 and highlight the importance of optimizing metabolic control as early as possible. 12
Other intervention studies (Action to Control Cardiovascular Risk in Diabetes [ACCORD], Action in Diabetes and Vascular Disease: Preterax and Diamicron Magnetic Resonance Controlled Evaluation [ADVANCE] and Veterans’ Administration Diabetes Trial [VADT]) that included patients with known CVD or multiple CV risk factors who had type 2 diabetes for a longer period than those enrolled in the UKPDS confirmed the benefits of intensive glycaemic control to reduce microvascular complications, predominantly nephropathy.13–15 However, results from these individual trials did not demonstrate a reduction in macrovascular complications with an intensive therapy regimen and, in fact, treating to a HbA1c target of <6% (<42 mmol/mol) (and achieving <6.5% [<48 mmol/mol]) in the ACCORD study was associated with an increase in mortality of 22%. 13
Given the neutral or negative outcomes on macrovascular complications in these studies despite the benefits on microvascular disease, the temptation to relax measures that reduce hyperglycaemia would seem understandable. It is vital, therefore, that these results are viewed in context, 16 and new analyses are considered that not only reinforce the need to reduce glycaemia effectively (in order to manage the risk of microvascular complications) but also clarify the benefit of early and intensive control of hyperglycaemia on macrovascular disease.
In ACCORD, ADVANCE and VADT, the overall CV mortality rate was actually substantially lower than originally expected, although this most likely reflects general improvements in type 2 diabetes management such as multifactorial therapeutic strategies. 7 The study duration of these trials (3.5–5.6 years) may have been too short for any significant benefits of tight glycaemic control on CV outcomes to become apparent (as opposed to the 10-year follow-up in the UKPDS). 16 Furthermore, the patients enrolled in the short-term studies had a history of poorly controlled long-standing diabetes together with either micro- or macrovascular complications,13–15 and were therefore at higher risk of negative outcomes than their counterparts in the UKPDS (newly diagnosed patients with no prior CV events). 7
A meta-analysis of the UKPDS, Prospective Pioglitazone Clinical Trial in Macrovascular Events (PROactive), ADVANCE, ACCORD and VADT studies found that intensive therapy achieving a modest 0.9% (10 mmol/mol) reduction in HbA1c was associated with a 17% reduction in non-fatal MI and a 15% reduction in coronary heart disease (CHD) compared with conventional therapy. 17 Further meta-analyses have also evaluated the effectiveness of good glycaemic control in preventing or delaying the onset of macrovascular complications and provide potential explanations for the lack of apparent benefits seen in landmark trials.18–20 A Cochrane review of intensive glycaemic control in type 2 diabetes (including 20 trials and ~30,000 patients) reported a significant 14% reduction in non-fatal MI with intensive glycaemic control, although no differences in all-cause or CV mortality were reported. 18 In this analysis, patients who had poor glycaemic control at baseline (higher fasting blood glucose or HbA1c) were less likely to benefit from intensive control in terms of all-cause mortality. 18 Pooled analyses have also suggested that the presence of CVD, 20 a higher degree of obesity and longer duration of diabetes 19 are factors associated with poorer outcomes in patients receiving intensive glycaemic control.
Retrospective analysis of ACCORD data suggested that episodes of severe hypoglycaemia may be associated with a high mortality rate, 21 possibly reflecting the intensive escalation of therapy in those patients failing to achieve the low HbA1c target of <6% (<42 mmol/mol). 16 However, the higher mortality rate in the intensive therapy arm was not related to the incidence of hypoglycaemia, 21 and a post hoc analysis later showed that the excess mortality actually occurred in patients who did not achieve their HbA1c target (or who had displayed substantial hyperglycaemia). 22 That is, intensive therapy in individuals with poorly controlled diabetes may, in fact, carry a higher risk of mortality than in patients with better controlled disease.
The ADVANCE investigators also pointed out that hypoglycaemia rather than being a trigger for CV events may represent a marker for vulnerability. 14 A post hoc analysis of data from ADVANCE reported that reducing HbA1c to less than 7% (53 mmol/mol) was not associated with an added benefit in terms of CV risk reduction but also showed that lowering HbA1c levels even further did not appear to be harmful. Moreover, the analysis found no evidence of interaction between severe hypoglycaemia, the assigned glucose-lowering treatment and mortality, 23 although the relationship between hypoglycaemia and MI remains unresolved.
As outlined here, new interpretations of data from large-scale clinical trials and assessment of meta-analyses and retrospective studies have provided the Global Partnership with the opportunity to further update and revise suggestions for the management of type 2 diabetes. In the following sections, we re-examine and expand upon the original sub-categories of type 2 diabetes patients for whom specific recommendations are necessary, with the aim of defining more closely the glycaemic targets and choice of treatments to achieve these goals.
Tailoring treatment to type 2 diabetes patients: glycaemic targets and individualized therapy
Definitions for patient types have been described in an earlier publication by the Global Partnership. 7 We stated that early and effective intervention is likely to confer the greatest benefits, 7 particularly bearing in mind the ‘legacy effect’ of early glycaemic control demonstrated in the UKPDS 11 and outcomes reported in meta-analyses.18–20 As part of management, lifestyle adjustment and a defined target HbA1c are required, with rapid treatment assessment should targets fail to be met – primarily to avoid periods of inadequately controlled hyperglycaemia.1,7 Table 3 contains an at-a-glance summary of treatment recommendations for the patient subtypes discussed in more detail below.
Treatment recommendations for subtypes of type 2 diabetes patients.

GLP-1: glucagon-like peptide-1; DPP-4: dipeptidyl peptidase-4; SGLT2: sodium glucose cotransporter-2; SUs: sulphonylureas; CVD: cardiovascular disease; CV: cardiovascular; FPG: fasting plasma glucose.
In addition to lifestyle measures (exercise, diet).
Or FPG < 7 mmol/L.
Newly diagnosed individuals with type 2 diabetes but no complications
Newly diagnosed overweight or obese adults
In overweight and obese individuals with type 2 diabetes, diet and exercise are essential components of therapy, as are agents that address the underlying pathophysiology of diabetes, including treatments directed at β-cell dysfunction 24 (owing to impaired β-cell function at diagnosis that declines thereafter) 24 and insulin resistance. 7 An additional consideration in this patient population is the impact of pharmacological therapies on weight, with sulphonylureas (SUs), thiazolidinediones (TZDs) and insulin associated with weight gain, 8 whereas weight neutral agents or those associated with weight loss 8 would appear more preferable. 24
Practical guidance for newly diagnosed overweight or obese individuals
Implement a multifactorial management plan, including diet and lifestyle intervention to achieve a healthy weight, and appropriate treatment to combat CV risk factors such as dyslipidaemia and hypertension.
Individuals with newly diagnosed type 2 diabetes and without CVD should aim for an HbA1c that is as near to normal as possible (<6.5% [<48 mmol/mol]), without causing hypoglycaemia or marked weight gain (metformin, glucagon-like peptide-1 [GLP-1] receptor agonists and dipeptidyl peptidase-4 [DPP-4] inhibitors).
HbA1c should be monitored on a 3-month basis, and a combination of glucose-lowering therapies should be considered if glycaemic targets are not met with one agent.
Among patients with mild-to-moderate hyperglycaemia (HbA1c <7.5% [<58 mmol/mol]), opt for agents that are not associated with hypoglycaemia (metformin) and address the underlying pathophysiology of diabetes (GLP-1 receptor agonists and DPP-4 inhibitors). In individuals on a diet rich in complex carbohydrates, an α-glucosidase inhibitor may offer benefit. To avoid weight gain or facilitate weight loss, the SGLT2 inhibitors may also be useful when available.
For patients with an HbA1c between 7.5% (58 mmol/mol) and 8.9% (74 mmol/mol), provide more intensive use of the agents in the category above, possibly considering combination therapy for patients in the upper end of the range but avoiding the risk of hypoglycaemia for patients in the lower end.
In patients with HbA1c approximately 9% (75 mmol/mol) or above, consider initiating combination therapy (e.g. metformin plus DPP-4 inhibitor), or temporarily insulin to reduce glucose toxicity, together with an increase in glucose monitoring. Use of a GLP-1 receptor agonist could also be an option, although experience is limited at the current time.
Newly diagnosed lean adults
In lean patients, the degree of β-cell dysfunction requires major consideration and may be more marked than in obese/overweight individuals.7,25 Insulin resistance in lean patients is generally comparatively lower, 25 risk factors for CVD are often less obvious and the risk of developing macrovascular complications may not be as great compared with their obese/overweight counterparts.7,26 It may be appropriate to measure antibodies, for example, for glutamic acid decarboxylase (GAD), in an effort to exclude or confirm latent autoimmune diabetes in adults (LADA).
Practical guidance for newly diagnosed lean adults
Individuals with newly diagnosed type 2 diabetes and no complications should aim for an HbA1c of 6.5%–7% (48–53 mmol/mol) (even in patients with mild hyperglycaemia) while avoiding hypoglycaemia or marked weight gain.
Agents that are not associated with hypoglycaemia or weight gain are preferable (DPP-4 inhibitors, GLP-1 receptor agonists and metformin), although SUs and meglitinides may be considered in suitable patients.
Where possible, choose anti-hyperglycaemic agents that support β-cell function (DPP-4 inhibitors and GLP-1 receptor agonists).
In patients with HbA1c > 9% (>75 mmol/mol), consider initiating combination therapy of oral agents or temporarily, insulin to reduce glucose toxicity, together with an increase in glucose monitoring to reduce the risk of hypoglycaemia.
Tailor the wider management plan to account for the less marked CV risk profile compared with their obese/overweight counterparts.
Reinforce the need for a healthy lifestyle.
Children and adolescents
The onset of type 2 diabetes at an early age points to a glycaemic legacy if the disease is uncontrolled for long periods of time. 7 Many of these patients are overweight at diagnosis and also have co-morbidities at a relatively early age (such as hypertension, dyslipidaemia and microalbuminuria). 27 Although lifestyle interventions do not appear as effective as in adults, an education programme involving family and friends can assist in improving glycaemic control. 7 An HbA1c target in the near-normal range of <7.0% (<53 mmol/mol) is appropriate in these patients. 27
Choice of pharmacotherapy should aim to preserve β-cell function and improve insulin sensitivity. At present, metformin is the only oral anti-hyperglycaemic agent approved for use in children and adolescents. 27 However, recent data from the Treatment Options for Type 2 Diabetes and Adolescents and Youth (TODAY) study showed that 50% of children and adolescents failed to maintain durable glycaemic control with metformin monotherapy and combination therapy or insulin was often necessary within a few years of diagnosis. 28
Practical guidance for newly diagnosed children/adolescents
Measure antibodies to GAD to exclude or confirm type 1 diabetes.
Implement a multidisciplinary approach to optimize care among young people with type 2 diabetes, emphasizing education of family and friends on the importance of lifestyle choices.
Agents that address insulin resistance (metformin) and potentially help to preserve β-cell function (DPP-4 inhibitors and GLP-1 receptor agonists) may be preferable given the disease will progress over many decades.
Accepting that clinical classification of diabetes is difficult in overweight or obese young people, consider insulin as a first-line treatment.
Long-term safety is of paramount importance in children and adolescents with type 2 diabetes – an HbA1c target as near to normal as possible, while minimizing the risk of hypoglycaemia is appropriate (HbA1c 6.5%–7% [48–53 mmol/mol]).
Newly diagnosed individuals with complications (micro- or macrovascular)
Type 2 diabetes can remain undiagnosed until an advanced stage of the disease, and micro- and macrovascular complications are often already present when a new patient is diagnosed. Indeed, up to 40% of adults with either diagnosed or undiagnosed type 2 diabetes have a degree of chronic kidney disease. 29 Moreover, the risk of CVD increases proportionally as glomerular filtration rate declines in this patient group. 29
Treatment choice should be based on the types of complications identified, with some medications contraindicated in specific patient groups. Metformin, for example, is contraindicated in patients with renal insufficiency (estimated glomerular filtration rate [eGFR] <50 mL/min) and heart failure. However, because of the accepted CV benefit of metformin, both National Institute for Health and Clinical Excellence (NICE) 30 and the ADA/EASD 8 position statements suggest that metformin should be avoided for eGFR < 30 mL/min, while dosage should be reduced for eGFR of 30–45 mL/min. TZDs should not be used in patients with moderate-to-severe heart failure, and risk of fracture should be taken into consideration. 3 In patients with renal insufficiency, medications that are eliminated by the kidneys, such as many SUs, increase the risk of hypoglycaemia and should be used with caution. 29 Dose of DPP-4 inhibitors should be reduced for eGFR < 60 mL/min with the exception of linagliptin, which has less than 5% renal elimination. More caution should be used for GLP-1 receptor agonists in cases of moderate renal failure. Careful control of blood pressure is recommended in patients with diabetic nephropathy and retinopathy.
Practical guidelines for newly diagnosed individuals with micro- or macrovascular complications
An HbA1c target of <7% (<53 mmol/mol) is recommended in those patients in whom the reduction in blood glucose can be achieved safely and without delay.
Multiple therapies may be required at an early stage to maintain control of HbA1c and reduce the risk of complication progression, and caution should be exercised with respect to drug interactions.
Less stringent glycaemic targets of HbA1c 7%–7.5% (53–58 mmol/mol) (or even up to 8.5% [69 mmol/mol]) may be indicated where co-morbidities impede use of, or full titration of, some therapies.
Ensure regular glucose monitoring owing to the risk of hypoglycaemia with some medications.
Remember that neuropathy and nephropathy are risk factors for severe hypoglycaemia.
Choice of medication is dependent on the nature and severity of complications, with several therapies contraindicated in renal insufficiency, hepatic impairment or CVD. 31
Individuals with a history of inadequate glycaemic control (lean or obese)
A key component of eliminating poor glycaemic control is determining the factors behind insufficient management, such as poor adherence with medications, delayed use of combination therapy or insulin to provide suitable control and failure to adequately assess β-cell function.1,7
No complications
In patients with a history of inadequate glycaemic control, the risk of a legacy effect on development of complications emphasizes the need to establish HbA1c levels to near normal (HbA1c 6.5%–7% [48–53 mmol/mol]) while also ensuring patient safety. 7 Long disease duration moderates the effects of rapid glycaemic control in this patient group, 19 and efforts to reduce blood glucose should not be delayed.
Practical guidelines for individuals with a history of inadequate glycaemic control but no complications
Address the underlying causes for poor glycaemic control, including adherence, inadequate therapy and failure to manage the underlying pathophysiology.
Aim for a target HbA1c near to normal levels (HbA1c 6.5%–7% [48–53 mmol/mol]) in those patients in whom the reduction in blood glucose can be achieved safely and without delay. Avoid hypoglycaemia and marked weight gain.
Do not delay the therapeutic decision and explain to the patient the potential risk of long-term unacceptable hyperglycaemia.
Consider a change of therapy (including insulin) or combination of medications to achieve control based on the patient’s response to initial treatment.
Implement additional education programmes to assist patients in taking a more active role in the management of their disease.
Individuals with a history of CVD
Results from the ADVANCE, ACCORD and VADT studies suggested that very intensive glycaemic control regimens may be inappropriate for some patients with more advanced diabetes who also have macrovascular complications.13–15 As discussed in more detail earlier, several potential explanations have been proposed to account for the outcomes observed in these studies,18–20 and meta-analyses have also suggested that intensive glycaemic regimens have positive effects if targeted at specific patient types.17,18 Improved glycaemic control may still have microvascular benefits in these patients if achieved safely, that is, without hypoglycaemia.
Avoiding hypoglycaemia in this group of patients is particularly important, and less strict glycaemic targets (HbA1c 7%–7.5% [53–58 mmol/mol] or even up to 8.5% [69 mmol/mol]) with a gradual decrease in HbA1c levels may be more appropriate, or, indeed, imposed by limitations in the choice of therapies. 24 In the Bypass Angioplasty Revascularization Investigation 2 Diabetes (BARI 2D) study, a regimen based on insulin sensitization (e.g. with metformin) had a similar effect on survival and incidence of major CV events as an insulin provision regimen. 32 However, the frequency of severe hypoglycaemic episodes in these type 2 diabetes patients with CHD was decreased in those patients receiving insulin-sensitizing agents. 32 In addition, insulin sensitization was associated with less weight gain, higher high-density lipoprotein cholesterol levels and improved glycaemic control. 32
Practical guidelines for individuals with a history of inadequate glycaemic control and CVD
A target of <7% (<53 mmol/mol) is desirable in those patients in whom the reduction in blood glucose can be achieved safely but less strict HbA1c targets (HbA1c 7–7.5% [53–58 mmol/mol] or even up to 8.5% [69 mmol/mol]) are recommended where safety is a concern.
Aim to avoid hypoglycaemic episodes (increased monitoring of glucose is also advised), marked weight gain and the burden of extra therapies with limited efficacy (treatment changes should be based on the patient’s response to initial therapy).
Insulin-sensitizing agents (e.g. metformin) should be employed where appropriate unless complications contraindicate their use or drug interactions are an issue (owing to the high frequency of multiple therapies in this patient group).
Implement an intensified comprehensive CV risk factor (dyslipidaemia, hypertension) management plan.
Safety and potential cardioprotective properties of incretin-based therapy are currently under assessment in large intervention trials.
Individuals with a history of microvascular complications
Intensive glycaemic control regimens have been shown to reduce the risk of microvascular disease development in people with type 2 diabetes.2–6 Strict glycaemic control is warranted in the presence of microalbuminuria without elevated serum creatinine levels or in patients with early stage retinopathy, to reduce the rate of disease progression. 33
As noted for people with a history of CVD, however, the risk of hypoglycaemia in patients with a history of microvascular complications has to be balanced against the benefits in terms of delaying progression of microvascular complications. 24 Moreover, type 2 diabetes patients with autonomic neuropathy are predisposed to hypoglycaemia unawareness (in addition to increased cardiac mortality), and therapy must therefore be implemented with appropriate care. 33 Options for managing inadequate glycaemic control in this patient group are further limited by contraindications, with several medications not suitable in patients with renal impairment or renal insufficiency.3,29
Practical guidance for individuals with a history of inadequate glycaemic control and microvascular complications
Individualized glycaemic targets and therapy are of paramount importance in this patient group.
Set initial glycaemic targets as close to normal as possible (HbA1c 6.5%–7% [48–53 mmol/mol]) without causing hypoglycaemia in those patients whose microvascular complications are at an early stage.
Be aware of contraindications, especially in individuals with late-stage microvascular complications, and be vigilant for drug interactions in patients receiving multiple medications.
Consider less stringent control where the risk of hypoglycaemia or the limited therapeutic choices are problematic.
Individuals at risk of hypoglycaemia
As discussed in an earlier publication, individuals with an increased susceptibility to hypoglycaemia have usually experienced a hypoglycaemic episode on previous occasions, have a wide fluctuation in their daily glucose levels and may already have impaired creatinine clearance. 7 In type 2 diabetes, the risk for hypoglycaemia is further enhanced with microvascular complications, such as autonomic neuropathy 33 and renal impairment, 29 and occurs more frequently in patients receiving therapy to achieve intensive glycaemic control.10,13–15
Hypoglycaemia can also exacerbate CVD risk among individuals who already have CV risk factors through increasing or fluctuating heart rate and rhythm, raising systolic blood pressure and increasing myocardial contractility. 34 This increase in cardiac stress has the potential to contribute to destabilization of existing atherosclerotic plaques in older patients with type 2 diabetes who have CHD.35,36
Practical guidance for individuals at risk of hypoglycaemia
Identify patients at risk of severe hypoglycaemia. Risks include older age, diabetic neuropathy, impaired kidney function, lower body weight, long duration of diabetes and prior episodes of severe hypoglycaemia.
Individualize glycaemic targets according to the level of hypoglycaemia risk and potential implications for that patient, for example, less stringent targets (HbA1c 7%–7.5% [53–58 mmol/mol] or even up to 8.5% [69 mmol/mol]) for patients at higher risk (increased glucose monitoring is also advised), or those who live alone or are elderly.
Agents that are not associated with an increased risk of hypoglycaemia are preferable, for example, agents that reduce insulin resistance or potentiate (but not initiate) glucose-dependent insulin release, or agents that are not contraindicated in significant renal insufficiency or liver dysfunction.
Educate patients on the symptoms of hypoglycaemia and the associated risks, in order to increase awareness and responsiveness to these symptoms.
If conventional insulin therapy (mixed formulations) is used, encourage regular eating patterns to lessen the risk of hypoglycaemia.
Counsel patients on regular self-monitoring of blood glucose where appropriate.
Type 2 diabetes and pregnancy
Women with pre-existing diabetes during pregnancy
Pre-existing diabetes is associated with numerous adverse outcomes for mother and child during and following pregnancy, 37 including a higher frequency of Caesarean section, hypertension, fetal congenital anomaly, pre-term birth and spontaneous abortion. 37 Pregnancy can also exacerbate the effects of diabetes on renal function and retinopathy,38,39 and has the potential to accelerate the development of these complications. 37 The occurrence of these adverse outcomes is related to the level of glycaemic control achieved during pregnancy. 37
Women planning or continuing pregnancy are recommended to substitute their oral anti-diabetic agent(s) with insulin, although oral anti-diabetic agents can still be considered on an individual basis. 35 Metformin may be an appropriate agent for women who refuse insulin therapy or who require an additional therapeutic agent. 35
Practical guidance for women with pre-existing diabetes during pregnancy
In pre-conception and antenatal care, management of maternal glucose concentrations should remain a priority.
Prior to conception, women with diabetes should receive multidisciplinary care, including an assessment of diabetes complications and advice/education on glycaemic control, diet (including folic acid and multivitamin supplementation), maternal diabetes complications and fetal risks.
Aim for normal glycaemia (HbA1c 6.5%–7% [48–53 mmol/mol], or fasting plasma glucose [FPG] <7 mmol/L) to minimize the risk of perinatal complications (use of fructosamine to assess glycaemic control may be helpful in pregnancy).
Insulin is recommended for glycaemic control during pregnancy; however, oral agents (e.g. metformin) may be considered on an individual basis.
Women with gestational diabetes
Gestational diabetes is associated with a number of complications and an increased long-term risk of type 2 diabetes and CVD in both mother and infant. 37 Balancing the benefits of therapy with potential adverse effects is essential: lifestyle intervention and insulin have been demonstrated to improve perinatal outcomes (although insulin is associated with hypoglycaemia and weight gain), 40 and metformin can be an option in those women who prefer not to use insulin. 40
Practical guidance for women with gestational diabetes
Women with gestational diabetes should receive multidisciplinary care, including advice/education on glycaemic control, diet (including folic acid and multivitamin supplementation), maternal diabetes complications and fetal risks.
Aim for normal glycaemia (HbA1c 6.5%–7% [48–53 mmol/mol], or FPG <7 mmol/L) to minimize the risk of perinatal complications.
Insulin is recommended for glycaemic control during pregnancy; however, oral agents such as metformin may be considered on an individual basis.
Additional considerations affecting glycaemic control
Setting and achieving glycaemic targets also relies on a number of additional patient characteristics or factors. 31 These include age and life expectancy, patient preference, degree of social support, adherence to therapy and ethnicity. 31 Frequently, poor adherence to medications is a significant cause of inadequate glycaemic control in type 2 diabetes. 31 Non-adherence can result from a series of contributing factors, including lack of confidence in effectiveness of therapy and fear of side effects such as hypoglycaemia or weight gain.
In addition, the absence of apparent symptoms in type 2 diabetes may have an impact on patient perception with respect to the seriousness of their condition and the need to take medication. 31 Complex dosing regimens, polypharmacy, difficulties with language and socio-economic issues may also impact adherence.6,31 Measures to maximize adherence through enhanced communication and education of patients on the benefits of treatment are vital. As part of this approach, physicians should aim to minimize the number of medications and doses per week, including the use of fixed-dose combinations where appropriate. 31
Conclusion
Good glycaemic control remains the cornerstone of managing type 2 diabetes and plays a vital role in preventing or delaying the onset and progression of diabetic complications. Treating a heterogeneous population of patients as a homogenous group is impractical and does not take into account the variety of factors that affect patients with type 2 diabetes. As such, the ‘10 steps to get more type 2 diabetes patients to goal’, developed by the Global Partnership, continue to offer a valuable way of ensuring patients achieve and maintain their glycaemic targets. This article builds on earlier publications, expanding the scope of practical guidance to account for newly diagnosed individuals with complications and for women with diabetes in pregnancy. Individualizing therapeutic goals and treatments to meet those goals remains paramount, as is the recognition that early and intensive glycaemic control to achieve near-normal HbA1c targets, while essential in some patient subgroups, is not applicable to every patient with type 2 diabetes.
Key messages
Good glycaemic control achieved safely and without delay remains the cornerstone of managing type 2 diabetes.
An individualized therapeutic approach rather than a ‘one-size-fits-all’ strategy is essential to ensure maximum success.
It is important to recognize that early and intensive glycaemic control to achieve near-normal HbA1c targets is not applicable to every patient with type 2 diabetes.
Footnotes
Acknowledgements
The authors acknowledge the editorial assistance of Dr Sheelagh Farrow, Dr Paola Accalai and Duncan Pennington, International Medical Press, in the development of this article.
Conflict of interest statement
Pablo Aschner is a member of the Global Partnership for Effective Diabetes Management and has received honoraria from GlaxoSmithKline for his participation in board meetings and other related scientific activities. He has also participated in advisory panels and provided ad hoc consultancy to AstraZeneca, Boehringer Ingelheim, Eli Lilly and Company, Merck Sharp & Dohme, Novartis and Sanofi-Aventis.
Clifford Bailey has received research support from AstraZeneca, Sanofi-Aventis and has received presentation support from, has participated in advisory panels and provided ad hoc consultancy to, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Merck, Novo Nordisk and Takeda.
Larry Blonde has received research support from Eli Lilly and Company, Novo Nordisk and Sanofi and has received speaker/consultant honoraria from Amylin Pharmaceuticals, Johnson & Johnson – Janssen, Johnson & Johnson Diabetes Institute, Merck & Co; Inc, Pfizer Inc, Novo Nordisk, Sanofi and Santarus. Dr Blonde’s late spouse’s estate contains shares of Pfizer.
Stefano Del Prato has received research support from Boehringer Ingelheim Pharmaceuticals, Merck Sharp & Dohme, Sanofi-Aventis, and has acted as a consultant for Bristol-Myers Squibb, GlaxoSmithKline, Novartis, Sanofi-Aventis, Takeda and Eli Lilly.
Anne-Marie Felton has participated in Bayer and GlaxoSmith-Kline advisory boards and symposia sponsored by Bayer, Novo Nordisk and Abbott.
Ramon Gomis has received fees for serving as a speaker, a consultant and an advisory board member for GlaxoSmithKline, Merck Sharp & Dohme, Novartis and Sanofi-Aventis.
Ed Horton has received fees for consulting and advisory boards from Abbott, AstraZeneca, Bristol-Myers Squibb, Daiichi-Sankyo, Janssen, Medtronic, Merck, Metabasis, Novartis, Roche, Sanofi-Aventis, Takeda and Tethys Bioscience, and is on the speakers bureau for Merck and has received research support from Amylin Pharmaceuticals and Eli Lilly.
Linong Ji has received fees for lectures and consulting from Abbott, AstraZeneca, Bristol-Myers Squibb, Merck, Metabasis, Novartis, Eli Lilly, Roche, Sanofi-Aventis and Takeda.
James LaSalle has received fees as an advisor and speaker from AstraZeneca, Bristol-Myers Squibb, Boehringer Ingelheim, GlaxoSmithKline, Eli Lilly and Novo Nordisk, and received research support from AstraZeneca, Bristol-Myers Squibb and Novo Nordisk.
Lawrence Leiter has received research funding and has acted as a consultant to Amgen, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen, Merck, Novartis, Novo Nordisk, Roche, Sanofi-Aventis, Servier and Takeda.
Stephan Matthaei has received research support from Sanofi-Aventis and Novo Nordisk, and honoraria for advisory boards and/or speaker engagements from AstraZeneca, Bayer, Bristol-Myers Squibb, Disetronic, Eli Lilly, GlaxoSmithKline, LifeScan, Merck, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, Roche, Sanofi-Aventis and Takeda.
Marg McGill has acted as a consultant for GlaxoSmithKline.
Neil Munro has received fees for serving as a speaker, a consultant or an advisory board member for Eli Lilly, Novo Nordisk, GlaxoSmithKline, Takeda, Bristol-Myers Squibb, AstraZeneca, Merck Sharp & Dohme, LifeScan, Medtronic, Novartis, Pfizer, Servier, Daiichi-Sankyo and Roche.
Richard Nesto is on the speakers bureau of, and is a consultant to, GlaxoSmithKline and Sanofi-Aventis.
Jay Skyler has received research funding from Bayhill Therapeutics, Halozyme Therapeutics, Osiris Therapeutics and Mesoblast; is on the Board of Directors of DexCom Corporation, Moerae Matrix and Paean Therapeutics and has received fees for serving as a consultant or an advisory board member from BD Technologies, Cebix, Gilead, Ideal Life, Merck, Sanofi Diabetes, Takeda, and Valeritas.
Paul Zimmet is currently involved in advisory work with Novartis, NovoNordisk and OM Pharma.
Funding
The Global Partnership for Effective Diabetes Management, including the development of this article, is supported by an unrestricted educational grant from Bristol-Myers Squibb and AstraZeneca LP, and also Novartis Pharma AG.
*
Global Partnership for Effective Diabetes Management Members: Pablo Aschner, Javeriana University School of Medicine, Bogota, Colombia; Clifford Bailey, Aston University, Birmingham, UK; Lawrence Blonde, Ochsner Medical Center, New Orleans, LA, USA; Stefano Del Prato, University of Pisa, Pisa, Italy (Chair); Anne-Marie Felton, Federation of European Nurses in Diabetes, London, UK; Ramon Gomis, Hospital Clinic, Barcelona, Spain; Edward Horton, Joslin Diabetes Center, Boston, MA, USA; Linong Ji, Peking University Peoples Hospital, Beijing, China; James LaSalle, Medical Arts Research Collaborative, Excelsior Springs, MO, USA; Lawrence A. Leiter, Keenan Research Center in the Li Ka Shing Knowledge Institute, St Michael’s Hospital, and Division of Endocrinology and Metabolism, University of Toronto, Toronto, Ontario, Canada; Stephan Matthaei, Diabetes Centre Quakenbrück, Quakenbrück, Germany; Marg McGill, Diabetes Centre, Royal Prince Alfred Hospital, Sydney, Australia; Neil Munro, Beta Cell Diabetes Centre, Chelsea and Westminster Hospital, London, UK; Richard Nesto, Lahey Clinic, Burlington, MA, USA; Jay Skyler, University of Miami, FL, USA; Paul Zimmet, Baker IDI Heart and Diabetes Institute, Caulfield, Australia.