Abstract
Aims/Hypothesis:
Hygiene hypothesis has been shown valid in respect of allergies and asthma but has not been, however, sufficiently studied in relation to type 1 diabetes mellitus. This study investigates the effect of childhood farm environment on the prevalence of type 1 diabetes mellitus in Finnish children and young adults.
Methods:
Data come from a questionnaire study carried out in 2007 (N = 5805). Prevalence of type 1 diabetes mellitus was analysed according to the type of the rural exposure below the age of 18 years. Associations were estimated and tested using Bayesian methods.
Results:
Consistent suggestive evidence of the inverse association of childhood’s farm environment with the occurrence of type 1 diabetes mellitus was shown, even though conclusive results were not reached.
Conclusion/Interpretation:
Exposure to a broad variety of non-pathogenic environmental microorganisms during childhood might have a protective effect on type 1 diabetes mellitus. Results clearly suggest that further research is well grounded with larger samples.
Introduction
Prevalence of type 1 diabetes mellitus (Type 1 DM) in Finland is the highest in the world. Incidence today is quadruple compared to the 1950s, and morbidity has increased rather steadily over the decades by some 3% per year. 1 Along with the greatly increased Type 1 DM rates, the societies have changed and depopulation of the countryside has become a common phenomenon in highly industrialized countries. The trend has been very alike in all major diabetes countries, such as Finland, Sweden, United States and Great Britain. Connection between the changed living environment and significantly increased Type 1 DM rates is striking and can only be explained by the combined effect of genetic susceptibility and altered environmental factors, as has been proved in several studies. 2,3
It has continuously been reported that there is an inverse relationship between the childhood living environment and prevalence of allergies and asthma. Studies show that incidences of allergies and asthma are lower among children who have been living in a farm environment than among children raised in an urban environment. 4 –6 This association has been explained by the positive effect of the child’s exposure to a broader variety of commensal, non-pathogenic microorganisms in early childhood. It has also been suggested that a smaller diversity of microbiota might play a role in prevalence of Type 1 DM. 7 Valid and reliable evidence on the association of living environment and occurrence of Type 1 DM is, however, still lacking. An extensive register-based and questionnaire-based study is under way; thus, this article may be seen as an introduction to the topic.
Material and methods
Data consist of a follow-up questionnaire for an allergy study conducted in 1995 among Finnish first-year university students. Data on lifetime prevalence of some chronic diseases such as Type 1 DM were collected by a postal questionnaire in 2007 from 5805 responders. The original study design by Kilpeläinen et al. 4 was approved by the Ethics Committee of the University of Turku.
The age of the responders at the time of the questionnaire varied from 30 to 37 years. Diagnoses of the self-reported Type 1 DM onset cases in the questionnaire were made over 10 years ago. Cases diagnosed less than 5 years ago or 5–10 years ago have not been included to reduce a possibility of type 2 diabetes incidences as different diabetes types were not specified in the questionnaire. Living environment was assessed until the age of 18 years, but the duration of the farm living was not considered in this study.
We analysed the prevalence of Type 1 DM according to the type of the rural exposure below the age of 18 years, and we classified subjects into four categories: subjects lived solely in an urban environment without any exposure to animal or plant farms (reference group), subjects with only plant exposure, subjects with only animal exposure and subjects with both plant and animal exposures.
To evaluate the effect of living in rural areas in more detail, the areas were categorized into seven different variables according to the production sector of the farm: grain, plants, cattle, pigs, poultry, horses and forestry with urban living as a referent category.
Variables were cross-tabulated, and proportional shares and prevalence ratios (PRs) were counted. Because all participants were aged between 30 and 37 years at the time of the questionnaire and no gender differences were observed in associations, we report crude PRs. Because Type 1 DM is a very rare event (<0.1%) in our cross-sectional sample, we estimated PRs with posterior median (odds ratios are interpreted as PRs) and report 95% highest posterior density credible intervals (HPD-CI) using Bayesian methods with beta prior distribution (0.5, 0.5). We prefer to use equally tailed prior as suggested by Brown et al. 8 to ensure better coverage rates for the HPD-CI of PRs.
Results
Data comprised 5805 questionnaires from former Finnish university students, of which 64% from women and 36% from men. From this population, 849 (15%) responders had lived in a farm environment at some stage before the age of 18 and 4956 (85%) had only lived in an urban environment. The prevalence of Type 1 DM was 3 (0.4%) among persons who had lived in a farm environment below the age of 18 and 35 (0.7%) among the responders who had only lived in an urban area at the same age. The PR of Type 1 DM in living in a farm environment versus urban living environment was 0.53 (95% confidence interval (CI) = 0.13–1.43, p = 0.23), taking all seven rural production sectors into account. There was no difference between male and female subjects in the effects of farm environment versus urban environment regarding prevalence of Type 1 DM.
When comparing only animal exposure (N = 307, 1 Type 1 DM case) to urban environment (N = 4950, 35 Type 1 DM cases), the PR of Type 1 DM was 0.54 (95% CI = 0.05–2.24, p = 0.46). When comparing both plant and animal exposures (N =217, 0 Type 1 DM cases) to urban environment, the PR of Type 1 DM was 0.15 (95% CI = 0.00–1.63, p = 0.16). When comparing only plant exposure (N = 202, 2 Type 1 DM cases) versus urban environment, the PR was 1.54 (95% CI = 0.28–4.87, p = 0.56). Results of more detailed analysis on prevalence of Type 1 DM in different production sectors are shown in Table 1.
Number of subjects, PRs, 95% CI and Bayesian p-values of Type 1 DM in a sample of children who have lived either in an urban or in a rural environment at some age below 18 years, stratified by production sector.
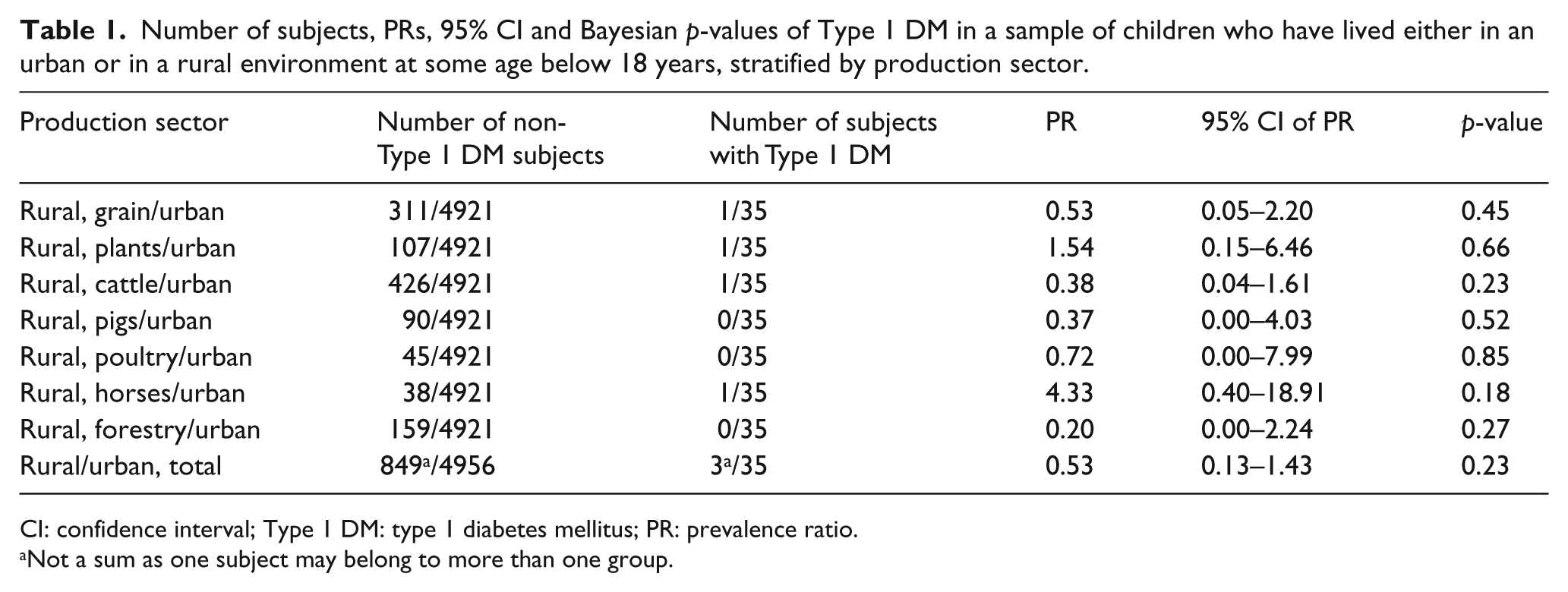
CI: confidence interval; Type 1 DM: type 1 diabetes mellitus; PR: prevalence ratio.
Not a sum as one subject may belong to more than one group.
Discussion
Even though we could not reach statistically significant results due to a small number of exposed cases and non-cases in our cross-sectional study design, the findings consistently show that the living environment may have a protective effect measured by the prevalence of Type 1 DM in children and adolescents. Particularly, farm animals seemed to play an important role since all comparisons between urban living environment and different animal farms produced PRs where protective effect was manifested. Correspondingly, when comparing subjects who had only lived in a plant farm with subjects who had only lived in an urban environment, living in an urban environment was more propitious (PR = 1.54). Thus, it appears that plant farm environments did not provide protective effects. This strengthens the above-mentioned suggestion that it is particularly the exposure to farm animals that might create beneficial circumstances regarding the occurrence of Type 1 DM. However, since previous research also suggest opposite findings, where incidence of Type 1 DM was higher in rural, low-population density areas, 9,10 further research with significantly larger datasets is encouraged. It should also be noted that there has been some indications on Type 1 DM possibly being a zoonosis, where an infectious agent from an animal could trigger the onset of the disease. 11 These counterarguments, among other research lines in the field, point out the need for large-scale studies to reliably and conclusively identify the environmental trigger or triggers involved in the onset of Type 1 DM.
Regarding our study, we do not currently know exactly how the broader microbial exposure creates the protective effect on the development of some autoimmune conditions. We know that bacteria and fungi activate the innate immune system through pattern recognition receptors, such as the toll-like receptors. Activation of these receptors in children exposed to farm environment has been demonstrated in previous studies. 12 In this study, we will not exploit the possible mechanisms of the effect of broader microbial exposure but instead put our focus in attesting the association of wider exposure to environmental microorganisms and the prevalence of Type 1 DM. Later, the challenge is to identify the specific microbes that are responsible for the possible protective effect of the farm environment.
In this study, the problem was the cross-sectional design and the relative rareness of the disease in focus, together with the relative rarity of children and young adults living in a farm environment in Finland. The number of responders who had lived in a farm environment was rather small, which also led to a rather small sample of exposed cases, leading to the loss of statistical power to explore the association. This is partially due to the fact that higher education rate among the urban population is generally greater than among the population living in the country side. 13 It is obvious that in future research, larger sample sizes are required.
In conclusion, the PRs obtained in this study support the general idea of hygiene hypothesis, where we assume that early exposure to infections and to a greater variety of environmental microorganisms protects from certain autoimmune diseases. Studies investigating the association between Type 1 DM and a broader environmental microbial exposure are very few in number, and the preliminary results obtained in this study entitle and enforce further research on this topic. If it would be conclusively proved that occurrence of Type 1 DM actually is, like asthma and allergies, to some extent dependent on the early exposure to immune modulating agents from the environment, it would greatly advantage the future research on the aetiology and treatment of Type 1 DM.
Footnotes
Authors’ contributions
M.J.K. and J.M.P. developed the study concept and supervised the research. M.L.K. prepared the database. S.M.M.H. and J.M.P. performed the statistical analyses of the data and with M.J.K. interpreted the findings. S.M.M.H. wrote the first draft of the report; all the co-authors contributed to writing and agreed the final draft.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by a grant (SH/6340) from the Yrjö Jahnsson Foundation, Finland.
