Abstract
Angiotensin II receptor blockers (ARB) are often co-administered with a calcium channel blocker (CCB) for treating hypertension. In this open-label randomised study, untreated diabetic hypertensive patients were randomised to receive either olmesartan 20 mg/day or candesartan 8 mg/day for 12 weeks. Patients with blood pressure exceeding 130/80 mm Hg received add-on 16 mg/day azelnidipine to ongoing olmesartan (OL group) or 5 mg/day amlodipine to ongoing candesartan (CA group) for 24 weeks. Home-measured and clinic-measured blood pressure decreased in both groups. Fasting blood glucose, haemoglobin A1c (HbA1c) and urinary albumin levels decreased significantly in the OL group but not in the CA group. In conclusion, this study revealed clinically relevant differences between two combinations of an ARB+CCB in diabetic hypertensive patients. Olmesartan and azelnidipine had a more persistent early morning antihypertensive effect and produced greater decreases in heart rate, fasting blood glucose and HbA1c (National Glycohemoglobin Standardization Program values) levels, and microalbuminuria than did candesartan and amlodipine.
Introduction
Hypertension is an important risk factor for cardiovascular and cerebrovascular diseases. The goal of antihypertensive treatment is to prevent the onset of these diseases by controlling blood pressure within appropriate levels. Hypertensive patients with diabetes mellitus are at even higher risk for cardiac and cerebrovascular disorders, as well as renal disorders. Therefore, more aggressive control of blood glucose and blood pressure is needed and the Japanese Society of Hypertension guidelines for the management of hypertension (JHS (2009)) have proposed target blood pressure values of 130/80 mm Hg. 1 However, many patients are unable to achieve such low blood pressure values. Therefore, for the management of blood pressure it is important that clinicians are aware of the rate of achievement of these antihypertensive targets and that they determine why many patients experience inadequate blood pressure lowering. In fact, in terms of antihypertensive drugs, blood pressure is often poorly controlled with one antihypertensive drug alone and at least two antihypertensive drugs may be required in many patients to achieve blood pressure targets.
Inhibitory effects of antihypertensive drugs on cardiovascular and cerebral events have been reported in hypertensive patients complicated with diabetes mellitus and, considering the antihypertensive effects and the effect on glucose and lipid metabolism, renin–angiotensin (RA) inhibitors are considered as first-line drugs in these patients. If RA inhibitors show inadequate efficacy, calcium channel blockers (CCB), and sometimes thiazide diuretics, are often used as second-line drugs.1–3 It was recently reported in the large-scale ACCOMPLISH (Avoiding Cardiovascular events through COMbination therapy In Patients LIving with Systolic Hypertension) study that co-administration of a RA inhibitor and a CCB inhibited cardiovascular events in all nondiabetic patients, diabetic patients and chronic kidney disease patients, unlike the use of a RA inhibitor plus a diuretic.4,5 It was also found that the combination of a RA inhibitor and a CCB confers greater organ protection. Since antihypertensive treatment is generally conducted for a long time, the tolerability of and long-term adherence to the treatment regimen are also important factors when selecting antihypertensive drugs. 6 Angiotensin II receptor blockers (ARB) are often co-administered with a CCB in clinical practice because of their relatively high tolerability. It has been found that the binding force to the angiotensin II type 1 receptor differs among ARBs. 7 Moreover, some CCBs stimulate the sympathetic nervous system while others inhibit it, and azelnidipine was reported to show renal protective effects by reducing proteinuria.8–11 In Japan, combinations of ARBs and CCBs have been launched by various companies, but there are few reports comparing the clinical effects of different combinations of drugs. Therefore, the purpose of this study was to compare the effects of two combinations of an ARB and a CCB on early-morning blood pressure, heart rate, renal function and glucose/lipid metabolism in hypertensive patients with type 2 diabetes mellitus.
Methods
Patients
Untreated hypertensive patients complicated with type 2 diabetes mellitus (with or without insulin treatment) who were attending the outpatient clinic for diabetes mellitus at Sakaide City Hospital were invited to participate in this two-arm, open-label, randomised controlled study. The study protocol was approved by the Institutional Ethical Committee at Sakaide City Hospital. All of the patients were given an explanation of the study and their written consent for participation and for the use of their data was obtained before enrolment. We also explained to the patients that no personal information would be disclosed during publication of the results.
Patients meeting the following criteria were eligible for inclusion: (i) hypertension in the presence of type 2 diabetes mellitus and systolic pressure or diastolic pressure at medical examination exceeding 130 mm Hg or 80 mm Hg respectively; and (ii) no planned changes in antidiabetic therapy. Patients with secondary hypertension or Grade 3 hypertension were excluded from the study.
Patients meeting any of the following criteria were also excluded: (i) contraindication for any of the test drugs; (ii) uncontrolled diabetes mellitus; (iii) diabetic nephropathy after the late stage of overt nephropathy (because blood insulin clearance may decrease after the late stage of overt nephropathy and may mask glucose metabolic status); (iv) history of acute coronary syndrome or cerebrovascular disorders within one year of enrolment; (v) severe infection, before or after surgery, or serious trauma; (vi) history of hypersensitivity to the study drugs; (vii) pregnant women or women with the possibility of being pregnant; and (viii) other reasons for ineligibility as determined by the investigator.
Study design and treatments
The study design is shown in Figure 1. Three-hundred hypertensive patients complicated with type 2 diabetes mellitus were randomly allocated (150 patients/group) to receive either olmesartan medoxomil (olmesartan) 20 mg/day or candesartan cilexetil (candesartan) 8 mg/day for 12 weeks. Based on the JSH (2009) guidelines, 1 115 patients who did not achieve blood pressure less than 130/80 mm Hg with olmesartan were also given azelnidipine once daily after breakfast at a dose of 16 mg/day (OL group). Meanwhile, 121 patients in the candesartan group who did not reach the antihypertensive target added amlodipine 5 mg/day once daily after breakfast (CA group). The doses used were chosen based on the doses commonly used in Japan, and were expected to achieve similar hypotensive effects. None of the patients dropped out after starting CCB therapy. Antidiabetic drugs (including insulin) used at the start of the study were to be continued without any change in type or dosage during the study.
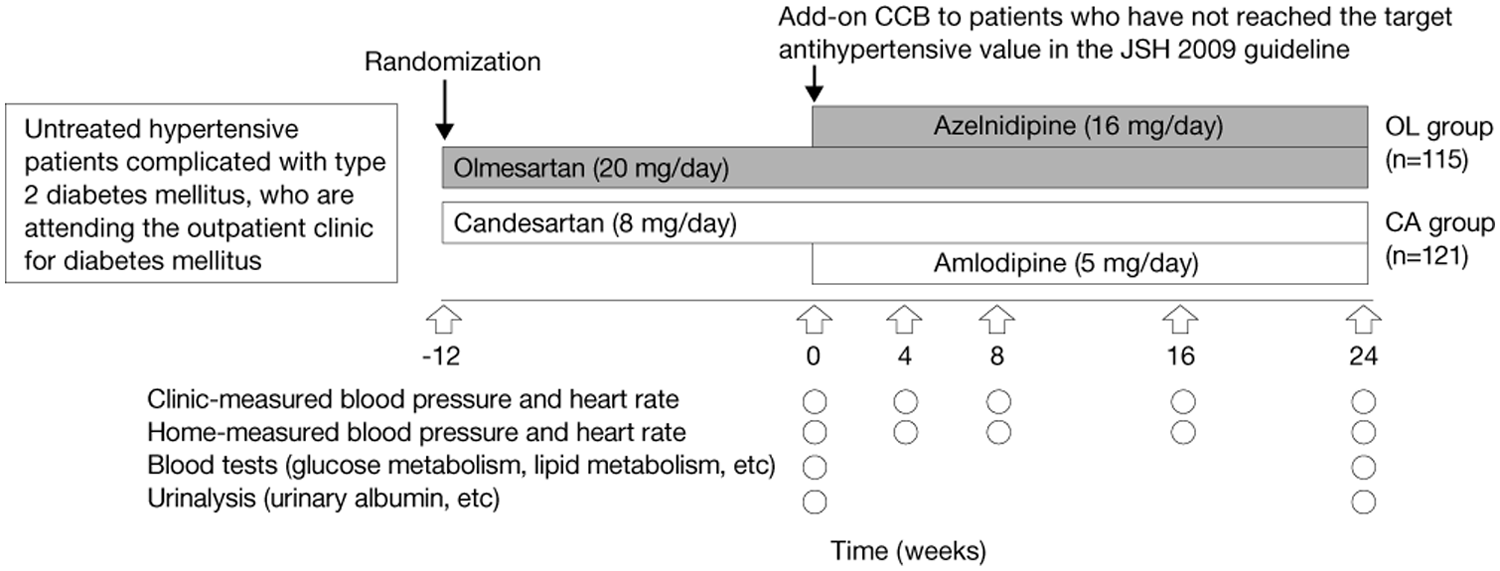
Study design.
In 236 patients in the two groups, blood pressure and heart rate were measured in the outpatient clinic and at home at weeks 4, 8, 16 and 24 after the start of combination therapy. Fasting blood glucose, haemoglobin A1c (HbA1c) (National Glycohemoglobin Standardization Program (NGSP)), low-density lipoprotein–cholesterol (LDL-C), high-density lipoprotein–cholesterol (HDL-C), triglyceride (TG), serum uric acid, blood urea nitrogen (BUN), serum creatinine, and serum electrolyte (Na, K and Cl) levels, urinary albumin levels and the estimated glomerular filtration rate (eGFR) were measured 24 weeks after starting combination therapy. The most common oral antidiabetic drugs used before starting the study were sulfonylureas (30%), biguanides (30%), α-glucosidase inhibitors (25%), dipeptidyl peptidase inhibitors (20%) and glinides (5%). Thirty-nine percent of patients were treated with insulin.
Blood pressure was measured in the clinic multiple times at one- or two-minute intervals with the patient in the resting and sitting position. The mean of two values differing by less than 5 mm Hg was recorded. Blood pressure was also measured at home, after waking in the morning, using a pressure measurement device for the upper arm. The patient was instructed to measure blood pressure while in the sitting position, with a 1–2-minute rest, after urination but before administration of hypotensive drugs, as recommended by the JSH. 1 Additional parameters were determined using blood and urine samples. Changes in blood pressure and heart rate measured in the clinic, early morning blood pressure and heart rate measured at home, urine albumin, eGFR, HbA1c (NGSP), fasting blood glucose, LDL-C, HDL-C and TG between the start and after 24 weeks of combination therapy were defined as endpoints in this study. HbA1c was measured by column chromatography at our institute. Urinary albumin levels were measured by a turbidimetric immunoassay (Shikoku Chuken Inc., Kagawa, Japan).
Values are expressed for all patients as means ± standard deviation (SD), except for the urinary albumin level, which is expressed as the mean ± standard error (SE). Unpaired t-tests and paired t-tests were used to determine the significance of differences between the two groups and within each group, respectively. The number of urinary albumin-positive patients was compared using the χ2-test. The level of significance was set at 5%. Intention-to-treat analytical procedures were applied. Based on data from previous studies,8,12 we assumed a difference in SBP of 4.8 mm Hg between the two groups. To achieve 90% power at a 5% significance level, and allowing for a 25% dropout rate, 150 patients were needed in each group.
Results
Comparing the clinical characteristics of 300 hypertensive patients complicated with diabetes mellitus, there were no differences between the two groups of patients (150 patients/group) randomised to either olmesartan 20 mg/day or candesartan 8 mg/day. Overall, 115 patients treated with olmesartan and 121 patients treated with candesartan did not achieve the antihypertensive goal within 12 weeks of treatment and received add-on CCB therapy. The characteristics of these patients before adding a CCB to ongoing ARB therapy (i.e. at week 0) are shown in Table 1. There were no significant differences between the OL and CA groups. Similar proportions of patients in both groups used insulin or oral antidiabetic drugs. There were no changes in antidiabetic therapy (type or dose) during the study.
Patient characteristics before starting combination therapy.
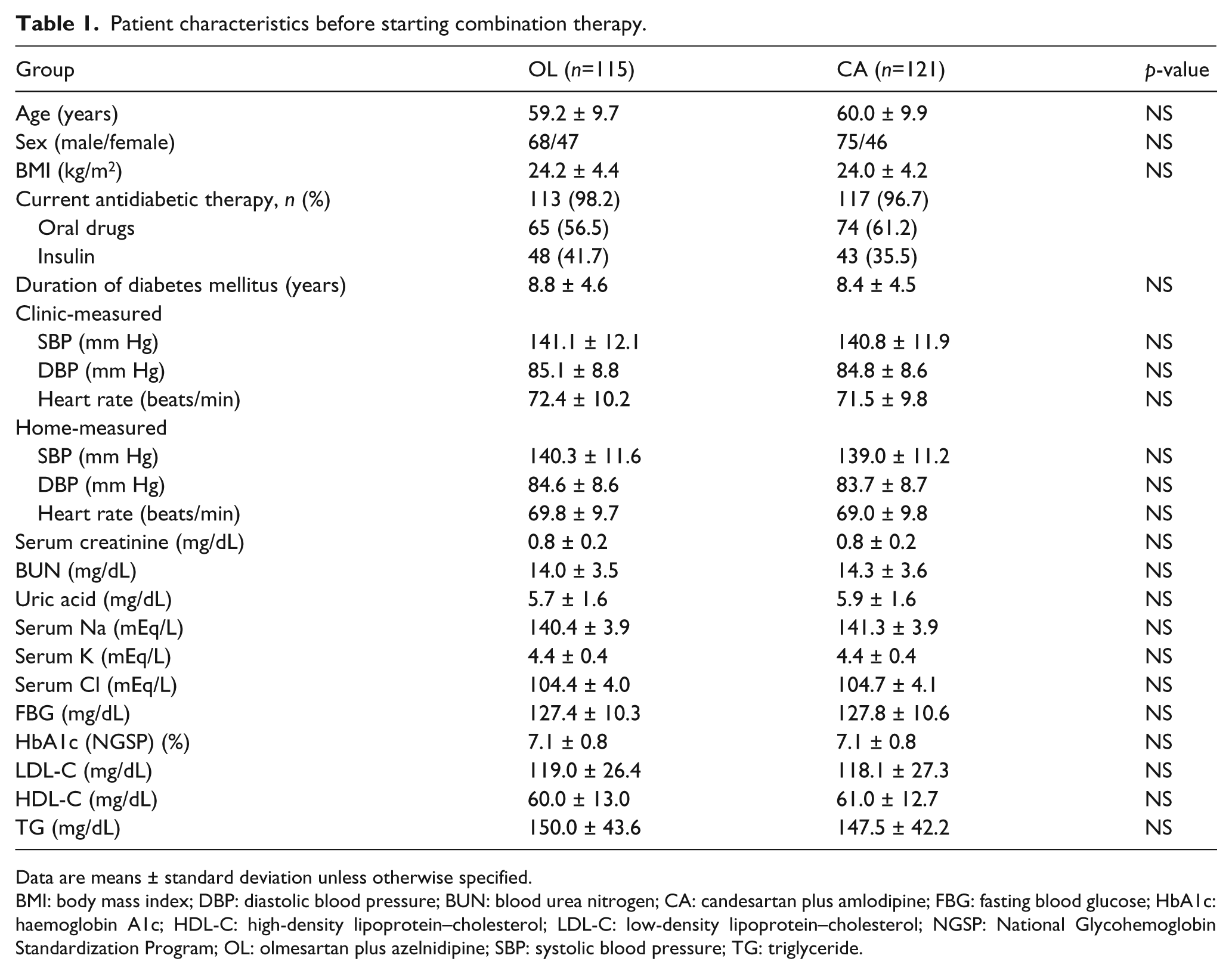
Data are means ± standard deviation unless otherwise specified.
BMI: body mass index; DBP: diastolic blood pressure; BUN: blood urea nitrogen; CA: candesartan plus amlodipine; FBG: fasting blood glucose; HbA1c: haemoglobin A1c; HDL-C: high-density lipoprotein–cholesterol; LDL-C: low-density lipoprotein–cholesterol; NGSP: National Glycohemoglobin Standardization Program; OL: olmesartan plus azelnidipine; SBP: systolic blood pressure; TG: triglyceride.
The changes in clinic-measured blood pressure and home-measured early morning blood pressure during the study period are shown in Figures 2 and 3. The changes in other parameters between weeks 0 and 24 of ARB and CCB combination therapy are shown in Figures 4–6. The mean change in clinic-measured blood pressure (systolic/diastolic) following 24 weeks of ARB and CCB combination therapy was −15.3 ± 4.8/−7.1 ± 3.2 mm Hg (both p < 0.01) in the OL group and −14.5 ± 4.6/−6.7 ± 3.1 mm Hg (both p < 0.01) in the CA group. The corresponding changes in home-measured early morning blood pressure were −16.7±5.0/−8.8 ± 3.0 mm Hg (both p < 0.01) and −13.0 ± 4.6/−6.4 ± 3.1 mm Hg (both p < 0.01). These results indicate that both OL and CA had significant antihypertensive effects, but that the lowering of home-measured early morning blood pressure was significantly greater in the OL group than in the CA group (p < 0.05) (see Figures 2 and 3).
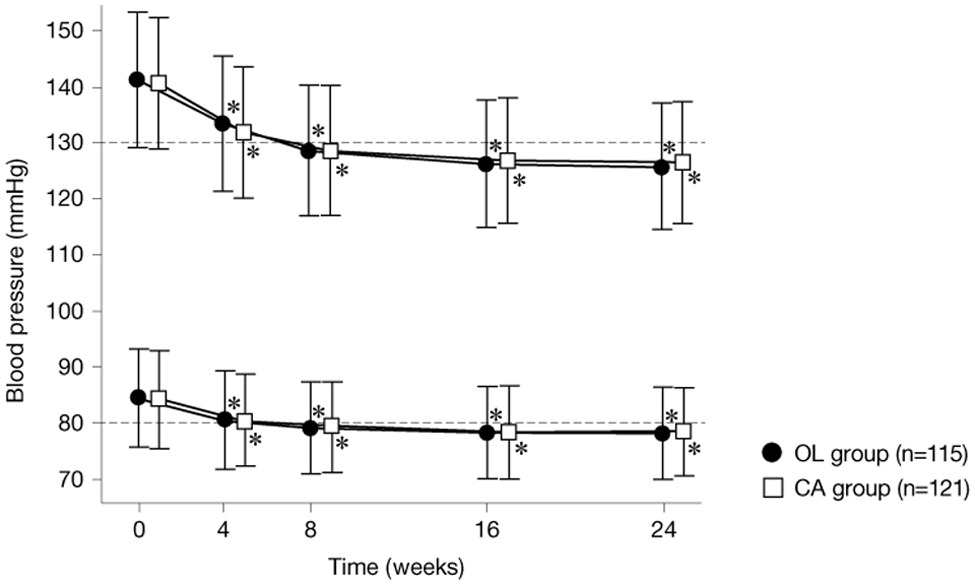
Changes in clinic-measured blood pressure.
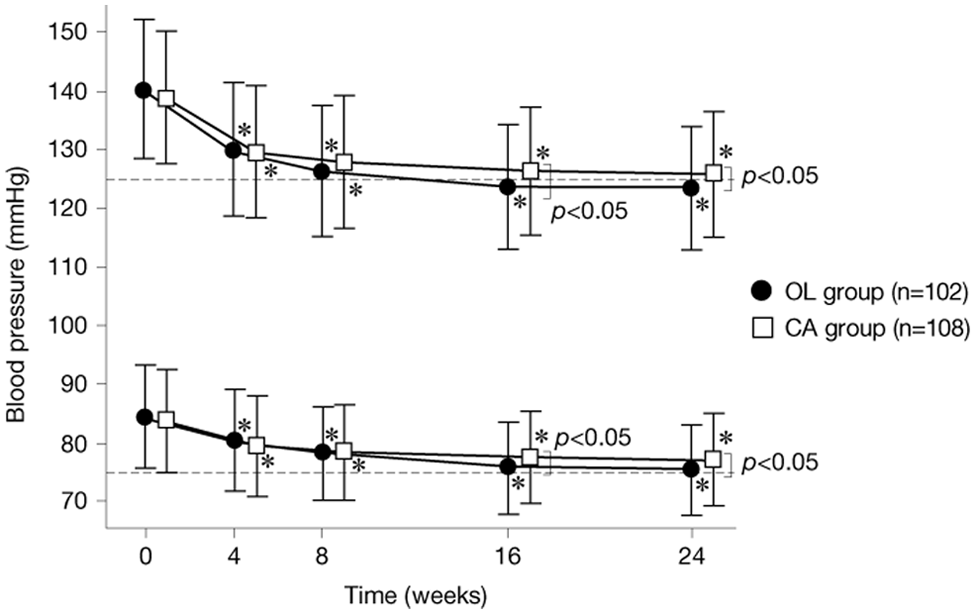
Changes in home-measured early morning blood pressure.
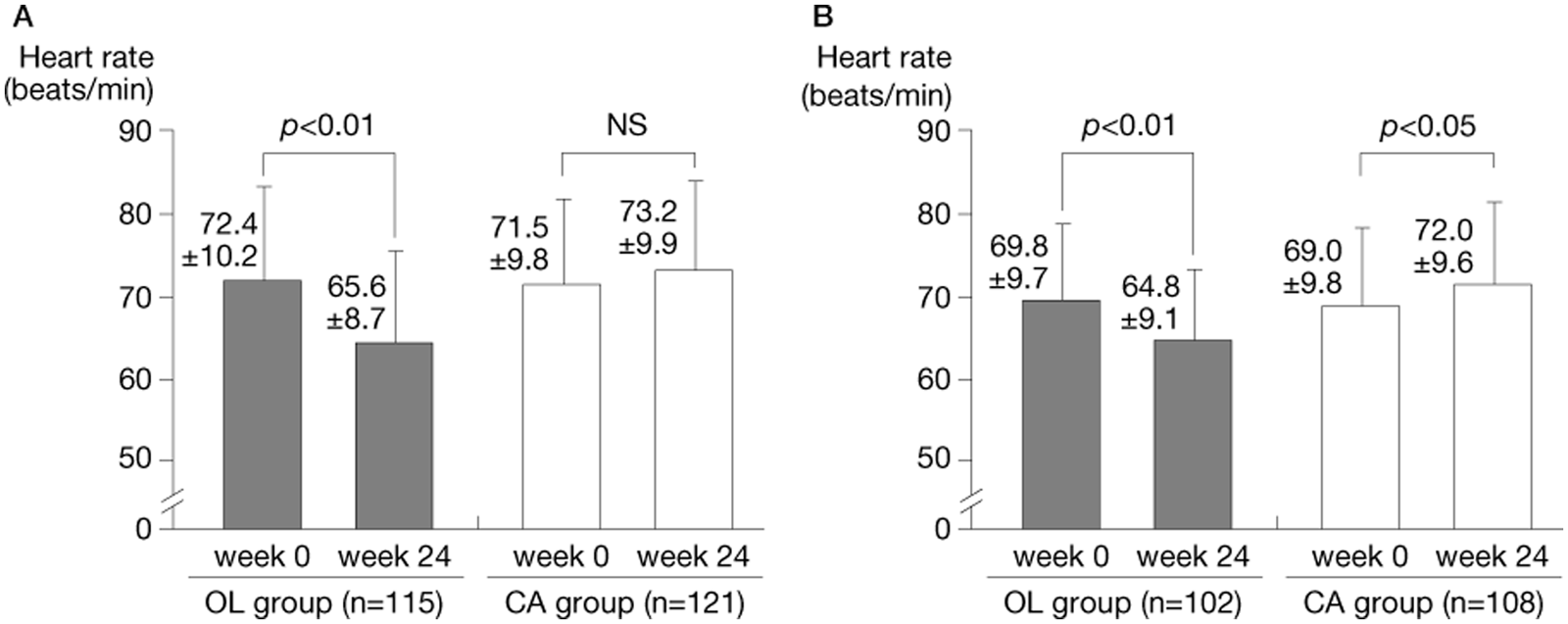
Changes in heart rate measured (A) in the clinic and (B) at home in the early morning.
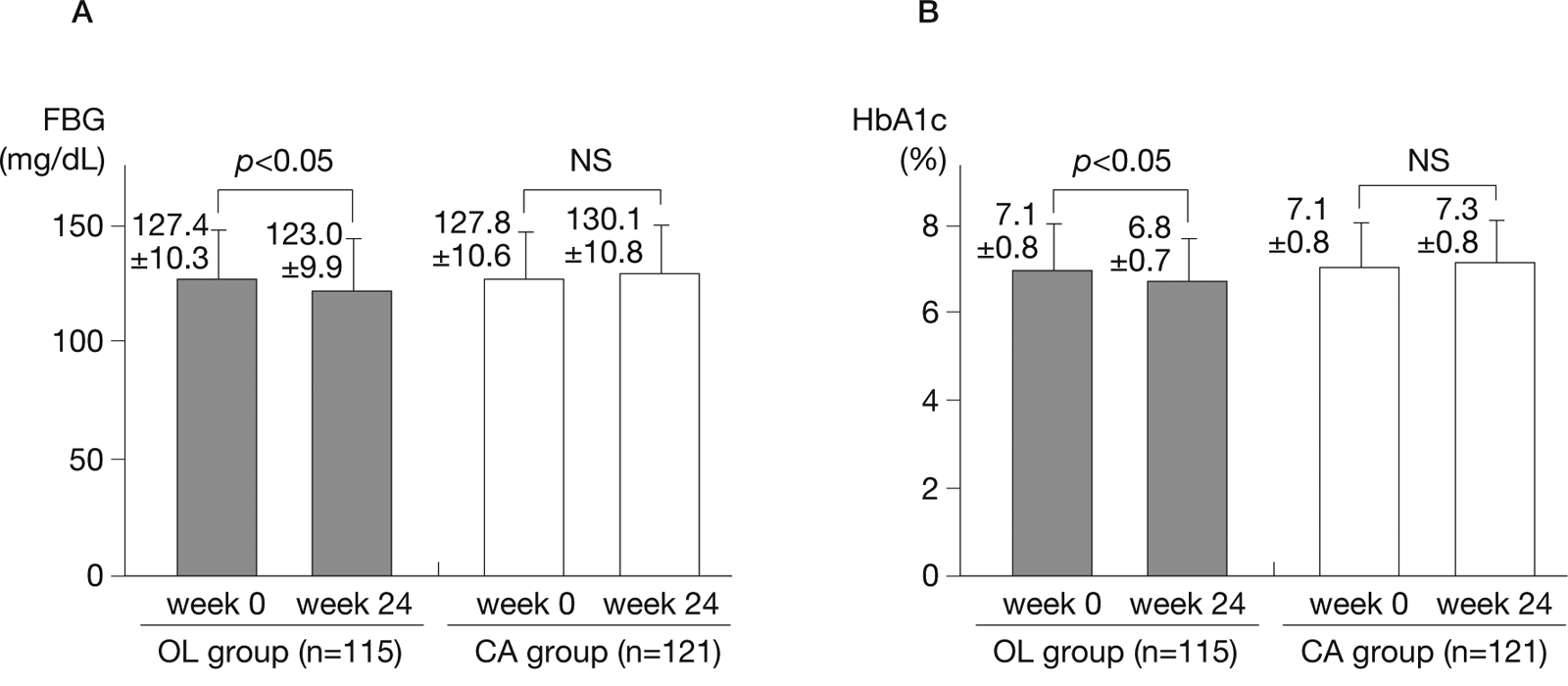
Changes in (A) fasting blood glucose (FBG) and (B) haemoglobin A1c (HbA1c) (NGSP) levels.
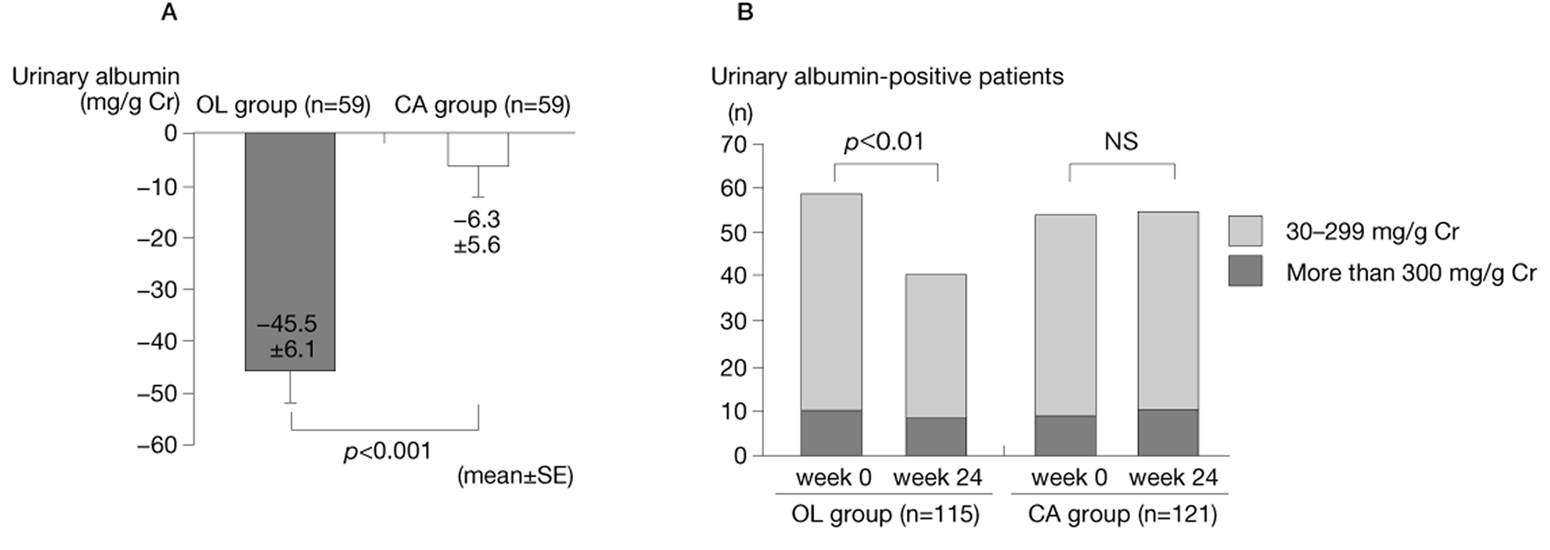
Changes in (A) urinary albumin levels and (B) the number of urinary albumin-positive patients.
Clinic-measured heart rate tended to increase while home-measured early morning heart rate increased significantly in the CA group. In contrast, these parameters decreased significantly in the OL group, with changes of −6.8 ± 2.7 beats/ minute (p < 0.01) and −5.0 ± 2.1 beats/minute (p < 0.01), respectively (see Figure 4.). In the OL group, the fasting blood glucose level decreased significantly from 127.4±10.3 mg/dL before the start of combination therapy to 123.0±9.9 mg/dL at week 24 (p < 0.05), as did the HbA1c (NGSP) level, which decreased from 7.1±0.8% to 6.8 ± 0.7% (p < 0.05) (see Figure 5). Although eGFR was not significantly different between the two groups, the urinary albumin level decreased significantly in the OL group from 175.5 ± 118.7 mg/gCr to 130.0 ± 107.4 mg/gCr (p < 0.001) and a significant decrease in the number of urinary albumin-positive patients was observed (see Figure 6).
Body weight increased very slightly in both groups, increasing from 62.2 ± 12.3 to 62.5 ± 12.7 kg in the OL group, and from 62.7 ± 12.6 to 63.0 ± 12.9 kg in the CA group. These changes were not significantly different between the two groups. Peripheral foot oedema was noted in one patient treated with OL and two patients treated with CA; however, the oedema was very mild and did not interfere with daily living.
There were no significant changes in the levels of metabolic markers, such as LDL-C, HDL-C, TG, serum uric acid, BUN, serum creatinine, Na, K or Cl in either group. Furthermore, there were no significant changes in other parameters during the study, and we detected no changes in clinical symptoms resulting from discontinuation of the study. Similarly, we noted no adverse reactions caused by administration of the study drugs and none of the patients withdrew because of adverse events.
Discussion
In this study, we found that the administration of a CCB in combination with an ARB elicited significant antihypertensive effects in hypertensive patients complicated with diabetes mellitus who did not achieve the antihypertensive targets despite usual doses of an ARB. Clinic-measured blood pressure showed similar antihypertensive effects in the OL and CA groups. However, the clinic-measured heart rate tended to increase in the CA group and it decreased significantly in the OL group. Meanwhile, patients in the OL group showed significant and persistent improvements in home-measured early morning blood pressure, compared with the CA group. Furthermore, home-measured early morning heart rate decreased significantly in the OL group.
It is known that home-measured early morning blood pressure is more strongly associated with cardiovascular events than is clinic-measured blood pressure. 13 Accordingly, antihypertensive drugs showing antihypertensive effects that persist until the early morning are desirable. In the REZALT study, the use of an automatic blood pressure meter (ABPM) showed that the effects of olmesartan in combination with azelnidipine persisted for 24 hours. 14 In other studies using an APBM, differences in blood pressure control were reported between ARBs and between CCBs. For example, in double-blind comparative studies, the 24-hour persistence of olmesartan was significantly longer than that of candesartan 12 while the persistence of azelnidipine was longer than that of amlodipine. 8 Based on these findings, it is likely that the reduction in early morning blood pressure was significantly greater in the OL group than in the CA group because of concomitant use of olmesartan and azelnidipine, which show excellent persistency.
Since the type and dosage of antidiabetic medications were not changed during the study period, the decreases in fasting blood glucose and HbA1c (NGSP) in the OL group were likely due to the effects of azelnidipine. This is because azelnidipine has been reported to improve insulin resistance 15 possibly by inhibiting sympathetic nerve activity. Palatini and Julius reported that tachycardia in hypertension results in sympathetic hyperactivity and that the heart rate itself influences cardiovascular risk factors such as hyperinsulinaemia and hyperglycaemia. 16 Moreover, an increased heart rate is considered a risk factor for type 2 diabetes mellitus because a high resting heart rate is significantly associated with obesity and diabetes mellitus. 17 The results of the present study suggest that inhibition of sympathetic nerve activity may positively influence glucose metabolism. Notably, there were no differences in clinic-measured blood pressure and heart rate, home-measured early morning blood pressure, blood glucose, HbA1c, urinary albumin, lipid or electrolytes between patients treated with or without insulin (data not shown).
It has also been reported that azelnidipine elicits significantly greater decreases in urinary protein compared with amlodipine. 10 In this study, we detected a significant decrease in the number of urinary albumin-positive patients in the OL group and renal function improved in the OL group but not in the CA group. These findings suggest that the renal disorders regressed from stage II to stage I because of the renal protective effects of OL, which normalised renal function. Since it was reported that the rate of cerebrovascular, cardiovascular and renal events decreases in patients with type 2 diabetes mellitus showing improvements in microalbuminuria, 18 the combination of olmesartan and azelnidipine is particularly appropriate for hypertensive patients complicated with diabetes mellitus in terms of cardiovascular and renal function protection. In Japan, tablets combining olmesartan with azelnidipine are now commercially available and are considered useful in terms of long-term adherence.
Conclusions
Two combinations of a CCB with an ARB elicited antihypertensive effects in hypertensive patients complicated with type 2 diabetes mellitus who did not achieve sufficient antihypertensive effects with the ARB at the usual dose. Our results suggest that the effects on the metabolic system may differ according to the combination of ARB and CCB. We observed persistent antihypertensive effects in terms of early morning hypertension in patients treated with olmesartan plus azelnidipine, in addition to decreases in heart rate, fasting blood glucose and HbA1c (NGSP), and a significant decrease in microalbuminuria. Taken together, these results indicate that, in hypertensive patients complicated with diabetes mellitus, olmesartan in combination with azelnidipine has beneficial effects in terms of blood pressure, glucose metabolism and renal protection.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest statement
None declared.
